Structural design strategies of polymer binders for Silicon-based anodes in lithium-ion batteries
Abstract
Silicon (Si) anodes have emerged as promising candidates for next-generation lithium-ion batteries owing to their high theoretical capacity. However, their practical application is hindered by severe volume expansion and unstable electrode interfaces during cycling. Polymer binders play a critical role in mitigating these issues by maintaining electrode integrity and enhancing interfacial stability. This review provides a systematic classification of polymer binders for Si-based anodes according to their binding mechanisms, including covalent, hydrogen-bonding, and supramolecular interactions. The discussion emphasizes how structural configurations, such as linear, branched, and cross-linked architectures, affect mechanical resilience, adhesion strength, and compatibility with high-loading electrodes. Recent advances in multifunctional and dynamic cross-linked binders are highlighted, with particular focus on strategies to accommodate large volume changes and suppress interfacial degradation. In addition, a comparative analysis of advantages and limitations for various binder systems is provided, along with perspectives on future development trends. This work aims to guide the rational design of polymer binders for achieving stable, high-energy-density Si-based anodes.
Keywords
INTRODUCTION
Lithium-ion batteries (LIBs), which are deeply integrated in our daily lives, are a type of secondary battery that operates based on redox electrochemical reactions involving lithium ions. Among all metals, lithium is the lightest and has a very low standard reduction potential, enabling lithium-based batteries to deliver not only high specific capacities but also high operating voltages exceeding 3 V. Owing to these advantages, LIBs have become essential for powering small electronic devices, such as mobile phones, and are also regarded as one of the most promising candidates for electric vehicles, a market expected to grow rapidly in the near future[1-4]. The most commonly used anode material in LIBs is graphite, which is cost-effective and offers excellent cycling stability. However, its practical capacity is limited to approximately
Si is well known to be abundant in nature, inexpensive, and possesses a remarkably high theoretical capacity of approximately 4,200 mAh g-1[18-35], which is more than ten times that of graphite. Nevertheless, Si undergoes drastic volume expansion up to 400% during lithiation and shrinkage during delithiation[36-41]. These extreme volume fluctuations induce mechanical failures, such as pulverization, electrode delamination, and layer separation[42-47], ultimately leading to capacity degradation caused by electrically isolated particles and irreversible lithium consumption. To address these challenges, polymer binders have emerged as critical components in Si-based electrodes. Typically, binders serve as additives that maintain the adhesion between active material particles and between the electrode and the current collector, thereby preserving the mechanical integrity of the electrode[48-51]. In the case of Si anodes, recent studies have emphasized that polymer binders play a far more critical role than previously considered, significantly influencing long-term cycling stability. A polymer binder is the only component in the electrode that ensures stable adhesion without inducing undesirable side reactions in organic electrolyte systems, even during repeated charge-discharge cycles. It can accommodate the extreme volume changes of Si, thereby maintaining mechanical stability and consistent electrochemical performance. Although the binder occupies only a small fraction of the total electrode mass, it plays a crucial role in maintaining cohesion among active materials, conductive additives, and the current collector, and in some cases contributes to enhanced ionic or electronic conductivity.
This review aims to systematically classify the various polymer binders reported to date based on their binding mechanisms, with particular emphasis on polymer-polymer and polymer-surface interactions, molecular structures, and mechanical properties. Through this analysis, we intend to provide a comprehensive understanding of how structural and interfacial design strategies affect the electrochemical performance of Si-based anodes. Over the past two decades, the development of polymer binders for Si anodes has progressed from the use of conventional PVDF binders to water-based CMC/SBR systems, and more recently to PAA-based formulations capable of accommodating high-Si loading. This evolution is summarized in Figure 1, which presents an industrially oriented timeline of binder development, highlighting representative milestones and their technological significance. By establishing this historical and structural context, the Introduction provides a clear framework for readers to follow the detailed discussions and critical perspectives presented in the subsequent sections of this review.
Figure 1. Roadmap of binder evolution for Si-based anodes. Conventional PVDF binders (2000-Present) exhibit high chemical stability but suffer from weak adhesion and poor elasticity. CMC/SBR systems (2005-Present) introduce water-based processing and improved adhesion, though stability is limited to low Si content. PAA binders (2010-Emerging) provide strong Si adhesion via -COOH anchoring and enable higher Si loading. Hybrid systems (2015-Future/End) feature cross-linked/branched architectures with improved toughness, self-healing behavior, and scalability toward industrial applications. The original figure was created by the authors.
WORKING FUNCTIONS OF BINDERS
This section outlines the essential roles of polymer binders in Si-based anodes, linking molecular-level interactions to macroscopic electrode behavior. We discuss how binders contribute to mechanical integrity, interfacial stability, and ion/electron transport, and identify key performance metrics that guide binder design for high-capacity applications.
Interaction mechanisms between polymer binders and electrode constituents
Polymer binders used in battery electrodes are applied in small quantities, typically below 3 wt% of the total electrode composition. Despite their limited proportion, significant research has been devoted to enhancing high-energy battery performance by engineering the chemical properties of polymeric binders. A comprehensive understanding of both the functional roles of binders in LIB electrodes and the operational mechanisms of Si anodes is essential for developing advanced binder systems that improve electrochemical performance. Various binder materials have been designed through structural tailoring and the incorporation of functional moieties to achieve high elasticity, strong interfacial adhesion, and mechanical flexibility. The following sections explore these roles in greater detail within the context of Si-based anodes. Although the adhesive mechanisms of binders are inherently complex and cannot be fully described by a single model, the most widely accepted binding mechanisms include mechanical interlocking, chemical bonding, van der Waals interactions, and electrostatic forces[52]. When binders interact with active material and conductive additive particles, the system typically forms three distinct interfacial layers[53,54]. First, upon dispersion in a solvent, the binder spreads uniformly across particle surfaces, forming a primary adhesion layer. Surrounding this, a secondary immobilized layer is established, where molecular-level interactions anchor adjacent polymer chains, reinforcing structural cohesion. Finally, residual free polymer chains give rise to an outer surplus layer, which contributes to the overall mechanical and chemical properties of the binder system. The specific configuration and properties of these interfacial layers are largely governed by the inherent characteristics of the polymer and the surface chemistry of the contacting materials. Variations in polymer composition, molecular weight, and functional groups can significantly influence how the binder interacts with different particle surfaces, thereby affecting interfacial stability and electrode performance
Figure 2. Schematic illustration of the functional roles of polymer binders in electrodes (A) Binding action; (B) Mitigation of volume expansion; (C) Dispersion of active materials; (D) Facilitation of ionic conductivity; (E) Enhancement of electronic conductivity; (F) Contribution to SEI formation. The original figure was created by the authors.
Maintaining structural integrity of Si electrodes with binders
The mechanical properties imparted by polymer binders play a critical role in accommodating the substantial volumetric fluctuations experienced by Si electrodes during cycling [Figure 2B]. To effectively mitigate the mechanical stress induced by expansion and contraction, binders must exhibit key characteristics, such as tensile strength, adhesive strength, flexibility, and elasticity[55]. When the interaction between the binder and Si particles is sufficiently strong, the resulting interfacial adhesion can exceed the stress generated by volume expansion, thereby stabilizing the electrode interface and maintaining structural integrity over repeated cycles. In addition to mechanical reinforcement, another essential function of polymer binders lies in their ability to disperse electrode components uniformly throughout the slurry. The primary role of a binder is to uniformly bind the active material and conductive agents via dispersion in solvent and to facilitate the formation of a homogeneous electrode slurry that coats the current collector evenly during the drying process [Figure 2C]. One of the critical challenges during slurry preparation is the propensity of Si particles to aggregate with conductive additives, which typically exhibit high specific surface area and fine particle size. This non-uniform dispersion can disrupt the uniformity of the electrode architecture and impair electrochemical performance. Therefore, polymer binders should be designed to promote homogeneous distribution of all electrode components throughout the mixing process.
In particular, polymers containing aromatic moieties are highly effective in dispersing carbon-based conductive agents via π-π stacking interactions between the aromatic rings of the binder and the graphitic surfaces of the additives. These noncovalent interactions suppress aggregation and promote uniform dispersion, thereby enhancing the formation of a continuous electronic network. As a result, both electronic and ionic transport within the electrode are improved [Figure 2D and E]. Given the importance of these functions, a wide range of polymeric materials has been employed as binders in LIB electrodes, each offering distinct structural and chemical properties.
Moreover, the solid electrolyte interphase (SEI) formed on the surface of active material critically influences the electrochemical behavior of LIBs [Figure 2F]. In Si anodes, continuous volume changes expose new surfaces, leading to ongoing SEI formation. The composition of the SEI layer can vary depending on the electrolyte used, and the resulting surface chemistry can affect the adhesion of the binder. The addition of binders plays a key role in promoting uniform dispersion of electrode components, which contributes not only to a more homogeneous SEI but also to a more stable interfacial structure. A uniformly distributed SEI is beneficial in reducing surface resistance during electrochemical reactions. In Si anodes, the SEI is primarily composed of reduction products such as Li2CO3, LiF, and Li2O[54]. Among these, LiF is particularly important for maintaining the mechanical and chemical stability of the SEI[56]. Therefore, the formation of a stable SEI of appropriate thickness on the active material surface is critical for maintaining electrode capacity and prolonging cycle life. In addition to mechanical stability and SEI formation, the chemical stability of polymer binders toward liquid electrolytes is a critical factor affecting the long-term performance of Si anodes. During extended cycling, binders may undergo swelling, partial dissolution, or degradation due to reactions with electrolyte components, such as LiPF6 salt and carbonate solvents. Such degradation can weaken interfacial adhesion and disrupt the electronic network within the electrode. Functional groups in the binder, including -OH, -COOH, and amide moieties, can interact with electrolyte additives, such as fluoroethylene carbonate (FEC), influencing SEI composition and stability. As reported in a recent study by An et al.,[57] optimizing the chemical compatibility between the binder and electrolyte is essential to suppress undesirable side reactions and maintain the mechanical integrity of the electrode. Therefore, designing binders with enhanced chemical resistance, reduced solubility in electrolyte, and the capability to maintain robust adhesion under electrochemical stress is critical for improving both cycling stability and coulombic efficiency of Si-based electrodes. Polymer binders aid in this process by providing mechanical support, preventing interfacial collapse during repeated volume changes of Si[58]. Following the discussion on the mechanical properties and dispersibility of polymer binders, it is also essential to examine how molecular-level interactions, particularly those involving functional groups, govern the performance of Si anodes.
For Si anodes, polymers have been actively engineered not only to improve electrolyte wettability and ionic/electronic conductivity, but also to reinforce mechanical robustness and electrochemical stability, both of which are crucial for managing drastic volume changes during cycling. Therefore, the molecular architecture of the binder polymer is a key determinant of electrode performance.
CLASSIFICATION OF POLYMER BINDERS BY ORIGIN
Si tends to be oxidized in air to form a surface layer of SiO2 (or SiOx), which contains hydroxyl (-OH) groups that impart hydrophilic properties to the surface[59-63]. Hydrophilic functional groups improve electrolyte wettability and dispersion, which can facilitate ion transport in the composite electrode. These polymers not only form stronger interfacial bonds with Si particles but also improve adhesion to the current collector, thereby contributing to improved structural stability of the electrode. Furthermore, the diversity and density of functional groups within the binder matrix give rise to various types of molecular interactions such as hydrogen bonding, electrostatic attraction, and van der Waals forces that collectively influence the electrode’s mechanical integrity and cycling performance. As these interactions are highly dependent on the chemical functionalities present in the polymer backbone, optimizing the binder composition to tailor such interactions is a key strategy in the design of next-generation binders for high-capacity Si anodes. While the chemical functionality of polymer binders governs interfacial behavior at the molecular scale, their three-dimensional (3D) structural architecture plays an equally important role in maintaining mechanical durability. The degree of polymer chain branching, cross-linking, and network formation determines the binder’s capacity to accommodate internal stress and to preserve the mechanical stability of the electrode during continuous cycling.
Polymer binders are commonly categorized into linear, branched, and cross-linked structures, each offering distinct mechanical responses to the significant volume changes associated with Si anodes [Figure 3][64,65]. Depending on their structural configuration, these binders exhibit varying capabilities to buffer or relieve mechanical stress during electrode expansion and contraction. To address the mechanical degradation associated with volumetric fluctuations of Si, recent research has focused on developing binders with networked polymer architectures. These systems introduce multiple interaction points through covalent bonds or combined covalent and noncovalent linkages, which limit polymer chain deformation and improve mechanical resilience[55]. Such reinforced polymer networks enable Si anodes to better tolerate repeated volume expansion and contractions, ultimately contributing to enhanced structural stability and prolonged cycle life.
Figure 3. Representative polymer structures formed by polymerization: linear, branched/grafted, and cross-linked networks. This original figure was created by the authors.
Natural polymer binders
In this subsection, we focus on naturally derived polysaccharide binders, which offer environmental benefits, aqueous processability, and abundant functional groups for Si adhesion. We discuss their advantages, performance limitations at high Si loading, and industrial considerations for large-scale implementation.
Naturally derived polysaccharides (e.g., CMC, chitosan, alginate, carrageenan) provide dense networks of
From a manufacturing viewpoint, we find that controlling substitution/oxidation levels and ionic strength is as important as choosing the polysaccharide family itself, because these parameters dictate slurry stability and coating uniformity at line scale. Representative examples include Carboxymethyl cellulose (CMC)
Figure 4. Chemical structures of polymer binders containing hydrophilic functional groups and their effects on Si anode performance. (A) CMC; (B) SA; (C) CS; (D) L-CGN; (E) Design of the binder material (left) and cycling stability (right) of a Si anode using a natural rubber/chitosan network binder. Adapted with permission from ref.[72]; (F) Chemical structures of the mannuronic and guluronic acid repeat units (left); cycling performance of nano-Si anodes with alginate (SA), CMC, and PVDF binders (right; 0.01-1.0 V vs. Li/Li+). Adapted from ref.[64]; (G) Catechol-conjugated polymer binders and cycling performance of Si electrodes using Alg-C, Alg, and PVDF binders. Adapted from ref.[64]; (H) Cycling performance of Si electrodes using different natural polymer binders (CGN, PAA, SA, CMC). The Si@CGN electrode demonstrated the highest capacity retention over 100 cycles, underscoring the superior binding and structural stabilizing effect of lambda carrageenan. Adapted with permission from ref.[71]; (I) Schematic illustration of the synthesis of Ox-L-CGN, comparison of its solubility in distilled water with that of L-CGN, and cycling performance of Si electrodes using different polymer binders. The optimized Ox L-CGN 5 binder demonstrated improved solubility and enhanced electrochemical performance, attributed to its optimized viscosity and formation of a stable polymer network. Adapted with permission from ref.[70].
In addition to polysaccharides, various other bio-based polymers have been explored as sustainable binder materials. Jang et al. designed a nature-inspired L-CGN binder derived from sulfated polysaccharides of red algae[71]. This binder contains abundant -OH and -SO3- functional groups, which enable strong hydrogen bonding with the -OH groups on the Si surface. As a result, even after 50 cycles, the CGN binder demonstrated superior interfacial adhesion to Si particles and internal cohesion within the electrode, as confirmed by SEM analysis, compared to conventional binders such as SA, CMC, and PAA. This case highlights the effective applicability of naturally derived functional groups in Si anode binder systems
Notably, excessive oxidation (Ox-L-CGN-10) hindered effective network formation, resulting in increased electrode pulverization and unstable SEI regeneration, which significantly degraded electrochemical performance. This degradation is presumed to result from reduced solubility, which compromised the structural integrity and stability of the water-processable binder system [Figure 4I][70]. Sulfated polysaccharides can enhance adhesion and internal cohesion via hydrogen bonding and electrostatic interactions; controlled oxidation lowers viscosity and improves coatability, yet over-oxidation weakens network formation and accelerates pulverization at high strain. Despite their appeal in terms of sustainability and processing advantages, natural polymer binders pose challenges, such as irregular molecular weights and complex chemical structures, which can lead to inconsistent performance. Nevertheless, the environmental and functional benefits of naturally derived materials continue to drive intensive research in this area. While polysaccharides, such as guar gum (GG) and gum arabic (GA), have been employed as components in certain binder systems, they are not considered key materials for Si anode applications and are thus excluded from the scope of this study.
Synthetic polymer binders
This subsection examines synthetic binders, highlighting their tunable architectures and ability to integrate functional groups not easily accessible in natural polymers. We outline how molecular design impacts mechanical resilience, adhesion to Si, and compatibility with high-solid-loading slurry processes.
Synthetic binders offer tunable architectures and functionalities that are difficult to achieve with natural polymers. For Si electrodes, the central design problem is to combine strong interfacial bonding with elastic stress dissipation and electrolyte compatibility while maintaining slurry processability at high solid loading. Here we synthesize cross-study evidence into three takeaways: linear fluorinated baselines are robust but adhesion-limited on Si; acrylic chemistries (e.g., PAA) add dense -COOH sites and stiffness for higher Si content; and architectural modifications (branched/cross-linked/dynamic) are required when areal loading and volumetric strain exceed the elastic window of linear binders. In the following discussion, we focus on representative synthetic binders and their performance-structure relationships, beginning with the widely used fluorinated baseline PVDF and progressing to non-fluorinated acrylic and nitrile-based systems. Notably, synthetic polymers, like polyvinylidene fluoride (PVDF) [Figure 5A][75,76], a fluoropolymer, as well as non-fluorinated options like polyacrylic acid (PAA) [Figure 5B][11,75,77-80], and Polyacrylonitrile (PAN)
Figure 5. Chemical structures of synthetic polymer-based binders and their effects on Si anode performance. (A) PVDF; (B) PAA; (C) PVA. PVDF-based binder strategies; (D) Cross-sectional SEM images of Si-graphite/PVDF composite electrodes before and after the first cycle at various temperatures, along with the corresponding cycling performance of the Si anode. Adapted with permission from ref.[82]; (E) Introduction of Si-functional groups via an azide-nitrile click reaction on a PAN-based polymer and the resulting electrochemical performance. Adapted with permission from ref.[87]; (F) Schematic illustration of ion transport pathways and structural evolution in PAA@Si and PPD PAA@Si electrodes during cycling. The long-term cycling performance of Si electrodes using PAA, CMC SBR, and PPD PAA binders at a current density of 1.2 A g-1 is also presented. Adapted with permission from ref.[39].
In our view, PVDF is an excellent baseline for graphite but structurally mismatched to high-strain Si. PAA-centered formulations currently offer the most pragmatic route to scale, provided viscosity and elasticity are co-optimized to avoid cracking at high Si loading. In addition to PAA, PAN has also been investigated as a binder, owing to the polar nature of its nitrile groups (-CN)[81]. PAN’s nitrile (-C≡N) groups confer high polarity, improving interfacial adhesion to Si and enhancing slurry rheology and electrolyte wettability rather than creating intrinsic ion-conducting pathways within the binder phase. High-molecular-weight PAN forms entangled chains that wrap and bridge Si particles, increasing mechanical integrity and adhesion; however, its intrinsic electronic conductivity remains low, and neat PAN binders may exhibit brittleness under large strains. In practice, PAN is less widely adopted than PVDF or PAA owing to higher cost and more complex processing, although targeted functionalization (e.g., silane coupling, click chemistry) has yielded hybrid organic-inorganic networks with improved durability. These modifications, while effective, must be balanced against synthesis complexity and solvent compatibility to ensure feasibility for large-scale manufacturing. The strong polarity of the nitrogen atom in the nitrile group improves both ionic conductivity and interfacial adhesion with the Si surface. One key advantage of high molecular weight PAN is its ability to form entangled polymer networks around Si particles, thereby improving mechanical integrity and adhesion. This structural advantage results in a notable improvement in capacity retention -37.95% for high-molecular-weight PAN compared with 17.5% for PVDF[86]. While the nitrile group imparts beneficial characteristics, the intrinsic conductivity of PAN remains limited. Consequently, further improvements can be achieved by compositing PAN with conductive additives or applying doping strategies. For example, a study utilizing an azide nitrile click reaction to enhance adhesion of PAN to Si showed a significant improvement in capacity retention, achieving 522 mAh g-1 after 50 cycles compared to just 152 mAh g-1 for unmodified PAN [Figure 5E][87]. This work highlights a promising approach wherein silane-treated PAN (Si-PAN) was integrated into Si alloy electrodes. The Si-PAN binder forms a hybrid organic-inorganic structure through silane functionalization, enhancing both electrode durability and electrochemical performance. Notably, Si alloy electrodes fabricated with the optimized Si-PAN binder and thermally annealed at 450 °C delivered an initial discharge capacity of approximately
Table 1 summarizes representative linear binders used for Si anodes, including their structural features and functional groups. These binders offer good processability and enable polar interactions with Si particles; however, their limited mechanical reversibility often restricts long-term cycling stability.
Summary of representative linear binders and their characteristics for Si anodes
| Binder | Feature | Structure | Interaction | Ref. |
| CMC | High dispersion High viscosity | Linear | Hydroxyl group Carboxylate | [110] |
| SA | High viscosity | Linear | Hydroxyl group Carboxylate | [69] |
| CS | Strong hydrogen bonding | Linear | Hydroxyl group Amino group | [111] |
| GA | High mechanical property | Linear | Hydroxyl group | [112] |
| GG | High viscosity | Linear | Hydroxyl group | [113] |
| PVDF | Poor elasticity | Linear | Fluorine atom | [114] |
| PAN | High polarity/intrinsically low electronic conductivity | Linear | Nitrile group | [86] |
| PVA | High adhesion | Linear | Hydroxyl group | [115] |
| PI | High mechanical characteristics | Linear | Carbonyl group | [17,116] |
| PAA | High mechanical characteristics High viscosity | Linear | Carboxyl group | [117] |
Linear binders, such as PVDF, PAA, and PAN, offer well-defined molecular structures, high processability, and strong polar interactions with Si surfaces, making them compatible with scalable electrode fabrication. However, their limited mechanical reversibility and susceptibility to structural degradation during repeated cycling remain critical challenges, often restricting long-term stability. In particular, PAA, while not delivering the highest electrochemical performance, has attracted significant attention in the domestic cell industry due to its low cost and compatibility with aqueous processing.
Current R&D efforts focus on optimizing slurry viscosity and improving adhesion to enhance electrode integrity without compromising manufacturability. Future developments are expected to focus on controlling molecular weight, diversifying functional groups, and developing hybridization strategies to balance mechanical resilience with ionic/electronic conductivity in next-generation Si-based anodes. Recent studies have also explored advanced linear synthetic binder architectures, including copolymer systems with zwitterionic functional groups and supramolecular host-guest assemblies, which have demonstrated improved Li+ transport, enhanced interfacial adhesion, and superior cycling stability under high-Si-loading and industrially relevant conditions[88]
STRUCTURALLY ENGINEERED BINDERS
This section compares binders engineered with branched, covalently cross-linked, and dynamically cross-linked architectures. We identify how each structural motif contributes to stress buffering, crack suppression, and cycling stability, and we assess the trade-offs between mechanical robustness and processing feasibility.
The molecular structure of the polymer plays a crucial role in determining the effectiveness of the binder. The binders discussed above consist of linear polymer chains, where interactions primarily arise from noncovalent bonding between functional groups. As an alternative design strategy, the incorporation of covalent bonds or dynamic physical interactions between polymer chains, often associated with self-healing capabilities, can markedly improve the electrochemical performance of Si anodes.
Branched or grafted polymer binders
Here, we examine branched and grafted binder architectures, which introduce multiple interaction sites for enhanced adhesion and stress dispersion. We assess their performance benefits, synthesis challenges, and potential as an intermediate strategy between linear and fully cross-linked systems.
Branched or grafted architectures increase the density and spatial distribution of interaction sites relative to linear analogs, improving multi-point anchoring and stress dispersion around Si particles. Across reported systems, elasticity and capacity retention benefit from side-chain mediated hydrogen bonding/ionic interactions, yet synthesis complexity and viscosity management remain practical bottlenecks. We therefore view branched motifs as effective intermediates especially when used to tune chain mobility and solvent uptake between simple linear baselines and fully cross-linked networks[32,89,90]. Recent studies have demonstrated that compared to linear binders, branched architectures or grafted systems composed of functional moieties can better withstand the mechanical stress associated with Si expansion. Among these approaches, grafting functional groups onto a polymer backbone to generate side chains has been shown to improve both mechanical strength and interfacial adhesion in binder systems. For instance, He et al. developed a branched-type water-soluble binder, poly(vinyl alcohol)-grafted poly(acrylic acid) (PVA-g-PAA), synthesized via esterification between the hydroxyl groups of PVA and the carboxyl groups of PAA
Figure 6. Electrochemical performance and structural stability of Si anodes employing polymeric branched binders. (A) Cycling performance of a Si electrode using the Si@PVA-g-10PAA binder over 1,000 cycles at a current density of 400 mA g-1, accompanied by rate performance comparisons with various binders including PVA, PVA-g-10PAA, PVA-g-15PAA, PAA, and CMC. Adapted with permission from ref.[89]; (B) Schematic illustration of the interaction between Si particles and the CA-g-PAA binder. Cross-sectional SEM images of Si electrodes with PAA and CA-g-PAA binders before cycling and after 5 and 50 cycles, along with corresponding cycling performance using different binders. Adapted with permission from ref.[32]; (C) Schematic illustration and long-term cycling performance of Si/graphite composite anodes using Na-PAA/SBR and Na-PAA-g-SBR binders, demonstrating the superior stability and capacity retention imparted by the grafted binder system. Adapted with permission from ref.[90].
Branched-like polymer architectures offer enhanced multi-point anchoring and network connectivity compared to their linear counterparts, enabling improved adhesion to Si surfaces and better stress distribution during cycling. The incorporation of multiple functional side chains can facilitate both mechanical stability and Li-ion transport pathways, thereby enhancing long-term capacity retention. However, the synthesis of branched polymers often requires more complex monomer feed ratios and reaction controls, which can lead to batch-to-batch variability. Additionally, higher viscosity in aqueous processing may limit slurry uniformity at a large scale. To advance industrial applicability, future work should prioritize scalable synthetic strategies, optimized branching density, and cost-efficient monomers that balance mechanical robustness with manufacturability.
Cross-linked polymer binders
In this section, the term “cross-linked” encompasses both covalent and physical network formations. While covalent cross-links (e.g., esterification, amidation) provide permanent structural reinforcement, physical cross-links (e.g., hydrogen bonding, ionic interactions, supramolecular assemblies) can also deliver significant mechanical and interfacial benefits. Both types are considered here due to their relevance in suppressing Si anode pulverization and delamination. To overcome the limitations of conventional linear polymer binders, cross-linked polymer systems have been extensively explored. These systems form strong covalent bonds between polymer chains, thereby achieving high mechanical strength, an essential property for preserving the structural integrity of Si anodes during repeated cycling. On a molecular level, covalent bonding between the binder and hydroxyl-terminated Si surfaces (Si-OH) helps mitigate stress from volumetric expansion. Covalently cross-linked binders form robust three-dimensional networks that effectively suppress active material pulverization and electrode delamination, but their structural irreversibility can limit long-term adaptability under extreme cycling. Hybrid designs incorporating partially dynamic cross-links have recently been explored to mitigate this trade-off. However, since these covalent bonds are typically irreversible, they cannot reform once broken, which may lead to a gradual deterioration in adhesion between the binder and Si particles. Despite this limitation, chemically cross-linked binders built on well-chosen polymer backbones have demonstrated excellent long-term cycling performance. Their interconnected polymer networks effectively accommodate the repeated volume changes of Si while maintaining mechanical cohesion and interfacial adhesion over extended cycles[93,94]. A notable challenge is that highly cross-linked binders are often insoluble in common solvents, necessitating thermal activation or specialized processing to form stable networks. A representative example is the thermally cross-linked PAA-PVA binder system, where esterification occurs between the hydroxyl groups of PVA and the carboxyl groups of PAA under elevated temperatures[69,89]. The resulting interpenetrating polymer network (IPN) significantly enhances mechanical robustness and provides abundant functional groups for strong interactions with Si surfaces. This architecture effectively stabilizes the electrode against Si expansion/contraction during lithiation and delithiation, while outperforming linear alternatives in both adhesion and elasticity. Notably, the PAA-PVA binder delivered a stable capacity of ~2,283 mAh g-1 over 100 cycles, surpassing PVDF and NaCMC systems [Figure 7A][95]. Cross-linked binders also excel in suppressing electrode thickness changes during cycling. For instance, electrodes using linear binders such as SA experienced irreversible thickness growth of ~16.5% after just five cycles. In contrast, PAA-PVA cross-linked binders limited this change to ~4.5%, owing to the rigidity of their IPN structure[95]. In another study, a cross-linked binder synthesized via esterification between PAA and CMC retained over 60% of its initial capacity after 100 cycles at 0.5 C, even under high mechanical stress[84], highlighting the structural resilience of the covalent network.
Figure 7. (A) Schematic illustration of the interaction between cross-linked PAA-PVA and the Si anode, and the cycling performance of Si electrodes using PAA-PVA, NaCMC, and PAA binders. Adapted from ref.[64]; (B) Schematic illustration of polymer interaction among PAA, NaCMC, and functionalized SCNT, enhancing mechanical integrity, and peeling test results and cycle performance comparison among μSi electrodes using PAA-NaCMC 1, PAA, and NaCMC binders. Adapted with permission from ref.[34]; (C) Schematic illustration of Si/C composite anodes incorporating the cross-linked binder P(SH-BA). Rate capability comparison of Si/C450 electrodes with various binder systems. Long-term cycling performance and Coulombic efficiency of coin-type Si/C600//Li half cells employing P(SH-BA3%) and PSH binders (tested at 0.2 C). Adapted with permission from ref.[13]; (D) Schematic representations of LiPAA, TA, and SS, followed by a depiction of the physicochemically cross-linked LPTS composite polymer binder and its functional integration into Si-based anodes during lithiation/delithiation. Initial galvanostatic charge-discharge profiles of Si/PTS, Si/LPT, Si/LPS, and Si/LPTS electrodes at 0.1 C are shown, along with their cycling stability at 0.5 C, highlighting the improved electrochemical performance enabled by the LPTS binder system. Adapted with permission from ref.[38]; (E) Schematic illustration of the Si/CPU-PAA anode architecture and hierarchical stress-dissipating polymeric network. Cycling performance of Si anodes in half cells at 1 A g-1 with a high Si mass loading of
Zhang et al. also introduced a chemically cross-linked binder system comprising PAA, CMC, and CNT-NH2 to improve mechanical and electrical properties [Figure 7B][34]. Spontaneous esterification and amidation reactions, which involve the formation of ester bonds between -COOH and -OH groups and amide bonds between -COOH and -NH2 groups, occurred during electrode fabrication and resulted in a stable three-dimensional polymer network. This structure buffered Si expansion while preserving electronic pathways, resulting in improved cycling stability and overall performance. Recent studies have further demonstrated the potential of multifunctional cross-linked networks for Si anodes[96]. For example, a CA@CMC binder system formed via esterification between the -COOH groups of citric acid and the -OH groups of CMC created a mechanically robust, three-dimensional covalent network that not only mitigated Si pulverization but also
Xiao et al. developed a multifunctional composite binder composed of LiPAA, tannic acid (TA), and silk sericin (SS), forming a physically cross-linked network via hydrogen bonding [Figure 7D][38]. TA acts as a molecular bridge between the carboxyl groups of LiPAA and the amino/hydroxyl groups of SS, forming an interpenetrating supramolecular network. This structure provided superior elasticity, cohesion, and adhesion. The Si/LPTS electrode showed excellent rate capability, delivering over 1,500 mAh g-1 even at 2 C, and nearly full capacity recovery at 0.1 C. The use of silk sericin, a biodegradable protein, enhances structural stability and environmental compatibility, offering a practical and sustainable solution for Si anodes. He et al. developed a dual cross-linked binder system composed of cross-linked polyurethane (CPU) and PAA, where covalent and hydrogen bonds synergistically construct a physicochemical network
Covalently cross-linked binders provide robust, permanent networks that effectively suppress Si pulverization and electrode delamination, delivering high mechanical strength and stable cycling even under high areal loadings. Their dense network structure minimizes irreversible deformation, maintaining electrical and ionic pathways over prolonged operation. Nonetheless, the irreversible nature of covalent bonds can limit adaptability to extreme volumetric changes, and the multi-step synthesis or thermal curing requirements may increase production cost and energy consumption. Future research should focus on hybrid cross-linking approaches that integrate partial reversibility, reduce curing temperatures, and employ eco-friendly, water-based processing routes to bridge the gap between laboratory performance and industrial-scale electrode fabrication.
Dynamically cross-linked polymer binders
Here, we review binders that employ reversible cross-links to enable autonomous healing and stress relaxation. We analyze their potential to combine mechanical adaptability with ionic conductivity, while addressing the challenges of bond lifetime control, rheology management, and scalability.
Self-healing polymers refer to materials in which physically separated chains can recombine through the formation of new bonds at reactive sites on adjacent chains via dynamic cross-linking mechanisms[97]. Unlike conventional cross-linking, in which broken bonds are irreversible, dynamic cross-linking enables the formation of new interactions at alternative active sites even after bond dissociation. This mechanism is driven by a multitude of noncovalent interactions, such as hydrogen bonding, ionic interactions, and π-π stacking, acting collectively like microscopic Velcro. These reversible bonds allow the material to adapt to mechanical deformation by temporarily dissociating and reassembling, thereby enabling flexible and self-healing behavior. In the context of Si anodes, the use of self-healing binders helps mitigate the mechanical stress induced by large volume expansion. Upon expansion, the cross-linked polymer network stretches to dissipate mechanical stress, while upon contraction, it reverts to its original configuration by re-establishing bonds and healing microcracks. One representative example is the dynamic host-guest system utilizing cyclodextrin (β-cyclodextrin polymer, β-CD) and adamantane groups by Kwon et al. [Figure 8A][97]. The hydrophobic interior and hydrophilic exterior of β-CD enable specific supramolecular binding with adamantane, and when combined with a multifunctional adamantine (AD) linker, the resulting network achieved ~90% capacity retention after 150 cycles. Another notable approach is the hydrogen-bond-based dynamic binder developed by Chen et al., which involves the reaction of TA and PAA[79]. The branched TA structure, rich in hydrogen bonding sites, forms a dynamic network that accommodates Si volume fluctuations and improves electrode integrity. Additionally, a PAA-free strategy combining CMC and cationic polyacrylamide (CPAM) was introduced to form an ionically cross-linked network through electrostatic interactions[98]. This dynamic yet physical network showed excellent adaptability to volume changes without relying on covalent bonding, leading to improved structural resilience. In a more recent study, Kang et al. proposed a dynamic covalently cross-linked network (xPAA-B-DA), in which boronic ester linkages undergo reversible exchange reactions [Figure 8B][28]. This imparts self-healing functionality, preserves ionic transport pathways, and alleviates structural disintegration under cycling stress. Importantly, the residual dopamine moieties enhance adhesion via catechol metal and hydrogen bonding, stabilizing SEI formation and reducing charge transfer resistance over time. In another advanced system, Kang et al. designed a multifunctional binder integrating multiple dynamic interactions - boronic ester (B-N) bonds, Al3+ carboxylate coordination, and hydrogen bonding - within a semi-interpenetrating polymer network of PAA and CMC [Figure 8C][99]. This design imparted robust mechanical strength, elasticity, and rapid autonomous healing under ambient conditions. The inclusion of Al3+ ions enhanced compressive resistance and interfacial adhesion. A particularly notable feature was the visible healing of separated hydrogel segments within 3 h without external stimuli. The binder (CMC-AAc-3) retained over 1,000 mAh g-1 after 300 cycles, exhibited a high initial Coulombic efficiency of 84.7%, suppressed swelling in electrolyte, and facilitated stable SEI formation. These results underscore the versatility of dynamic hydrogel binders for Si anode applications. Building on such multifunctional dynamic architectures, recent work has reported a supramolecular polymer binder featuring hard/soft phase interactions[100]. In this design, the hard domain integrates reversible disulfide exchange (dynamic covalent bonds), cooperative hydrogen bonding, and π-π interactions, imparting self-healing capability and robust mechanical strength. Meanwhile, the soft polyether domain establishes Li+ ion-hopping pathways, enhancing stretchability and ionic conductivity. When applied to Si electrodes, this binder effectively suppressed volumetric expansion, promoted the formation of a dense and homogeneous SEI, and achieved a high initial coulombic efficiency of 85.2%, along with superior rate capability and cycling stability in both half-cell and NCM523||Si/C full-cell configurations. This synergy between dynamic covalent bonding and supramolecular interactions exemplifies a promising pathway for enhancing both mechanical adaptability and electrochemical performance in next-generation Si anode binders. Lastly, Xu et al. developed a fast self-healing and highly adhesive aqueous binder (PAA-LS) by incorporating lignosulfonate into PAA, utilizing dynamic Fe3+ catechol coordination[29]. This system enables efficient healing of microcracks and stress redistribution during lithiation/delithiation, while providing mechanical robustness and strong interfacial adhesion. The SiOx@PAA-LS electrode demonstrated an impressive capacity of 997.3 mAh g-1 after 450 cycles at 0.5 A g-1, outperforming conventional binders such as PAA and CMC [Figure 8D]. In full-cell tests (SiOx||NCM622), the binder achieved 82.72% capacity retention after 100 cycles. The superior performance stems from the synergy of dynamic coordination bonding, polar functional groups enhancing Li-ion transport, and the optimized LS content (0.5 wt%) ensuring network integrity. This work validates the practical potential of dynamic cross-linked binders for next-generation high-capacity Si oxide anodes[29].
Figure 8. Electrochemical performance and structural stability of Si anodes incorporating polymeric dynamically cross-linked binders. (A) Schematic illustration of the self-healing mechanism in a polymer binder and the cycling performance of Si electrodes using dynamically cross-linked polymer binders based on CD and AD host-guest interactions. Adapted with permission from ref.[97]; (B) Schematic illustration of the self-healing 3D polymeric binder (xPAA-B-DA), constructed via boronic ester and dopamine-based cross-linking chemistry, demonstrating its integration within Si anodes during lithiation/delithiation. Also shown are the electrochemical impedance spectra of Si@xPAA-B-DA and Si@PAA electrodes measured at the 1st, 50th, and 100th cycles under a current density of
Table 2 categorizes various polymeric binders according to their structural architectures and bonding mechanisms, including linear, branched, covalently cross-linked, and dynamically cross-linked systems. These binders employ diverse interactions, such as hydrogen bonding, ionic interactions, covalent ester linkages, and coordination bonding to improve mechanical resilience and interfacial stability in Si anodes. Table 3 provides a concise overview of the representative binder types covered in Sections "Branched or grafted polymer binders"-"Dynamically cross-linked polymer binders", including linear, branched, covalently cross-linked, and dynamically cross-linked systems, highlighting their respective advantages and limitations. This comparative summary offers a basis for the subsequent discussion in Section "MECHANICAL PROPERTIES AND THEIR IMPACT ON SI ANODE PERFORMANCE", which focuses on the mechanical properties of these binder systems.
Polymeric binders with diverse structures and interactions for Si anodes
| Binder | Feature | Structure | Ref. |
| Ox-L-CGN LGN | Hydrogen bonding, Electrostatic interaction | Linear Linear | [70] [71] |
| POWPU | Multiple Hydrogen bonding | Linear | [12] |
| API | Multiple Hydrogen bonding | Linear | [17] |
| PA2X1 | Multiple Hydrogen bonding | Linear | [10] |
| PEaMAc | Hydrogen bonding, Ionic interactions | Linear | [78] |
| MW PAA | Hydrogen bonding, Ionic interactions | Linear | [11] |
| PIBDA | Noncovalent interactions | Linear | [118] |
| β-CDp | Multi-hydrogen-bonding interactions | Branched | [119] |
| PAA-NH3 | Ionic bonding, Strong adhesion | Branched | [120] |
| GA-g-PAA | Covalent interaction | Branched | [121] |
| CA-g-PAA | Covalent interaction | Branched | [32] |
| PVA-g-PAA | Hydrogen bonding | Branched | [89] |
| PAA-g-SBR | Covalent-Noncovalent bonding, | Branched | [90] |
| PAA/PVA | Ester bonding, Strong adhesion | Cross-linked | [95] |
| PAA-CMC | Ester bonding, Strong adhesion | Cross-linked | [117] |
| b-Li0.5PAA@SA | Ester bonding, Mechanical Strength | Cross-linked | [122] |
| SBR/CMC-PEG Im-Zn2+ | Ester bonding, Zinc ion | Cross-linked | [123] |
| SA/Ca2+ | Ester bonding, Calcium ion | Cross-linked | [124] |
| cxPAA-PEI(x) | Condensation reaction | Cross-linked | [93] |
| PAA-CMC-Na | Covalent-Noncovalent bonding | Cross-linked | [34] |
| AGE binder | Covalent bonding | Cross-linked | [9] |
| CPU-PAA | Covalent-Noncovalent bonding | Cross-linked | [22] |
| LPTS | Covalent-Noncovalent bonding | Cross-linked | [38] |
| Alg(Ni)-PAM-TA | Covalent, Coordination bonding, Hydrogen bonding | Cross-linked | [40] |
| SiO_P(SH-BA) | Covalent bonding | Cross-linked | [13] |
| PPD-PAA | Covalent-Noncovalent bonding | Cross-linked | [39] |
| PVA-Malonic Acid | Covalent ester bonding | Cross-linked | [14] |
| PEI-PVA | Covalent ester bonding Multi noncovalent interactions | Cross-linked | [43] |
| HM | Covalent ester bonding, Noncovalent interactions | Cross-linked | [27] |
| FP2SA | Ionic bonding, Covalent interactions | Dynamic cross-linked | [63] |
| β-CDp-6AD | Multi Noncovalent interactions | Dynamic cross-linked | [97] |
| TA-c-PAA | Multi Noncovalent interactions | Dynamic cross-linked | [79] |
| CMC-CPAM | Ionic bonding | Dynamic cross-linked | [98] |
| xPAA-B-DA | Dynamic covalent bonding Multi Noncovalent interactions | Dynamic cross-linked | [28] |
| PEI@PAA@CMC | Dynamic covalent bonding, Multi Noncovalent interactions | Dynamic cross-linked | [15] |
| PAA-LS | Coordination bonding Multi Noncovalent interactions | Dynamic cross-linked | [29] |
| CMC-AAc-Al3+ | Coordination bonding Multi Noncovalent interactions | Dynamic cross-linked | [99] |
| SA@Borax | Dynamic covalent bonding Multi Noncovalent interactions | Dynamic cross-linked | [26] |
Comparative advantages and disadvantages of representative polymer binder types for Si-based anodes
| Binder type | Advantages | Limitations |
| Linear polymer binders | - Film-forming; uniform coating - Simple synthesis; water-processable - Interfacial bonding (H-bonding/polar) - Low cost; commodity-grade | - Limited mechanical strength - Irreversible fracture; electrical contact loss |
| Branched or grafted polymer binders | - Multifunctional groups; multi-point adhesion - Improved toughness, crack suppression - Tunable modulus, branch/graft density control - Interface stabilization; SEI stability | - Synthesis complexity, batch variability - Higher slurry viscosity, solids cap - Cost ↑ (functional monomers/initiators) - Adhesion-elasticity trade-off |
| Covalently cross-linked polymer binders | - Robust 3D network; high mechanical strength - Suppress particle pulverization; crack control - Maintain structural integrity (Long-term) | - Irreversible network; limited self-healing - Harsh synthesis/curing (temperature/time) - Curing shrinkage; residual stress |
| Dynamically cross-linked polymer binders | - Reversible bonding; self-healing - Expansion-contraction tolerance - Mechanical resilience; interfacial stability - Extended cycle life | - Electrolyte-sensitive; bond weakening - Narrow processing window - Healing-rate mismatch |
Dynamic cross-linked architectures combine adaptive mechanical buffering with autonomous healing, making them particularly well-suited for mitigating the extreme volume changes of high-capacity Si-based anodes. Dynamic networks leverage reversible bonds to enable autonomous crack healing and stress relaxation, though industrial deployment requires careful control of bond lifetime, mechanical stability, and slurry rheology under high-solid-loading conditions.
Their versatility in incorporating multiple reversible interactions ranging from hydrogen bonding and π-π stacking to metal-ligand coordination enables simultaneous improvement of structural resilience, interfacial adhesion, and ionic conductivity. Nevertheless, the complexity of multi-component synthesis, the inherently high cost of functionalized precursors, potential variability in dynamic bond lifetimes, and uncertainties in long-term stability under industrial processing conditions remain barriers to commercialization. Future efforts will need to focus on cost-effective precursor selection, large-scale aqueous synthesis routes, and rheological optimization to fully leverage the advantages of dynamic cross-linked binders in mass-produced next-generation LIB electrodes. While these dynamic cross-linked architectures have been primarily investigated in conventional liquid electrolyte systems, similar design principles can be extended to solid-state battery (SSB) configurations. In SSBs, the absence of liquid electrolyte eliminates swelling and dissolution issues, but introduces challenges related to maintaining intimate solid-solid interfacial contact and ensuring sufficient ionic conductivity across the electrode-electrolyte interface[101-103]. Binders in this context must combine strong adhesion to both the Si anode and solid electrolyte with mechanical compliance to accommodate substantial volume changes during cycling. Incorporating ion-conductive polymer segments, such as polyethylene oxide (PEO) or polycarbonate-based backbones, can facilitate Li+ transport through the binder phase and reduce interfacial resistance[104-106]. Furthermore, hybrid binder systems containing ceramic fillers, such as Li7La3Zr2O12 (LLZO), have been explored to enhance both mechanical robustness and ionic conductivity in SSB environments[107]
MECHANICAL PROPERTIES AND THEIR IMPACT ON SI ANODE PERFORMANCE
This section links the structural and chemical design of binders to measurable mechanical parameters such as tensile strength, elongation, and modulus. We explore how these properties influence Si anode cycling stability, and outline testing methods relevant for both laboratory studies and industrial qualification.
Building on the comparative analysis in Table 3, which outlined the structural architectures and bonding mechanisms of various polymer binder systems, this section focuses on how these design differences translate into mechanical behavior. In particular, we discuss why the ability to accommodate substantial volume changes and maintain electrode integrity is a decisive factor for the practical deployment of Si-based anodes, especially in the context of high-loading and micrometer-sized Si particles.
Previous research on Si anode binders has predominantly focused on the functional groups and structural configurations of polymers. However, relatively few studies have comprehensively examined how the intrinsic material properties, particularly the mechanical characteristics of polymeric binders, influence their overall performance. Moreover, most investigations on Si anodes have utilized nanoparticles or engineered nanostructures. While these approaches are effective in mitigating volume change issues, they remain impractical for large-scale commercial implementation due to cost-related limitations. In this context, Si microparticles (SiMPs) have emerged as a more economically viable alternative. Yet, the use of micrometer-sized Si presents significant challenges. During cycling, these particles undergo severe volume expansion, leading to particle fracture, loss of contact with the binder, electrical isolation, and eventual failure of the conductive network. As particle size increases, these issues become more pronounced, underscoring the urgent need to develop binder systems capable of maintaining electrode integrity in SiMP-based electrodes. To address this challenge, recent research has explored elastic cross-linked polymers as promising binder candidates. Elastic binders are designed to encase SiMPs and buffer the mechanical stress induced by their repeated expansion and contraction, thereby preserving electrical pathways and structural coherence. For such systems to be effective, the binder must possess sufficient elasticity and mechanical resilience to withstand the internal stresses imposed by SiMP deformation. Inspired by the mechanical principle of a pulley system, a novel binder architecture known as the molecular pulley binder (PR-PAA) was developed[47,108]. In this design, polyrotaxane (PR) molecules are covalently bonded to a PAA backbone, forming a cross-linked polymer network where PR acts as a dynamic “cord”. The PR consists of polyethylene glycol (PEG) chains threaded through cyclodextrin (α-CD) rings, which serve as movable pulleys. This design enables the distribution of mechanical stress across the network and allows for reversible deformation under volumetric changes. The repulsive interactions between the α-CD rings provide additional restorative force, contributing to the elasticity required to return to the original conformation during Si contraction. This unique architecture enables the PR-PAA binder to maintain electrode integrity even when using SiMPs, which are inherently difficult to stabilize due to their large volume fluctuations [Figure 9A][47,108]. Remarkably, with just 10 wt% binder content relative to the total electrode mass, the PR-PAA-SiMP electrode achieved an initial Coulombic efficiency exceeding 90%, and retained 91% of its capacity after 150 cycles [Figure 9B and C][47,109]. This indicates that the stability of SiMPs is influenced not only by the adhesive properties or structural features of the polymer but also by its physical characteristics, such as elasticity. As such, the mechanical properties of the polymer are crucial in determining the effectiveness of binders for Si anodes. Key attributes, such as elasticity, are vital to how well the binder maintains its structure under the stresses caused by the expansion and contraction of Si during cycling. These findings demonstrate that the mechanical properties of the polymer, particularly elasticity, are just as critical as chemical adhesion or structural design in determining the effectiveness of the binder. Attributes such as flexibility, resilience, and stress dissipation directly influence electrochemical stability, long-term durability, and capacity retention of Si-based anodes in LIBs. Therefore, future binder development must integrate mechanical considerations alongside chemical functionalities to meet the demands of practical high-energy-density battery systems. Among the various binder systems exhibiting comparable electrochemical performance, the PR-PAA binder stands out due to its uniquely engineered mechanical adaptability. Compared to conventional cross-linked polymers, the molecular pulley architecture of PR-PAA incorporates mechanically sliding polyrotaxane units that enable dynamic stress redistribution throughout the binder matrix. This design not only buffers the internal strain induced by the volumetric expansion of SiMPs but also allows the network to reversibly recover its structure during contraction. Such a mechanically adaptive response is critical for maintaining long-term electrode integrity under practical conditions. Therefore, the distinction of PR-PAA lies not merely in its cycle life metrics, but in its rational structural design that integrates elasticity, reversibility, and stress tolerance, essential qualities for scalable and durable Si anode systems.
Figure 9. Self-healing polymer binders based on PR-grafted PAA (PRPAA) structures and their application to Si anodes, highlighting enhanced mechanical reversibility and interfacial stability during repeated lithiation/delithiation. (A) Chemical structure and schematic illustration of a molecular pulley-type binder using a polyrotaxane backbone. Adapted with permission from ref.[108]; (B and C) Initial charge-discharge profiles of Si electrodes with the molecular pulley binder and cycling performance of the Si anode using the PR-based binder. Adapted with permission from ref.[109].
Table 4 provides a comparative overview of various polymeric binders for Si anodes in terms of specific capacity, cycle life, and binder content. Conventional binders such as PVDF exhibit limited cycling performance, whereas advanced systems incorporating functional groups, grafted architectures, or dynamic cross-linking mechanisms demonstrate significantly improved capacity retention over extended cycling. These results highlight the importance of binder design in achieving stable electrochemical performance in high-capacity Si-based electrodes.
Electrochemical performance of reported polymer binders for Si anodes
| Binder | Capacity/Cycles (mAh g-1/cycles) | Binder mass (wt%) | Ref. |
| PVDF | 600/50 | 8 | [114] |
| CMC | 2,733/55 | 15 | [125] |
| GG (Gellan Gum) | 2,222/100 | 15 | [125] |
| SA (Na-Alg) | 1,700/50 | 15 | [69] |
| CS | 950/50 | 8 | [126] |
| CS/rubber | 1,350/50 | 15 | [72] |
| PAA | 2,500/100 | 25 | [80] |
| PAN | 1,639/50 | 10 | [86] |
| PAA/PVA | 2,283/100 | 20 | [95] |
| PAA-UPy | 2,638/110 | 20 | [111] |
| b-Li0.5PAA@SA | 1,584/150 | 20 | [122] |
| PAA-NH3 | 711/100 | 10 | [120] |
| Alg-C | 1,700/400 | 20 | [74] |
| AA(xx)UR(yy) | 2,334/100 | 15 | [33] |
| PI-BIDA | 1,350/150 | 10 | [118] |
| Si@API | 1,444/200 | 20 | [17] |
| Ox-L-CGN-5 | 2,534/100 | 20 | [70] |
| Si/POWPU | 1,204/400 | 20 | [12] |
| HAMN-based Si/C | 1,386/600 | 10 | [127] |
| C-Alg-g-PAAm | 836/100 | 15 | [94] |
| β-CDp | 1,471/200 | 20 | [119] |
| β-CDp-6AD | 1,542/100 | 10 | [119] |
| c-PAA-CMC | 2,100/100 | 20 | [117] |
| cxPAA-PEI(x) | 2,606/100 | 20 | [93] |
| CS-grafted-PANI | 1,091/200 | 20 | [67] |
| PAA-CMC-Na | 950/400 | 20 | [34] |
| PR-PAA | 2,971/100 | 10 | [47] |
| CMC-CPAM | 1,906/100 | 20 | [98] |
| PVA-g-PAA | 2,001/150 | 10 | [89] |
| Na-PAA-g-SBR | 673/130 | 10 | [90] |
| AGE binder | 2,000/100 | 20 | [9] |
| CPU-PAA | 1,500/400 | 10 | [22] |
| LPTS | 2,082/400 | 5 | [38] |
| Alg(Ni)-PAM-TA | 1,863/200 | 10 | [40] |
| SiO-P(SH-BA) | 500/600 | 3 | [13] |
| PPD-PAA | 2,400/200 | - | [39] |
| PVA-Malonic Acid | 2,267/100 | 20 | [14] |
| PEI-PVA | 2,126/200 | 20 | [43] |
| Si-HM | 1,426/100 | 10 | [27] |
| SiOx-PAA-Dex₉ | 1,200/100 | 10 | [41] |
| xPAA-B-DA | 1,674/200 | 20 | [28] |
| PEI@PAA@CMC | 1,310/400 | 6.5 | [15] |
| SiOx-PAA-LS | 997/450 | 10 | [29] |
| CMC-AAc-Al3+ | 1,028/300 | 15 | [99] |
| SA@Borax | 1,655/500 | 10 | [26] |
From our perspective, the next stage of mechanical binder design should focus on scalable synthesis routes that preserve elasticity and stress-dissipating features, while balancing cost and process compatibility to enable widespread adoption in industrial SiMP-based anodes.
INDUSTRIAL PERSPECTIVES AND FUTURE RESEARCH DIRECTIONS
This section discusses binder development from an industrial perspective, focusing on scalability, cost efficiency, and process compatibility. Despite numerous reports of effective binders for Si-based electrodes, their utilization has remained largely limited to the SBR/CMC binder system for negative electrodes, reflecting the persistence of unresolved technical approaches in alternative approaches. For high-volume production lines, the most impactful binder strategy is one that supports incremental Si loading increases from low percentages (≤ 5 wt%) for initial validation, toward mid-to-high fractions (> 15 wt%) without requiring disruptive changes to coating, drying, or calendaring processes. Industrial practice shows that stable scale-up is more often achieved through evolutionary integration (e.g., gradual Si ratio increase) rather than step-change adoption of high-Si electrodes. Binder technology must also align with particle size strategy. Micrometer-scale Si, despite higher volumetric expansion than nanoscale Si, offers advantages in raw material cost, supplier diversity, and easier quality control during slurry preparation. This shifts binder requirements toward stronger elastic recovery and fracture suppression, ensuring mechanical stability under high strain while maintaining slurry compatibility at industrial speeds. From a cost perspective, the binder is a minor mass fraction but a critical cost-control lever. Reducing binder content from typical research levels (≥ 10 wt%) to commercial targets (≈ 2-3 wt%) not only improves energy density but also cuts raw material expenses and reduces solvent usage, thereby lowering overall electrode manufacturing cost per kWh. Cost modeling by several manufacturers indicates that binder-related savings can directly offset the higher cost of Si powders, especially in early high-Si pilot lines. Commercial LIB manufacturing lines still rely heavily on the CMC/SBR system for graphite-rich electrodes. However, PAA-based systems are increasingly favored for Si-rich blends due to their superior interfacial adhesion and mechanical stiffness. Challenges such as brittleness at high modulus, electrolyte sensitivity, and functionalized monomer cost must be addressed through molecular weight tuning, controlled branching, or hybridization. Hybrid binder architectures that combine adhesive and elastic domains, while maintaining aqueous processability, are emerging as the most viable pathway to bridge lab-scale performance with mass-production reliability. In the industrial context, the binder that succeeds will not necessarily be the one delivering the highest lab-measured capacity, but the one that integrates seamlessly into high-throughput coating lines, demonstrates reproducible performance across batches, and enables cost reductions through simplified formulations, reduced loading, and compatibility with existing equipment.
For manufacturers, the most competitive binders will be those that align with key industrial goals:
(i) Cost per kWh reduction through minimized binder loading and inexpensive feedstocks;
(ii) Robust process compatibility and yield stability, ensuring reliable performance during slurry preparation, coating, drying, and calendaring at industrial scales;
(iii) Scalability across multiple anode chemistries, from graphite-Si blends to full Si and emerging solid-state formats;
(iv) Environmentally responsible processing, supported by water-based formulations and sustainable manufacturing practices.
By framing binder development in terms of these manufacturing and economic imperatives, research directions can be better aligned with the realities of commercial cell production, ensuring a smoother transition from promising laboratory concepts to profitable mass-market solutions. By embedding these industrial considerations into the binder selection and design framework, this review aims to bridge the gap between academic insights and manufacturable, cost-effective solutions, an aspect often missing in conventional literature but essential for successful Si anode commercialization.
CONCLUSION
The concluding section synthesizes key insights from the review, summarizing the trade-offs, design principles, and industrial considerations for Si anode binders. We provide a consolidated outlook on the most promising directions for future development, integrating both scientific and manufacturing perspectives.
Polymer binders play a crucial role in maintaining the structural integrity and electrochemical stability of Si anodes. They not only mitigate the detrimental effects of volume expansion, such as pulverization and electrode delamination, but also stabilize the interface between the active material and the electrolyte. In particular, the incorporation of functional groups capable of forming secondary interactions with Si surfaces enhances adhesion, while supramolecular and mechanical characteristics of the binder directly influence cycling performance. This review has outlined key considerations in the rational design of polymer binders for Si anodes, emphasizing the importance of both chemical and physical interactions. First, the formation of strong interfacial adhesion through hydrophilic functional groups is essential to anchor the binder to both the active material and the current collector. Second, an optimal balance between bonding strength and flexibility achieved through covalent cross-linking or hydrogen bonding helps the binder accommodate the mechanical strain of Si during cycling. Notably, recent studies suggest that the inclusion of elastic properties in the binder design correlates positively with long-term cycling stability, especially in systems using micrometer-sized Si. Moreover, from a practical application standpoint, minimizing the binder content within the electrode is imperative for achieving higher energy densities. While many existing studies have used binder contents exceeding 10 wt%, commercial viability typically requires ≤ 3 wt%. Therefore, future research must continue to explore binder systems that maintain high performance even at significantly reduced loading levels. A particular focus should be placed on enhancing mechanical adaptability and reducing binder content, while preserving strong interfacial and cohesive properties, which are key aspects for the development of scalable, high-performance binder systems for next-generation Si anodes in LIBs.
In conclusion, we outline the key future directions for the rational design of polymer binders for Si anodes. These perspectives are schematically summarized in Figure 10. The figure illustrates future strategies for commercial Si-based binders, highlighting three core directions: (i) adhesion engineering to enhance interfacial binding and mechanical cohesion; (ii) electrolyte optimization to improve interfacial compatibility and suppress undesirable side reactions; and (iii) binder engineering to reduce binder content, accommodate volume fluctuations, facilitate ionic transport, and ensure an aqueous slurry process. These approaches collectively aim to enhance mechanical durability and electrochemical performance, while also supporting cost-effectiveness and environmentally sustainable processing. As Si continues to gain traction as a high-capacity anode material, the development of multifunctional, structurally robust, and scalable binder systems will remain a critical component in realizing practical next-generation LIBs.
Figure 10. Schematic representation of future design strategies for commercial Si anode binders. Key directions include: (i) adhesion engineering to enhance interfacial binding and mechanical cohesion; (ii) electrolyte optimization to improve compatibility and suppress undesirable side reactions; and (iii) binder engineering to reduce binder content, accommodate volume fluctuations, facilitate ionic transport, and ensure water solubility. These strategies collectively aim to enhance mechanical durability and electrochemical performance, while supporting cost-effectiveness and environmentally sustainable processing. The original figure was created by the authors.
From an industrial perspective, manufacturing strategies for Si anode binders can largely be divided into two approaches: replacing CMC with PAA in the conventional CMC/SBR dual binder system, or substituting SBR with PAA altogether. The adoption of PAA is driven by the need to increase Si content for high-energy and high-power batteries, while mitigating volumetric expansion through its high rigidity. Current development efforts focus on optimizing PAA binders with a viscosity range of 20,000-40,000 cps and tailoring molecular weight to balance slurry rheology, particle dispersion, coating uniformity, electrode density, and mechanical strength. PAA also offers notable formulation flexibility, being compatible with other binders and conductive additives such as CMC, SBR, and CNT.
In industrial practice, the preferred binder is not necessarily the one with the highest laboratory performance, but the one that consistently operates reliably on a production line. In this regard, PAA meets key requirements including cost-effectiveness, process stability, reproducibility, and established performance data. It also offers potential for cost reduction through simplified formulations, reduced binder content, and compatibility with existing aqueous processing lines. Looking ahead, the development of single-component binder systems that can simultaneously provide mechanical robustness, strong adhesion, and ionic conductivity is an emerging yet technically challenging research direction. Such systems often face difficulties in achieving optimal coating quality and sufficient adhesion under high Si-loading conditions, due to the challenge of balancing multiple functional requirements within a single polymer matrix. Overcoming these limitations will require advanced molecular design to improve slurry wettability, enhance interfacial bonding with active materials and current collectors, and maintain uniform particle dispersion. If successfully realized, these systems could simplify electrode formulations, reduce processing complexity, and improve production efficiency. They also hold the potential to significantly lower manufacturing costs for both lithium-ion and solid-state batteries, representing a transformative pathway toward the commercialization of high-capacity Si anode technologies across multiple battery platforms. Looking across the evolution of binder technologies, it is clear that progress has followed a stepwise trajectory from PVDF toward water-based CMC/SBR systems, then to PAA-centered formulations, and most recently to dynamic cross-linked and multifunctional architectures. This pathway, summarized in the industrially oriented roadmap of Figure 1, highlights how binder design has continuously adapted to balance adhesion, mechanical robustness, ionic conductivity, and processing feasibility. Importantly, the “Future” stage in the roadmap underscores the shift from academic demonstrations toward scalable solutions such as multifunctional and solid-state binders. By situating these perspectives at the conclusion of the review, we aim to emphasize not only the scientific advances achieved so far but also the practical milestones and challenges that lie ahead for commercialization. Looking ahead, the development of single-component multifunctional binder systems that can combine mechanical robustness, strong adhesion, and ionic conductivity represents a particularly promising yet technically demanding research direction. If realized, such systems could greatly simplify electrode formulations and accelerate the commercialization of high-capacity Si anode technologies.
In summary, this review has synthesized recent advances in natural, synthetic, branched, covalently cross-linked, and dynamically cross-linked binders for Si-based anodes, with an emphasis on linking molecular design strategies to electrochemical performance and manufacturability. By integrating cross-study analysis and author perspectives, we have identified key trade-offs between adhesion, elasticity, ionic conductivity, and processing compatibility. Industrially, while PAA-centered systems currently offer a pragmatic path for higher Si loadings, hybrid and multifunctional binders hold promise for long-term scalability. Continued innovation in binder chemistry, coupled with environmentally responsible manufacturing, will be essential for realizing the full potential of Si-based anodes in both liquid electrolyte and solid-state battery systems. Beyond material-level advances, the next phase of binder research will benefit from system-level integration, where binder design is coupled with conductive additives, electrode architecture, and electrolyte formulations. Such holistic approaches are expected to maximize the practical impact of binder innovation, ensuring that progress in molecular design translates effectively to full-cell stability, scalability, and cost competitiveness.
DECLARATIONS
Authors’ contributions
Characterization, original draft writing: Lim, S. Y.; Kim, J. H.
Review, editing, and supervision: Mun, J.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the Ministry of Trade, Industry & Energy/Korea Evaluation Institute of Industrial Technology (MOTIE/KEIT) (20022514 & RS-2024-00449747).
Conflicts of interest
Kim, J. H. is a Senior Editorial Board member of the journal Microstructures. Kim, J. H. was not involved in any steps of editorial processing, notably including reviewer selection, manuscript handling, or decision making, while the other authors have declared that they have no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026
REFERENCES
1. Jung, H. W.; Ko, S. M.; Lee, J. T. Sustainable and robust biomass-based binder for silicon anodes in lithium-ion batteries: cross-linked sodium alginate and chondroitin sulfate. Sci. Technol. Adv. Mater. 2025, 26, 2523243.
2. Park, S.; Choi, M.; Lee, J.; et al. Architecting sturdy Si/graphite composite with lubricative graphene nanoplatelets for high-density electrodes. Small 2025, 21, e2404949.
3. Zeng, X.; Dai, S.; Huang, F.; Chen, C.; Liu, L.; Hong, S. H. A novel gelatin binder with helical crosslinked network for high-performance Si anodes in lithium-ion batteries. Small 2024, 20, e2403754.
4. Kang, D.; Im, J.; Chae, S.; Jang, H.; Ko, M. Efficient recycling of spent LiCoO2 cathodes via confined pore-assisted simplified direct carbothermic reduction without external reducing agents. Energy. Environ. Mater. 2025, 9, e70037.
5. Samridh, A.; Gopinadh, S. V.; John, B.; Sarojiniamma, S.; Devassia, M. T.; Joseph, M. G. Sustainable binder system: cross-linked tamarind gum-polyacrylic acid for silicon-graphite anodes in future lithium-ion batteries. Energy. Technol. 2025, 13, 2401837.
6. Han, S. A.; Suh, J. H.; Kim, J.; et al. 3D pathways enabling highly-efficient lithium reservoir for fast-charging batteries. Small 2024, 20, e2310201.
7. Suh, J. H.; Han, S. A.; Yang, S. Y.; et al. Toward fast-charging and dendritic-free Li growth on natural graphite through intercalation/conversion on MoS2 nanosheets. Adv. Mater. 2025, 37, e2414117.
8. Feng, J.; Wu, X.; Amzil, S.; et al. Slurry additive approach enables a mechanically robust binder for silicon-carbon anodes in lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2025, 17, 18339-50.
9. Xiang, Y.; Xu, H.; Deng, J.; Li, J.; Nazir, M. A.; Bao, S. Spiderweb-like three-dimensional cross-linked AGE binder for high performance silicon-based lithium battery. ACS. Appl. Energy. Mater. 2025, 8, 2973-81.
10. Li, X.; Yuan, Q.; Rajapaske, G.; et al. A rigid-flexible network binder with multiple hydrogen bond interactions for high-performance silicon anodes. ACS. Appl. Energy. Mater. 2025, 8, 5199-208.
11. Jolley, M. J.; Pathan, T. S.; Jenkins, C.; Loveridge, M. J. Exploration of high and low molecular weight polyacrylic acids and sodium polyacrylates as potential binder system for use in silicon graphite anodes. ACS. Appl. Energy. Mater. 2025, 8, 1647-60.
12. Kim, J.; Kim, E.; Lim, E. Y.; et al. Stress-dissipative elastic waterborne polyurethane binders for silicon anodes with high structural integrity in lithium-ion batteries. ACS. Appl. Energy. Mater. 2024, 7, 1629-39.
13. Sun, C.; Zhang, H.; Mu, P.; et al. Covalently cross-linked chemistry of a three-dimensional network binder at limited dosage enables practical Si/C composite electrode applications. ACS. Nano. 2024, 18, 2475-84.
14. Lee, T.; Reddy, B.; Na, H.; et al. Cross-linked poly(vinyl alcohol)-malonic acid water-soluble binder for high-performance silicon anodes in lithium-ion batteries. ACS. Appl. Energy. Mater. 2025, 8, 332-42.
15. Liu, H.; Kang, T.; Li, S.; Ma, Z.; Nan, J. A Temperature-controlled chemoswitching aqueous binder and in situ binding strategy for stabilizing SiOx anodes of lithium-ion batteries. ACS. Sustain. Chem. Eng. 2024, 12, 17855-68.
16. Yoon, J.; Lee, J.; Kim, H.; Kim, J.; Jin, H. J. Polymeric binder design for sustainable lithium-ion battery chemistry. Polymers 2024, 16, 254.
17. Tan, W.; Liang, B.; Chen, M.; et al. Bio-based adenine-derived polyimide as a dual-functional binder for the silicon anode in lithium-ion batteries. ACS. Appl. Energy. Mater. 2023, 6, 10723-33.
18. Kang, Y.; Liu, F.; Yin, J.; et al. Structurally adaptive branched polyimide binder with synergistic elastic buffering and ionic transport for long-term stability of silicon anodes. Small 2025, e07023.
19. Lee, J.; Moon, J.; Han, S. A.; et al. Everlasting living and breathing gyroid 3D network in Si@SiOx/C nanoarchitecture for lithium ion battery. ACS. Nano. 2019, 13, 9607-19.
20. Han, D.; Han, I. K.; Son, J.; et al. Molecular engineering of coacervate network binders for stable silicon-based anodes in lithium-ion batteries. Adv. Funct. Mater. , 2025, e09445.
21. Wang, W.; Li, X.; Chen, X.; Sun, M.; Wang, G. Aqueous binder with self-emulsifying characteristics for practical Si/C anode in lithium-ion batteries. Chemistry 2025, 31, e202403924.
22. He, Y.; Zhou, F.; Zhang, Y.; Lv, T.; Chu, P. K.; Huo, K. High-toughness and hierarchical stress-dissipating binder based on physicochemical dual-cross-linking for high-performance silicon anodes. ACS. Appl. Mater. Interfaces. 2025, 17, 21206-14.
23. Sun, Z.; Zhu, J.; Yang, C.; et al. N-type polyoxadiazole conductive polymer binders derived high-performance silicon anodes enabled by crosslinking metal cations. ACS. Appl. Mater. Interfaces. 2023, 15, 12946-56.
24. Xiao, H.; Qiu, J.; Wu, S.; et al. Cross-linked γ-polyglutamic acid as an aqueous SiOx anode binder for long-term lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2022, 14, 18625-33.
25. Zhang, Z.; Ma, X.; Li, Y.; et al. Highly ion-conductive Si anode binder with optimized tensile strength and large deformation for lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2025, 17, 43034-47.
26. He, T.; Ding, Y.; Zhang, H.; et al. Self-healing SA@borax binder for in situ tuning of the solid electrolyte interfaces for silicon anodes. ACS. Sustain. Chem. Eng. 2025, 13, 4093-107.
27. Ai, W.; Yang, C.; Wang, Q.; et al. Stable silicon anodes enabled by innovative biobased binder with cross-linking network in lithium-ion batteries. Langmuir 2025, 41, 9469-79.
28. Kang, Y.; Kim, S.; Han, J.; Yim, T.; Kim, T. Poly(acrylic acid) grafted with a boronic ester and dopamine as a self-healable and highly adhesive aqueous binder for Si anodes. ACS. Appl. Energy. Mater. 2024, 7, 2436-50.
29. Xu, M.; Wei, X.; Yan, Z.; et al. A fast self-healing binder for highly stable SiOx anodes in lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2024, 16, 55353-61.
30. Kim, S.; Kang, Y.; Han, J. H.; Yeon, S. J.; Min, K.; Kim, T. A polysaccharide binder with carbon quantum dots for improved flexibility of Si anodes in lithium-ion batteries. ACS. Appl. Energy. Mater. 2024, 7, 9806-15.
31. Yang, L.; Meng, T.; Zheng, W.; et al. Advanced binder design for high-performance silicon anodes. Energy. Storage. Mater. 2024, 72, 103766.
32. Shen, J.; Wang, H.; Mei, Y.; et al. Multidimensional branched composite binder via covalent grafting for high-performance silicon-based anodes. J. Phys. Chem. C. 2023, 127, 14613-22.
33. Preman, A. N.; Vo, T. N.; Choi, S.; et al. Self-healable poly(acrylic acid) binder toward optimized electrochemical performance for silicon anodes: importance of balanced properties. ACS. Appl. Energy. Mater. 2024, 7, 749-59.
34. Zhang, B.; Li, Z.; Xie, H.; et al. Cross-linking chemistry enables robust conductive polymeric network for high-performance silicon microparticle anodes in lithium-ion batteries. J. Power. Sources. 2023, 556, 232495.
35. Guo, S.; Li, H.; Li, Y.; et al. SiO2-enhanced structural stability and strong adhesion with a new binder of konjac glucomannan enables stable cycling of silicon anodes for lithium-ion batteries. Adv. Energy. Mater. 2018, 8, 1800434.
36. Shang, Y.; Li, H.; Ma, T.; Yang, Y.; Jiang, Y.; Yu, W. Suppression strategies for Si anode volume expansion in Li-ion batteries based on structure design and modification: a review. ACS. Appl. Mater. Interfaces. 2025, 17, 31730-53.
37. Tatara, R.; Tomoi, Y.; Yamazaki, S.; Komaba, S. Impact of maturation on Si-based negative electrodes for LIBs: contributions of binder migration and Cu corrosion. J. Electrochem. Soc. 2025, 172, 030519.
38. Xiao, Y.; Hao, X.; Li, T.; et al. Multifunctional cross-linking composite binder enables the stable performance of Si-based anodes for high-energy-density lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2024, 16, 41036-47.
39. Sun, B.; Jiao, X.; Liu, J.; et al. Neural network inspired binder enables fast Li-ion transport and high stress adaptation for Si anode. Nano. Lett. 2024, 24, 7662-71.
40. Wu, F.; Liu, J.; Yang, Z.; et al. Highly stable silicon anode enabled by a water-soluble tannic acid functionalized dual-network binder. ACS. Appl. Mater. Interfaces. 2024, 16, 23396-405.
41. Long, J.; He, W.; Liao, H.; Ye, W.; Dou, H.; Zhang, X. In Situ prepared three-dimensional covalent and hydrogen bond synergistic binder to boost the performance of SiOx anodes for lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2023, 15, 10726-34.
42. Wu, Z.; Ma, Y.; Li, S.; et al. Damage-tolerant and self-repairing web-like borate type binder enable stable operation of efficient Si-based anodes. Small 2024, 20, e2401345.
43. Ba, Z.; Wang, Z.; Xu, K.; et al. Dual cross-linked poly(ether imide)/poly(vinyl alcohol) network binder with improved stability for silicon based anodes in lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2025, 17, 20197-208.
44. Jin, J. H.; Lee, D. G.; Kim, J. H.; et al. Graphene-assisted interfacial engineering to develop binder- and dispersant-free cast Si Alloy/nanocarbon anodes for high-performance Li-ion batteries. Adv. Funct. Mater. 2025, 35, e11352.
45. An, W.; Gao, B.; Mei, S.; et al. Scalable synthesis of ant-nest-like bulk porous silicon for high-performance lithium-ion battery anodes. Nat. Commun. 2019, 10, 1447.
46. Kwon, T. W.; Choi, J. W.; Coskun, A. The emerging era of supramolecular polymeric binders in silicon anodes. Chem. Soc. Rev. 2018, 47, 2145-64.
47. Choi, S.; Kwon, T. W.; Coskun, A.; Choi, J. W. Highly elastic binders integrating polyrotaxanes for silicon microparticle anodes in lithium ion batteries. Science 2017, 357, 279-83.
48. Mun, J.; Song, T.; Park, M. S.; Kim, J. H. Paving the way for next-generation all-solid-state batteries: dry electrode technology. Adv. Mater. 2025, 37, e2506123.
49. Salehabadi, M.; Medhi, K.; Werner-Zwanziger, U.; Obrovac, M. N. Citric acid as a small molecule binder for Si-based Li-ion battery anode materials. J. Electrochem. Soc. 2025, 172, 020520.
50. Oh, H.; Kim, J. T.; Shin, H.; et al. Stable performance for pouch-type all-solid-state batteries enabled by current collector with optimized primer layer. Mater. Sci. Eng. R. Rep. 2025, 164, 100970.
51. Kim, T.; Kim, K. H.; Kim, H.; et al. New suggestion of highly durable electrode design for ordered mesoporous Ni-Mn binary transition metal oxide anode material in lithium-ion batteries. Small 2025, 21, e2406243.
52. Chen, H.; Ling, M.; Hencz, L.; et al. Exploring chemical, mechanical, and electrical functionalities of binders for advanced energy-storage devices. Chem. Rev. 2018, 118, 8936-82.
53. Shen, H.; Wang, Q.; Chen, Z.; Rong, C.; Chao, D. Application and development of silicon anode binders for lithium-ion batteries. Materials 2023, 16, 4266.
54. Yang, Y.; Wu, S.; Zhang, Y.; et al. Towards efficient binders for silicon based lithium-ion battery anodes. Chem. Eng. J. 2021, 406, 126807.
55. Kwon, T.; Choi, J. W.; Coskun, A. Prospect for supramolecular chemistry in high-energy-density rechargeable batteries. Joule 2019, 3, 662-82.
56. Lee, J.; Park, M. S.; Kim, J. H. Stabilizing Li-metal host anode with LiF-rich solid electrolyte interphase. Nano. Converg. 2021, 8, 18.
57. An, Y.; Tian, Y.; Liu, C.; Xiong, S.; Feng, J.; Qian, Y. One-step, vacuum-assisted construction of micrometer-sized nanoporous silicon confined by uniform two-dimensional N-doped carbon toward advanced Li ion and MXene-based Li metal batteries. ACS. Nano. 2022, 16, 4560-77.
58. Browning, K. L.; Sacci, R. L.; Doucet, M.; Browning, J. F.; Kim, J. R.; Veith, G. M. The study of the binder poly(acrylic acid) and its role in concomitant solid-electrolyte interphase formation on Si anodes. ACS. Appl. Mater. Interfaces. 2020, 12, 10018-30.
59. Hu, Z.; Zhao, R.; Lv, M.; et al. Bio-inspired toughening elastomer as an innovative self-healing binder for Si-based electrode. Adv. Energy. Mater. 2025, 15, 2501991.
60. Chen, W.; Li, W.; Jiang, J.; Yao, C.; Li, L.; Gao, H. Effect of binder types on performance of spherical and flaky silicon electrodes. J. Solid. State. Electrochem. 2025, 29, 2623-37.
61. Li, K.; Zhang, X.; Qiao, S.; et al. Fatigue resistant polyimide binders for high-performance SiOx/C anodes in lithium-ion batteries. ACS. Appl. Polym. Mater. 2025, 7, 278-86.
62. Magasinski, A.; Zdyrko, B.; Kovalenko, I.; et al. Toward efficient binders for Li-ion battery Si-based anodes: polyacrylic acid. ACS. Appl. Mater. Interfaces. 2010, 2, 3004-10.
63. Wang, Y.; Yang, X.; Meng, Y.; et al. Fluorine chemistry in rechargeable batteries: challenges, progress, and perspectives. Chem. Rev. 2024, 124, 3494-589.
64. Choi, N.; Ha, S.; Lee, Y.; et al. Recent progress on polymeric binders for silicon anodes in lithium-ion batteries. J. Electrochem. Sci. Technol. 2015, 6, 35-49.
65. Liu, Y.; Shao, R.; Jiang, R.; Song, X.; Jin, Z.; Sun, L. A review of existing and emerging binders for silicon anodic Li-ion batteries. Nano. Res. 2023, 16, 6736-52.
66. Vogl, U. S.; Das, P. K.; Weber, A. Z.; Winter, M.; Kostecki, R.; Lux, S. F. Mechanism of interactions between CMC binder and Si single crystal facets. Langmuir 2014, 30, 10299-307.
67. Rajeev, K.; Kim, E.; Nam, J.; Lee, S.; Mun, J.; Kim, T. Chitosan-grafted-polyaniline copolymer as an electrically conductive and mechanically stable binder for high-performance Si anodes in Li-ion batteries. Electrochim. Acta. 2020, 333, 135532.
68. Chai, L.; Qu, Q.; Zhang, L.; Shen, M.; Zhang, L.; Zheng, H. Chitosan, a new and environmental benign electrode binder for use with graphite anode in lithium-ion batteries. Electrochim. Acta. 2013, 105, 378-83.
69. Kovalenko, I.; Zdyrko, B.; Magasinski, A.; et al. A major constituent of brown algae for use in high-capacity Li-ion batteries. Science 2011, 334, 75-9.
70. Kim, S. W.; K,
71. Jang, W.; K,
72. Lee, S. H.; Lee, J. H.; Nam, D. H.; et al. Epoxidized natural rubber/chitosan network binder for silicon anode in lithium-ion battery. ACS. Appl. Mater. Interfaces. 2018, 10, 16449-57.
73. Ko, M.; Chae, S.; Ma, J.; et al. Scalable synthesis of silicon-nanolayer-embedded graphite for high-energy lithium-ion batteries. Nat. Energy. 2016, 1, 16113.
74. Ryou, M. H.; Kim, J.; Lee, I.; et al. Mussel-inspired adhesive binders for high-performance silicon nanoparticle anodes in lithium-ion batteries. Adv. Mater. 2013, 25, 1571-6.
75. Zhang, Q.; Zhang, F.; Zhang, M.; Yu, Y.; Yuan, S.; Liu, Y. A highly efficient silicone-modified polyamide acid binder for silicon-based anode in lithium-ion batteries. ACS. Appl. Energy. Mater. 2021, 4, 7209-18.
76. Wang, X.; Liu, S.; Zhang, Y.; et al. Highly elastic block copolymer binders for silicon anodes in lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2020, 12, 38132-9.
77. Jiang, H.; Wei, C.; Yasmin, S.; Obrovac, M. N. Deconvoluting slurry rheology from binder performance in Si-based anodes. J. Electrochem. Soc. 2023, 170, 120522.
78. Jagodziński, K.; Boaretto, N.; Casado, N.; Martínez-Ibañez, M. Poly(ethylene-alt-maleic acid)-based binders for silicon anodes. ACS. Appl. Polym. Mater. 2025, 7, 2973-85.
79. Chen, J.; Li, Y.; Wu, X.; et al. Dynamic hydrogen bond cross-linking binder with self-healing chemistry enables high-performance silicon anode in lithium-ion batteries. J. Colloid. Interface. Sci. 2024, 657, 893-902.
80. Parikh, P.; Sina, M.; Banerjee, A.; et al. Role of polyacrylic acid (PAA) binder on the solid electrolyte interphase in silicon anodes. Chem. Mater. 2019, 31, 2535-44.
81. Rahaman, M.; Ismail, A.; Mustafa, A. A review of heat treatment on polyacrylonitrile fiber. Polym. Degrad. Stab. 2007, 92, 1421-32.
82. Hwang, S. S.; Sohn, M.; Park, H.; Choi, J.; Cho, C. G.; Kim, H. Effect of the heat treatment on the dimensional stability of Si electrodes with PVDF binder. Electrochim. Acta. 2016, 211, 356-63.
83. Liu, D.; Zhao, Y.; Tan, R.; et al. Novel conductive binder for high-performance silicon anodes in lithium ion batteries. Nano. Energy. 2017, 36, 206-12.
84. Li, J.; Wu, Z.; Lu, Y.; et al. Water soluble binder, an electrochemical performance booster for electrode materials with high energy density. Adv. Energy. Mater. 2017, 7, 1701185.
85. Yoon, J.; Han, G.; Cho, S.; et al. Microbial-copolyester-based eco-friendly binder for lithium-ion battery electrodes. ACS. Appl. Polym. Mater. 2023, 5, 1199-207.
86. Luo, L.; Xu, Y.; Zhang, H.; et al. Comprehensive understanding of high polar polyacrylonitrile as an effective binder for Li-ion battery nano-Si anodes. ACS. Appl. Mater. Interfaces. 2016, 8, 8154-61.
87. Umirov, N.; Moon, S.; Park, G.; Kim, H.; Lee, K. J.; Kim, S. Novel silane-treated polyacrylonitrile as a promising negative electrode binder for LIBs. J. Alloys. Compd. 2020, 815, 152481.
88. Yang, J.; Qu, Y.; Li, B.; et al. Zwitterionic polymer binder networks with structural locking and ionic regulation functions for high performance silicon anodes. ACS. Appl. Mater. Interfaces. 2024, 16, 70544-54.
89. He, J.; Zhang, L. Polyvinyl alcohol grafted poly (acrylic acid) as water-soluble binder with enhanced adhesion capability and electrochemical performances for Si anode. J. Alloys. Compd. 2018, 763, 228-40.
90. Jolley, M. J.; Pathan, T. S.; Wemyss, A. M.; et al. Development and application of a poly(acrylic acid)-grafted styrene-butadiene rubber as a binder system for silicon-graphite anodes in Li-ion batteries. ACS. Appl. Energy. Mater. 2023, 6, 496-507.
91. He, J.; Zhang, L.; Zhong, H. Enhanced adhesion and electrochemical performance of Si anodes with gum arabic grafted
92. Yang, C.; Jiang, Y.; Chen, F.; Zhao, J. Highly elastic hyperbranched polymer binder for silicon anodes in lithium-ion batteries. Electrochim. Acta. 2023, 442, 141805.
93. Chuang, Y.; Lin, Y.; Wang, C.; Hong, J. Dual cross-linked polymer networks derived from the hyperbranched poly(ethyleneimine) and poly(acrylic acid) as efficient binders for silicon anodes in lithium-ion batteries. ACS. Appl. Energy. Mater. 2021, 4, 1583-92.
94. Gendensuren, B.; Oh, E. Dual-crosslinked network binder of alginate with polyacrylamide for silicon/graphite anodes of lithium ion battery. J. Power. Sources. 2018, 384, 379-86.
95. Song, J.; Zhou, M.; Yi, R.; et al. Interpenetrated gel polymer binder for high-performance silicon anodes in lithium-ion batteries. Adv. Funct. Mater. 2014, 24, 5904-10.
96. Lou, X.; Zhang, Y.; Zhao, L.; Zhang, T.; Zhang, H. Cross-linked multifunctional binder in situ tuning solid electrolyte interface for silicon anodes in lithium ion batteries. Sci. Rep. 2023, 13, 18560.
97. Kwon, T. W.; Jeong, Y. K.; Deniz, E.; AlQaradawi, S. Y.; Choi, J. W.; Coskun, A. Dynamic cross-linking of polymeric binders based on host-guest interactions for silicon anodes in lithium ion batteries. ACS. Nano. 2015, 9, 11317-24.
98. Zhang, J.; Wang, N.; Zhang, W.; et al. A cycling robust network binder for high performance Si-based negative electrodes for lithium-ion batteries. J. Colloid. Interface. Sci. 2020, 578, 452-60.
99. Kang, E.; Hwang, J.; Kim, S.; Seo, J.; Cho, K. Y.; Chang, Y. W. A tough and self-healable semi-IPN hydrogel binder based on multiple dynamic bonds for Si anodes in lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2025, 17, 30054-66.
100. Zhang, J.; Yan, L.; Zhao, Y.; et al. A multifunctional supramolecular polymer binder with hard/soft phase interaction for Si-based lithium-ion batteries. Nano. Energy. 2024, 125, 109573.
101. Boorboor, Ajdari. F.; Asghari, P.; Molaei, Aghdam. A.; et al. Silicon solid state battery: the solid-state compatibility, particle size, and carbon compositing for high energy density. Adv. Funct. Mater. 2024, 34, 2314822.
102. Sun, Z.; Yin, Q.; Chen, H.; et al. Building better solid-state batteries with silicon-based anodes. Interd. Mater. 2023, 2, 635-63.
103. Nelson, D. L.; Sandoval, S. E.; Pyo, J.; et al. Fracture dynamics in silicon anode solid-state batteries. ACS. Energy. Lett. 2024, 9, 6085-95.
104. Grill, J.; Popovic-Neuber, J. Cation conducting binders: from liquid to solid-state batteries. ACS. Energy. Lett. 2024, 9, 4465-74.
105. Hong, S.; Lee, Y.; Kim, U.; et al. All-solid-state lithium batteries: Li+-conducting ionomer binder for dry-processed composite cathodes. ACS. Energy. Lett. 2022, 7, 1092-100.
106. Shin, D. O.; Kim, H.; Jung, S.; et al. Electrolyte-free graphite electrode with enhanced interfacial conduction using Li+-conductive binder for high-performance all-solid-state batteries. Energy. Storage. Mater. 2022, 49, 481-92.
107. Boaretto, N.; Meabe, L.; Lindberg, S.; et al. Hybrid ceramic polymer electrolytes enabling long cycling in practical 1 Ah-class high-voltage solid-state batteries with Li metal anode. Adv. Funct. Mater. 2024, 34, 2404564.
108. Shi, Y.; Zhou, X.; Yu, G. Material and structural design of novel binder systems for high-energy, high-power lithium-ion batteries. Acc. Chem. Res. 2017, 50, 2642-52.
109. Qin, T.; Yang, H.; Li, Q.; Yu, X.; Li, H. Design of functional binders for high-specific-energy lithium-ion batteries: from molecular structure to electrode properties. Ind. Chem. Mater. 2024, 2, 191-225.
110. Lestriez, B.; Bahri, S.; Sandu, I.; Roue, L.; Guyomard, D. On the binding mechanism of CMC in Si negative electrodes for Li-ion batteries. Electrochem. Commun. 2007, 9, 2801-6.
111. Li, M.; Lu, J.; Chen, Z.; Amine, K. 30 years of lithium-ion batteries. Adv. Mater. 2018, 30, e1800561.
112. Ling, M.; Xu, Y.; Zhao, H.; et al. Dual-functional gum arabic binder for silicon anodes in lithium ion batteries. Nano. Energy. 2015, 12, 178-85.
113. Liu, J.; Zhang, Q.; Zhang, T.; Li, J.; Huang, L.; Sun, S. A robust ion-conductive biopolymer as a binder for Si anodes of lithium-ion batteries. Adv. Funct. Mater. 2015, 25, 3599-605.
114. Li, J.; Christensen, L.; Obrovac, M. N.; Hewitt, K. C.; Dahn, J. R. Effect of heat treatment on Si electrodes using polyvinylidene fluoride binder. J. Electrochem. Soc. 2008, 155, A234.
115. Park, H.; Kong, B.; Oh, E. Effect of high adhesive polyvinyl alcohol binder on the anodes of lithium ion batteries. Electrochem. Commun. 2011, 13, 1051-3.
116. Kim, J. S.; Choi, W.; Cho, K. Y.; Byun, D.; Lim, J.; Lee, J. K. Effect of polyimide binder on electrochemical characteristics of surface-modified silicon anode for lithium ion batteries. J. Power. Sources. 2013, 244, 521-6.
117. Koo, B.; Kim, H.; Cho, Y.; Lee, K. T.; Choi, N. S.; Cho, J. A highly cross-linked polymeric binder for high-performance silicon negative electrodes in lithium ion batteries. Angew. Chem. Int. Ed. 2012, 51, 8762-7.
118. Li, K.; Wang, Z.; Zhang, X.; et al. Dual-function polyimide binders containing benzimidazole/benzoxazole moieties for high-performance SiOx/C anodes in lithium-ion batteries. ACS. Appl. Polym. Mater. 2025, 7, 7267-74.
119. Jeong, Y. K.; Kwon, T. W.; Lee, I.; Kim, T. S.; Coskun, A.; Choi, J. W. Hyperbranched β-cyclodextrin polymer as an effective multidimensional binder for silicon anodes in lithium rechargeable batteries. Nano. Lett. 2014, 14, 864-70.
120. Shi, Z.; Jiang, S.; Robertson, L. A.; et al. Restorable neutralization of poly(acrylic acid) binders toward balanced processing properties and cycling performance for silicon anodes in lithium-ion batteries. ACS. Appl. Mater. Interfaces. 2020, 12, 57932-40.
121. Zhong, H.; He, J.; Zhang, L. Crosslinkable aqueous binders containing Arabic gum-grafted-poly (acrylic acid) and branched polyols for Si anode of lithium-ion batteries. Polymer 2021, 215, 123377.
122. Hu, Y. Y.; You, J. H.; Zhang, S. J.; et al. Li0.5PAA domains filled in porous sodium alginate skeleton: a 3D bicontinuous composite network binder to stabilize micro-silicon anode for high-performance lithium ion battery. Electrochim. Acta. 2021, 386, 138361.
123. Kim, J.; Park, K.; Cho, Y.; et al. Zn2+-imidazole coordination crosslinks for elastic polymeric binders in high-capacity silicon electrodes. Adv. Sci. 2021, 8, 2004290.
124. Zhao, Y.; Yue, F.; Li, S.; et al. Advances of polymer binders for silicon-based anodes in high energy density lithium-ion batteries. InfoMat 2021, 3, 460-501.
125. Wu, C.; Li, C. Distribution uniformity of water-based binders in Si anodes and the distribution effects on cell performance. ACS. Sustain. Chem. Eng. 2020, 8, 6868-76.
126. Liu, J.; Zhang, Q.; Wu, Z. Y.; et al. A high-performance alginate hydrogel binder for the Si/C anode of a Li-ion battery. Chem. Commun. 2014, 50, 6386-9.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data






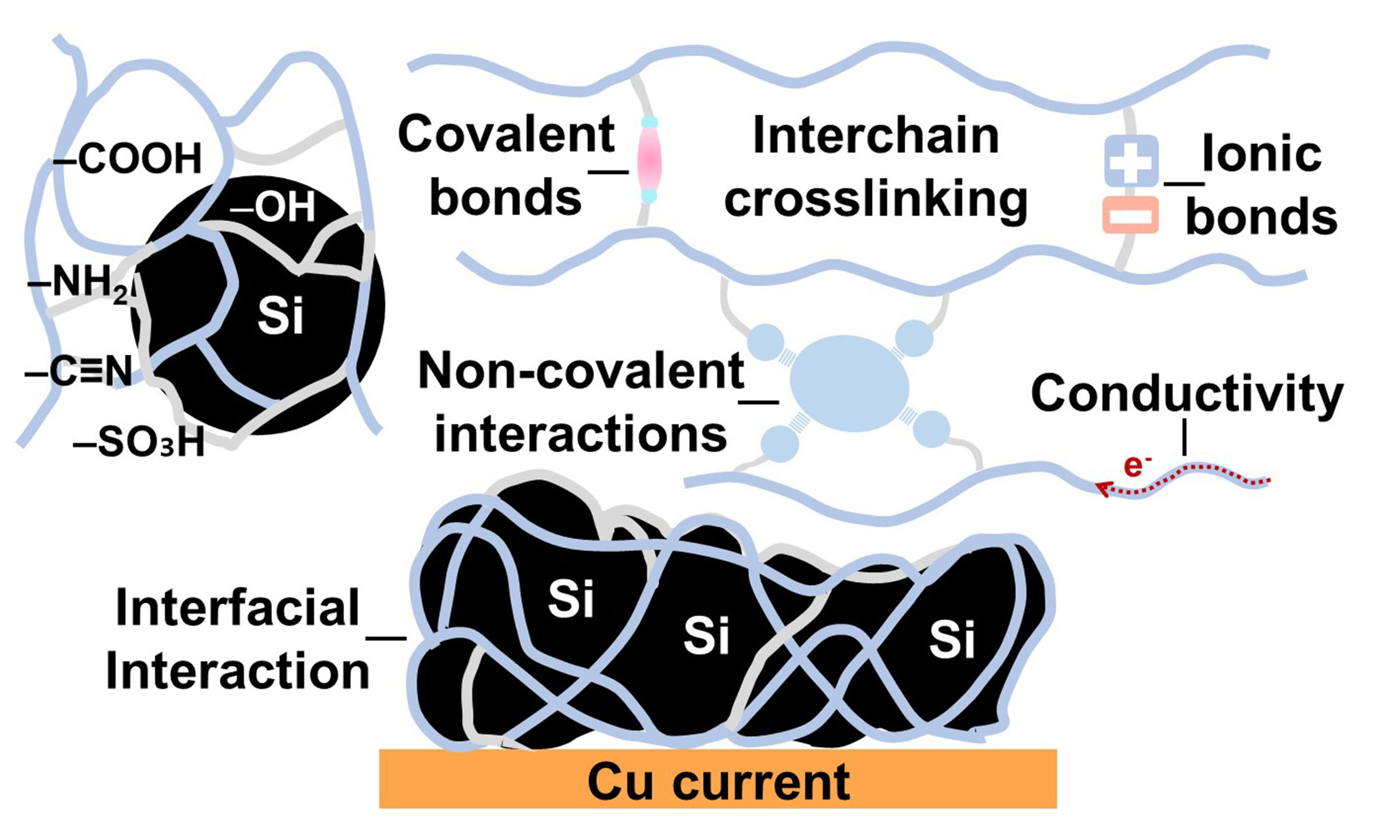
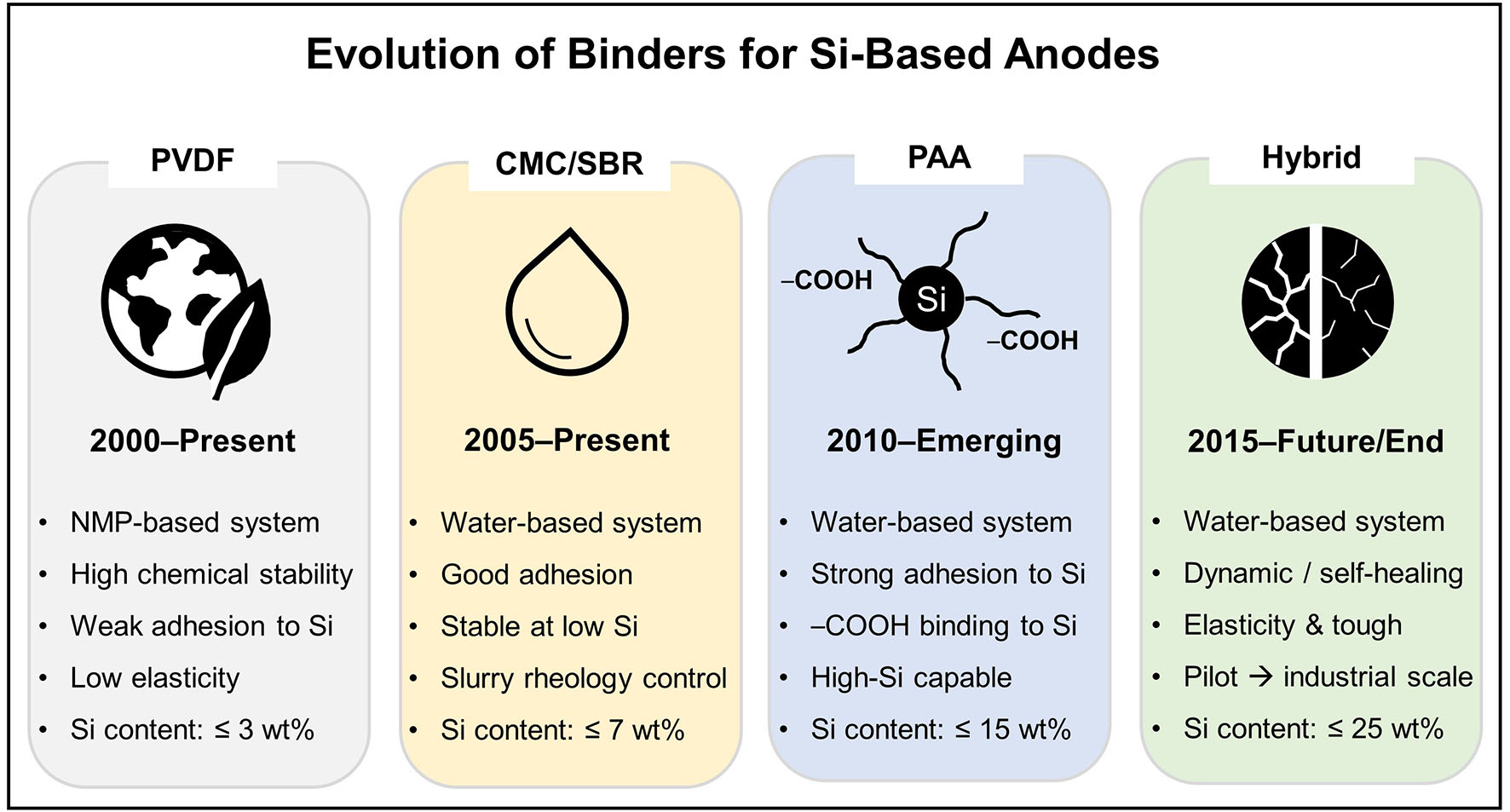
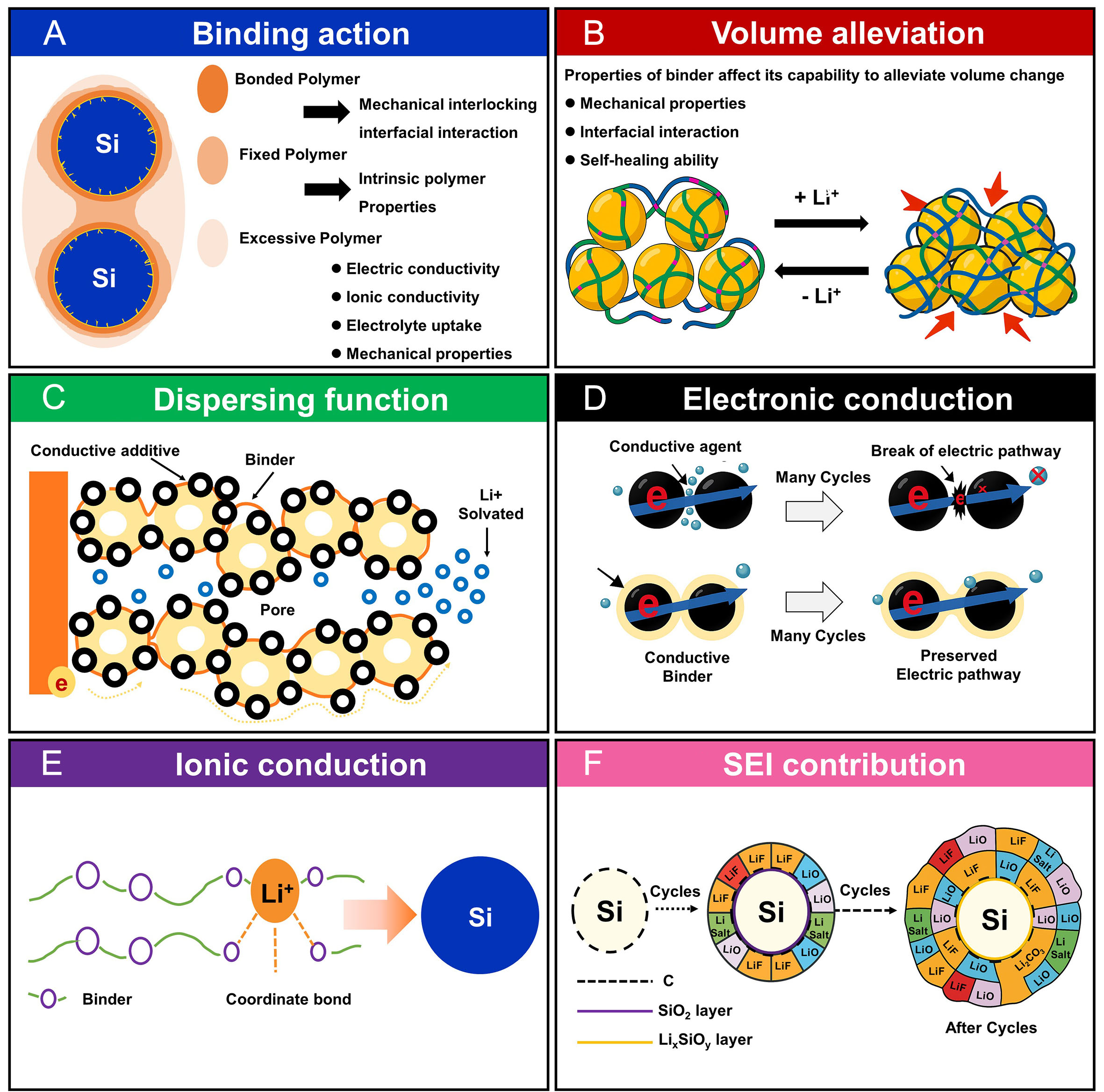
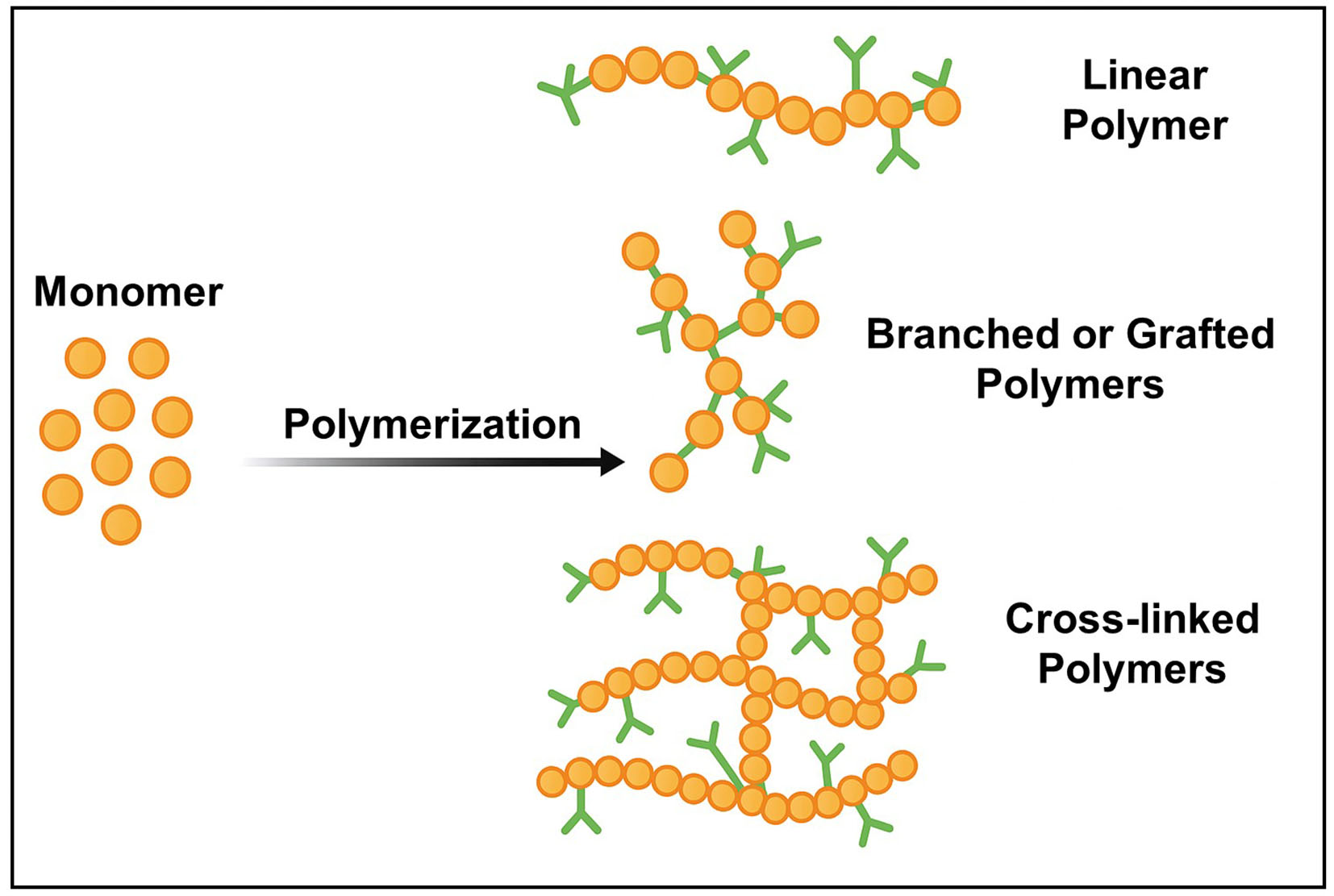
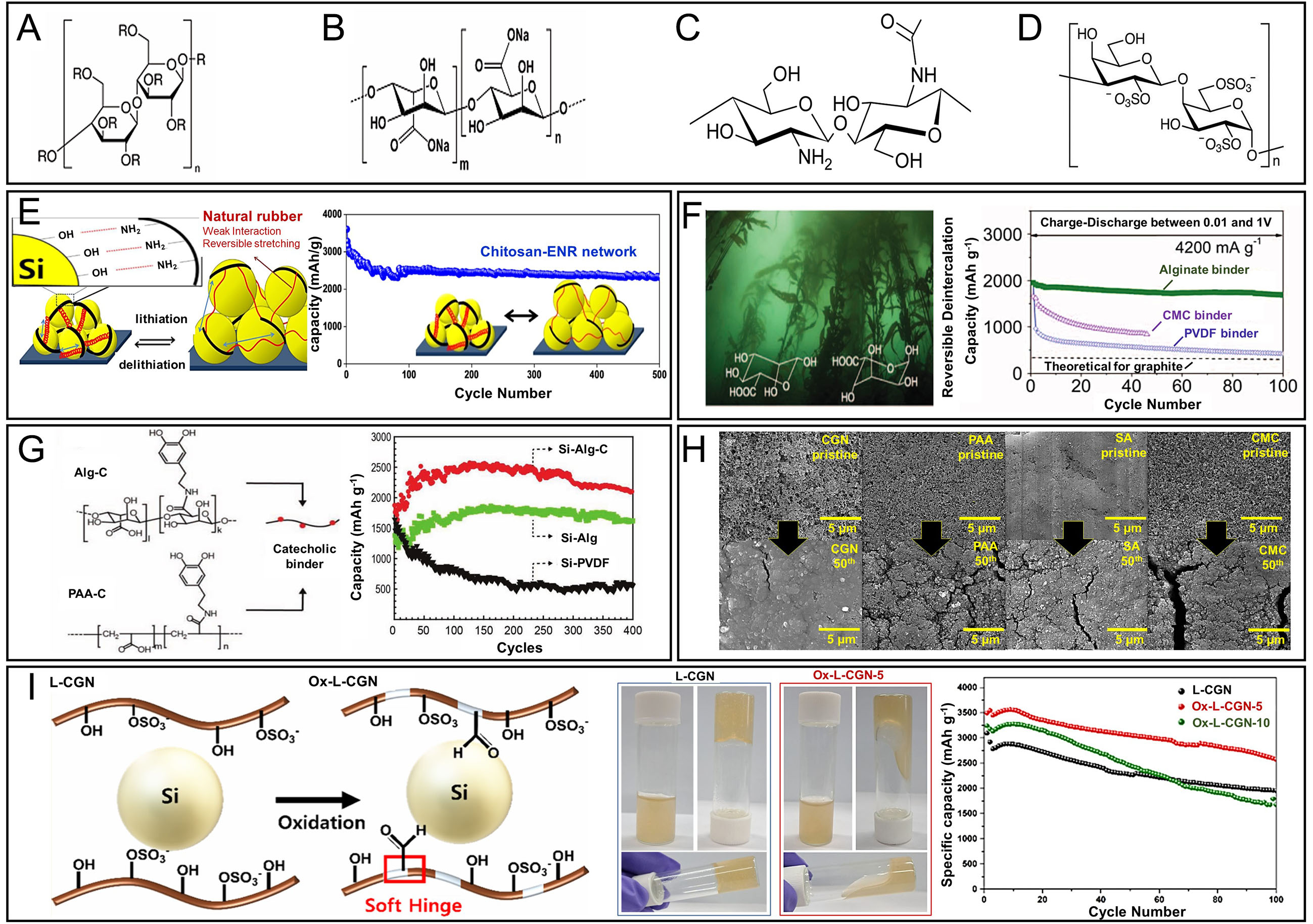
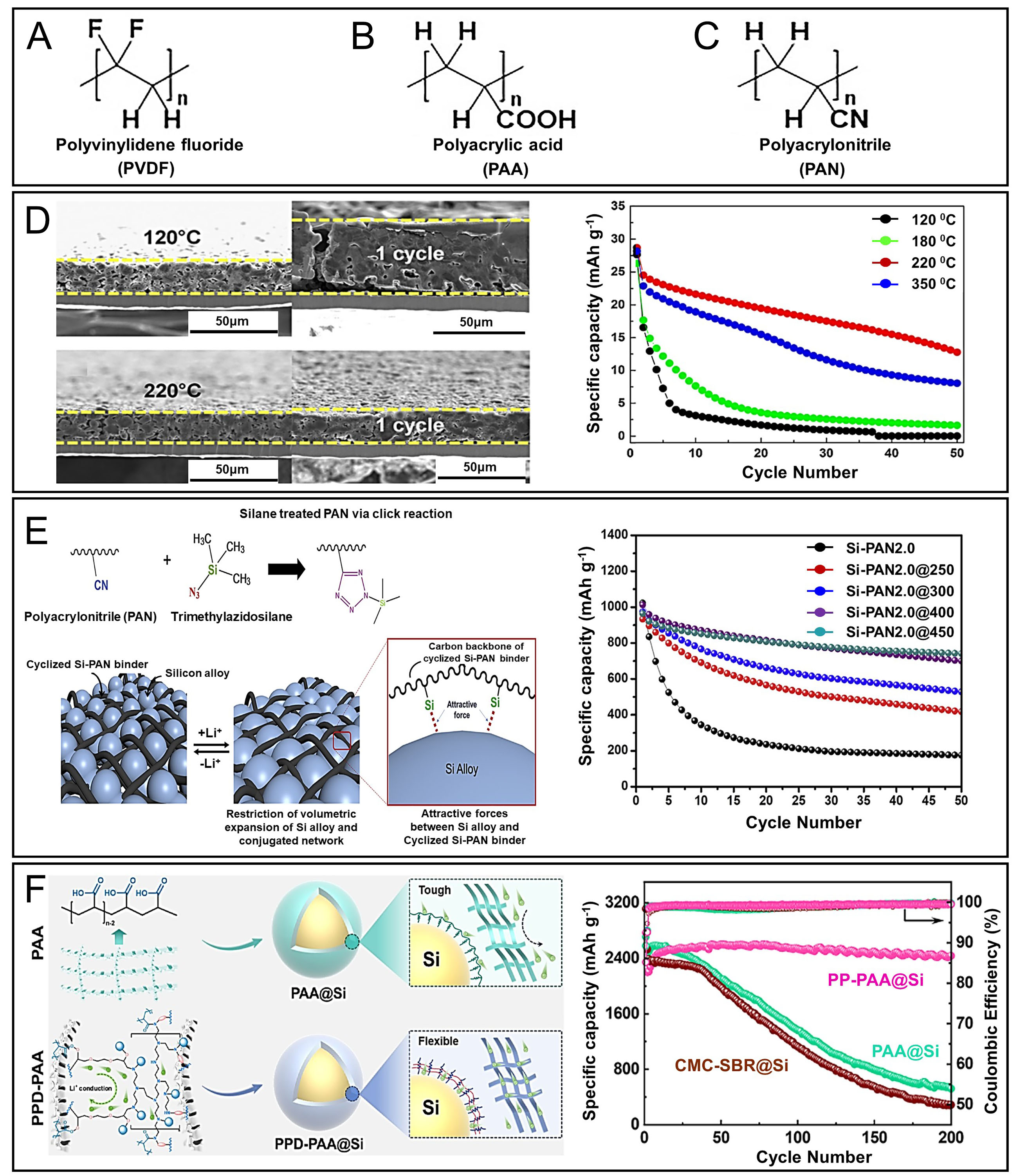
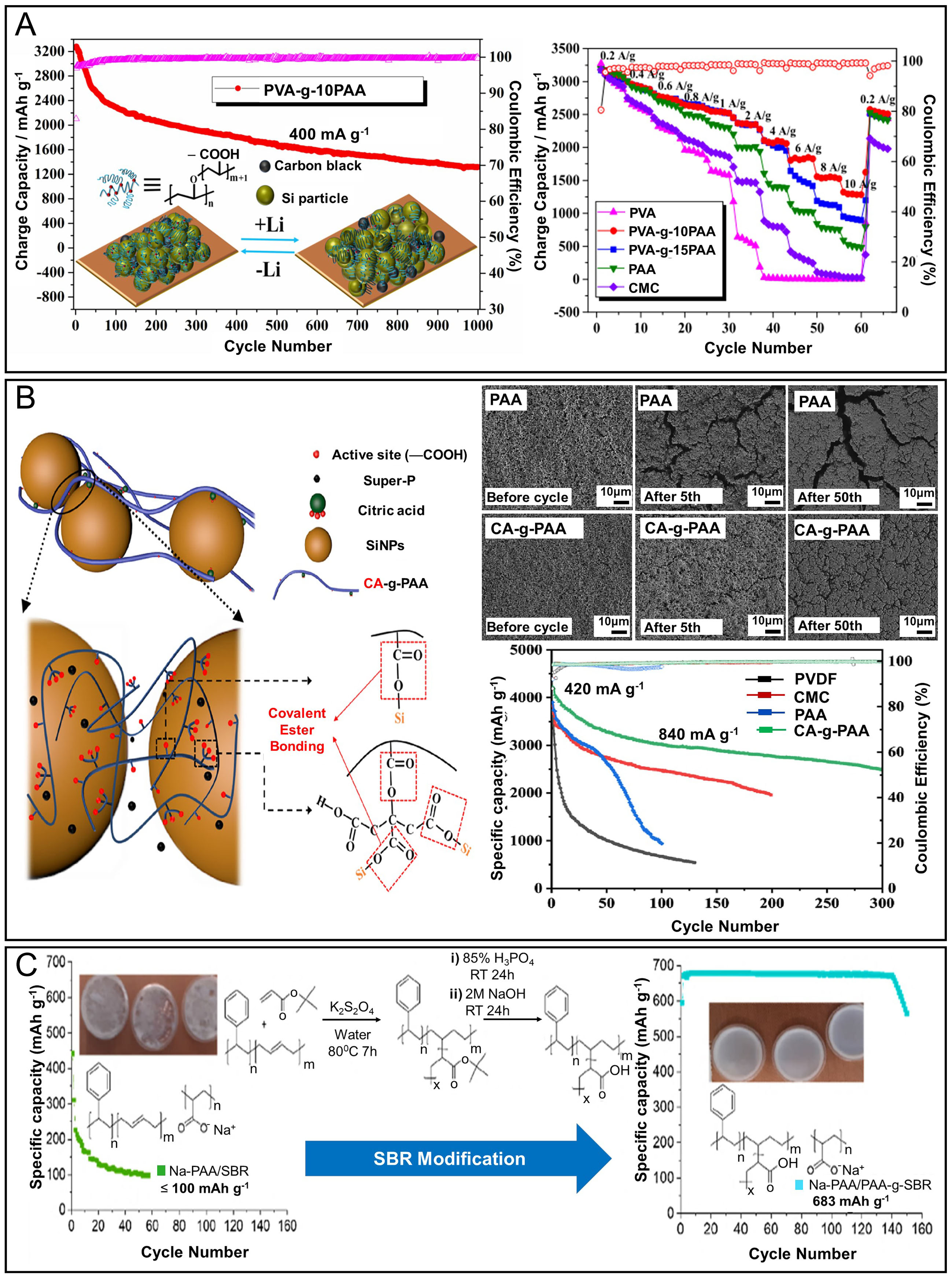
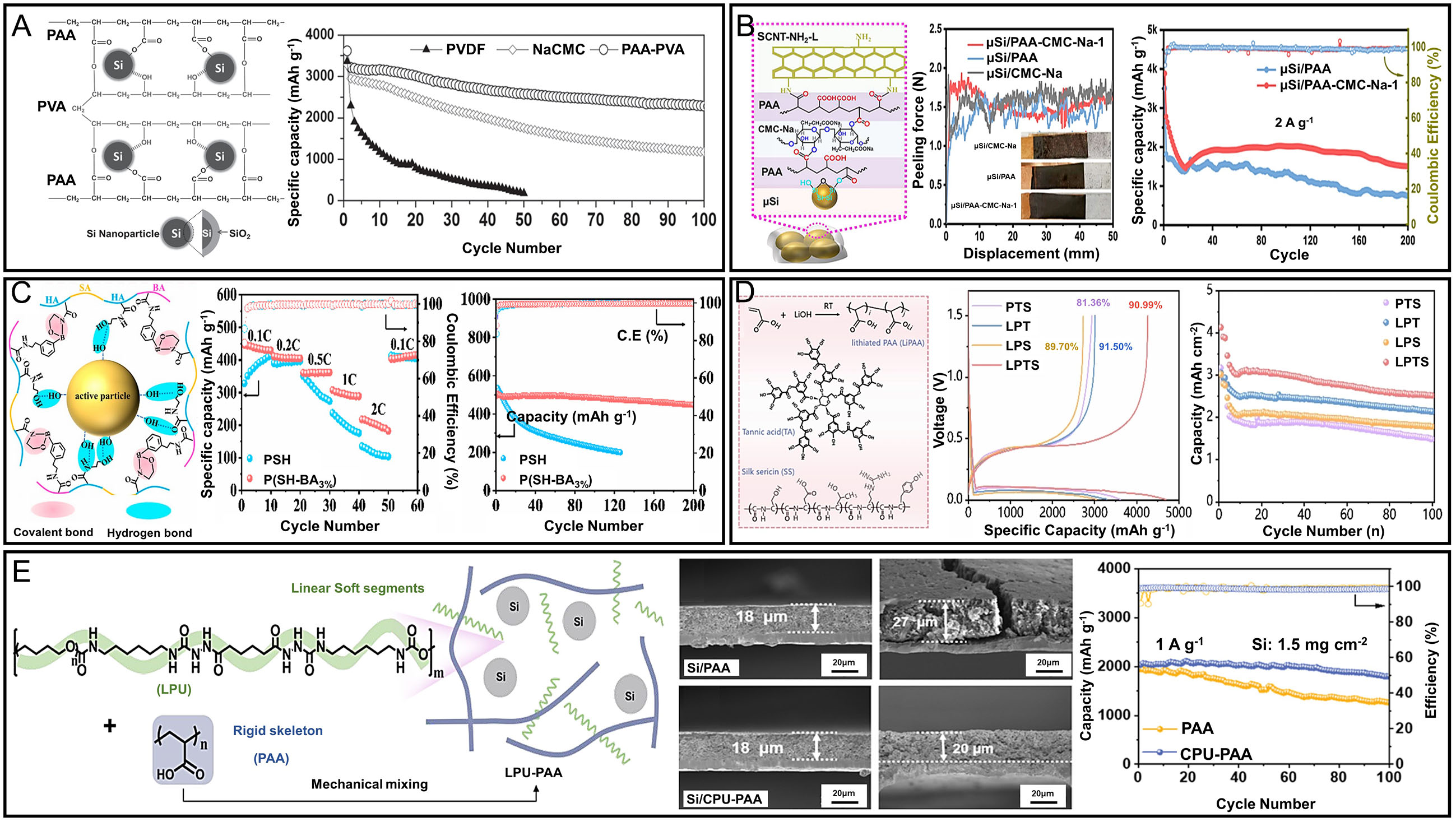
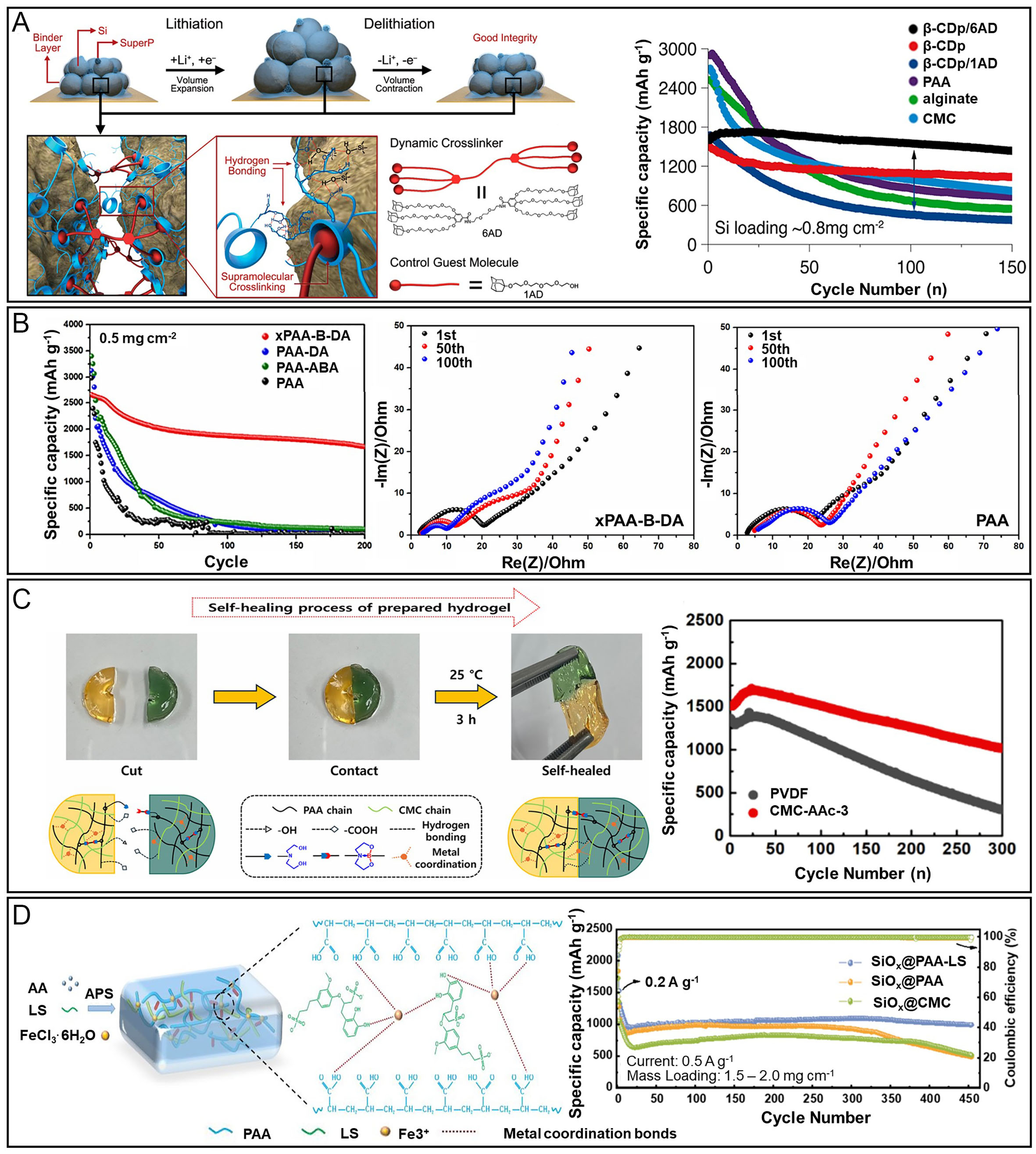
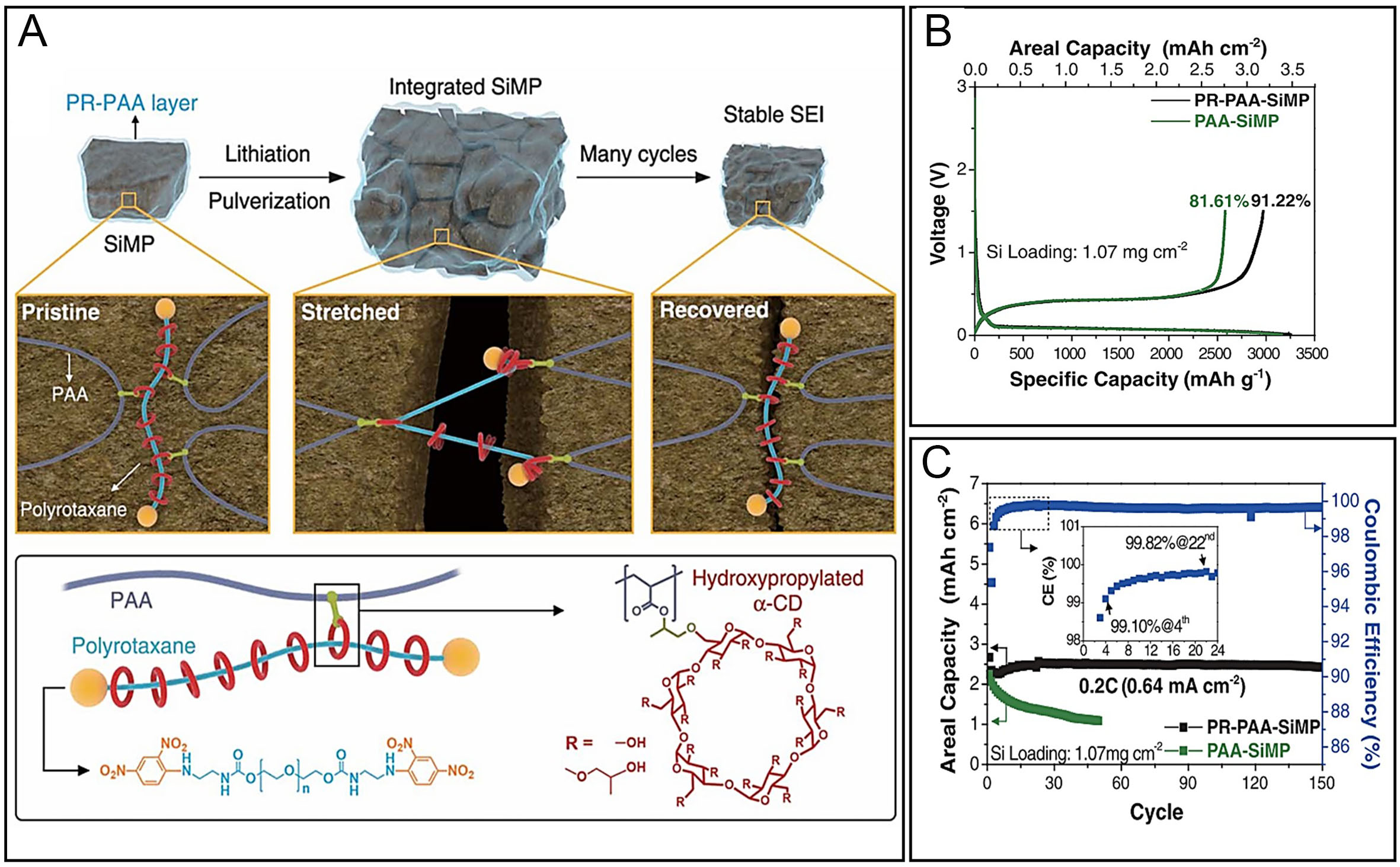
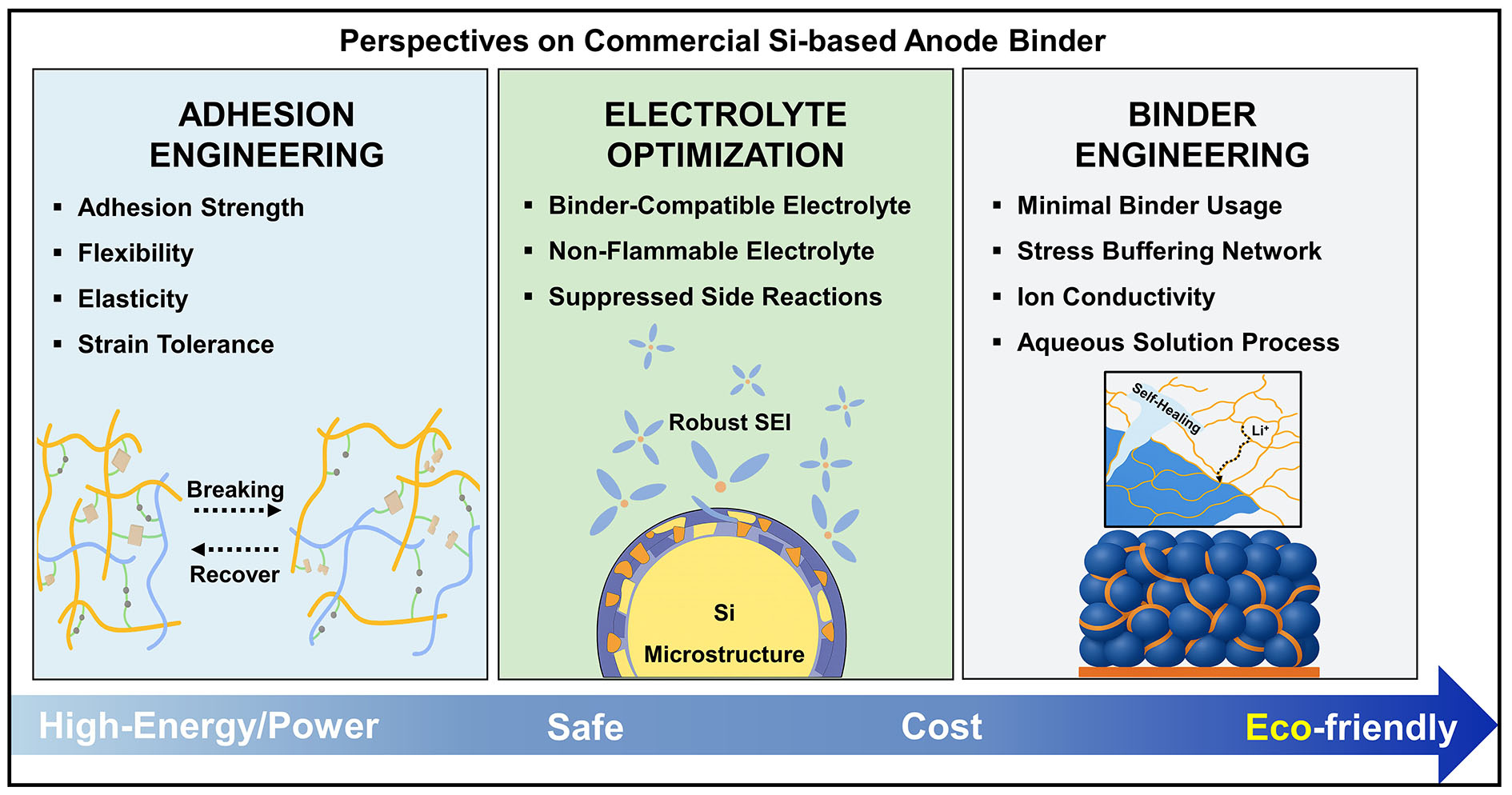












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.