Insights on electrolyte engineering toward aqueous zinc-ion batteries
Abstract
The pursuit of safety and efficiency in electrochemical energy storage and conversion systems has long been a central theme. Among these systems, aqueous zinc-ion batteries (AZIBs) are considered promising candidates for next-generation energy storage devices due to their high safety, low cost, and high capacity. However, several critical issues associated with Zn2+ ion transport, including dendrite formation and side reactions at zinc (Zn) metal anodes, severely restricted their practical applications. As the “blood” of AZIBs, electrolytes play a crucial role in stabilizing Zn metal anodes by introducing various components or optimizing the liquid environment. Therefore, a comprehensive understanding of electrolyte engineering for AZIBs is of great significance. In this review, the development of electrolytes is first discussed. Then, the roles of electrolytes in AZIBs are summarized based on recent advances, including regulation of the solvation process, optimization of the solid electrolyte interphase layer, and modulation of ionic transport. Finally, perspectives on the further development of electrolytes for AZIBs are provided. This review may offer valuable insights for the design of functional electrolytes for advanced electrochemical energy storage and conversion systems.
Keywords
INTRODUCTION
In recent years, environmental pollution and the climate change crisis stemming from excessive reliance on fossil fuels have become increasingly prominent, resulting in a significant surge in global demand for clean and sustainable energy sources[1-3]. This trend has driven continuous growth in the proportion of electricity generated from renewable energy sources, such as wind, wave, and solar power[4-6]. However, the inherently intermittent nature of these energy sources leads to substantial fluctuations in power output, thereby necessitating advancements in energy storage technologies[7-9]. In this context, energy storage systems represented by lithium-ion batteries (LIBs) have emerged, creating new opportunities for the development of clean energy technologies[10-12]. Nevertheless, their intrinsic safety concerns, as well as the gradual limitation in achievable energy density, have limited further development[13-15]. Therefore, exploring novel battery systems remains a key direction for the effective utilization and expansion of clean energy.
Rechargeable aqueous batteries represent an advanced class of energy storage systems and have attracted considerable attention due to their non-toxicity and fast ion-transfer kinetics[16-20]. Among these, aqueous zinc-ion batteries (AZIBs) have been widely investigated because of their promising overall performance in meeting current high-level application requirements[21]. First, zinc (Zn) is readily available and cost-efficient[22-24]. Compared with alkali metals such as lithium and sodium, Zn demonstrates superior chemical stability and environmental compatibility in aqueous media[25,26]. Moreover, AZIBs offer competitive energy density and have broad prospects for large-scale energy storage applications[27-29]. In recent years, numerous studies have focused on the design and construction of high-performance AZIBs to advance energy storage technologies.
Despite these advantages, the development of AZIBs still faces numerous obstacles[30]. Among them, the formation of Zn dendrites is one crucial bottleneck, as these dendrites can penetrate the separator and cause internal short circuits within the battery[31-33]. In addition, the hydrogen evolution reaction (HER) and surface corrosion further restrict the practical application of AZIBs. In mildly acidic electrolytes, Zn typically exists in a thermodynamically unstable state, which can induce parasitic HER due to the reduction of solvated water during Zn2+ ion plating[34-40]. The generation of H2 gas can increase the local pH, thereby facilitating the formation of insulating byproducts[34,41-43]. Consequently, significant challenges remain in achieving the practical application of AZIBs[44-48]. Electrolytes, often regarded as the “blood” of AZIBs, play a critical role in stabilizing Zn metal anodes. First, the development of functional electrolytes can regulate the solvation structure of Zn2+ ions, weakening Zn-H2O interactions. This process helps redistribute Zn2+ ion flux on the anode surface and promotes uniform zinc deposition[49,50]. In addition, rational electrolyte design could decrease the content of free and solvated water molecules while facilitating the formation of a robust solid electrolyte interphase (SEI)[51,52]. Furthermore, the transport kinetics of Zn2+ ions could be boosted by modulating the electrolyte structure[53,54]. Therefore, a comprehensive understanding of electrolyte mechanisms at Zn metal anodes is essential for advancing aqueous battery technologies.
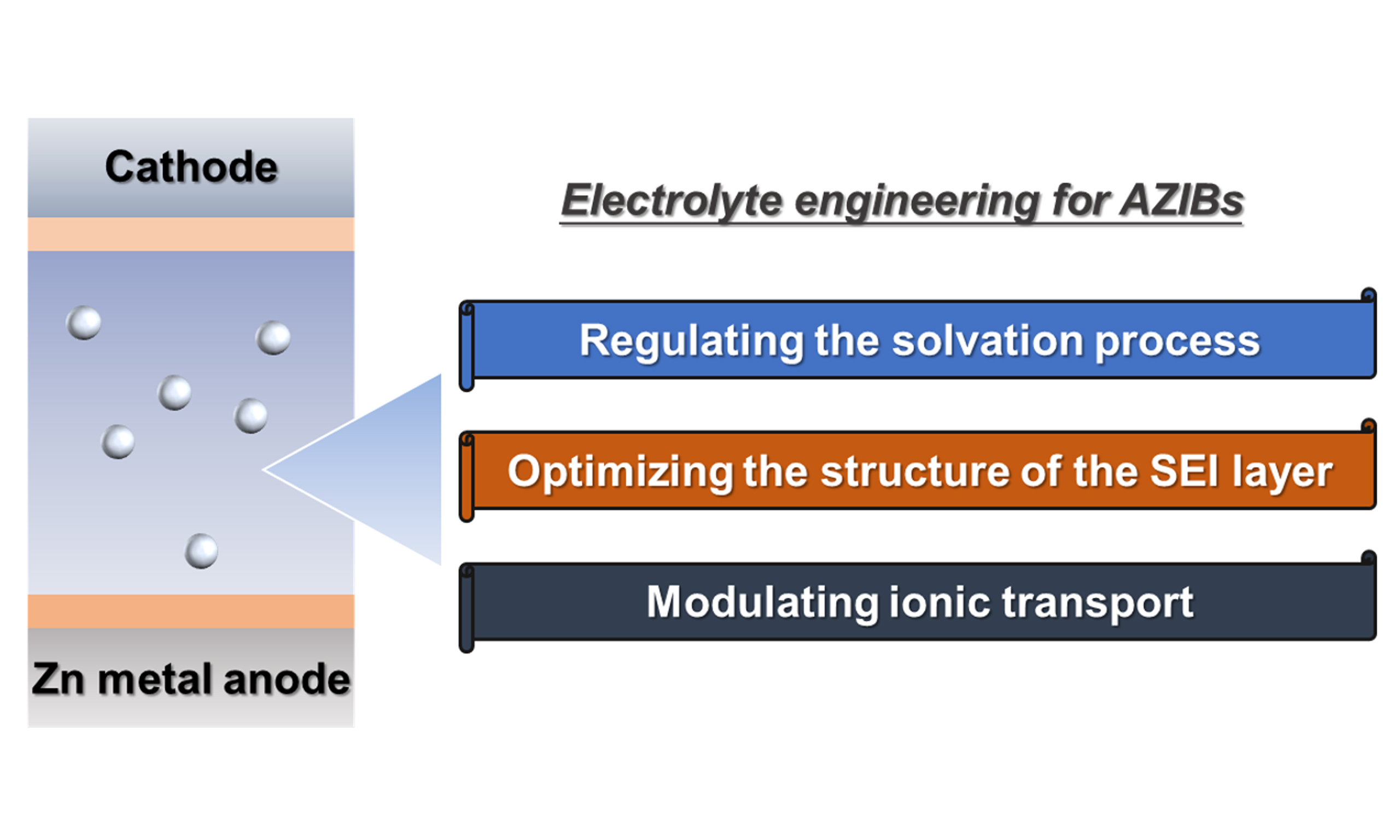
In this paper, recent developments in electrolytes for AZIBs are first reviewed. Then, the mechanisms of electrolyte engineering are discussed, focusing on three main aspects: regulation of the solvation structure, optimization of the SEI layer, and modulation of ionic transport. Finally, perspectives are offered to boost the development of the electrolytes for AZIBs.
DEVELOPMENT, MECHANISM, AND MAIN TYPES OF ELECTROLYTES FOR AZIBS
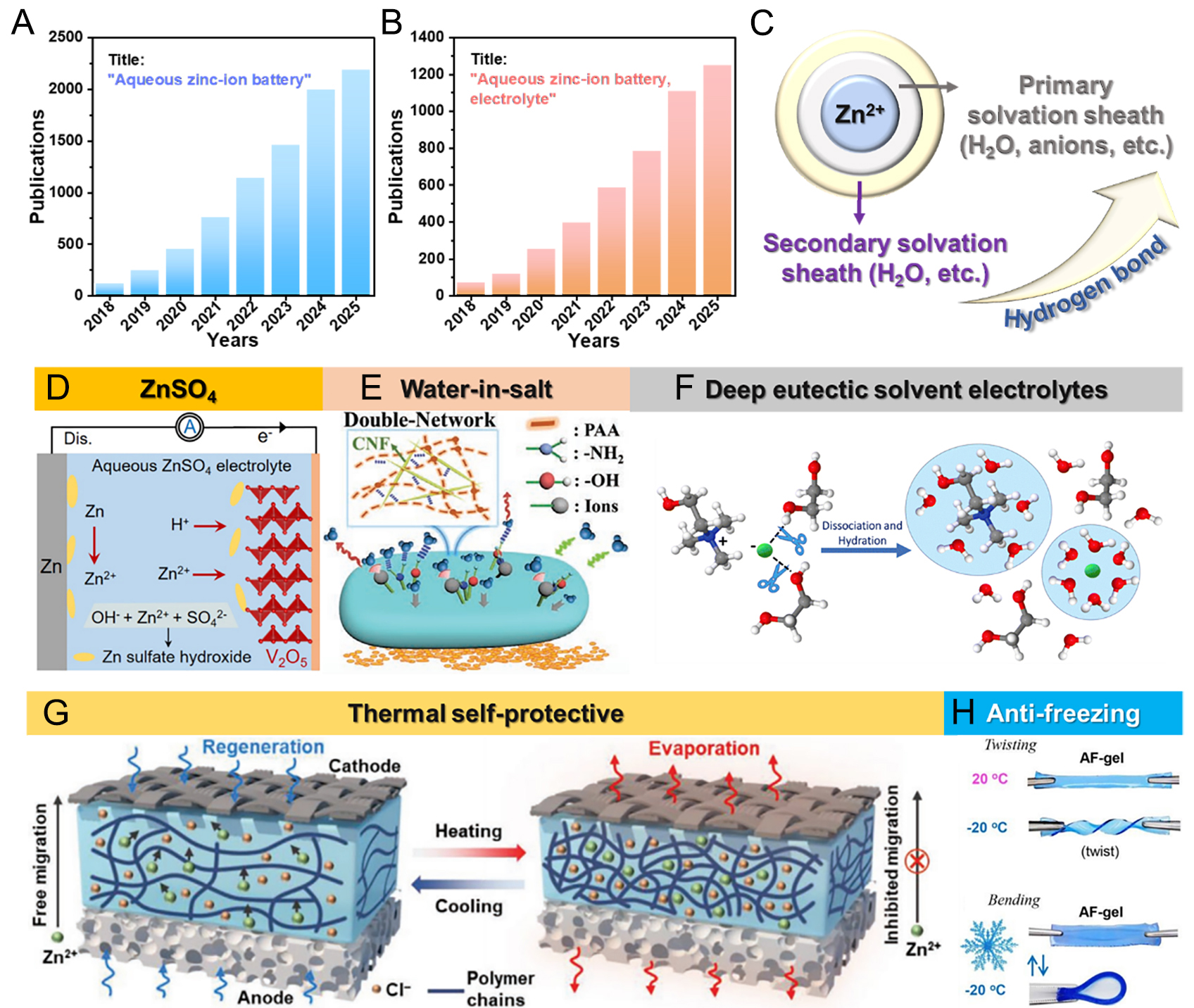
Among numerous electrochemical energy storage and conversion devices, AZIBs have experienced rapid development in recent years, as reflected by the increasing number of publications with the title “aqueous zinc-ion battery,” as shown in Figure 1A. Among the various components of AZIBs, electrolytes have received increasing attention, demonstrating their critical role in determining battery performance
Figure 1. Number of publications per year from the Web of Science for titles containing (A) Aqueous zinc-ion battery” and (B) “Aqueous zinc-ion battery, electrolyte”. (C) Schematic illustration of the solvation structure of Zn2+ ions. Typical examples of electrolytes for AZIBs: (D) ZnSO4-based electrolyte (Reproduced with permission from[58]. Copyright 2020, American Chemical Society); (E) water-in-salt electrolyte (Reproduced with permission from[61]. Copyright 2022, Wiley-VCH GmbH); (F) deep eutectic solvent electrolyte (Reproduced with permission from[62]. Copyright 2025, The Royal Society of Chemistry). Temperature-responsive electrolytes: (G) thermal self-protection (Reproduced with permission from[64]. Copyright 2022, Wiley-VCH GmbH); (H) anti-freezing electrolyte (Reproduced with permission from[65]. Copyright 2019, The Royal Society of Chemistry).
Regarding the underlying mechanism, solvation sheaths typically consist of two layers [Figure 1C]. The primary solvation sheath includes H2O molecules, anions, and other coordinating species, whereas the secondary solvation sheath mainly comprises H2O molecules and other species[55-57]. Continuous optimization of this solvation structure is an important strategy for improving the performance of AZIBs.
With respect to electrolyte composition, most commonly used electrolytes for AZIBs are based on ZnSO4 [Figure 1D][58-60]. In recent years, “water-in-salt” electrolytes and “deep eutectic solvent electrolytes” have become research hotspots due to their desirable electrochemical performance in AZIBs
MECHANISM OF ELECTROLYTE ENGINEERING FOR AZIBS
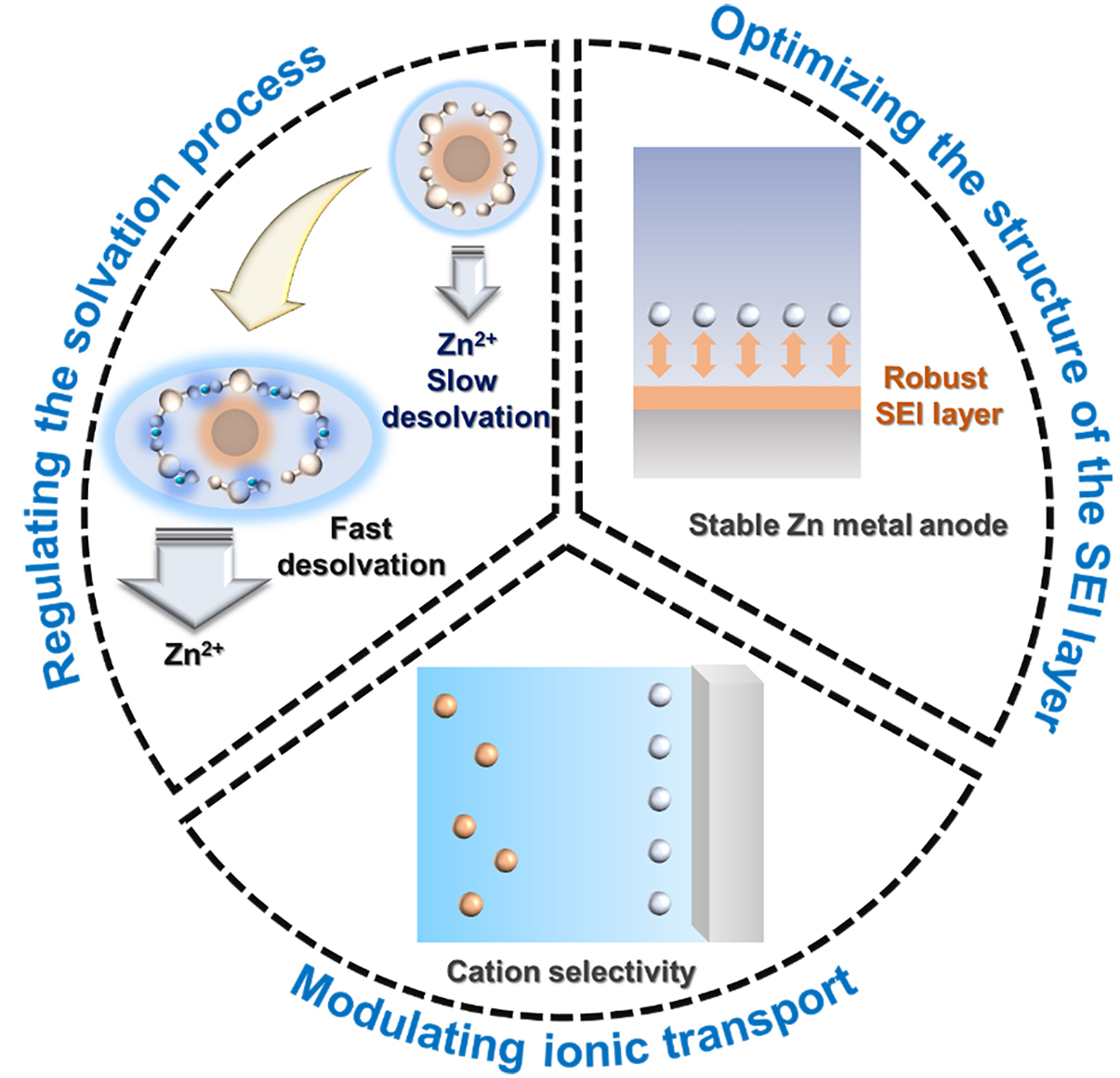
The formation of Zn dendrites, HER, and surface corrosion are closely related to the migration behavior of Zn2+ ions in aqueous electrolytes. First, the desolvation process occurs in the electrolyte. Optimizing the component of the electrolyte can realize multiple functions, such as modifying the solvation sheath to reduce the energy barrier of the desolvation process[67]. Subsequently, the structure of SEI could also be influenced by the use of different electrolytes[68]. In addition, the transport properties of Zn2+ ions can affect dendritic formation. On one hand, promoting Zn2+ ion transport while inhibiting the transport of free anions can suppress the nucleation of Zn dendrites[69-71]. On the other hand, a homogeneous distribution of Zn2+ ions can suppress their growth process[27,72,73]. Therefore, regulating Zn2+ ion transport in AZIBs is of great significance. Among various approaches, electrolyte engineering is considered an effective strategy due to its intrinsic influence on ionic transport properties. Thus, electrolyte engineering plays a critical role in achieving stable AZIBs [Figure 2].
Table 1 summarizes representative examples of the three main strategies, including electrolyte materials and their corresponding electrochemical performance.
Typical examples of electrolyte engineering strategies, including electrolyte materials and their typical electrochemical performance
| Strategies | Electrolyte materials | Typical performance | Ref. |
| Regulating the solvation process | Tripropylene glycol electrolyte additive | Zn-MnO2 full cell (specific capacity of 124.48 mAh g-1 after 1,000 cycles at a current density of 4 A g-1) | [74] |
| Regulating the solvation process | Eutectic electrolyte consisting of ethylene glycol (EG) and ZnCl2 | Polyaniline-Zn cell (approximately 78% capacity retention after 10,000 cycles) | [75] |
| Regulating the solvation process | Aqueous Zn2+ electrolyte with a high-concentration Na salt as a supporting electrolyte | Na2V6O16·nH2O-Zn batteries (negligible capacity decay over 2,000 cycles at 4 A g-1) | [76] |
| Regulating the solvation process | Choline glycerophosphate (CGP) zwitterionic electrolyte additive | Zn//NH4V4O10 battery (217 mAh g-1 over 14,000 cycles at 5 A g-1) | [77] |
| Regulating the solvation process | Butanone (low dielectric constant (ε) and donor number (DN)) as an additive to 0.5 M ZnSO4 | Rechargeable AA-type Zn/NaV3O8·1.5H2O (NVO) cells (capacity of 101.7 mAh and 96.1% retention at 0.66 C after 30 cycles) | [78] |
| Regulating the solvation process | Disodium succinate (SADS) added to 2 M ZnSO4 electrolyte | Zn||MnO2 full cell (80% capacity retention after 1,000 cycles) | [79] |
| Regulating the solvation process | Biocompatible hydrogel electrolyte based on hyaluronic acid | Zn//LiMn2O4 pouch cell (82% capacity retention after 1,000 cycles at 3 C) | [80] |
| Regulating the solvation process | Fire-retardant triethyl phosphate-coordinated electrolyte | Zn/V2O5 full cell (250 mAh g-1 for over 1,000 cycles at 5 A g-1) | [81] |
| Regulating the solvation process | Dual-network hydrogel composed of modified polysaccharides (carboxymethyl chitosan) and polyacrylamide | Zn/polyaniline battery (reversible capacity of 70 mAh g-1 at 5 A g-1 for 2,500 cycles) | [82] |
| Optimizing the structure of the SEI layer | Zincophobic electrolyte containing succinonitrile | Soft-pack battery (energy density of | [83] |
| Optimizing the structure of the SEI layer | Bio-inspired polyanionic electrolytes | Pouch cells (capacity retention of 96.9 %) | [84] |
| Optimizing the structure of the SEI layer | Acetamide-Zn(TFSI)2 eutectic electrolyte | Zn//V2O5 cell (stable operation over 600 cycles at 8.43 mA cm-2 with only 0.0035% cycle-1 capacity fading per cycle) | [85] |
| Modulating ionic transport | Inorganic high-concentration colloidal electrolyte | Zn/α-MnO2 cells (89% capacity retention at 500 mA g-1 after 1,000 cycles) | [86] |
| Modulating ionic transport | Nanocellulose-carboxymethylcellulose hydrogel electrolyte | Zn||MnO2 batteries (over 500 cycles at 8 C) | [87] |
| Modulating ionic transport | Rigid hydrogel electrolytes | Zn||V2O5 pouch batteries (excellent cycling life and stable operation even under extreme conditions) | [88] |
| Modulating ionic transport | Recyclable and biodegradable hydrogel electrolyte based on natural biomaterials (chitosan and polyaspartic acid) | Zn/MnO2 battery (negligible capacity decay over 5,000 cycles at 5 A g-1) | [89] |
| Modulating ionic transport | Anti-freezing hydrogel electrolyte based on zinc tetrafluoroborate and polyacrylamide | Anti-freezing flexible ZIBs (stable performance under bending and at -70 °C) | [90] |
Regulating the solvation process
The solvation structure of electrolytes is closely related to the desolvation ability of metal ions and plays a critical role in stabilizing overall electrochemical performance[91-93]. The interactions among Zn2+ ions, anions, solvents, and electrolyte additives directly determine the solvation structure in the electrolyte[94,95]. Regulating the solvation process to promote the transport of charge carriers is a promising strategy for electrolyte modification.
In 2012, Xu et al.[96] illuminated that during cycling, two distinct peaks appeared at approximately 1.3 and
Figure 3. (A) Cyclic voltammograms of the Zn anode (red line) and the amorphous manganese dioxide (a-MnO2) cathode (blue line) at
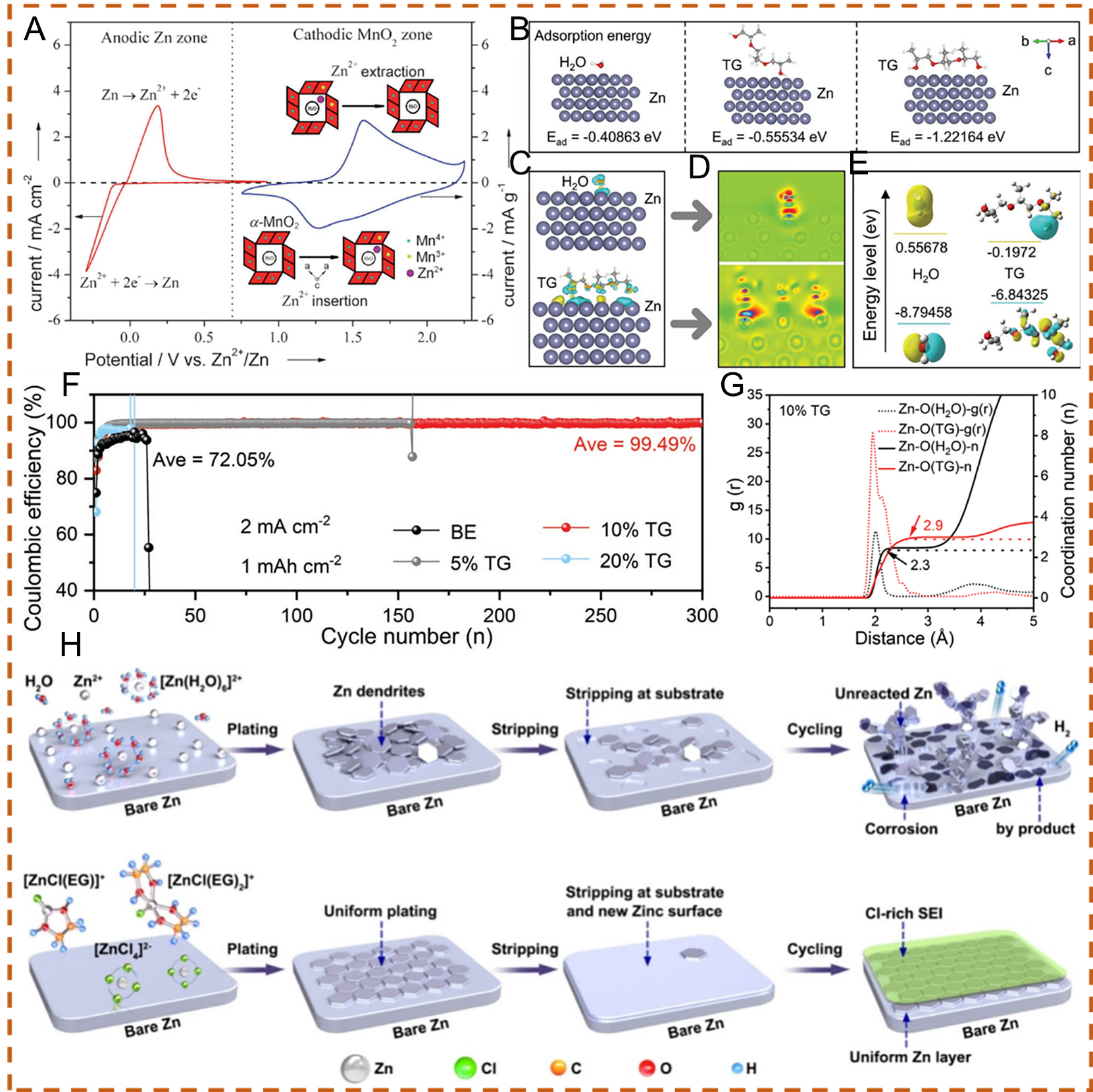
Participating in the Zn2+ ions solvation via electrolytes modulation can optimize the stability of zinc metal anodes. Based on this concept, Geng et al.[75] developed a eutectic electrolyte composed of ethylene glycol (EG) and ZnCl2. Compared with aqueous electrolytes that cause dendritic formation, EG molecules participate in the Zn2+ solvation structure within deep eutectic solvents (DESs), forming cations such as [ZnCl(EG)]+ and [ZnCl(EG)2]+. The dissociation and reduction of these complex cations facilitate the formation of an organic-inorganic hybrid SEI (Cl-rich) during the initial Zn plating process. This stable SEI layer acts as a protective barrier that regulates Zn2+ ion flux toward uniform Zn plating [Figure 3H][75,98]. This study provides a platform for the development of eutectic electrolytes for high-safety and practical AZIBs.
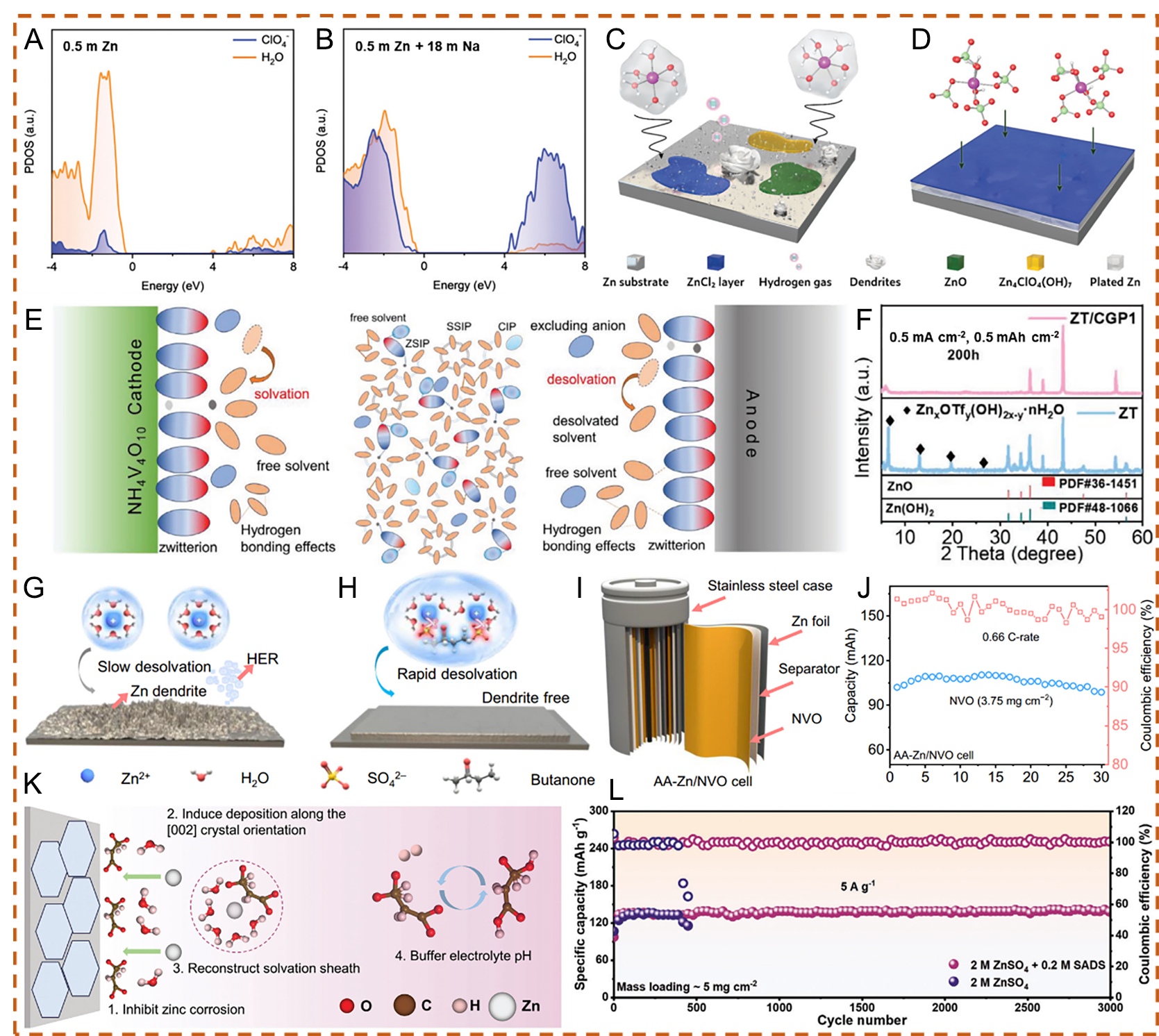
Further regulation of the solvation process can also be achieved by tailoring the cation solvation shell, thereby suppressing water activity. For instance, Zhu et al.[76] applied a high-concentration Zn-Na perchlorate mixture in water. The influence of solute concentration on the electronic structure of the electrolytes was investigated using density functional theory (DFT) [Figure 4A and B]. For the dilute electrolyte (0.5 m Zn(ClO4)2), the lowest unoccupied molecular orbital (LUMO) was primarily contributed by water, leading to preferential water reduction on the Zn metal surface and rapid capacity degradation[76]. In such dilute electrolytes, the presence of unstable [Zn(H2O)6]2+ complexes could result in various irreversible parasitic reactions, which compete with Zn plating/stripping at low potentials and promote Zn dendrite formation. In the highly concentrated electrolyte, the primary solvation shell of Zn2+ ions is predominantly occupied by ClO4- ions. The reduced affinity between H2O and Zn2+ mitigates parasitic reactions at the Zn metal anode due to the strong coordination between ClO4- to Zn2+. The synergistic effects of the ZnCl2 passivation layer formed via perchlorate reduction and the strong Zn2+-ClO4- interactions, which increase the desolvation energy of Zn2+, enable highly efficient and stable Zn plating/stripping
Figure 4. Projected density of states (PDOS) plots of 0.5 m Zn(ClO4)2 (A) and 0.5 m Zn(ClO4)2 + 18 m NaClO4 (B). Schematic illustration of Zn2+ solvation structures and the corresponding interfacial reactions in (C) 0.5 m (ZnClO4)2 and (D) 0.5 m Zn(ClO4)2 + 18 m NaClO4. (A-D) Reproduced with permission from[76]. Copyright 2021, The Royal Society of Chemistry. (E) Schematic illustration of solvent-related decomposition in conventional electrolytes for AZIBs. (F) X-ray diffraction (XRD) patterns after 100 cycles in ZT and ZT/CGP1 electrolytes. (E and F) Reproduced with permission from[77]. Copyright 2024, Wiley-VCH GmbH. Schematic diagrams of Zn anode behavior in (G) localized concentrated electrolyte (LCE) and (H) WSE. (I) Schematic illustration of AA-type Zn/NVO cells. (J) Cycling stability at 0.66 C for AA-type Zn/NVO cells. (G-J) Reproduced with permission from[78]. Copyright 2024, The Author(s). (K) Illustration of the protection mechanism of the SADS additive on the Zn anode. (L) Long-term cycling performance of Zn||I2 full cells at 5 A g-1.
Similarly, to reduce the solvating power of H2O and strengthen the coordination of SO42- with Zn2+ over H2O, Shi et al.[78] proposed a weakly solvating electrolyte (WSE) for practical rechargeable AZIBs. When using WSE with a Zn metal anode, a water-deficient Zn2+ solvation structure is formed, which suppresses HER-induced corrosion as well as other side reactions. In addition, this solvation structure facilitates rapid Zn2+ migration and desolvation, thereby promoting uniform nucleation and growth [Figure 4G and H]. As a result, AA-type batteries employing WSE demonstrate stable long-term cycling performance
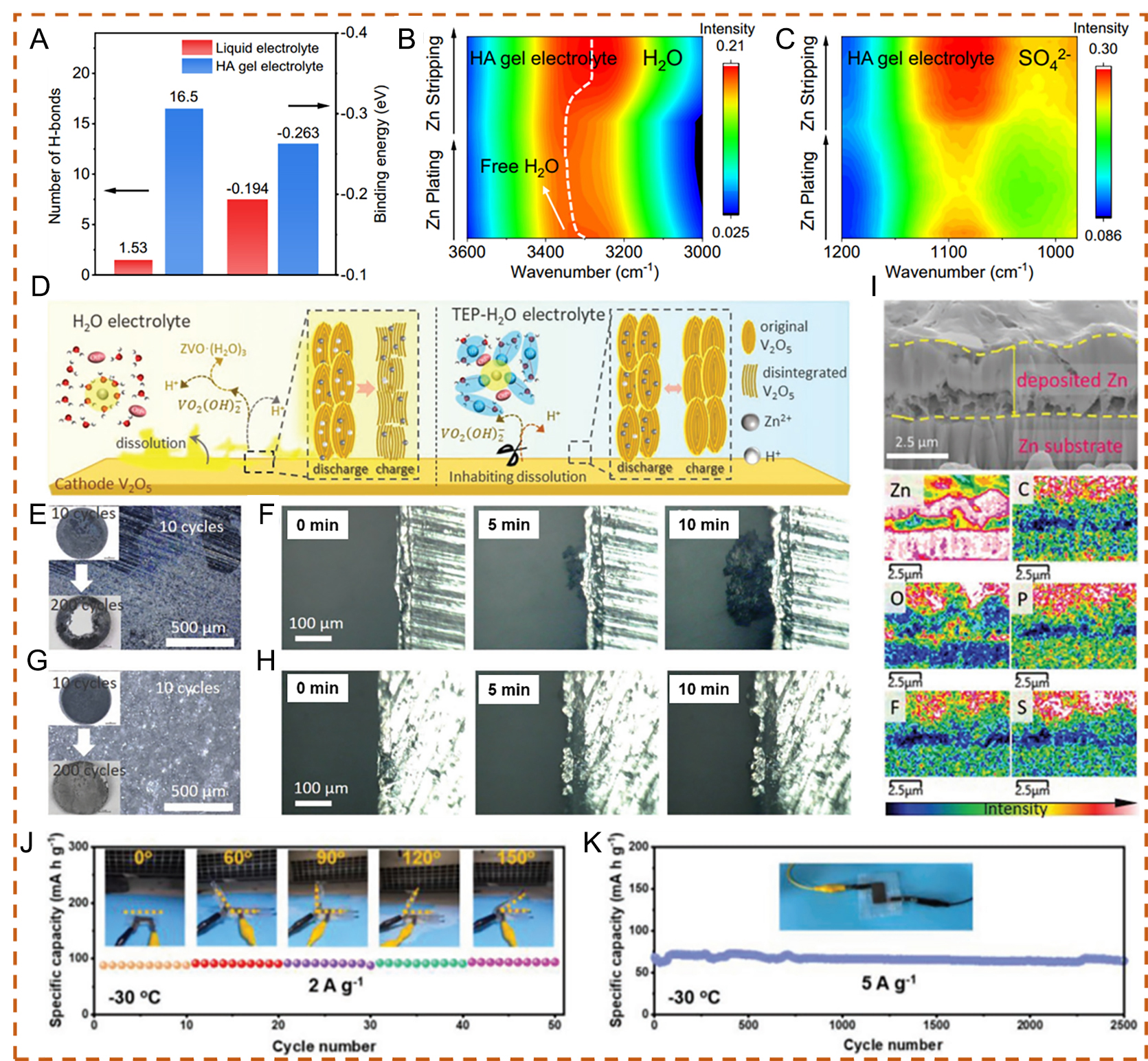
The regulation of hydrogen bonding is another strategy for achieving stable AZIBs via electrolyte engineering. Li et al.[80] designed a biocompatible hydrogel electrolyte using hyaluronic acid (HA), which contains abundant hydrophilic functional groups. Molecular dynamics (MD) simulations showed that a single HA monomer can form up to 16.5 hydrogen bonds, significantly more than a single H2O molecule (1.53), as illustrated in Figure 5A. Additionally, DFT calculations indicated that the binding energy of the H2O-HA interaction is more negative than that of H2O-H2O, suggesting stronger interactions between HA and H2O. During in situ synchrotron Fourier transform infrared (FTIR) spectroscopy measurements, the HA gel electrolyte exhibited a clear blueshift of the O-H stretching vibration toward shorter wavelengths, along with a decrease in the intensity of SO42- anions. These results confirm that both H2O and SO42- could be effectively removed from the solvation shell of Zn2+ ions during Zn deposition. This process promotes a more uniform Zn2+ ion flux and enables uniform nucleation and growth of Zn during the plating/stripping processes [Figure 5B and C][80,100,101]. To evaluate biocompatibility and potential applications, Zn//LiMn2O4 (LMO) cells were constructed with a low N/P ratio (around 3.3) and a thin Zn metal anode (16 µm), achieving desirable electrochemical performance. Similarly, Liu et al.[81] proposed triethyl phosphate (TEP) as a flame-retardant co-solvent with high solvating ability in a nonaqueous-aqueous hybrid electrolyte. In the pristine Zn(OTf)2-H2O electrolyte, adverse effects on the electrochemical performance of V2O5 were observed, including irreversible structural transformation and severe vanadium oxide dissolution, leading to continuous loss of active material and structural degradation. In contrast, the unique solvation structure in the Zn(OTf)2-TEP-H2O electrolyte effectively suppresses water activity and mitigates cathode dissolution under various current densities, enabling stable hosting of V2O5 with highly reversible intercalation
Figure 5. (A) Calculated number of hydrogen bonds for single H2O and HA molecules, and binding energies of H2O-H2O and H2O-HA. (B) ν (OH) spectral of the HA gel electrolyte. (C) ν (SO42-) spectra of the HA gel electrolyte. (A-C) Reproduced with permission from[80]. Copyright 2023, The Author(s). (D) Schematic illustration of electrolyte effects on the V2O5 electrode during electrochemical reactions. Images of cycled Zn foil under (E) Zn(OTf)2-H2O and (G) Zn(OTf)2-TEP-H2O electrolytes after 10 and 200 cycles. Real-time images of Zn2+ plating on the Zn substrate under (H) Zn(OTf)2-H2O and (F) Zn(OTf)2-TEP-H2O electrolytes within 10 min. (I) FIB cross-sectional image of the cycled Zn anode and corresponding elemental distribution from energy-dispersive X-ray spectroscopy (EDS) after cycling in Zn(OTf)2-TEP-H2O electrolyte. (D-I) Reproduced with permission from[81]. Copyright 2021, Wiley-VCH GmbH. (J) Demonstration of the battery under different bending angles at 2 A g-1 and -30 °C. (K) Cycling performance of the flexible battery with CSAM-C hydrogel electrolyte at 5 A g-1 and -30 °C. (J and K) Reproduced with permission from[82]. Copyright 2022, Wiley-VCH GmbH.
To further promote the development of new-generation flexible AZIBs, Huang et al.[82] in situ constructed a highly flexible polysaccharide hydrogel electrolyte. In this system, chaotropic anions (ClO4-), water molecules, and polymer chains form weak ternary hydrogen-bonding interactions. These interactions enhance the mechanical properties of the polymer network, disrupt hydrogen bonding among water molecules to significantly lower the freezing point of the electrolyte, and reduce the amount of free water, thereby suppressing side reactions and dendrite formation. As a result, flexible Zn/polyaniline (PANI) full batteries using this electrolyte exhibited negligible capacity variation when tested at -30 °C under different bending angles (0° to 150°), demonstrating high ionic conductivity and mechanical flexibility under subzero conditions [Figure 5J]. Furthermore, the flexible battery maintained stable long-term cycling performance, delivering a specific capacity of 64 mA h g-1 at -30 °C and a current density of 5 A g-1 [Figure 5K]. This study provides a facile and universal strategy for developing low-temperature-tolerated electrolytes.
Optimizing the structure of the SEI
In typical batteries, electrochemical instability of the electrolyte can lead to reactive decomposition at the anode surface within the Helmholtz double layer, resulting in the accumulation of reductive products and the formation of the SEI[102,103]. In other words, the SEI layer is formed through the reductive decomposition of electrolytes, accompanied by a dynamic desolvation process[103-105]. Therefore, electrolyte engineering is an important strategy for optimizing the structure of the SEI layer.
The SEI in AZIBs can be classified into several types, including inorganic SEI, organic SEI, and inorganic-organic composite SEI[60,68,106]. In general, inorganic SEI typically consists of dense inorganic components with high mechanical strength and good chemical stability. However, it is usually brittle and prone to cracking due to volume fluctuations during Zn deposition and dissolution. In contrast, organic SEI generally exhibits good flexibility and interfacial compatibility but suffers from relatively low mechanical strength and poor chemical stability, and may dissolve during long-term cycling in AZIBs. Based on these considerations, inorganic-organic composite SEI shows great potential for addressing the above issues.
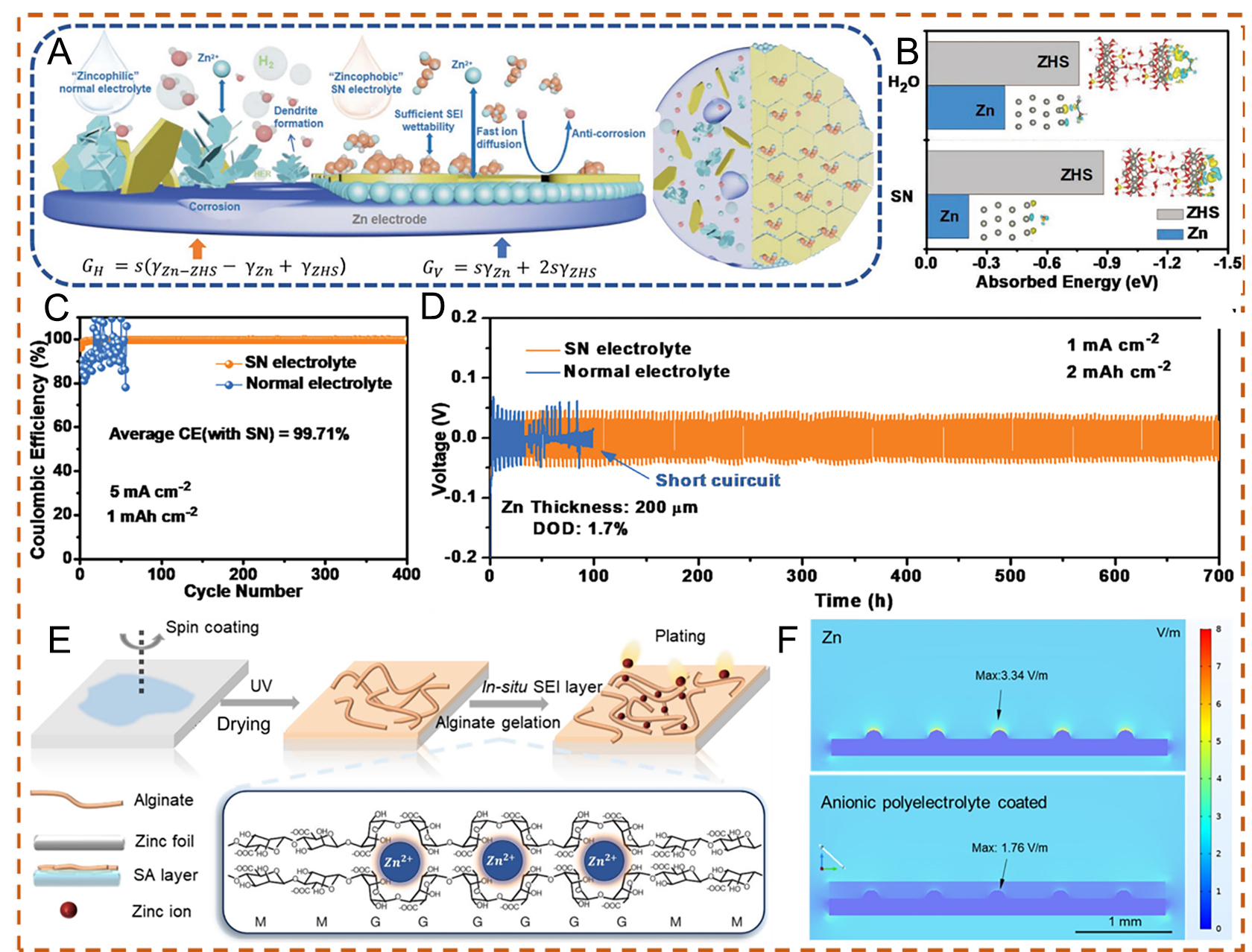
To construct a dense SEI, Wang et al.[83] synthesized a zincophobic electrolyte containing succinonitrile (SN) as an additive. As illustrated in Figure 6A, Zn in conventional aqueous electrolytes is susceptible to multiple side reactions, including HER and corrosion, because the Zn electrode remains in continuous contact with the electrolyte after cell assembly. The intermediate phase, defined as vertically grown zinc hydroxide sulfate SEI (V-SEI), exhibits a loose nanostructure and cannot protect Zn from further corrosion. With continued plating cycles, zinc hydroxide sulfate accumulates, leading to continuous consumption of both electrolyte and anode, which cannot be effectively mitigated. As a result, battery performance deteriorates and eventually fails[72,83,107]. In contrast, organic molecular additives such as SN can chemically adsorb onto the Zn surface, thereby modifying the interfacial affinity between the electrolyte and Zn[83]. Moreover, this electrolyte shows strong affinity for an SEI formed by horizontally stacked Zn hydroxide sulfate (H-SEI), which facilitates fast ionic transfer and suppresses corrosion. DFT calculations further prove that the SN-containing electrolyte exhibits stronger zincophobicity and more favorable interaction with the SEI [Figure 6B]. Accordingly, the SN additive enables high CE at 5 mA cm-2/1 mAh cm-2 and improved long-term cycling stability compared with the baseline electrolyte [Figure 6C and D]. This study offers support for the development of Zn-based electrochemistry in mild electrolyte environments for practical applications.
Figure 6. (A) Schematic illustration of a zincophobic yet SEI-affinitive electrolyte. (B) Absorption energies of H2O and SN on Zn (001) and ZHS. (C) Comparison of long-term CE in two electrolytes. (D) Cycling stability of symmetric cells at low depth of discharge (DOD). (A-D) Reproduced with permission from[83]. Copyright 2023, Wiley-VCH GmbH. (E) Schematic diagram of in situ SEI layer formation. (F) Simulated electric field distribution for bare Zn and SA-coated Zn with uniformly distributed nucleation sites. (E and F) Reproduced with permission from[84]. Copyright 2023, The Authors. Angewandte Chemie published by Wiley-VCH GmbH.
Constructing an anionic polyelectrolyte can be another route for the optimization of SEI. To enable the in situ formation of a high-performance SEI layer, Dong et al.[84] applied an anionic polyelectrolyte, alginic acid (sodium alginate, SA), inspired by biomass derived from seaweed. As presented in Figure 6E, Zn2+ ions are coordinated by the carboxylate groups (-COO-) in the G (guluronic acid) monomers, which induces in situ crosslinking and gelation of a Zn-ion-conductive alginate network. The resulting polyanionic layer functions as an in situ SEI, providing effective protection for the Zn anode. Simulations of the electric field distribution for the anionic-coated Zn anode show a more uniform electric field compared with bare Zn, which typically exhibits a “tip effect” associated with localized high current density during discharge. The hydrogel layer helps to shield Zn nucleation sites, resulting in more uniform plating and reduced corrosion [Figure 6F]. The beneficial effects are further demonstrated in cells with high mass-loading cathodes and low N/P ratios.
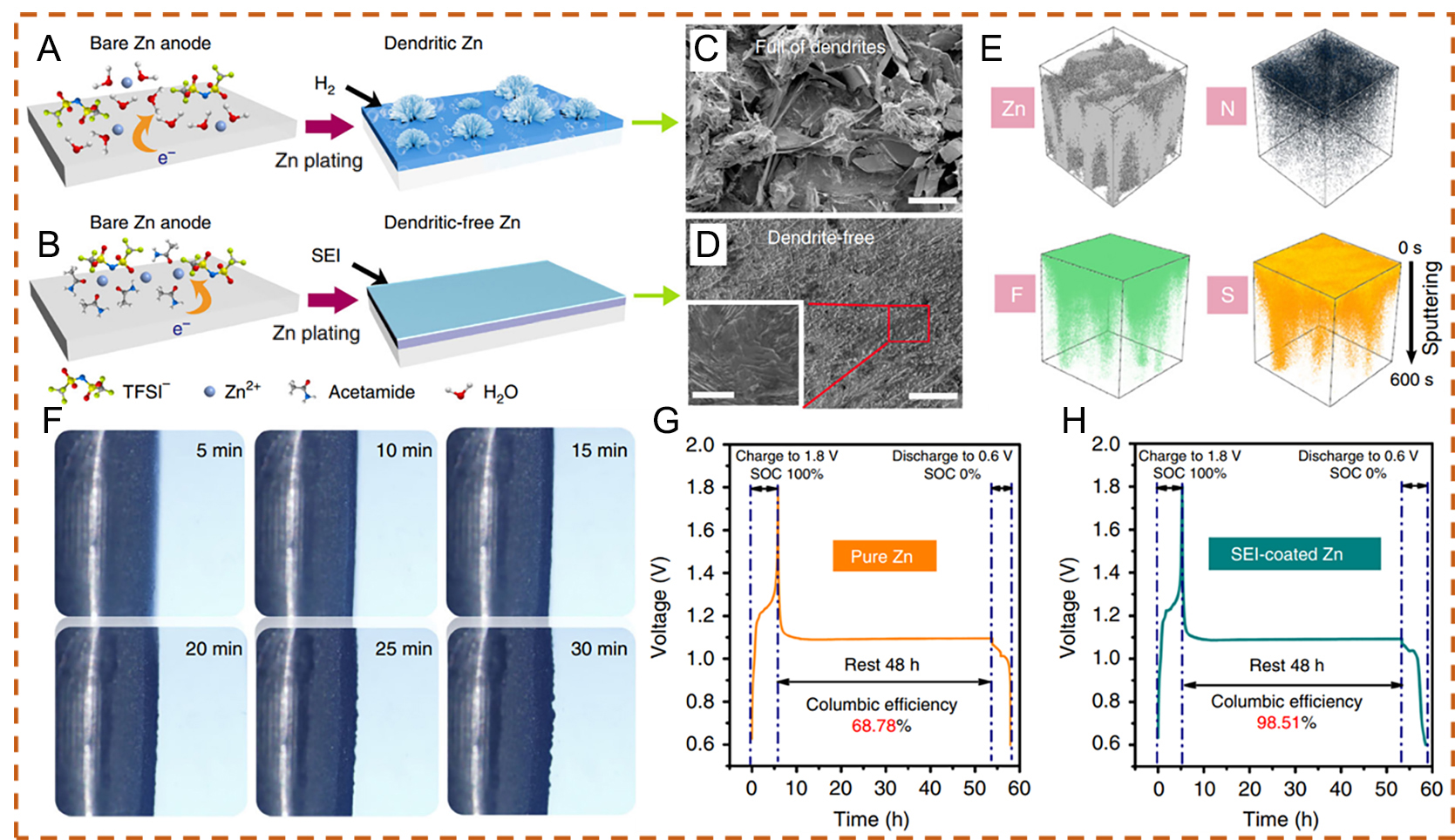
The eutectic electrolyte can regulate the formation of a protective SEI layer on the Zn metal anode. For instance, Qiu et al.[85] proposed an acetamide-Zn(TFSI)2 eutectic electrolyte and observed the formation of a zinc fluoride-rich organic-inorganic hybrid SEI. Compared with a 1 M Zn(TFSI)2 electrolyte, which led to the formation of Zn dendrites, the Zn(TFSI)2-based eutectic solvent (ZES) electrolyte enabled dendrite-free and smooth Zn deposition, as presented in Figure 7A-D. Time-of-flight secondary ion mass spectrometry (TOF-SIMS) further confirmed the uniformity of the SEI layer [Figure 7E]. The SEI-coated Zn anode was obtained after 20 cycles of galvanostatic plating/stripping in the ZES electrolyte (Zn/Zn cells at 0.5 mA cm-2 with a capacity of 1 mAh cm-2 per half-cycle). The homogeneous Zn deposition was further verified by in situ optical visualization [Figure 7F]. The influence of the SEI on parasitic reactions was studied by monitoring the open-circuit voltage decay of fully charged Zn/V2O5 cells in 1 M Zn(TFSI)2 electrolyte. After storage for 48 h at 100% stage of charge (SOC) for 48 h, the batteries were fully discharged. The cell with the SEI-protected Zn anode retained 97.8% of its original capacity, which is significantly higher than that of the untreated Zn anode (68.78%), as presented in Figure 7G and H. The rigid-flexible coupled SEI formed on the Zn surface could inhibit direct contact between the electrolyte and the active Zn anode. Therefore, it effectively suppresses interfacial side reactions, including passivation and H2 evolution during storage. In addition, the coated Zn surface facilitates stable Zn2+ ion transport with a low diffusion barrier once the SEI is formed, thereby promoting reversible Zn stripping and plating, even in aqueous electrolytes. Similarly,
Figure 7. (A) Zn dendrite growth accompanied by H2 evolution in 1 M Zn(TFSI)2 electrolyte and (B) SEI-regulated uniform Zn deposition in ZES electrolyte. Scanning electron microscopy (SEM) images of Zn deposits using (C) 1 M Zn(TFSI)2 and (D) ZES electrolytes at 1 mA cm-2 (0.5 mAh cm-2). (E) Three-dimensional (3D) distributions of Zn, N, F, and S elements in the SEI, obtained from time-of-flight secondary ion mass spectrometry (TOF-SIMS) sputtered volumes. (F) In situ optical microscopy observations of Zn deposition in Zn/Zn cells, showing the Zn-electrolyte interface in ZES. Zn/V2O5 cells in 1 M Zn(TFSI)2 using (G) a pristine Zn anode and (H) an SEI-coated Zn anode were first fully charged to 1.8 V at 20 mA g-1 (based on the cathode active material), respectively. (A-H) Reproduced with permission from[85]. Copyright 2019, The Author(s).
Modulating ionic transport
In addition to the functions discussed above, electrolyte engineering is also related to the modulation of ionic transport in AZIBs[30,109,110]. In general, the transport of charge carriers enables the transfer of charge during the charge-discharge process, while other ions typically make negligible contributions. Therefore, achieving selective transport of charge carriers is a critical objective[111] [Figure 8]. In conventional battery systems, critical objective modification could enhance the transport of charge carriers, particularly by promoting selective and homogeneous ionic transport. For instance, Bai et al. proposed a metal-organic framework (MOF)-modified electrolyte[112]. Compared with the pristine electrolyte, which results in uneven Li+ ion flux and dendrite formation, the MOF-modified electrolyte facilitates ordered ionic transport, thereby enabling stable Li electrodeposition[112]. Similarly, such mechanisms may also play a positive role in the electrochemical deposition of Zn2+ ions.
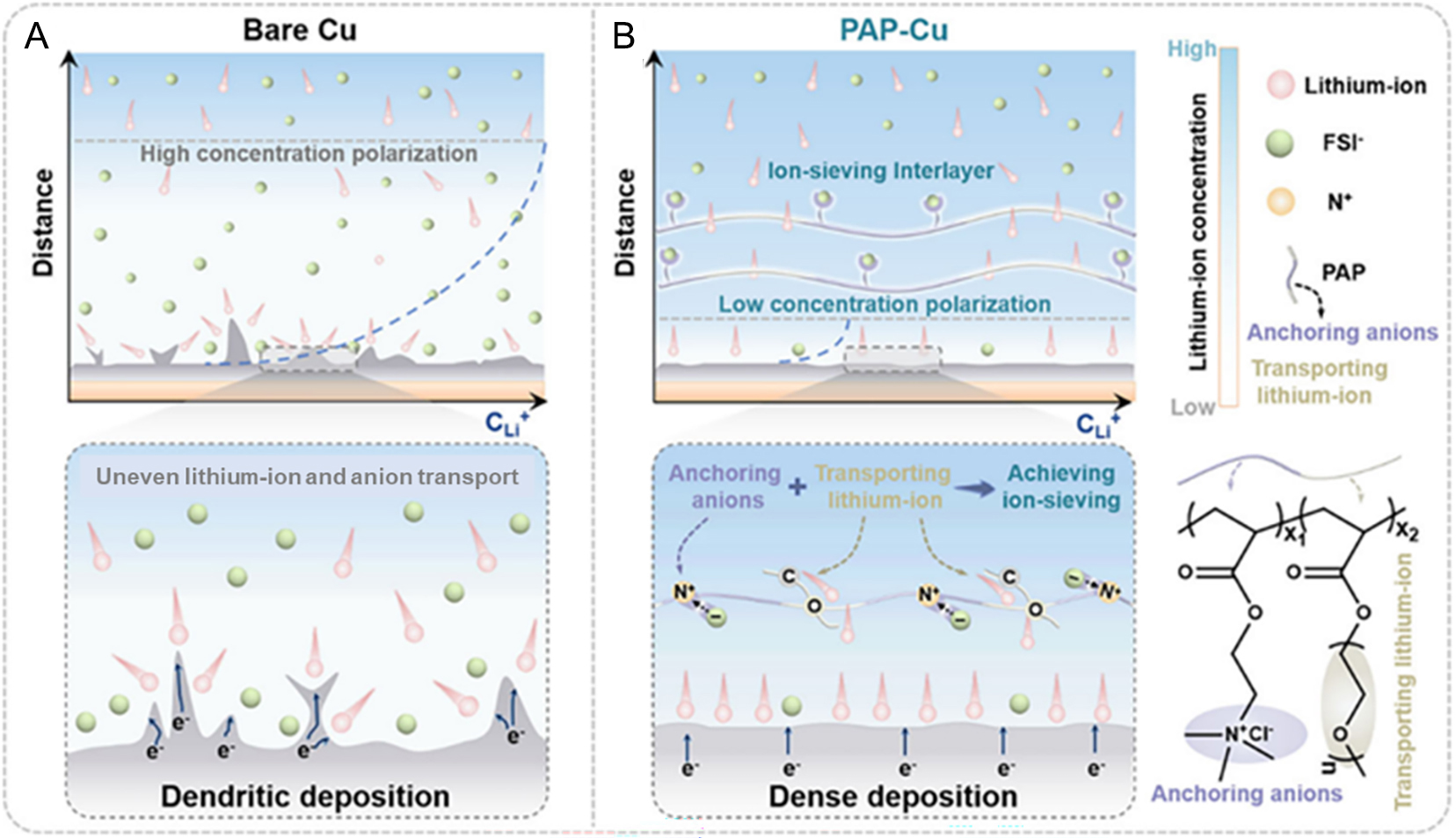
Figure 8. Schematic illustration of lithium plating behavior on Cu foil: (A) without and (B) with a PAP layer. (A and B) Reproduced with permission from[111]. Copyright 2026, Wiley-VCH GmbH.
Toward simultaneously enhancing the Zn2+ ion transference number (tZn2+) and ionic conductivity,
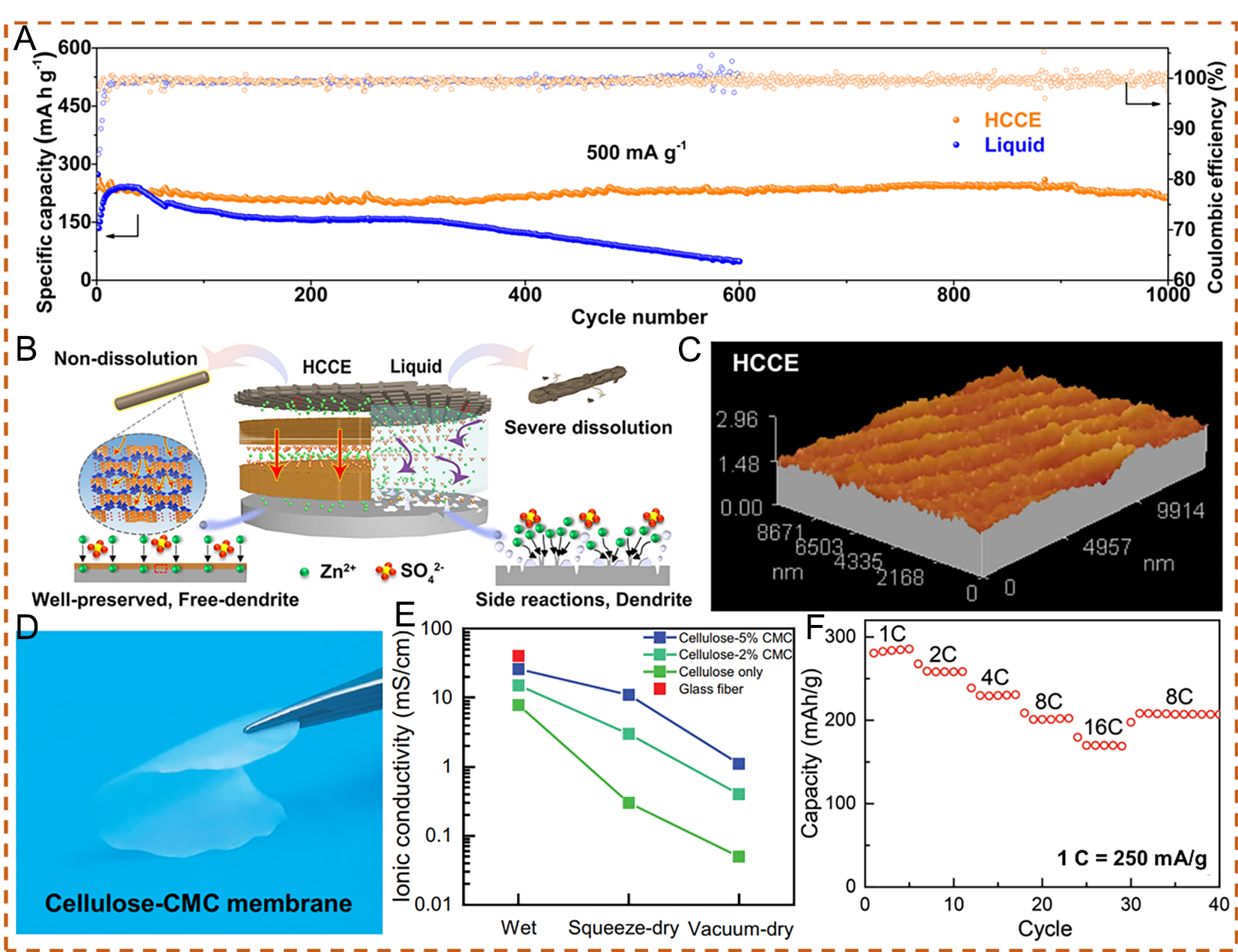
Figure 9. (A) Long-term cycling performance of the cell with HCCE and liquid electrolyte at 500 mA g-1. (B) Schematic diagram of the interfacial protection effect in HCCE and liquid electrolyte. (C) Atomic force microscopy (AFM)images of the Zn anode cycled for 200 cycles at 1,000 mA g-1 with HCCE. (A-C) Reproduced with permission from[86]. Copyright 2021, The Author(s). (D) Digital image of the cellulose-CMC electrolyte. (E) Ionic conductivity of a wet glass fiber separator and various cellulose-CMC membranes (0-5 wt.% CMC) under different drying conditions: wet, squeezed dry using Kimwipes at 0.5 MPa pressure, and vacuum dried for 30 s. (F) Rate performance of the Zn||MnO2 cell using cellulose-5 wt.% CMC electrolyte from 1 to 16 C. (D-F) Reproduced with permission from[87]. Copyright 2023, The Authors. Advanced Functional Materials published by Wiley-VCH GmbH.
For further promoting practical applications, Chen et al.[88] designed a hydrogel electrolyte composed of poly(vinyl alcohol) (PVA), polyacrylamide (PAM), polyacrylonitrile (PAN), and ZnSO4 (Zn) , which exhibited an ultrahigh modulus capable of overcoming the growth stress of Zn dendrites through a mechanical suppression effect. The calculation of the migration energy barrier of Zn2+ ions demonstrated their facile migration [Figure 10A]. In addition, quasi-solid-state Zn||V2O5 cells were prepared to evaluate the superiority and applicability of rigid electrolytes in AZIBs [Figure 10B], in which excellent electrochemical performance was obtained [Figure 10C]. This study offers a promising approach for designing mechanically reliable hydrogel electrolytes. Based on natural biomaterials, Lu et al.[89] proposed a biodegradable and recyclable hydrogel electrolyte composed of chitosan (CS), polyaspartic acid (PASP), and ZnSO4, referred to as CPZ-H. As illustrated in Figure 10D, the zincophilic functional groups (hydroxyl and N-acetylamino groups) in CS reduced the desolvation energy barrier. Therefore, homogeneous Zn deposition under a vertical electric field was facilitated. In the Zn/CPZ-H//MnO2 full cell, the promotion of the two-electron reduction process (Mn4+ to Mn2+) enabled the MnO2 cathode to enhance its Zn2+ storage capability during the electrochemical process, thereby contributing additional energy storage in the Zn/MnO2 system [Figure 10E]. Furthermore, the sustainable biomass characteristics of the CPZ-H electrolyte were evaluated by examining its degradability and recyclability in the Zn/MnO2 cell. First, the aged electrolyte was dried and ground into powder. Subsequently, it was mixed with a small amount of water to form a solution, which was then transferred into molds. The molds were heated to obtain the regenerated CPZ-H (rCPZ-H). The biomass-based hydrogel exhibited strong reprocessability and could therefore be recovered via a simple process, demonstrating its feasibility for large-scale recycling applications [Figure 10F]. Additionally, the rCPZ-H electrolyte showed promising practical application potential, as demonstrated by its ability to power a small elevator [Figure 10G]. This work provides a platform for the development of multifunctional and sustainable biomass-based electrolytes.
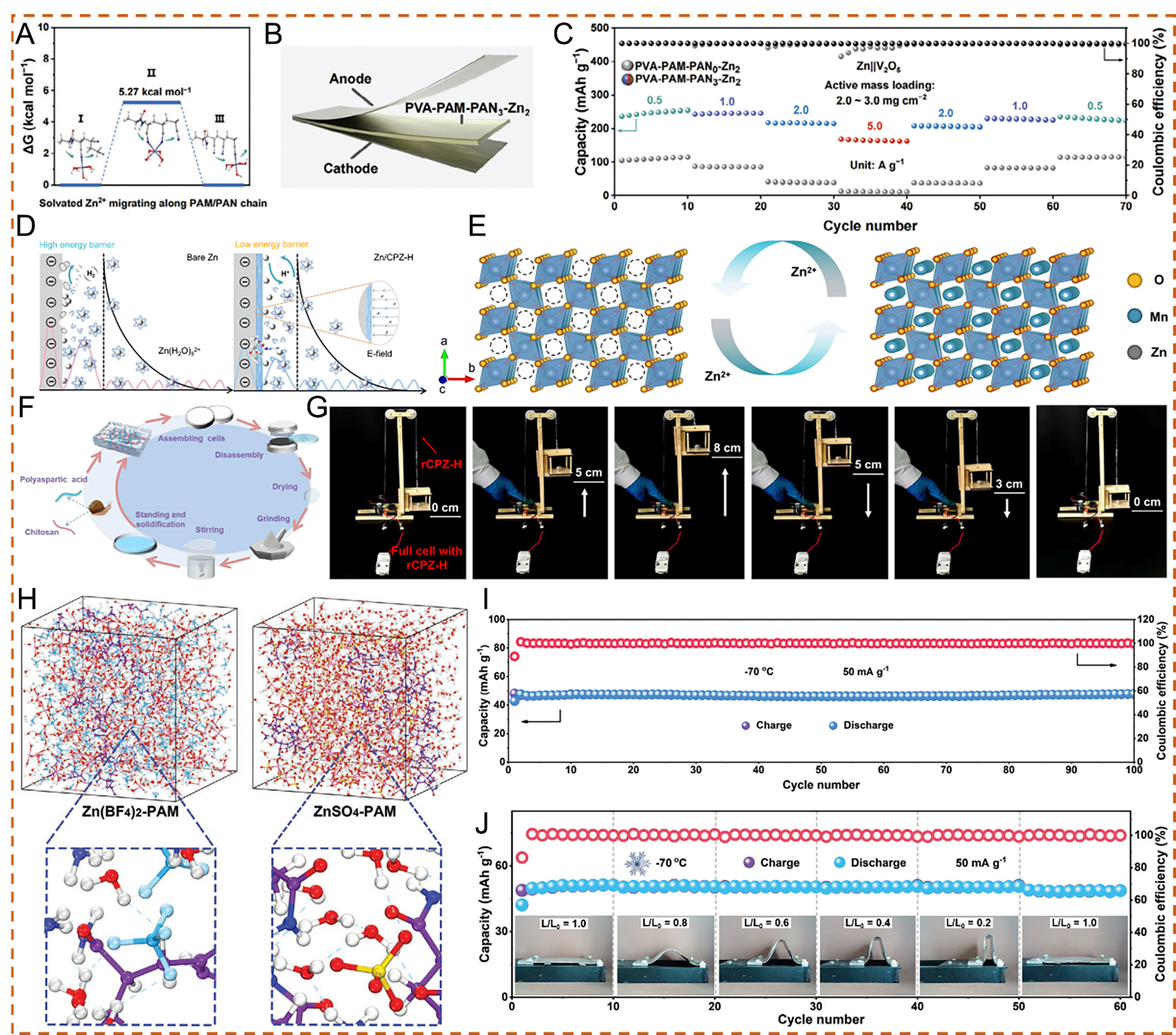
Figure 10. (A) Relative migration energy of solvated Zn2+ along the polyacrylamide/polyacrylonitrile (PAM/PAN) chain. (B) Schematic diagram of the Zn||PVA-PAM-PAN3-Zn2||V2O5 battery. (C) Rate performance of Zn||PVA-PAM-PAN0-Zn2||V2O5 and Zn||PVA-PAMPAN3-Zn2||V2O5 batteries. (A-C) Reproduced with permission from[88]. Copyright 2024, Wiley-VCH GmbH. (D) Schematic illustration of Zn2+ deposition processes on bare Zn and Zn/CPZ-H anodes. (E) Schematic illustration of reversible Zn2+ intercalation/de-intercalation in a MnO2 cathode during the electrochemical process. (F) Schematic diagram of the CPZ-H electrolyte recycling process and optical photographs of different stages during the recycling process. (G) Photographs of an elevator device powered by a Zn/rCPZ-H//MnO2 full cell. (D-G) Reproduced with permission from[89]. Copyright 2023, The Author(s). (H) MD simulation snapshots of saturated Zn(BF4)2-PAM hydrogel electrolyte and saturated ZnSO4-PAM hydrogel electrolyte. (I) Long-term cycling performance at 1 A g-1. (J) Cycling performance of anti-freezing flexible ZIBs at a current density of 50 mA g-1 under different bending states at -70 °C. (H-J) Reproduced with permission from[90]. Copyright 2023, Wiley-VCH GmbH. PVA: Poly(vinyl alcohol).
To improve low-temperature performance, Shi et al.[90] developed an anti-freezing hydrogel electrolyte composed of zinc tetrafluoroborate (Zn(BF4)2) and polyacrylamide (PAM). MD simulations indicated that the introduction of Zn(BF4)2 significantly disrupts hydrogen bonding among water molecules and promotes the formation of hydrogen bonds between BF4- and water molecules [Figure 10H]. The amount of free water molecules in the Zn(BF4)2-PAM hydrogel was markedly reduced compared with that in the ZnSO4-PAM hydrogel, resulting in improved resistance to subzero temperatures. Based on this, anti-freezing flexible AZIBs incorporating this hydrogel presented favorable long-term cycling stability at -70 °C, with no obvious capacity loss after repeated cycles under different bending states at -70 °C [Figure 10I and J]. This study opens new opportunities for constructing ultralow-temperature aqueous battery systems.
CONCLUSIONS AND PERSPECTIVES
As a representative energy storage system beyond conventional LIBs, AZIBs present enormous potential for high-safety, low-cost energy storage and conversion. Electrolytes, as an indispensable part of AZIBs, are highly correlated with their electrochemical performance. This review discusses electrolyte engineering for AZIBs. However, the three strategies still present several aspects that warrant attention. First, regulating the solvation process can effectively suppress Zn dendrite formation, HER, and corrosion, thereby significantly improving the overall electrochemical performance of AZIBs. However, this approach may also result in issues such as reduced ionic conductivity, increased interface impedance, high cost, and limited adaptability to practical working conditions. Therefore, it is necessary to comprehensively consider ionic transport kinetics, interfacial stability, cost, and practicality when developing mild, efficient, and low-cost strategies. Second, strategies for optimizing the structure of the SEI layer may encounter similar challenges. A comprehensive trade-off should be made among ionic conductivity, electrochemical stability, mechanical properties, cost-effectiveness, and practical applicability of the newly constructed SEI. This balance could promote the development of SEI engineering strategies that are mild, efficient, and compatible with practical requirements. Lastly, strategies for modulating ionic transport need to achieve a reasonable balance among ionic transport efficiency, interfacial stability, electrochemical kinetics, and practical application potential.
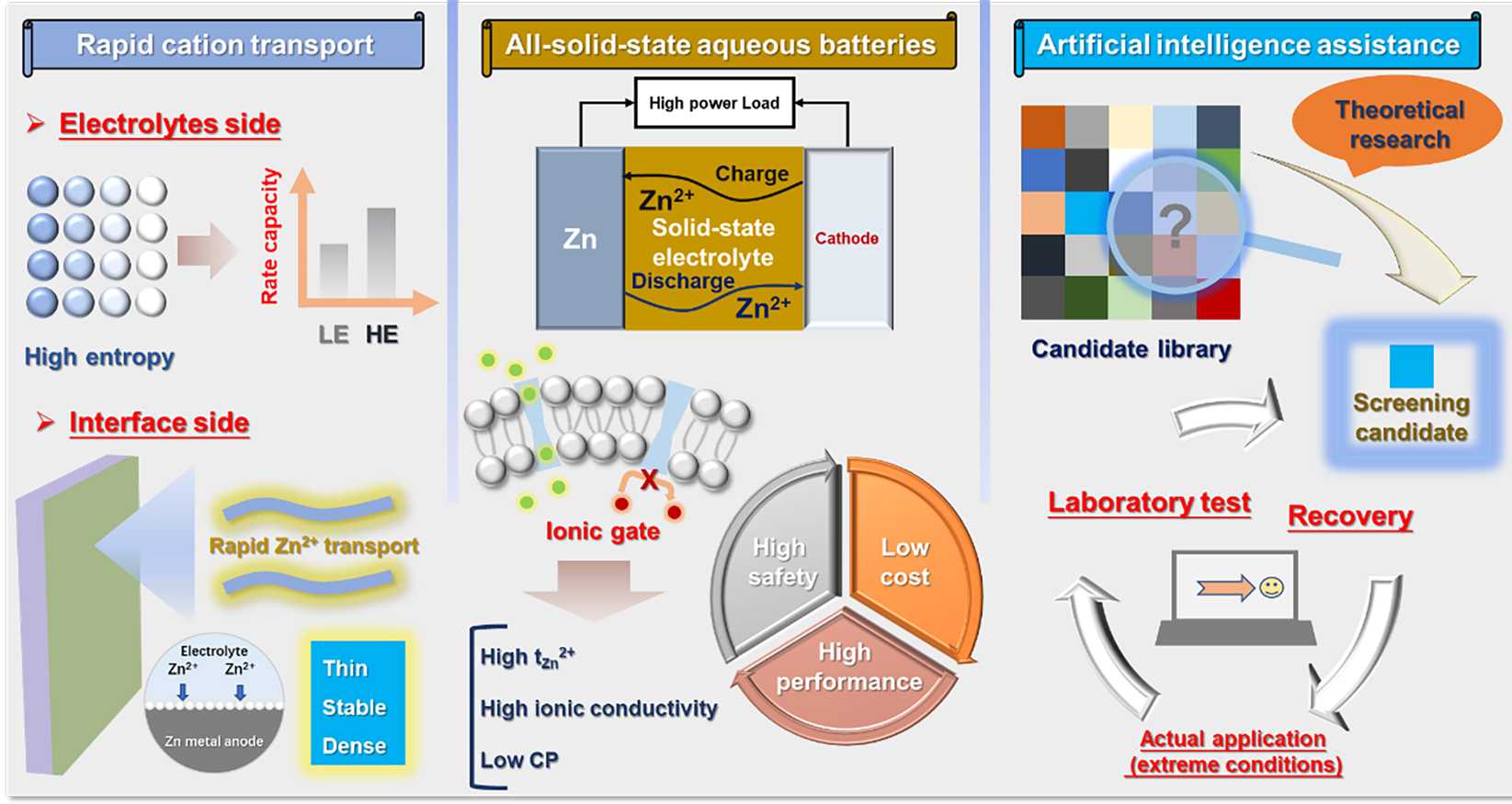
Some remaining challenges associated with electrolytes for AZIBs can be summarized as follows [Figure 11].
First, improving ionic conductivity to enable fast-charging batteries has always been an important research direction. High-entropy systems represent a promising approach. High-entropy materials possess several advantages in battery applications, such as suppressing short-range order and reducing volume change[113-116]. By introducing various cations or anions to construct high-entropy systems, it is possible to disrupt the hydrogen-bond network of water molecules and form diverse and smaller solvation structures, thereby enabling rapid ionic transport in the electrolyte. Additionally, the interface between the electrolyte and the Zn anode is vital for ionic transport. By designing and optimizing electrolyte components, a thin, stable, and dense solvation layer could be formed on the surface of the Zn anode, which helps reduce the energy barrier for the desolvation of Zn2+ ions and accelerates interfacial ionic transport, thereby significantly improving interfacial stability and electrochemical performance. High-concentration salt systems and eutectic electrolytes are effective strategies for regulating the solvation process. Considering cost, toxicity, and the challenges of large-scale production for practical applications, the addition of small amounts of multifunctional additives has great potential.
In terms of developing all-solid-state aqueous batteries for practical applications, the ionic transport capability of solid-state electrolytes (SSEs) is a crucial factor[117,118]. Designing customized nanochannels could significantly enhance cation transport. In biological systems, ion channels play a vital role in regulating ionic transport (e.g., ion gating) to maintain normal physiological functions. Inspired by this, the introduction of charged nanochannels with an electrical double layer has been demonstrated as an effective strategy for regulating ionic transport and shows great potential for energy conversion applications[119-121]. When applied to SSEs for AZIBs, biomimetic nanochannel structures may achieve high tZn2+ and ionic conductivity, as well as low concentration polarization (CP). With continuous advancements in material diversity and cost reduction, such SSEs may facilitate the development of all-solid-state aqueous batteries. On this basis, improving the compatibility of these SSEs with various high-performance cathode materials is necessary, such as V2C/VO2-N-doped carbon[122], which will further promote the practical application of full batteries.
To meet the aforementioned requirements for material screening, artificial intelligence (AI) may serve as an effective tool[123,124]. Through machine learning and deep learning, a comprehensive candidate material database can be constructed. Based on this, theoretical methods such as DFT and MD simulations could be employed to identify suitable electrolyte materials from the candidate library. Additionally, AI could be utilized to predict performance under specific extreme operating conditions. From the candidate database, optimal electrolyte materials could be further selected. This approach may not only reduce the cost of testing under extreme conditions but also accelerate the practical implementation of batteries. Furthermore, electrolyte materials used under extreme conditions could be intelligently recovered through the establishment of a mature and comprehensive recycling model. This would enable the recycling of high-performance electrolytes, especially those with high cost and complex processing requirements, thereby further promoting the practical application of aqueous batteries.
DECLARATIONS
Authors’ contributions
Writing-original draft, writing-review & editing: Hao, Z.
Investigation: Yao, W.; Li, W.
Visualization: Qi, S.; Tang, Y.; Liu, G.
Supervision: Dai, Y.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the Natural Science Foundation of Jiangsu Province (BK20230701), the National Natural Science Foundation of China (52401282), and the Jiangsu Provincial Young Scientific and Technological Talents Support Project (JSTJ-2025-681).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Chu, S.; Majumdar, A. Opportunities and challenges for a sustainable energy future. Nature 2012, 488, 294-303.
2. Yao, Z.; Lum, Y.; Johnston, A.; et al. Machine learning for a sustainable energy future. Nat. Rev. Mater. 2023, 8, 202-15.
3. Fuso, Nerini. F.; Tomei, J.; To, L. S.; et al. Mapping synergies and trade-offs between energy and the sustainable development goals. Nat. Energy. 2017, 3, 10-5.
4. Karijadi, I.; Chou, S.; Dewabharata, A. Wind power forecasting based on hybrid CEEMDAN-EWT deep learning method. Renew. Energy. 2023, 218, 119357.
5. Naik, N.; Gayathri, R.; Behera, H.; Tsai, C. Wave power extraction by a dual OWC chambers over an undulated bottom. Renew. Energy. 2023, 216, 119026.
6. Zhang, H.; Baeyens, J.; Degrève, J.; Cacères, G. Concentrated solar power plants: review and design methodology. Renew. Sustain. Energy. Rev. 2013, 22, 466-81.
7. Sun, J.; Wang, Y.; He, Y.; et al. The energy security risk assessment of inefficient wind and solar resources under carbon neutrality in China. Appl. Energy. 2024, 360, 122889.
8. Ayodele, T.; Ogunjuyigbe, A. Mitigation of wind power intermittency: storage technology approach. Renew. Sustain. Energy. Rev. 2015, 44, 447-56.
9. Rahman, F.; Rehman, S.; Abdul-Majeed, M. A. Overview of energy storage systems for storing electricity from renewable energy sources in Saudi Arabia. Renew. Sustain. Energy. Rev. 2012, 16, 274-83.
10. Kim, T.; Song, W.; Son, D.; Ono, L. K.; Qi, Y. Lithium-ion batteries: outlook on present, future, and hybridized technologies. J. Mater. Chem. A. 2019, 7, 2942-64.
11. Scrosati, B.; Hassoun, J.; Sun, Y. Lithium-ion batteries. A look into the future. Energy. Environ. Sci. 2011, 4, 3287.
12. Fu, K.; Li, X.; Sun, K.; et al. Thick electrode design for lithium-ion batteries from an ion-electron coupled transport perspective: from independent regulation to cooperative design. Energy. Z. 2026, 2, 200002.
14. Hao, Z.; Zhao, Q.; Tang, J.; et al. Functional separators towards the suppression of lithium dendrites for rechargeable high-energy batteries. Mater. Horiz. 2021, 8, 12-32.
15. Ma, L.; Cui, J.; Yao, S.; et al. Dendrite-free lithium metal and sodium metal batteries. Energy. Storage. Mater. 2020, 27, 522-54.
16. Zhao, Q.; Huang, W.; Luo, Z.; et al. High-capacity aqueous zinc batteries using sustainable quinone electrodes. Sci. Adv. 2018, 4, eaao1761.
17. Shang, Y.; Chen, S.; Chen, N.; et al. A universal strategy for high-voltage aqueous batteries via lone pair electrons as the hydrogen bond-breaker. Energy. Environ. Sci. 2022, 15, 2653-63.
18. Nian, Q.; Sun, T.; Liu, S.; Du, H.; Ren, X.; Tao, Z. Issues and opportunities on low-temperature aqueous batteries. Chem. Eng. J. 2021, 423, 130253.
19. Li, T. C.; Fang, D.; Zhang, J.; et al. Recent progress in aqueous zinc-ion batteries: a deep insight into zinc metal anodes. J. Mater. Chem. A. 2021, 9, 6013-28.
20. Hao, Z.; Dai, Y.; Xu, X.; et al. Strategies for addressing the challenges of aqueous zinc batteries enabled by functional separators. J. Mater. Chem. A. 2023, 11, 11031-47.
21. Luo, C.; Lei, H.; Xiao, Y.; et al. Recent development in addressing challenges and implementing strategies for manganese dioxide cathodes in aqueous zinc ion batteries. Energy. Mater. 2024, 4, 400036.
22. Xue, R.; Zou, Y.; Wang, Z.; et al. Enhancing temperature adaptability of aqueous zinc batteries via antifreezing electrolyte and site-selective ZnSe-Ag interface layer design. ACS. Nano. 2023, 17, 17359-71.
23. Cui, F.; Wang, D.; Hu, F.; et al. Deficiency and surface engineering boosting electronic and ionic kinetics in NH4V4O10 for high-performance aqueous zinc-ion battery. Energy. Storage. Mater. 2022, 44, 197-205.
24. Hu, Y.; Wang, P.; Li, M.; Liu, Z.; Liang, S.; Fang, G. Challenges and industrial considerations towards stable and high-energy-density aqueous zinc-ion batteries. Energy. Environ. Sci. 2024, 17, 8078-93.
25. Shin, J.; Lee, J.; Park, Y.; Choi, J. W. Aqueous zinc ion batteries: focus on zinc metal anodes. Chem. Sci. 2020, 11, 2028-44.
26. Wang, K.; Baule, N.; Jin, H.; et al. Multifunctional zinc silicate coating layer for high-performance aqueous zinc-ion batteries. Energy. Mater. 2025, 5, 500012.
27. Hao, Z.; Tang, J.; Cao, S.; et al. Functional ion-sieving separators based on covalent organic frameworks toward stable aqueous energy storage and conversion. ACS. Mater. Lett. 2025, 7, 3634-41.
28. Shang, Y.; Kundu, D. A path forward for the translational development of aqueous zinc-ion batteries. Joule 2023, 7, 244-50.
29. Cao, J.; Yuan, Z.; Li, C.; et al. Achieving high current density, high areal capacity, and high DOD AZIBs by screening amino acids. J. Mater. Chem. A. 2024, 12, 29869-85.
30. Wei, J.; Zhang, P.; Sun, J.; et al. Advanced electrolytes for high-performance aqueous zinc-ion batteries. Chem. Soc. Rev. 2024, 53, 10335-69.
31. Higashi, S.; Lee, S. W.; Lee, J. S.; Takechi, K.; Cui, Y. Avoiding short circuits from zinc metal dendrites in anode by backside-plating configuration. Nat. Commun. 2016, 7, 11801.
32. Han, D.; Wu, S.; Zhang, S.; et al. A corrosion-resistant and dendrite-free zinc metal anode in aqueous systems. Small 2020, 16, e2001736.
33. Kao, C. C.; Ye, C.; Hao, J.; Chen, Y.; Zhang, S. J.; Qiao, S. Z. Achieving high energy density in aqueous zinc-ion batteries. Adv. Energy. Mater. 2025, 15, 2501201.
34. Rana, A.; Paul, S.; Bhadouria, A.; et al. Interfacial pH gradients suppress HER at high currents in zinc metal batteries. Joule 2025, 9, 102167.
35. Roy, K.; Rana, A.; Heil, J. N.; Tackett, B. M.; Dick, J. E. For zinc metal batteries, How many electrons go to hydrogen evolution? An electrochemical mass spectrometry study. Angew. Chem. Int. Ed. 2024, 63, e202319010.
36. Tang, B.; Shan, L.; Liang, S.; Zhou, J. Issues and opportunities facing aqueous zinc-ion batteries. Energy. Environ. Sci. 2019, 12, 3288-304.
37. Yang, H.; Yang, Y.; Yang, W.; Wu, G.; Zhu, R. Correlating hydrogen evolution and zinc deposition/dissolution kinetics to the cyclability of metallic zinc electrodes. Energy. Environ. Sci. 2024, 17, 1975-83.
38. Rana, A.; Roy, K.; Heil, J. N.; et al. Realizing the kinetic origin of hydrogen evolution for aqueous zinc metal batteries. Adv. Energy. Mater. 2024, 14, 2402521.
39. Dong, N.; Zhang, F.; Pan, H. Towards the practical application of Zn metal anodes for mild aqueous rechargeable Zn batteries. Chem. Sci. 2022, 13, 8243-52.
40. Chen, J.; Zhao, W.; Jiang, J.; et al. Challenges and perspectives of hydrogen evolution-free aqueous Zn-ion batteries. Energy. Storage. Mater. 2023, 59, 102767.
41. Li, X.; Liu, P.; Han, C.; et al. Corrosion of metallic anodes in aqueous batteries. Energy. Environ. Sci. 2025, 18, 2050-94.
42. Yang, J.; Yin, B.; Sun, Y.; et al. Zinc anode for mild aqueous zinc-ion batteries: challenges, strategies, and perspectives. Nanomicro. Lett. 2022, 14, 42.
43. Blanc, L. E.; Kundu, D.; Nazar, L. F. Scientific challenges for the implementation of Zn-ion batteries. Joule 2020, 4, 771-99.
44. Kao, C. C.; Ye, C.; Hao, J.; Shan, J.; Li, H.; Qiao, S. Z. Suppressing hydrogen evolution via anticatalytic interfaces toward highly efficient aqueous Zn-ion batteries. ACS. Nano. 2023, 17, 3948-57.
45. Wang, Z.; Hu, S.; Wang, D.; et al. A HER‐inhibiting layer based on M‐H bond regulation for achieving stable zinc anodes in aqueous zinc-ion batteries. Adv. Funct. Mater. 2025, 35, 2502186.
46. Xing, Z.; Sun, Y.; Xie, X.; et al. Zincophilic electrode interphase with appended proton reservoir ability stabilizes Zn metal anodes. Angew. Chem. Int. Ed. 2023, 62, e202215324.
47. Yao, R.; Zhao, Y.; Wang, L.; et al. A corrosion-free zinc metal battery with an ultra-thin zinc anode and high depth of discharge. Energy. Environ. Sci. 2024, 17, 3112-22.
48. Huang, H.; Xie, D.; Zhao, J.; et al. Boosting reversibility and stability of Zn anodes via manipulation of electrolyte structure and interface with addition of trace organic molecules. Adv. Energy. Mater. 2022, 12, 2202419.
49. Lu, H.; Zheng, S.; Wei, L.; Zhang, X.; Guo, X. Manipulating Zn2+ solvation environment in poly(propylene glycol)‐based aqueous Li+/Zn2+ electrolytes for high‐voltage hybrid ion batteries. Carbon. Energy. 2023, 5, e365.
50. Zhang, Y.; Fu, X.; Ding, Y.; Liu, Y.; Zhao, Y.; Jiao, S. Electrolyte solvation chemistry for stabilizing the Zn anode via functionalized organic agents. Small 2024, 20, e2311407.
51. Lin, L.; Shao, Z.; Liu, S.; et al. High-entropy aqueous electrolyte induced formation of water-poor Zn2+ solvation structures and gradient solid-electrolyte interphase for long-life Zn-metal anodes. Angew. Chem. Int. Ed. 2025, 64, e202425008.
52. Wang, Y.; Kong, Y.; Qi, L.; Yang, W.; Li, G.; Liu, T. Regulating additive electroactivity for self-assembled multifunctional SEI in aqueous zinc batteries. Energy. Storage. Mater. 2025, 81, 104544.
53. Zheng, Q.; Liu, L.; Hu, Z.; et al. Altering the Zn2+ Migration mechanism enables the composite hydrogel electrolytes with high Zn2+ conduction and superior anti-dehydration. Adv. Funct. Mater. 2025, 35, 2504782.
54. Zhang, Q.; Yang, Z.; Ji, H.; et al. Issues and rational design of aqueous electrolyte for Zn‐ion batteries. SusMat 2021, 1, 432-47.
55. Zhu, Y.; Hao, J.; Huang, Y.; Jiao, Y. A new insight of anti-solvent electrolytes for aqueous zinc-ion batteries by molecular modeling. Small. Struct. 2023, 4, 2200270.
56. Liu, Z.; Sun, J.; Li, X.; et al. Constructing eutectic solvation sheath by weak solvation effect for stabilizing Zn-ion batteries with low-temperature adaptability. Energy. Storage. Mater. 2025, 83, 104727.
57. Yang, M.; Zhu, J.; Bi, S.; et al. The construction of anion-induced solvation structures in low-concentration electrolyte for stable zinc anodes. Angew. Chem. Int. Ed. 2024, 63, e202400337.
58. Dong, Y.; Jia, M.; Wang, Y.; et al. Long-life zinc/vanadium pentoxide battery enabled by a concentrated aqueous ZnSO4 electrolyte with proton and zinc ion co-intercalation. ACS. Appl. Energy. Mater. 2020, 3, 11183-92.
59. Dai, Y.; Li, J.; Zhang, C.; et al. Fluorinated interphase enables reversible Zn2+ storage in aqueous ZnSO4 electrolytes. ACS. Energy. Lett. 2023, 8, 4762-7.
60. He, Y.; Chen, Z.; Gao, Q.; Feng, J.; Hao, Z. Dynamic interface modulation of aqueous zinc-ion batteries by rational design of organic additives. Small 2025, 21, e06244.
61. Zhang, Y.; Wu, D.; Huang, F.; et al. “Water‐in‐salt” nonalkaline gel polymer electrolytes enable flexible zinc‐air batteries with ultra-long operating time. Adv. Funct. Mater. 2022, 32, 2203204.
62. Emanuele, E.; Batignani, G.; Cerullo, G.; et al. Solving ZIB challenges: the dynamic role of water in deep eutectic solvents electrolyte. J. Mater. Chem. A. 2025, 13, 9778-90.
63. Xie, J.; Lin, D.; Lei, H.; et al. Electrolyte and interphase engineering of aqueous batteries beyond "water-in-salt" strategy. Adv. Mater. 2024, 36, e2306508.
64. Mo, F.; Liang, G.; Meng, Q.; et al. A flexible rechargeable aqueous zinc manganese-dioxide battery working at -20 °C. Energy. Environ. Sci. 2019, 12, 706-15.
65. Yang, P.; Feng, C.; Liu, Y.; et al. Thermal self‐protection of zinc‐ion batteries enabled by smart hygroscopic hydrogel electrolytes. Adv. Energy. Mater. 2020, 10, 2002898.
66. Jiang, C.; Zhang, Y.; Jia, M.; et al. Anti-freezing hydrogel electrolytes: from molecular engineering to applications in aqueous zinc-ion batteries. Mater. Today. 2025, 90, 706-23.
67. Dong, J.; Cheng, X.; Yang, H.; et al. Suspension electrolytes with catalytically self-expediating desolvation kinetics for low-temperature zinc metal batteries. Adv. Mater. 2025, 37, e2501079.
68. Xie, D.; Sang, Y.; Wang, D. H.; et al. ZnF2 -riched inorganic/organic hybrid SEI: in situ-chemical construction and performance-improving mechanism for aqueous zinc-ion batteries. Angew. Chem. Int. Ed. 2023, 62, e202216934.
69. Sand, H. J. III. On the concentration at the electrodes in a solution, with special reference to the liberation of hydrogen by electrolysis of a mixture of copper sulphate and sulphuric acid. London. Edinburgh. Dublin. Philo. Mag. J. Sci. 2010, 1, 45-79.
70. Su, Y.; Liu, B.; Zhang, Q.; et al. Printing‐Scalable Ti3C2Tx MXene‐decorated janus separator with expedited Zn2+ flux toward stabilized Zn anodes. Adv. Funct. Mater. 2022, 32, 2204306.
71. Bai, P.; Li, J.; Brushett, F. R.; Bazant, M. Z. Transition of lithium growth mechanisms in liquid electrolytes. Energy. Environ. Sci. 2016, 9, 3221-9.
72. Jiao, S.; Fu, J.; Wu, M.; Hua, T.; Hu, H. Ion sieve: tailoring Zn2+ desolvation kinetics and flux toward dendrite-free metallic zinc anodes. ACS. Nano. 2022, 16, 1013-24.
73. Lin, C.; Zeng, L.; Liu, M.; et al. Dynamic regulation for the well-distribution of electrons and Zn2+ ions achieving uniform Zn redox in Ah-scale pouch cells. Adv. Mater. 2025, 37, e11484.
74. Liu, Z.; Wang, R.; Ma, Q.; et al. A dual-functional organic electrolyte additive with regulating suitable overpotential for building highly reversible aqueous zinc ion batteries. Adv. Funct. Mater. 2023, 34, 2214538.
75. Geng, L.; Meng, J.; Wang, X.; et al. Eutectic electrolyte with unique solvation structure for high-performance zinc-ion batteries. Angew. Chem. Int. Ed. 2022, 61, e202206717.
76. Zhu, Y.; Yin, J.; Zheng, X.; et al. Concentrated dual-cation electrolyte strategy for aqueous zinc-ion batteries. Energy. Environ. Sci. 2021, 14, 4463-73.
77. Deng, S.; Sun, Y.; Yang, Z.; et al. Zwitterion‐separated ion pair dominated additive‐electrolyte structure for ultra‐stable aqueous zinc ion batteries. Adv. Funct. Mater. 2024, 34, 2408546.
78. Shi, X.; Xie, J.; Wang, J.; Xie, S.; Yang, Z.; Lu, X. A weakly solvating electrolyte towards practical rechargeable aqueous zinc-ion batteries. Nat. Commun. 2024, 15, 302.
79. Ding, Y.; Yin, L.; Du, T.; et al. A trifunctional electrolyte enables aqueous zinc ion batteries with long cycling performance. Adv. Funct. Mater. 2024, 34, 2314388.
80. Li, G.; Zhao, Z.; Zhang, S.; et al. A biocompatible electrolyte enables highly reversible Zn anode for zinc ion battery. Nat. Commun. 2023, 14, 6526.
81. Liu, S.; Mao, J.; Pang, W. K.; et al. Tuning the electrolyte solvation structure to suppress cathode dissolution, water reactivity, and Zn dendrite growth in zinc‐ion batteries. Adv. Funct. Mater. 2021, 31, 2104281.
82. Huang, S.; Hou, L.; Li, T.; Jiao, Y.; Wu, P. Antifreezing hydrogel electrolyte with ternary hydrogen bonding for high-performance zinc-ion batteries. Adv. Mater. 2022, 34, e2110140.
83. Wang, N.; Chen, X.; Wan, H.; et al. Zincophobic electrolyte achieves highly reversible zinc-ion batteries. Adv. Funct. Mater. 2023, 33, 2300795.
84. Dong, H.; Hu, X.; Liu, R.; et al. Bio‐inspired polyanionic electrolytes for highly stable zinc‐ion batteries. Angew. Chem. Int. Ed. 2023, 135, e202311268.
85. Qiu, H.; Du, X.; Zhao, J.; et al. Zinc anode-compatible in-situ solid electrolyte interphase via cation solvation modulation. Nat. Commun. 2019, 10, 5374.
86. Gao, J.; Xie, X.; Liang, S.; Lu, B.; Zhou, J. Inorganic colloidal electrolyte for highly robust zinc-ion batteries. Nanomicro. Lett. 2021, 13, 69.
87. Xu, L.; Meng, T.; Zheng, X.; et al. Nanocellulose‐carboxymethylcellulose electrolyte for stable, high‐rate zinc‐ion batteries. Adv. Funct. Mater. 2023, 33, 2302098.
88. Chen, Z. J.; Shen, T. Y.; Xiao, X.; et al. An ultrahigh-modulus hydrogel electrolyte for dendrite-free zinc ion batteries. Adv. Mater. 2024, 36, e2413268.
89. Lu, H.; Hu, J.; Wei, X.; et al. A recyclable biomass electrolyte towards green zinc-ion batteries. Nat. Commun. 2023, 14, 4435.
90. Shi, Y.; Wang, R.; Bi, S.; Yang, M.; Liu, L.; Niu, Z. An anti‐freezing hydrogel electrolyte for flexible zinc‐ion batteries operating at -70 °C. Adv. Funct. Mater. 2023, 33, 2214546.
91. Tian, Z.; Zou, Y.; Liu, G.; et al. Electrolyte solvation structure design for sodium ion batteries. Adv. Sci. 2022, 9, e2201207.
92. Ma, W.; Wang, S.; Wu, X.; et al. Tailoring desolvation strategies for aqueous zinc-ion batteries. Energy. Environ. Sci. 2024, 17, 4819-46.
93. Liang, Y.; Qiu, M.; Sun, P.; Mai, W. Comprehensive review of electrolyte modification strategies for stabilizing Zn metal anodes. Adv. Funct. Mater. 2023, 33, 2304878.
94. Dai, C.; Hu, L.; Jin, X.; Zhao, Y.; Qu, L. The emerging of aqueous zinc-based dual electrolytic batteries. Small 2021, 17, e2008043.
95. Deng, W.; Li, G.; Wang, X. Zinc‐ion battery chemistries enabled by regulating electrolyte solvation structure. Adv. Funct. Mater. 2024, 34, 2405012.
96. Xu, C.; Li, B.; Du, H.; Kang, F. Energetic zinc ion chemistry: the rechargeable zinc ion battery. Angew. Chem. Int. Ed. 2012, 51, 933-5.
97. Zhang, L.; Miao, L.; Xin, W.; Peng, H.; Yan, Z.; Zhu, Z. Engineering zincophilic sites on Zn surface via plant extract additives for dendrite-free Zn anode. Energy. Storage. Mater. 2022, 44, 408-15.
98. Nian, Q.; Zhang, X.; Feng, Y.; et al. Designing electrolyte structure to suppress hydrogen evolution reaction in aqueous batteries. ACS. Energy. Lett. 2021, 6, 2174-80.
99. Chodankar, N. R.; Patil, S. J.; Lee, S.; et al. High energy superstable hybrid capacitor with a self‐regulated Zn/electrolyte interface and 3D graphene‐like carbon cathode. InfoMat 2022, 4, e12344.
100. Yan, H.; Han, C.; Li, S.; et al. Adjusting zinc ion de-solvation kinetics via rich electron-donating artificial SEI towards high columbic efficiency and stable Zn metal anode. Chem. Eng. J. 2022, 442, 136081.
101. Xie, X.; Liang, S.; Gao, J.; et al. Manipulating the ion-transfer kinetics and interface stability for high-performance zinc metal anodes. Energy. Environ. Sci. 2020, 13, 503-10.
102. Adenusi, H.; Chass, G. A.; Passerini, S.; Tian, K. V.; Chen, G. Lithium batteries and the solid electrolyte interphase (SEI) - progress and outlook. Adv. Energy. Mater. 2023, 13, 2203307.
103. Lin, C.; Yang, X.; Xiong, P.; et al. High-rate, large capacity, and long life dendrite-free Zn metal anode enabled by trifunctional electrolyte additive with a wide temperature range. Adv. Sci. 2022, 9, e2201433.
104. Wang, J.; Luo, J.; Wu, H.; et al. Visualizing and regulating dynamic evolution of interfacial electrolyte configuration during de‐solvation process on lithium-metal anode. Angew. Chem. Int. Ed. 2024, 136, e202400254.
105. Chen, Z.; Feng, J.; Yao, P.; Cai, J.; Hao, Z. Insight into aqueous electrolyte additives: unraveling functional principles, electrochemical performance, and beyond. Green. Chem. 2024, 26, 9939-56.
106. Liu, X.; Wang, X.; Yao, J.; et al. Endogenous organic-inorganic hybrid interface for reversible Zn electrochemistry. Adv. Energy. Mater. 2024, 14, 2400090.
107. Wang, N.; Wan, H.; Duan, J.; et al. A review of zinc-based battery from alkaline to acid. Mater. Today. Adv. 2021, 11, 100149.
108. Jiang, X.; Xiao, K.; Hu, T.; Yuan, K.; Chen, Y. Aqueous eutectic electrolyte-derived organic/inorganic hybrid interphase towards reversible zinc electrochemistry for long-life zinc ion batteries. Sci. China. Mater. 2025, 68, 1946-58.
109. Wang, D.; Tang, Y.; Peng, H.; Ma, G. Functional electrolyte additives for aqueous zinc-ion batteries: progress and perspectives. ChemSusChem 2025, 18, e202501387.
110. Dai, Q.; Li, L.; Tu, T.; Zhang, M.; Song, L. An appropriate Zn2+/Mn2+concentration of the electrolyte enables superior performance of AZIBs. J. Mater. Chem. A. 2022, 10, 23722-30.
111. Zhang, Y.; Liu, J.; Sun, B.; et al. Mitigating concentration polarization via ion-sieving interlayer towards long-life 510 Wh Kg-1 lithium metal pouch cells. Angew. Chem. Int. Ed. 2026, 65, e24476.
112. Bai, S.; Sun, Y.; Yi, J.; He, Y.; Qiao, Y.; Zhou, H. High-power Li-metal anode enabled by metal-organic framework modified electrolyte. Joule 2018, 2, 2117-32.
114. Lun, Z.; Ouyang, B.; Kwon, D. H.; et al. Cation-disordered rocksalt-type high-entropy cathodes for Li-ion batteries. Nat. Mater. 2021, 20, 214-21.
115. Zhao, X.; Tian, Y.; Lun, Z.; et al. Design principles for zero-strain Li-ion cathodes. Joule 2022, 6, 1654-71.
116. Stallard, J. C.; Wheatcroft, L.; Booth, S. G.; et al. Mechanical properties of cathode materials for lithium-ion batteries. Joule 2022, 6, 984-1007.
117. Yu, C.; Ganapathy, S.; Eck, E. R. H. V.; et al. Accessing the bottleneck in all-solid state batteries, lithium-ion transport over the solid-electrolyte-electrode interface. Nat. Commun. 2017, 8, 1086.
118. He, B.; Zhang, F.; Xin, Y.; et al. Halogen chemistry of solid electrolytes in all-solid-state batteries. Nat. Rev. Chem. 2023, 7, 826-42.
119. Huang, D.; Zou, K.; Wu, Y.; et al. TRPM4-inspired polymeric nanochannels with preferential cation transport for high-efficiency salinity-gradient energy conversion. J. Am. Chem. Soc. 2024, 146, 16469-77.
120. Zou, K.; Ling, H.; Wang, Q.; et al. Turing-type nanochannel membranes with extrinsic ion transport pathways for high-efficiency osmotic energy harvesting. Nat. Commun. 2024, 15, 10231.
121. Huang, Y.; Wu, C.; Cao, Y.; et al. Scalable integration of photoresponsive highly aligned nanochannels for self-powered ionic devices. Sci. Adv. 2024, 10, eads5591.
122. Ranjith, K. S.; Mohammadi, A.; Raju, G. S. R.; Huh, Y. S.; Han, Y. V2CTx-MXene/winery waste derived carbon-VO2/V2C-MXene aerogel based high-performance cathode for quasi-solid-state Zn-ion batteries. Carbon 2026, 248, 121152.
123. Tran, H.; Gurnani, R.; Kim, C.; et al. Design of functional and sustainable polymers assisted by artificial intelligence. Nat. Rev. Mater. 2024, 9, 866-86.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.