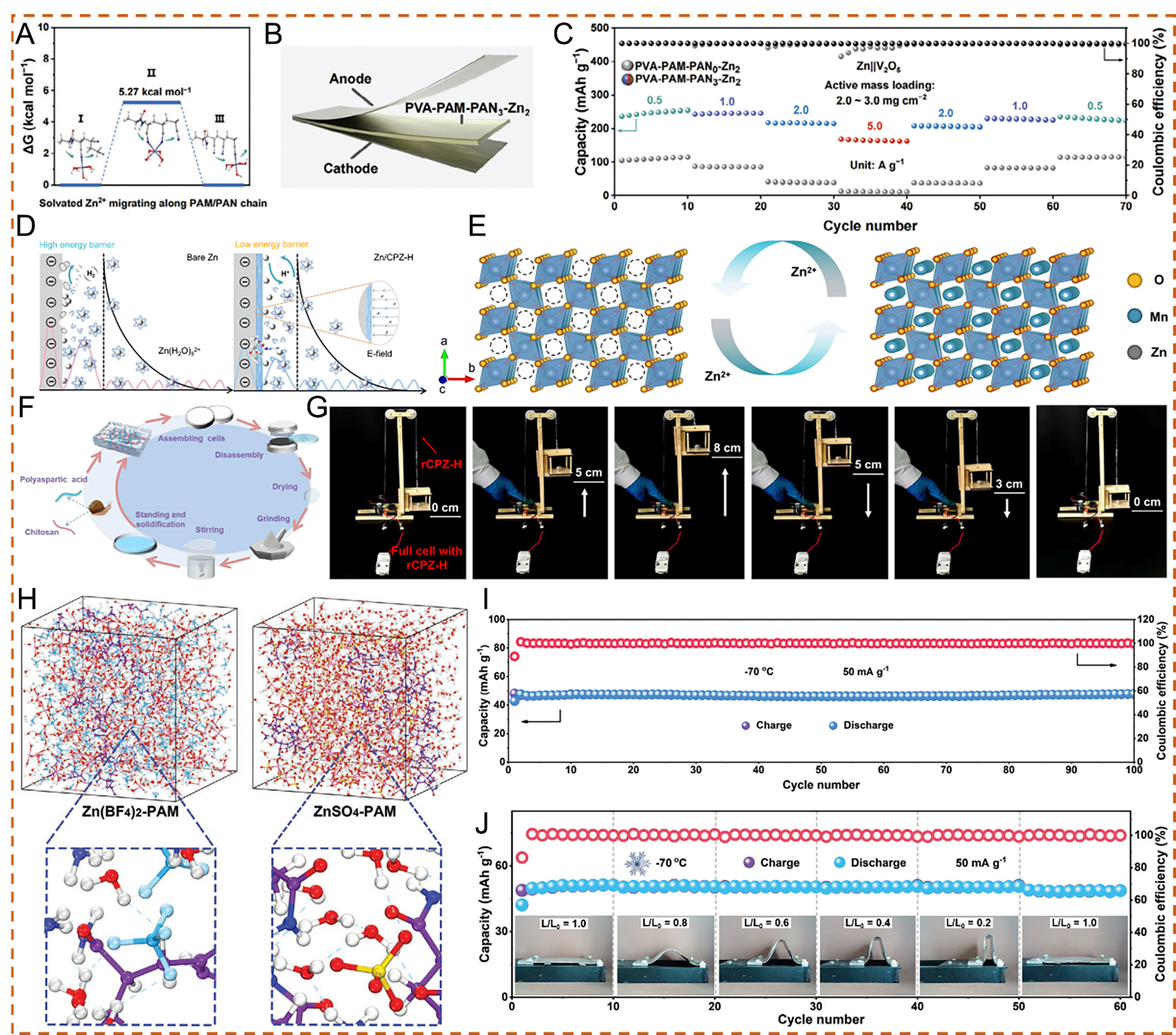
fig10
Figure 10. (A) Relative migration energy of solvated Zn2+ along the polyacrylamide/polyacrylonitrile (PAM/PAN) chain. (B) Schematic diagram of the Zn||PVA-PAM-PAN3-Zn2||V2O5 battery. (C) Rate performance of Zn||PVA-PAM-PAN0-Zn2||V2O5 and Zn||PVA-PAMPAN3-Zn2||V2O5 batteries. (A-C) Reproduced with permission from[88]. Copyright 2024, Wiley-VCH GmbH. (D) Schematic illustration of Zn2+ deposition processes on bare Zn and Zn/CPZ-H anodes. (E) Schematic illustration of reversible Zn2+ intercalation/de-intercalation in a MnO2 cathode during the electrochemical process. (F) Schematic diagram of the CPZ-H electrolyte recycling process and optical photographs of different stages during the recycling process. (G) Photographs of an elevator device powered by a Zn/rCPZ-H//MnO2 full cell. (D-G) Reproduced with permission from[89]. Copyright 2023, The Author(s). (H) MD simulation snapshots of saturated Zn(BF4)2-PAM hydrogel electrolyte and saturated ZnSO4-PAM hydrogel electrolyte. (I) Long-term cycling performance at 1 A g-1. (J) Cycling performance of anti-freezing flexible ZIBs at a current density of 50 mA g-1 under different bending states at -70 °C. (H-J) Reproduced with permission from[90]. Copyright 2023, Wiley-VCH GmbH. PVA: Poly(vinyl alcohol).