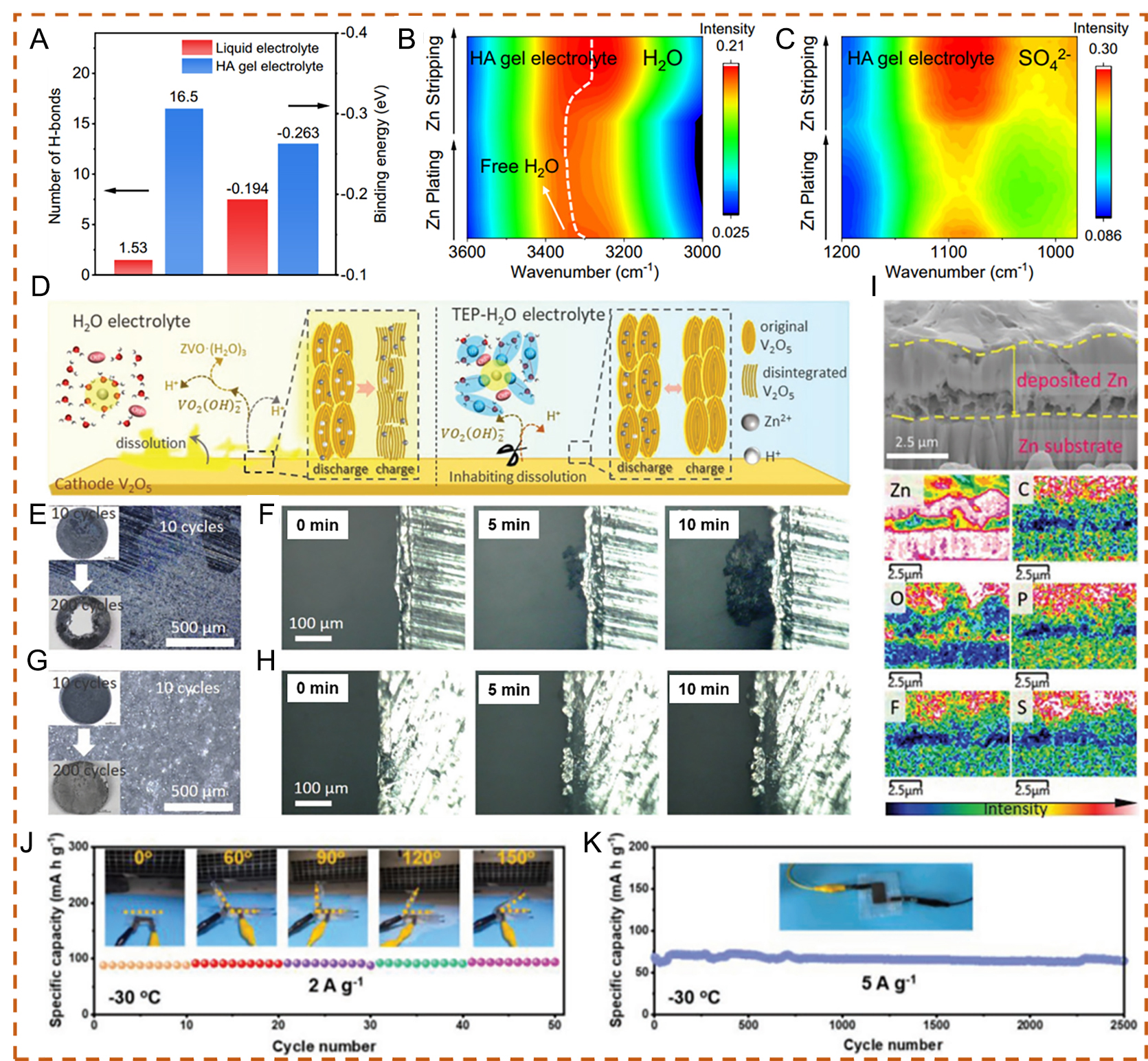
fig5
Figure 5. (A) Calculated number of hydrogen bonds for single H2O and HA molecules, and binding energies of H2O-H2O and H2O-HA. (B) ν (OH) spectral of the HA gel electrolyte. (C) ν (SO42-) spectra of the HA gel electrolyte. (A-C) Reproduced with permission from[80]. Copyright 2023, The Author(s). (D) Schematic illustration of electrolyte effects on the V2O5 electrode during electrochemical reactions. Images of cycled Zn foil under (E) Zn(OTf)2-H2O and (G) Zn(OTf)2-TEP-H2O electrolytes after 10 and 200 cycles. Real-time images of Zn2+ plating on the Zn substrate under (H) Zn(OTf)2-H2O and (F) Zn(OTf)2-TEP-H2O electrolytes within 10 min. (I) FIB cross-sectional image of the cycled Zn anode and corresponding elemental distribution from energy-dispersive X-ray spectroscopy (EDS) after cycling in Zn(OTf)2-TEP-H2O electrolyte. (D-I) Reproduced with permission from[81]. Copyright 2021, Wiley-VCH GmbH. (J) Demonstration of the battery under different bending angles at 2 A g-1 and -30 °C. (K) Cycling performance of the flexible battery with CSAM-C hydrogel electrolyte at 5 A g-1 and -30 °C. (J and K) Reproduced with permission from[82]. Copyright 2022, Wiley-VCH GmbH.