fig8
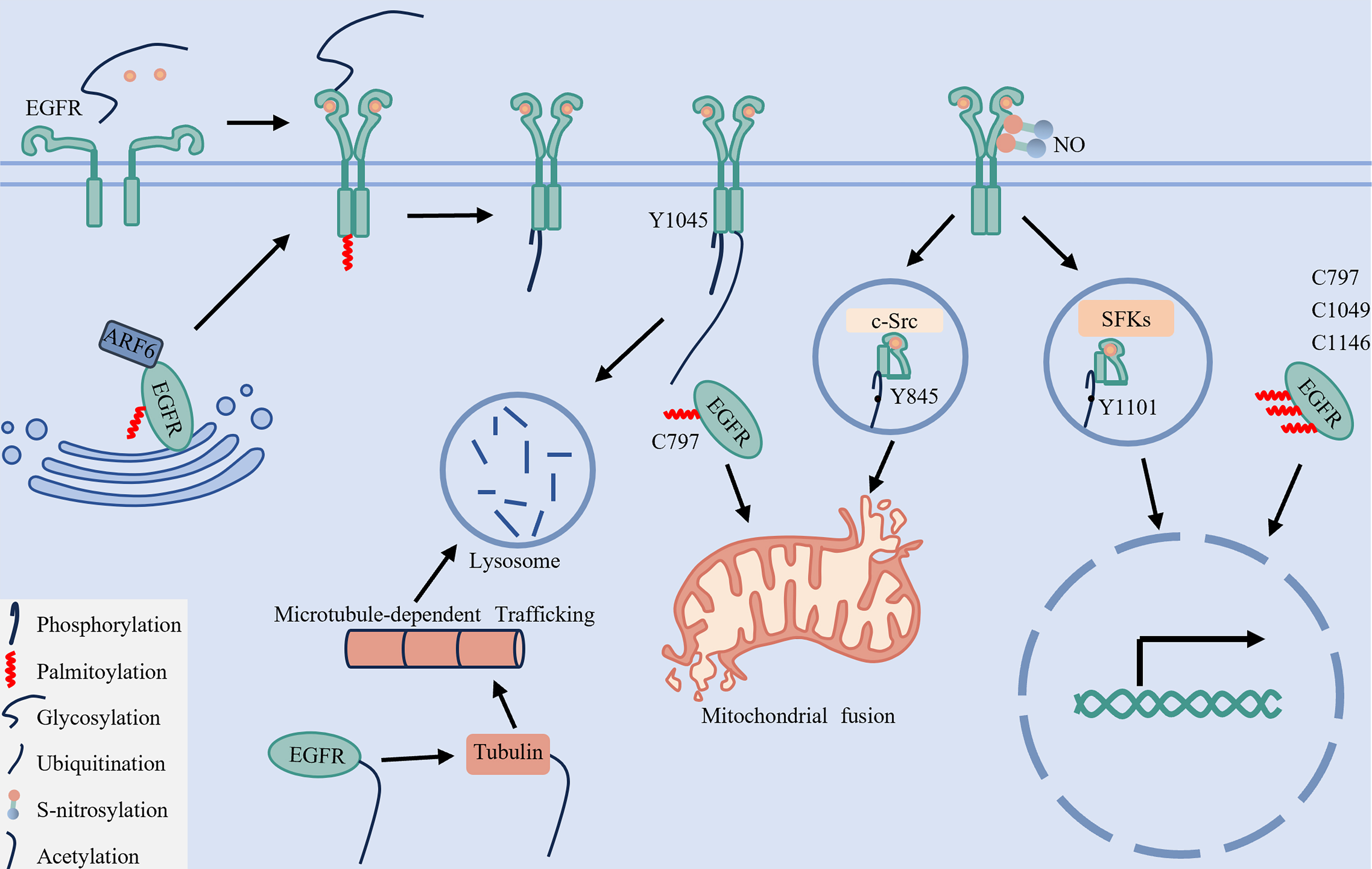
Figure 8. The PTM crosstalk regulatory network and spatial-fate model of EGFR. This diagram summarizes the cooperative regulation of EGFR subcellular localization and signaling through six key PTMs: phosphorylation, palmitoylation, glycosylation, ubiquitination, acetylation, and S-nitrosylation. ARF6-mediated palmitoylation facilitates EGFR trafficking from the Golgi apparatus to the plasma membrane. Glycosylation ensures proper receptor folding and dimerization, while together with palmitoylation, it supports stable membrane localization and enables downstream signaling. Phosphorylation at Y1045 serves as a molecular switch to initiate ubiquitination, whereas acetylation of EGFR promotes α-tubulin acetylation, which ultimately facilitates lysosomal degradation and signal termination. S-nitrosylation enhances Src binding to EGFR, promoting phosphorylation at Y845 and Y1101, thereby driving mitochondrial and nuclear translocation, respectively. Concurrently, palmitoylation at C797 together with phosphorylation at Y845 maintains mitochondrial localization, while phosphorylation at Y1101 combined with palmitoylation at C797, C1049, and C1146 sustains nuclear localization. EGFR: Epidermal growth factor receptor; ARF6: ADP-ribosylation factor 6; Src: SRC proto-oncogene, non-receptor tyrosine kinase; PTMs: post-translational modifications; SFKs: SRC family kinases; NO: nitric oxide.