fig7
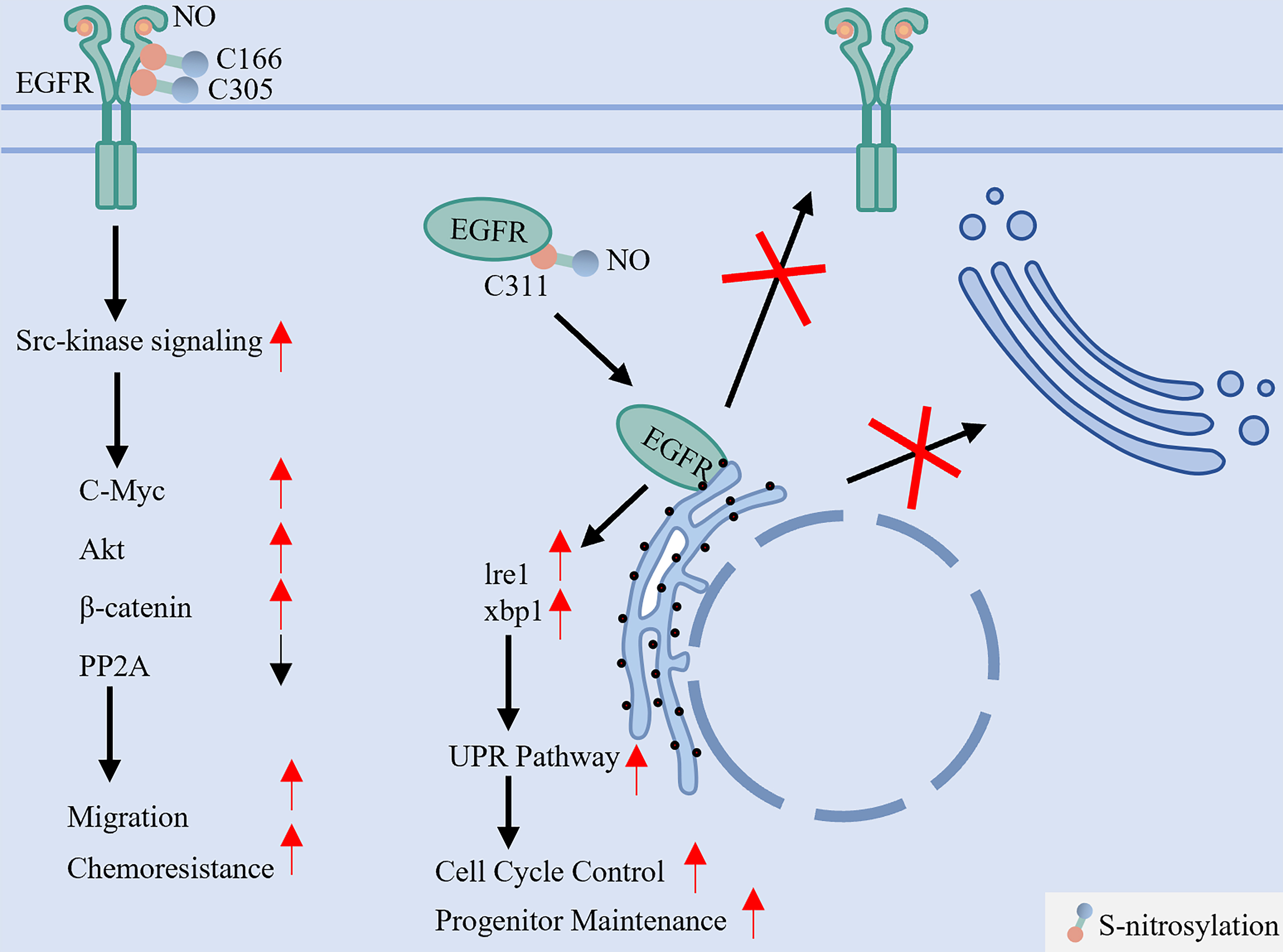
Figure 7. S-nitrosylation dichotomously regulates EGFR activation and maturation. This reversible modification, mediated by NO derived from NOS2, targets specific cysteine residues with opposing outcomes. S-nitrosylation at C166 and C305 enhances EGFR interaction with Src, activating downstream oncogenic pathways (e.g., Akt, MYC) that promote cell migration and chemoresistance. In stark contrast, S-nitrosylation at the extracellular C311 residue disrupts native disulfide bond formation, leading to protein misfolding, ER retention, and impaired membrane localization. EGFR: Epidermal growth factor receptor; NO: nitric oxide; NOS2: nitric oxide synthase 2; Src: SRC proto-oncogene, non-receptor tyrosine kinase; Akt: protein kinase B (PKB); MYC: MYC proto-oncogene; ER: endoplasmic reticulum; PP2A: protein phosphatase 2A; Ire1: inositol-requiring enzyme 1; xbp1: X-box binding protein 1; UPR: unfolded protein response.