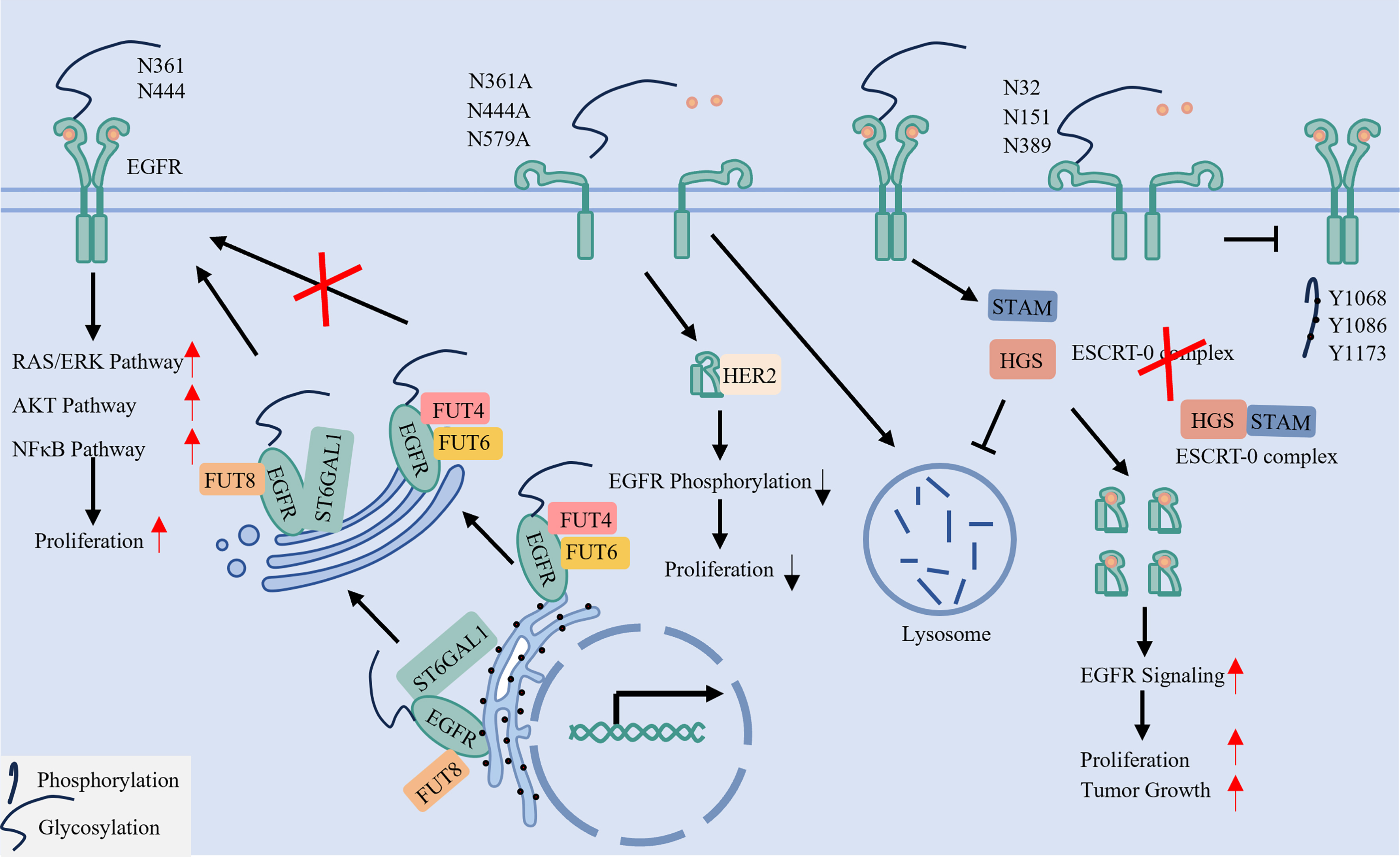
fig5
Figure 5. N-glycosylation ensures proper EGFR conformation, membrane positioning, and stability. N-glycosylation in the endoplasmic reticulum and Golgi apparatus, mediated by glycosyltransferases (e.g., FUT8, ST6GAL1), is essential for EGFR function. Glycans attached at specific sites (e.g., N361, N444) lift the extracellular domain for proper ligand binding and dimerization. In contrast, sialylation at other sites (e.g., N32, N151) can inhibit activation. Furthermore, glycosylation, particularly O-GlcNAcylation of the ESCRT-0 component HGS, can impede receptor degradation, thereby stabilizing EGFR. EGFR: Epidermal Growth Factor Receptor; FUT: Fucosyltransferase; ST6GAL1: ST6 Beta-Galactoside Alpha-2,6-Sialyltransferase 1; O-GlcNAcylation: O-linked β-N-acetylglucosaminylation; ESCRT: Endosomal Sorting Complex Required for Transport; HGS: Hepatocyte growth factor-regulated tyrosine kinase substrate. EGFR: Epidermal growth factor receptor; FUT: fucosyltransferase; O-GlcNAcylation: O-linked β-N-acetylglucosaminylation; ESCRT: endosomal sorting complex required for transport; HGS: hepatocyte growth factor-regulated tyrosine kinase substrate; STAM: signal-transducing adaptor molecule; RAS: rat sarcoma virus; ERK: extracellular signal-regulated kinase; AKT: protein kinase B; NF-κB: nuclear factor kappa-light-chain-enhancer of activated B cells.