Wearable multimodal sensing for geriatric healthcare
Abstract
Aging populations face growing multimorbidity, while episodic clinical assessments fail to capture gradual physiological changes unfolding during daily life. Although wearable technologies enable continuous monitoring, single-modality systems provide incomplete and context-limited insight. This Perspective focuses on hybrid wearable sensors that integrate physical and chemical sensing for geriatric healthcare. Hybrid wearable sensing provides a pathway toward continuous, predictive, and personalized geriatric health management. By monitoring continuously multiple health parameters, such multimodal systems have distinct advantages for real-time monitoring, including early risk detection and more personalized health assessment through the integration of complementary physical and biochemical signals. We discuss recent advances in wearable physical sensors, alongside with emerging wearable chemical sensors, then argue that chem-phys hybrid integration enables more interpretable and clinically actionable assessment of aging trajectories than single-modality wearable systems. Finally, we discuss translational requirements and future prospects, including robust real-world operation, AI-driven inference, and integration with telemedicine and home-based care.
Keywords
INTRODUCTION
As life expectancy rises, multimorbidity increases, including diabetes, cardiovascular, renal, and neurodegenerative diseases. However, intermittent clinic-based care provides limited longitudinal insight and often misses early physiological changes, particularly in older adults with mobility constraints and fragmented follow-up. Decisions based on discrete tests, such as blood panels or brief electrocardiogram (ECG) recordings, fail to reflect daily-life physiology, allowing deterioration to go undetected[1].
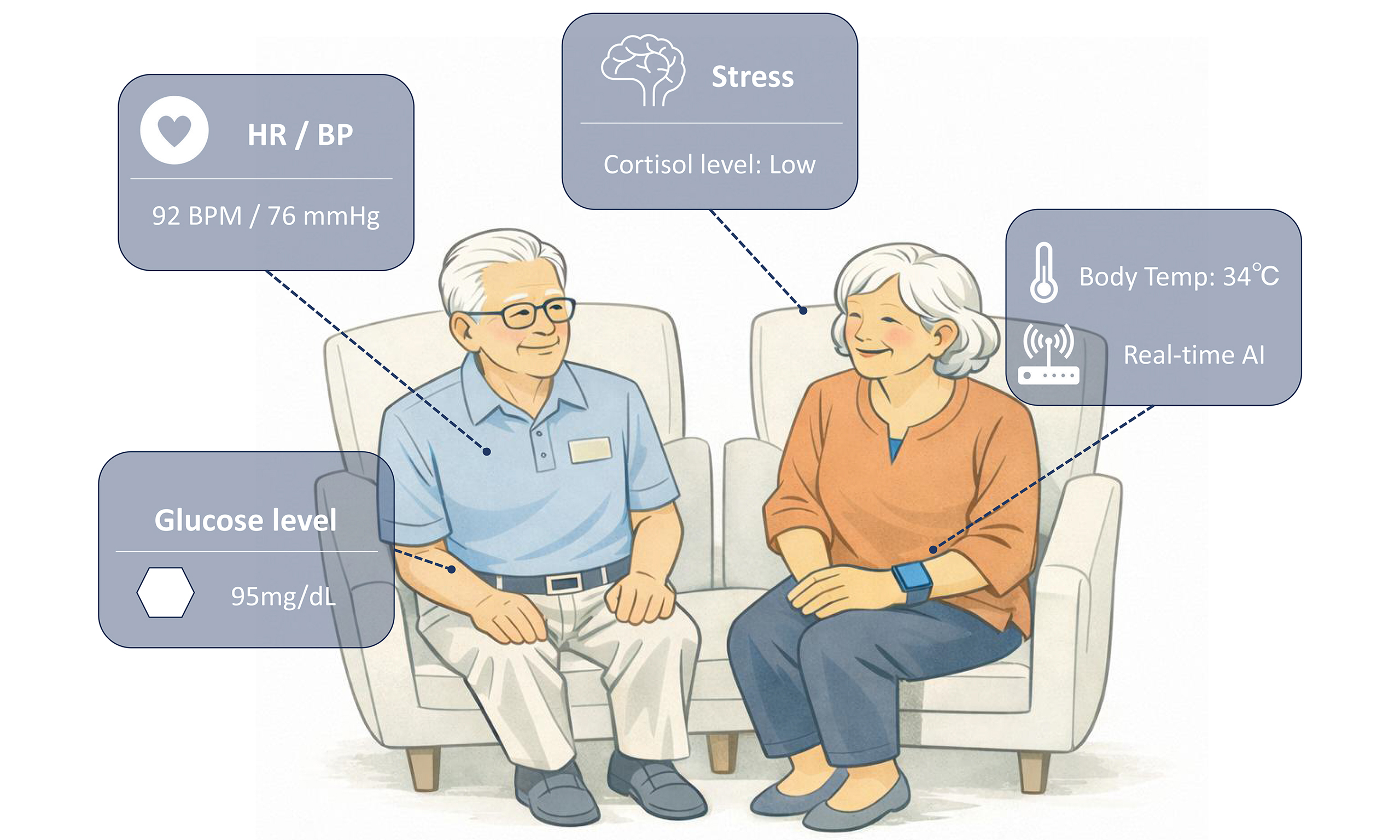
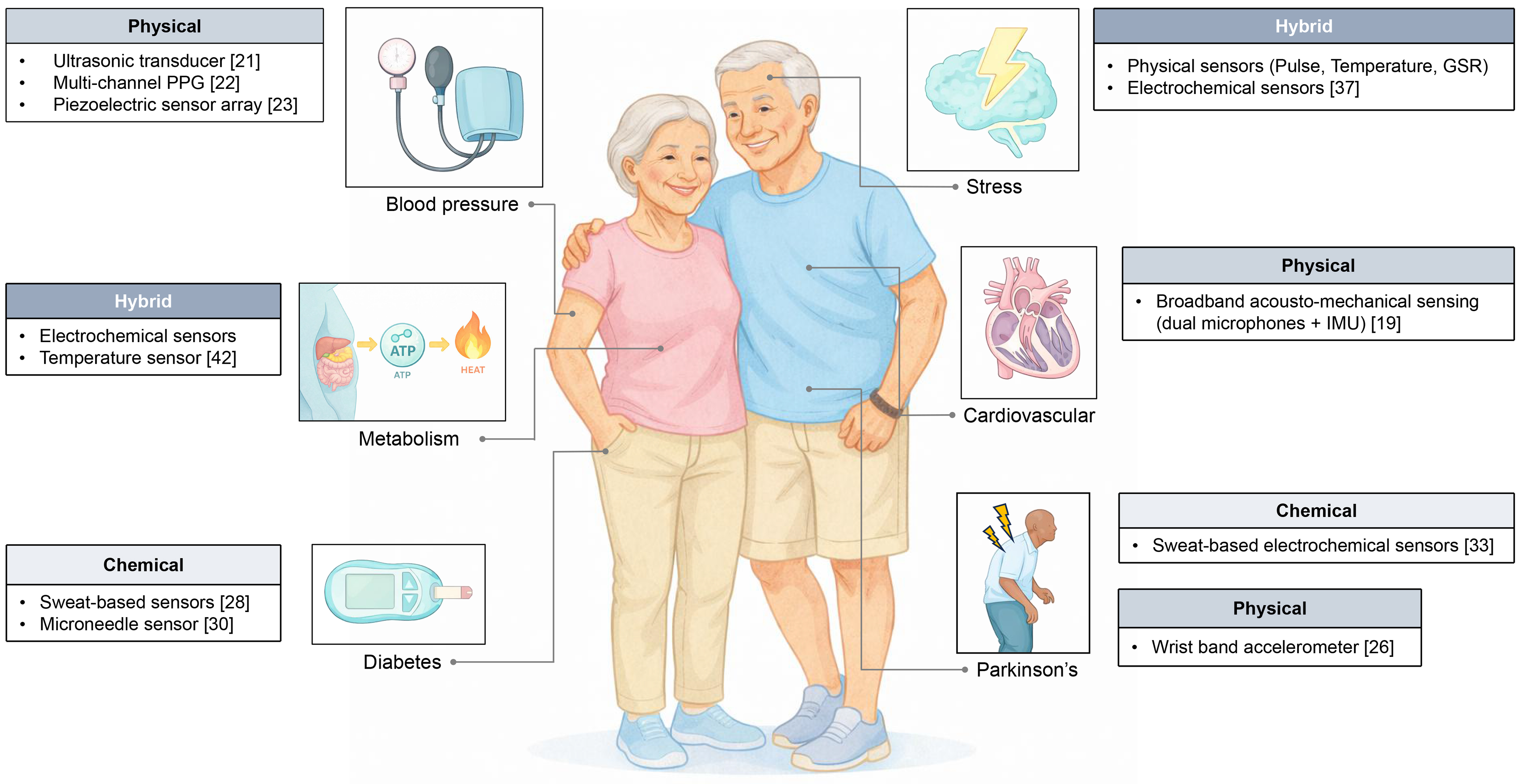
These gaps have motivated advanced sensing technologies and biomedical devices that measure health continuously as well as achieve personalized healthcare and treatment interventions. Wearable sensors that interface directly with the body using diverse formfactors, such as epidermal patches, smartwatches and ring-type devices, enabled by advances in skin-compatible and mechanically robust materials, offer continuous, user-friendly physiological monitoring in daily life[2-11]. Early generations of wearable devices have primarily focused on measurements of physical signals, such as electrocardiography, heart rate, blood pressure, body temperature, or motion, supporting detection of arrhythmias, fall risk, monitoring gait instability, and tracking rehabilitation outcomes in older adults[12,13]. In parallel, wearable biochemical sensors have emerged to capture molecular level information by continuously monitoring key biomarkers in biofluids, such as sweat or interstitial fluid. However, such single modality wearable sensors fall short of capturing the full spectrum of diverse symptoms experienced by old adults. By integrating well-established physical and chemical sensing modalities into a single unified platform, hybrid wearable systems enable synergistic physiological interpretation that extends beyond isolated signal acquisition and facilitates mechanistic insights into dynamic, system-level health states. Recently developed hybrid wearable devices track multiple biochemical and physiological signals on a single platform, enabling comprehensive monitoring of chronic conditions in older adults [Figure 1][14]. These integrated systems provide real-time data streams that support early detection and tailored intervention.
Figure 1. Integration of physical, chemical, and hybrid wearable sensors for holistic monitoring of aging health. PPG: Photoplethysmography; GSR: galvanic skin response; IMU: inertial measurement unit.
The true clinical value of continuous monitoring depends on translating high-dimensional data streams into interpretable and actionable endpoints. By establishing individualized baselines from longitudinal hybrid wearable data and integrating multi-sensor temporal trends, artificial intelligence (AI)-driven models can identify subtle physiological deviations that precede overt clinical deterioration[15-17]. By shifting from threshold-based alerts to probabilistic forecasting, AI enables earlier, lower-intensity interventions tailored to individual aging trajectories.
In this Perspective, we map a path towards hybrid wearable sensing for geriatric healthcare, focusing on older adults living with multimorbidity and the need for longitudinal health monitoring. We first summarize recent advances in biophysical wearables that capture cardiorespiratory and mobility-related dynamics, as well as chemical and minimally invasive platforms that access sweat or interstitial fluid for molecular monitoring of metabolic state and therapy response. We then argue that hybrid-based simultaneous tracking of biochemical and biophysical trends, combined with AI-analysis of longitudinal data addresses the elderly multifactorial health trajectories, enhances diagnostic accuracy and enables more clinically actionable assessment. Finally, we discuss key translational requirements, including real-world robustness, interpretable multimodal inference, and integration with telemedicine and home-based care.
THE WAY TO WEARABLE HYBRID SENSORS
Wearable physical and chemical sensors
The widespread adoption of consumer wearables, including smartwatches and rings, enables continuous physiological and behavioral monitoring during daily life[18]. Beyond these consumer devices, research-grade skin-interfaced systems expand the scope of cardiorespiratory monitoring by integrating soft strain sensors, acoustic sensors, and inertial measurement units (IMUs). By capturing body motion and deformation linked to respiration and cardiac activity, these platforms reveal mechanical–physiological interactions in real-world settings[19]. Age-adaptive polymeric skin electronics further address geriatric-specific challenges by accommodating age-related skin changes and preserving signal fidelity across diverse older populations[20].
In parallel, physical sensing research is increasingly focused on high-impact geriatric risks and chronic conditions. Wearable blood-pressure monitoring now spans ultrasound-based arterial tracking[21], cuffless optical strategies using multi-channel photoplethysmography (PPG) with finger pressing and learning-based estimation[22], thin, soft piezoelectric systems designed for continuous wireless monitoring of arterial pulse pressure[23], and stretchable inductor-capacitor (LC) resonant contact-lens sensors enable intraocular pressure monitoring relevant to glaucoma risk[24]. Mobility and falls risk are addressed through IMU-based sensing combined with machine learning enables automated fall-risk detection and monitoring from wearable inertial signals[25]. Physical sensing also contributes to neurodegenerative care, as wrist accelerometry combined with machine learning can detect early motor abnormalities predictive of prodromal Parkinson’s disease[26].
Compared with wearable physical sensing, wearable chemical monitoring faces challenges including biofluid access, biofouling, and signal instability, yet continues to advance rapidly due to the clinical value of continuous molecular monitoring[27]. Diabetes has driven the rapid progress in wearable chemical sensing, especially through the continuous glucose monitoring (CGM) systems that provide substantial clinical benefits. Additional approaches have demonstrated continuous wearable glucose sensing using passive sweat collection enabled by hydrogel-based interfaces and electrochemical readout strategies[28]. Beyond glucose, sweat-based platforms broaden the range of clinically relevant metabolites and electrolytes in real time[29]. Multiplexed electrochemical sensing systems, particularly wearable microneedle platforms have demonstrated continuous simultaneous detection of multiple ISF biomarkers[30,31]. Sweat has emerged as a noninvasive, information-rich biofluid enabling access to molecular health indicators beyond glucose alone. Early fully integrated sweat sensing systems established the feasibility of simultaneous, real-time analysis of multiple metabolites and electrolytes, during daily activities[32]. More recently, fingertip bioelectronic systems have shown that passively collected sweat can be used for non-invasive tracking of Parkinson disease levodopa drug following its oral administration, highlighting the potential of sweat analysis for personalized drug response assessment in neurological disorders[33,34]. Building on these developments, autonomous fingertip wearable microgrid platforms have unified sweat-powered energy harvesting, storage, and multi-analyte biochemical sensing within a miniaturized system for continuous, self-sustained monitoring[28]. Subsequent platforms further integrated wireless transmission and algorithm-assisted analysis for 24-hour biomarker monitoring, including uric acid[35]. Collectively, these advances demonstrate how sweat-based wearable chemical sensors are evolving toward longitudinal system-level monitoring of metabolism and therapy response in everyday life.
Multi-modal wearable sensor systems
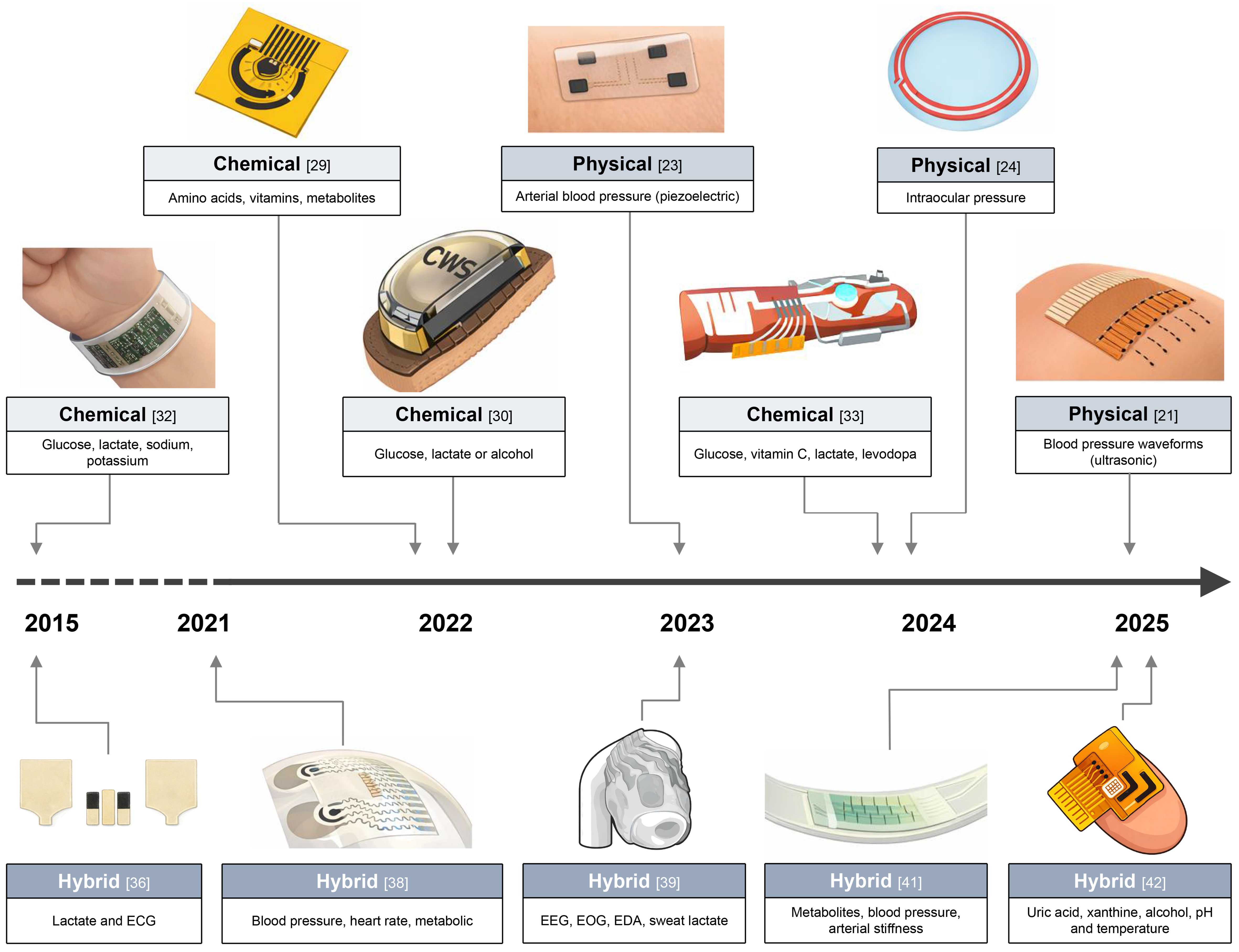
Hybrid wearable systems that simultaneously integrate biochemical sensing with physical vital-sign monitoring have emerged as a practical solution for continuous, home-based health surveillance in aging populations [Figure 2]. Early work demonstrated the feasibility of co-locating chemical and electrophysiological sensors within a single epidermal patch, enabling simultaneous sweat lactate monitoring and ECG recording without signal crosstalk[36]. This proof-of-concept established that metabolic and cardiac domains can be jointly monitored in a skin-conformal format suitable for prolonged wear. More recently, an AI-assisted physicochemical electronic skin expanded this concept by simultaneously tracking multiple sweat metabolites and electrolytes alongside pulse waveform, skin temperature, and galvanic skin response, enabling robust long-term multimodal monitoring of systemic stress responses[37].
Figure 2. Roadmap of wearable sensing technologies from chem-phys modalities toward integrated hybrid systems. ECG: Electrocardiogram; EEG: electroencephalography; EOG: electrooculography; EDA: electrodermal activity.
Subsequent advances extended hybrid sensing platforms toward clinically relevant hemodynamic and metabolic endpoints. An epidermal patch integrating ultrasonic transducers for blood pressure and heart rate with electrochemical sensors for glucose, lactate, caffeine, and alcohol, demonstrated simultaneous monitoring of cardiovascular and biochemical dynamics during daily activities[38]. Such combined monitoring is particularly relevant for older adults, where dehydration, infection, medication intake, or metabolic imbalance often manifests as coordinated rather than isolated changes in a single parameter.
Hybrid sensing has also diversified into unobtrusive form factors optimized for long-term adherence. Accordingly, different wearable form factors can be employed depending on the anatomical locations best suited for acquiring specific physiological signals. For example, the ear offers a rich source of brain and body biosignals towards continuous cognitive and metabolic health monitoring. An in-ear multimodal sensor, integrating brain electrophysiology with electrochemical lactate sensing, enabled continuous assessment of neurophysiological state alongside metabolic dynamics in real-world settings[39]. When combined with stimulation mechanisms, such systems open opportunities for personalized neurotherapeutic strategies[40]. This brain–body integration is particularly compelling in geriatric care, where cognitive decline and metabolic dysregulation frequently co-evolve.
Wrist-worn and minimally invasive hybrid platforms further extend multimodal monitoring for chronic disease management. A flexible wristband integrating microneedle-based interstitial sensing with cardiovascular monitoring enabled simultaneous metabolic and hemodynamic tracking in individuals with diabetes[41], addressing limitations of single-parameter glucose monitoring in multimorbid older adults.
Finally, advances in sweat interfacing and microfluidics have improved the reliability of biochemical sensing in hybrid systems. A bioinspired microfluidic wearable enabled multiday, multiplexed metabolic monitoring under low-sweat conditions[42], providing infrastructure for hybrid integration.
Collectively, these studies demonstrate a shift from isolated wearable measurements to integrated chem–phys platforms for longitudinal monitoring. In geriatric populations, hybrid wearables improve interpretability and early detection beyond episodic care.
POTENTIAL OF WEARABLE HYBRID PLATFORMS FOR GERIATRIC CARE
A defining challenge in geriatric medicine is that many age-associated diseases - including neurodegenerative disorders, cardiometabolic dysfunction, and frailty - progress gradually and manifest as coupled changes across physical function, biochemical regulation, and therapeutic response during daily life. Because these transitions unfold continuously outside clinical settings, episodic assessments often fail to capture meaningful dynamics. In the perspective of continuous monitoring, unlike single-modality wearables, hybrid sensors simultaneously measure multiple physiological and biochemical parameters, improving the reliability and predictive accuracy of health assessment.
In conditions, such as Parkinson’s disease, the symptom severity fluctuates with medication timing, activity level, stress, and sleep, while disease progression itself is slow and heterogeneous. Wearable hybrid platforms that combine motion sensing, electrophysiology, and biochemical monitoring of therapeutic drugs or stress-related biomarkers can capture these dynamics. By preserving ecological validity and providing high temporal resolution, wearable systems allow clinicians to interpret motor symptoms in the context of concurrent biochemical state rather than relying on sparse clinical snapshots.
The clinical impact of hybrid sensors will depend on their capacity to support personalized and adaptive care. By linking biochemical indicators with physical signals, hybrid systems can inform closed-loop or semi–closed-loop therapeutic strategies. This is particularly important for older adults, who exhibit altered pharmacokinetics, increased sensitivity to dosing, and multimorbidity that complicates standardized treatment. Hybrid sensing thus offers a pathway toward optimal tailored therapy that balances efficacy and safety.
Translational success depends on long-term wearability, usability, mitigation of biofouling, signal instability[43], and data security. Modular architectures that allow periodic replacement of chemical components while maintaining long-term physical monitoring address mismatched sensor lifetimes[44]. To mitigate signal overlap and data confounding in multimodal monitoring, hybrid systems should adopt multi-level data fusion strategies, enabling computational separation of overlapping physiological signatures[45]. Protecting sensitive biometric information will require secure transmission, edge processing, and privacy-aware frameworks[46]. Lastly, cost-effectiveness will be a critical determinant for the large-scale adoption of hybrid wearable systems across diverse healthcare settings.
As healthcare shift toward home-centered and preventive models, wearable hybrid sensors are poised to function as a central biological interface between older adults and digital health ecosystems. Integrated with telemedicine platforms and smart home systems, they provide body-level physiological and biochemical ground truth that complements ambient sensing and reduces diagnostic ambiguity. Recent precision health models have illustrated how continuous wearable data, when integrated with telemedicine and smart-home infrastructure, can support individualized risk assessment and proactive care beyond conventional clinic-centered management[47].
Ultimately, the promise of wearable hybrid sensors in geriatric healthcare lies in seamless integration into daily life. Human-centered design emphasizing unobtrusiveness, intuitive interfaces, minimal maintenance, and energy-efficient operation will be essential for sustained engagement. When coupled with scalable manufacturing and advanced analytics [e.g., AI/machine learning (ML)], hybrid wearable systems offer a foundation for transforming geriatric care from reactive management toward continuous, predictive, and personalized health support. Finally, this work highlights both technological opportunities and the need for large-scale longitudinal validation in multimorbid elderly populations to translate hybrid sensing into clinically meaningful solutions.
DECLARATIONS
Acknowledgments
The authors acknowledge helpful discussions within the Aiiso Yufeng Li Family Department of Chemical and Nano Engineering at UC San Diego. This work was supported by the UCSD Center for Wearable Sensors.
Authors’ contributions
Conceptualization: Kim, B.; Ding, S.
Writing - original draft: Kim, B.
Writing - review and editing: Kim, B.; Ding, S.; Wang, J.
Supervision: Ding, S.; Wang, J.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool ChatGPT (version 5.1, released 2026-11-12) was used for language and graphic editing. The tool did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Financial support and sponsorship
None.
Conflicts of interest
Wang, J. is the Advisory Editor Member in the Soft Science. Ding, S. is the Guest Editor of the Special Topic “Integrated Wearable Devices for Energy Management and Health Monitoring” in the Soft Science. They had no involvement in the review or editorial process of this manuscript, including but not limited to reviewer selection, evaluation, or the final decision, while the other authors have declared that they have no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
2. Kurul, F.; Aydoğan, D.; Janat, S.; Aydın Kırlangıc, I.; Kaya, H. O.; Topkaya, S. N. Wearable sensors for health monitoring: current applications, trends, and future directions. Biosens. Bioelectron. X. 2026, 28, 100727.
3. Ferreira, R. G.; Silva, A. P.; Nunes-Pereira, J. Current on-skin flexible sensors, materials, manufacturing approaches, and study trends for health monitoring: a review. ACS. Sens. 2024, 9, 1104-33.
4. Ates, H. C.; Nguyen, P. Q.; Gonzalez-Macia, L.; et al. End-to-end design of wearable sensors. Nat. Rev. Mater. 2022, 7, 887-907.
5. Kim, B.; Lee, S.; Kim, J. I.; et al. Liquid metal‐based multimodal wearable sensor platform enabled by highly accessible microfabrication of PDMS with tuned mechanical properties. Adv. Mater. Technol. 2025, 10, 2400859.
6. Sel, K.; Osman, D.; Huerta, N.; Edgar, A.; Pettigrew, R. I.; Jafari, R. Continuous cuffless blood pressure monitoring with a wearable ring bioimpedance device. NPJ. Digit. Med. 2023, 6, 59.
7. Keeler Bruce, L.; González, D.; Dasgupta, S.; Smarr, B. L. Biometrics of complete human pregnancy recorded by wearable devices. NPJ. Digit. Med. 2024, 7, 207.
8. Butler, P. M.; Yang, J.; Brown, R.; et al. Smartwatch- and smartphone-based remote assessment of brain health and detection of mild cognitive impairment. Nat. Med. 2025, 31, 829-39.
9. Perez, M. V.; Mahaffey, K. W.; Hedlin, H.; et al.; Apple Heart Study Investigators. Large-scale assessment of a smartwatch to identify atrial fibrillation. N. Engl. J. Med. 2019, 381, 1909-17.
10. Zhou, B.; Su, Y.; Wu, B.; Gao, B.; He, B.; Lim, C. T. Amorphous region modifications enhance genetically engineered spider silk for advanced wearable applications. Adv. Funct. Mater. 2026, 36, e07170.
11. Song, Y.; Li, Y.; Wang, Y.; Ye, C.; Zhu, C.; Yang, R. Artificial intelligence powered smart multimodal hand‐wearable systems for healthcare and human–machine interaction. Adv. Funct. Mater. 2026, e32014.
12. Teymourian, H.; Tehrani, F.; Longardner, K.; et al. Closing the loop for patients with Parkinson disease: where are we? Nat. Rev. Neurol. 2022, 18, 497-507.
13. Bayoumy, K.; Gaber, M.; Elshafeey, A.; et al. Smart wearable devices in cardiovascular care: where we are and how to move forward. Nat. Rev. Cardiol. 2021, 18, 581-99.
14. Mahato, K.; Saha, T.; Ding, S.; Sandhu, S. S.; Chang, A.; Wang, J. Hybrid multimodal wearable sensors for comprehensive health monitoring. Nat. Electron. 2024, 7, 735-50.
15. Jain, A.; Bhullar, S. AI-driven wearable health devices with health-aware control and secure prognostics: experimental and simulation-based validation. Array 2025, 28, 100532.
16. van der Vegt, A. H.; Campbell, V.; Mitchell, I.; et al. Systematic review and longitudinal analysis of implementing artificial intelligence to predict clinical deterioration in adult hospitals: what is known and what remains uncertain. J. Am. Med. Inform. Assoc. 2024, 31, 509-24.
17. Uwakwe, C. K.; Rangan, E. S.; Kumar, S.; et al. Longitudinal wearable sensor data enhance precision of long COVID detection. PLOS. Digit. Health. 2025, 4, e0001093.
18. Jamieson, A.; Chico, T. J. A.; Jones, S.; Chaturvedi, N.; Hughes, A. D.; Orini, M. A guide to consumer-grade wearables in cardiovascular clinical care and population health for non-experts. NPJ. Cardiovasc. Health. 2025, 2, 44.
19. Yoo, J. Y.; Oh, S.; Shalish, W.; et al. Wireless broadband acousto-mechanical sensing system for continuous physiological monitoring. Nat. Med. 2023, 29, 3137-48.
20. Mottini, V.; Xing, L.; Cai, Y.; et al. Age-adaptive polymeric skin electronics enhance neural data and machine learning accuracy. Device 2026, 4, 100987.
21. Zhou, S.; Park, G.; Longardner, K.; et al. Clinical validation of a wearable ultrasound sensor of blood pressure. Nat. Biomed. Eng. 2025, 9, 865-81.
22. Kyung, J.; Yang, J. Y.; Choi, J. H.; et al. Deep-learning-based blood pressure estimation using multi channel photoplethysmogram and finger pressure with attention mechanism. Sci. Rep. 2023, 13, 9311.
23. Li, J.; Jia, H.; Zhou, J.; et al. Thin, soft, wearable system for continuous wireless monitoring of artery blood pressure. Nat. Commun. 2023, 14, 5009.
24. An, H.; Wang, X.; Liao, Z.; et al. LC contact lens sensor for ultrasensitive intraocular pressure monitoring. npj. Flex. Electron. 2024, 8, 341.
25. Choi, A.; Kim, T. H.; Yuhai, O.; et al. Deep learning-based near-fall detection algorithm for fall risk monitoring system using a single inertial measurement unit. IEEE. Trans. Neural. Syst. Rehabil. Eng. 2022, 30, 2385-94.
26. Schalkamp, A. K.; Peall, K. J.; Harrison, N. A.; Sandor, C. Wearable movement-tracking data identify Parkinson’s disease years before clinical diagnosis. Nat. Med. 2023, 29, 2048-56.
27. Kim, J.; Campbell, A. S.; de Ávila, B. E.; Wang, J. Wearable biosensors for healthcare monitoring. Nat. Biotechnol. 2019, 37, 389-406.
28. Saha, T.; Khan, M. I.; Sandhu, S. S.; et al. A passive perspiration inspired wearable platform for continuous glucose monitoring. Adv. Sci. 2024, 11, e2405518.
29. Wang, M.; Yang, Y.; Min, J.; et al. A wearable electrochemical biosensor for the monitoring of metabolites and nutrients. Nat. Biomed. Eng. 2022, 6, 1225-35.
30. Tehrani, F.; Teymourian, H.; Wuerstle, B.; et al. An integrated wearable microneedle array for the continuous monitoring of multiple biomarkers in interstitial fluid. Nat. Biomed. Eng. 2022, 6, 1214-24.
31. Freeman, D. M. E.; Ming, D. K.; Wilson, R.; et al. Continuous measurement of lactate concentration in human subjects through direct electron transfer from enzymes to microneedle electrodes. ACS. Sens. 2023, 8, 1639-47.
32. Gao, W.; Emaminejad, S.; Nyein, H. Y. Y.; et al. Fully integrated wearable sensor arrays for multiplexed in situ perspiration analysis. Nature 2016, 529, 509-14.
33. Ding, S.; Saha, T.; Yin, L.; et al. A fingertip-wearable microgrid system for autonomous energy management and metabolic monitoring. Nat. Electron. 2024, 7, 788-99.
34. Moon, J. M.; Teymourian, H.; De la Paz, E.; et al. Non-invasive sweat-based tracking of L-dopa pharmacokinetic profiles following an oral tablet administration. Angew. Chem. Int. Ed. Engl. 2021, 60, 19074-8.
35. Li, J.; Xie, G.; Zhao, X.; Luo, X.; Chen, C.; Su, Y. A self-powered wearable uric acid sensor for prolonged monitoring and intervene of hyperuricemia. Chem. Eng. J. 2026, 530, 173622.
36. Imani, S.; Bandodkar, A. J.; Mohan, A. M.; et al. A wearable chemical-electrophysiological hybrid biosensing system for real-time health and fitness monitoring. Nat. Commun. 2016, 7, 11650.
37. Xu, C.; Song, Y.; Sempionatto, J. R.; et al. A physicochemical-sensing electronic skin for stress response monitoring. Nat. Electron. 2024, 7, 168-79.
38. Sempionatto, J. R.; Lin, M.; Yin, L.; et al. An epidermal patch for the simultaneous monitoring of haemodynamic and metabolic biomarkers. Nat. Biomed. Eng. 2021, 5, 737-48.
39. Xu, Y.; De la Paz, E.; Paul, A.; et al. In-ear integrated sensor array for the continuous monitoring of brain activity and of lactate in sweat. Nat. Biomed. Eng. 2023, 7, 1307-20.
40. Xu, Y.; Uppal, A.; Lee, M. S.; et al. Earable multimodal sensing and stimulation: a prospective towards unobtrusive closed-loop biofeedback. IEEE. Rev. Biomed. Eng. 2024, 18, 5-25.
41. Chang, A. Y.; Lin, M.; Yin, L.; et al. Integration of chemical and physical inputs for monitoring metabolites and cardiac signals in diabetes. Nat. Biomed. Eng. 2026, 10, 94-109.
42. Shin, S.; Liu, R.; Yang, Y.; et al. A bioinspired microfluidic wearable sensor for multiday sweat sampling, transport, and metabolic analysis. Sci. Adv. 2025, 11, eadw9024.
43. Xie, X.; Doloff, J. C.; Yesilyurt, V.; et al. Reduction of measurement noise in a continuous glucose monitor by coating the sensor with a zwitterionic polymer. Nat. Biomed. Eng. 2018, 2, 894-906.
44. Bandodkar, A. J.; Gutruf, P.; Choi, J.; et al. Battery-free, skin-interfaced microfluidic/electronic systems for simultaneous electrochemical, colorimetric, and volumetric analysis of sweat. Sci. Adv. 2019, 5, eaav3294.
45. Celik, Y.; Godfrey, A. Bringing it all together: wearable data fusion. NPJ. Digit. Med. 2023, 6, 149.
46. Zhang, B.; Chen, C.; Lee, I.; Lee, K.; Ong, K. A survey on security and privacy issues in wearable health monitoring devices. Comput. Secur. 2025, 155, 104453.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.