Spin-selection in L-chiral Ag-doped WS2 nanoflower for photocatalysis
Abstract
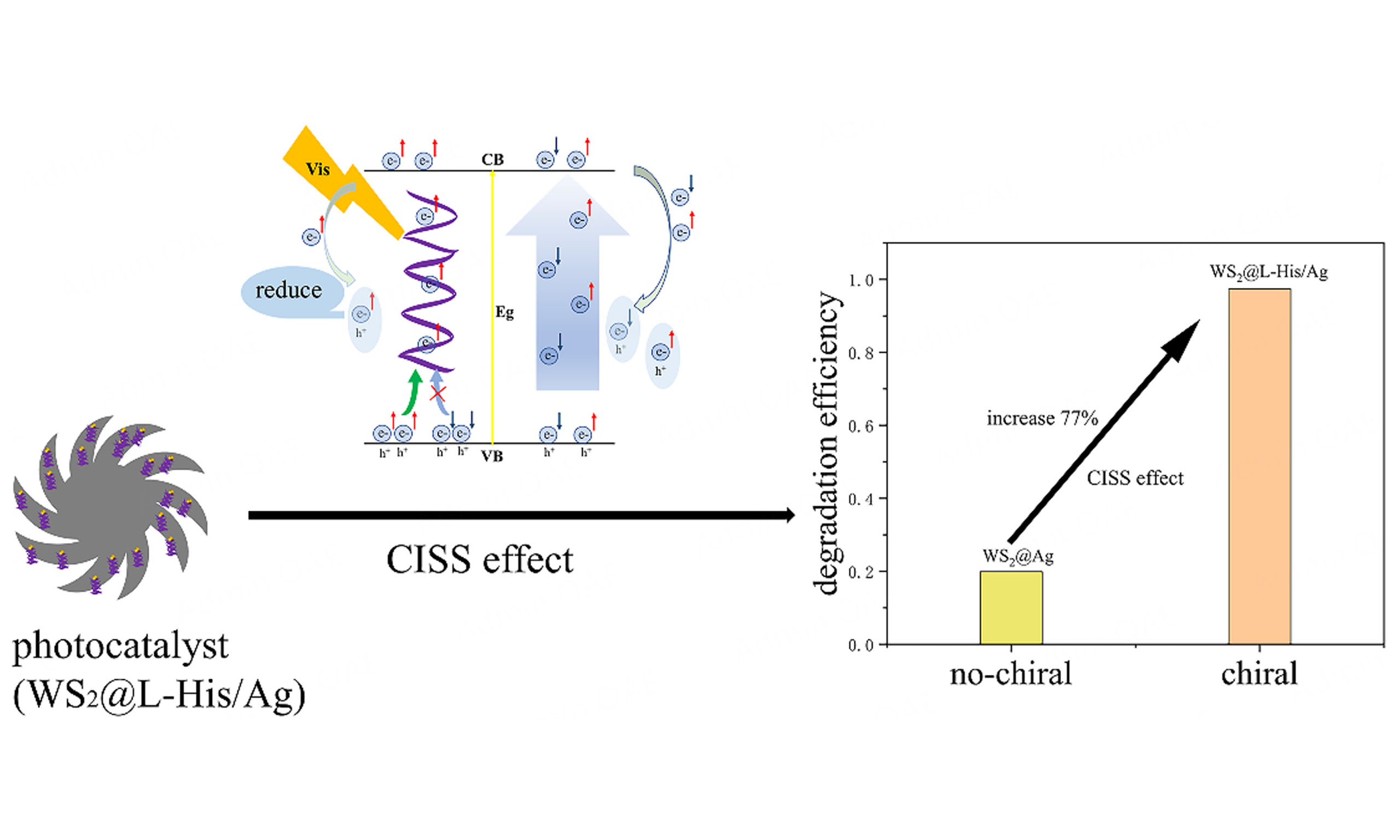
In this study, we synthesized a novel heterostructure comprising two-dimensional tungsten sulfide nanoflowers and chiral silver nanoparticles. Leveraging the chiral-induced spin selection (CISS) effect, our WS2@L-His/Ag(chiral) system achieved a remarkable 77% enhancement in photocatalytic degradation efficiency over the WS2@Ag(achiral) counterpart. The chiral nanostructures selectively facilitate the transport of electrons with defined spin orientations, thereby suppressing the recombination of electrons and holes and augmenting the generation of hydroxyl radicals, which significantly boosts degradation efficiency. Our findings underscore the potential of electron spin manipulation and herald novel avenues for chiral material applications in spin-dependent photoelectric chemical processes.
Keywords
INTRODUCTION
Photocatalysis is considered one of the best strategies for environmental remediation[1,2]. However, the efficacy of most photocatalysts is hindered by limitations in light absorption range and photogenerated carrier recombination rates, leading to suboptimal photocatalytic activity and efficiency[3,4]. Overcoming the primary obstacle of recombination experienced by photoinduced electron-hole pairs has become crucial for enhancing overall photocatalytic performance.
Two-dimensional materials exhibit excellent photocatalytic activity on account of their abundant surface active sites, distinctive geometric structures, and adjustable electronic structures[5,6]. In light of this, researchers have carried out extensive research endeavors, encompassing methods such as element doping, heterojunction design, defect creation, single-atom modification, and plasma metal loading, achieving promising outcomes[7,8]. One such approach involves utilizing black phosphorus, which possesses self-healing properties and exhibits unsaturated dangling bonds formed by double vacancies of phosphorus atoms. This unique characteristic effectively retards charge recombination, bolstering photocatalytic performance[9]. Another avenue lies in the utilization of two-dimensional van der Waals heterojunctions, exemplified by the MoSe2/Ti2CO2 system. This heterojunction demonstrates remarkable resistance against electron-hole recombination and photo corrosion, commonly observed in transition metal dichalcogenides. By employing this heterojunction, efficient electron-hole separation is achieved, elevating photocatalytic efficiency[10]. These cutting-edge strategies demonstrate that by precisely regulating the electronic structure of photocatalysts, their photocatalytic activity can be greatly enhanced[11].
However, spin, which is an intrinsic and fundamental property of electrons, is seldom contemplated in photocatalytic processes. The transfer of electrons must adhere to principles such as Hund's rules, Pauli's exclusion principle, and conservation laws for angular momentum. Consequently, considering the impact of electron spin on photocatalysis becomes imperative. Recent studies have revealed that controlling the electron spin polarization in TiO2 through metal defects can effectively suppress recombination between excited electrons and holes. This strategy shows promise in significantly boosting the overall performance of photocatalysts[12]. Similarly, photoexcitation in chiral semiconductors can induce spin splitting through structural asymmetry. For example, Jnawali et al. demonstrated ultrafast photoinduced band splitting in chiral tellurium nanosheets, highlighting the correlation between chirality, strain, and spin-selective carrier dynamics[13]. Chiral molecules, with their unique structural properties, offer a compelling avenue to manipulate electron spins through the remarkable phenomenon of chiral-induced spin-selective (CISS) effects[14,15]. This effect endows chiral structures with the ability to act as selective filters, enabling the transportation of electrons with specific spin orientations[16-18]. Recent findings have unveiled the potential of harnessing the CISS effect in photochemical water decomposition processes aimed at hydrogen production. By leveraging this effect, precise control over the spin direction of the process is achieved by effectively inhibiting the spin polarization of the hydroxyl group[19-22].
So herein, we present the successful development of a novel photocatalytic system involving the adsorption of a colloidal solution of chiral histidine-modified nano-Ag onto the surface of transition metal dichalcogenides. This innovative approach results in the creation of chiral semiconductor materials
MATERIALS AND METHODS
Materials
The following chemicals were used: sodium tungstate dihydrate (Na2WO4·2H2O; ACS reagent,
Preparation of the WS2 nanoflowers
The WS2 nanoflowers were synthesized via the hydrothermal method. 8 g of sodium tungstate was added to a beaker containing 100 mL of deionized water; the mixture was then stirred to dissolve sodium tungstate, and the pH of the solution was adjusted to be approximately 1 by adding 3 mol/L hydrochloric acid. 6.3 g of oxalic acid was added to the solution, which was then stirred and diluted to 250 mL. 3 g of thioacetamide was added to 80 mL of the above solution, which was again stirred. The solution was transferred to a 50 mL PTFE reactor, which was sealed, placed in a blast drying box (DHG-9053A, Jinghong, China), and heated to 180 °C. This temperature was kept for 24 h. Subsequently, the solution was left to cool down naturally to room temperature. The reaction liquid was centrifuged (H1850, Xiangyi, China); the upper layer of the liquid was discarded, while the black solid was retained. This was washed several times with ethanol and dried in an oven at 60 °C. The WS2 nanoflowers were obtained.
Preparation of the chiral Ag nanoparticles
An aqueous solution with a molar ratio of Ag nitrate to histidine of 1:6 was prepared. The solution was stirred quickly in an ice bath (Low-temperature circulating cooling pump, CCA-20, Yuhua Instrument, China), and 150 µL of a 6 mmol/L sodium hydroxide solution was added to adjust its pH (5 °C, 300 rpm,10 min). After stirring for 10 min, 250 µL of the newly obtained 1.5 mmol/L sodium borohydride aqueous solution was rapidly added to the solution; the obtained solution was then stirred vigorously for 30 min. The solution was initially colorless and then became pale yellow or brownish yellow; at this point, the nano-Ag colloid was formed.
Loading the chiral Ag nanocolloid on WS2
70 mg WS2 powder was added to 140 mL (D/L/D, L) histidine nano-Ag colloidal solution, which was placed in a rotating water bath in a constant temperature oscillator (SHY-2, Dadi Automation Instruments, China) at room temperature. A given oscillation speed was maintained for 10 min. Finally, a microporous membrane was used to filtrate the mixed solution (Jinteng, aqueous MCE; 50 mm, 0.2 µm), and then it was dried.
Characterization of WS2@chiral-His/Ag
In our comprehensive investigation, we employed state-of-the-art characterization techniques to analyze the properties and behavior of the WS2 and WS2@chiral-His/Ag nanosheets. Powder X-ray diffraction (XRD) patterns were acquired using a Rigaku Smart Lab X-ray diffractometer (Japan) in the range 2θ = 5°-70°. To gain insights into the elemental composition and chemical states, X-ray photoemission spectroscopy (XPS) was performed using a Thermo Scientific K-Alpha instrument (USA). Microstructural analysis was conducted using transmission electron microscopy (TEM) with a FEI Talos F200s instrument (Japan) and field emission scanning electron microscopy (FESEM) using a Thermo Scientific G4 UC instrument (USA). These techniques allowed us to observe and characterize the microstructure of WS2 and the WS2@chiral-His/Ag nanosheets, respectively. To investigate the optical properties, ultraviolet-isible-near-infrared (UV-Vis-NIR) spectrophotometry was carried out using a Lambda 750s instrument (USA), measuring the absorption of the WS2@chiral-His/Ag composite material in the wavelength range of 200-800 nm. Electron paramagnetic resonance (EPR) spectroscopy, utilizing a Bruker EMX pius A30 instrument (Germany), was employed to assess the free radical content. Furthermore, the concentration of the pollutant was determined using a UV-Vis spectrophotometer (Thermo Fisher Evolution220, USA). Raman spectra of the powder samples were acquired using a HORIBA LabRAM HR (Japan) Evolution Raman microscope, employing a laser excitation wavelength of 532 nm. The chiral characteristics of the samples were characterized using a circular dichroism (CD) spectrometer (Applied Photophysics Chirascan V100, UK). The photoluminescence (PL) spectroscopy of the samples was characterized using a steady-state transient fluorescence spectrometer (Edinburgh Instruments Fls 1000, UK). By employing this comprehensive suite of advanced characterization techniques, we gained valuable insights into the structural, optical, and chemical properties of the WS2 and WS2@chiral-His/Ag nanosheets.
Photocatalytic performance evaluation
To evaluate the exceptional photocatalytic degradation performance of the synthesized WS2@chiral-His/Ag nanosheets, we conducted a series of experiments focusing on the degradation of an organic Rhodamine B (RhB) solution. A solar simulator (Perfect Light PLS-SXE 330+UV, China) was used to act as a light source; this simulator is equipped with a 50% filter. The optical density of the simulated sunlight was carefully adjusted using an optical power meter. In a typical reaction, 30 mg of the catalyst was added to a double-walled beaker containing 15 mg/L RhB under dark conditions. The beaker was agitated for 30 min to achieve adsorption and desorption equilibrium between the catalyst and the RhB molecules. Then, the light source was turned on, and 1 mL of H2O2 (30 wt%) was added to the reactor. A catalytic reaction was performed for 30 min. During the reaction, one serving of 1 mL was taken from the mixture every 5 min and immediately centrifuged at a speed of 5,000 rpm to remove the catalyst particles; the supernatant was retained to determine the RhB concentration. The change in the RhB concentration was determined by the UV-Vis absorption intensity of the supernatant sampled at different times. By employing this rigorous experimental approach, we were able to quantitatively evaluate the degradation efficiency of the WS2@chiral-His/Ag nanosheets through the photocatalytic degradation of RhB.
RESULTS AND DISCUSSION
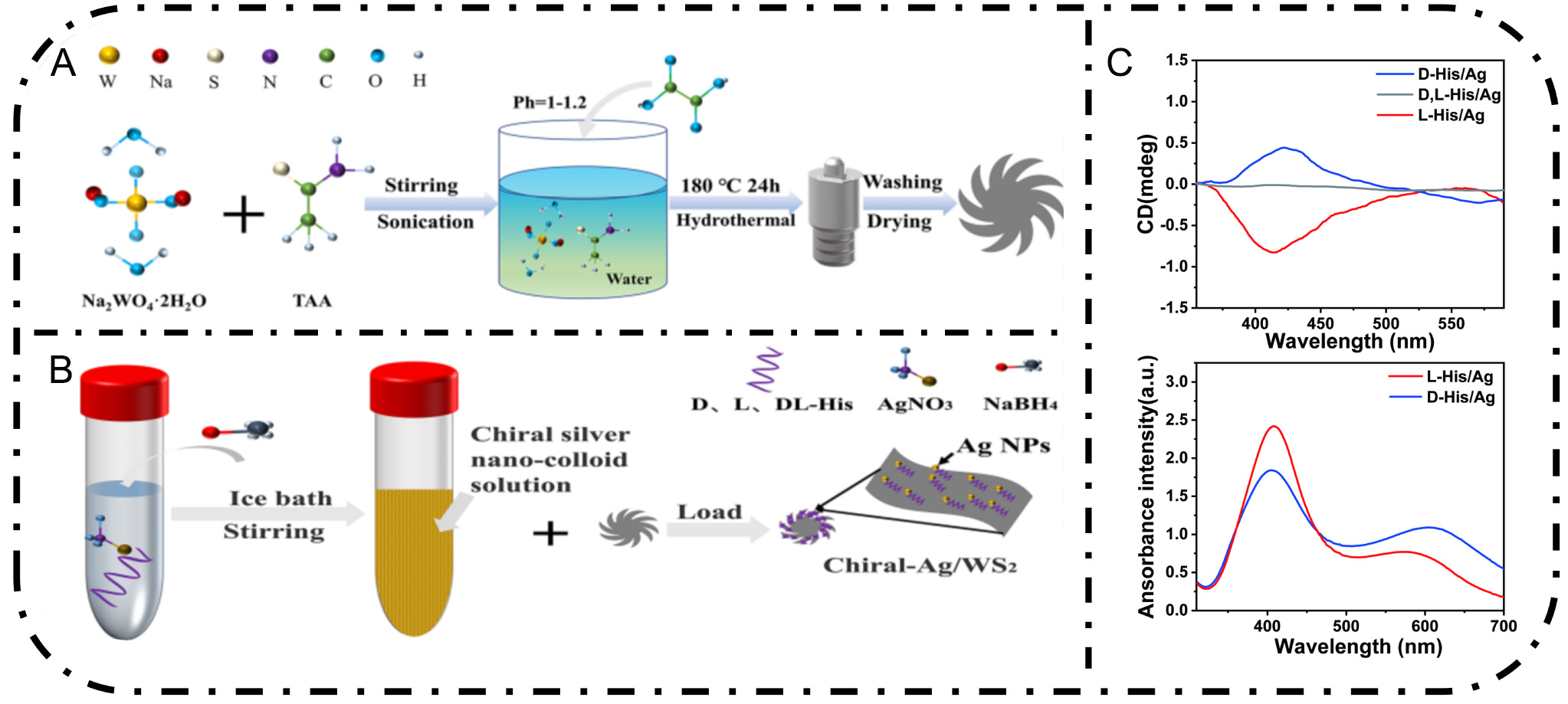
In this study, we employed hydrothermal synthesis and liquid-phase reduction methods to fabricate WS2 nano-flower-like particles and chiral Ag colloids, respectively. Figure 1A and B presents the schematic diagram of the preparation process for WS2 nanoflowers and the chiral Ag nanocolloid. The chiral silver nanocolloids were investigated by transmission electron microscopy (TEM) and circular dichroism (CD) spectrometer. The TEM images, which reveal the detailed structure of the nanocolloid, are shown in Supplementary Figure 1. CD spectroscopy can directly determine whether a sample has a chiral structure and provide detailed insights into its asymmetric structure and chiral properties. As depicted in Figure 1C, the CD spectra of D,L-His/Ag are nearly zero, indicating the absence of significant optical activity. In contrast, the CD spectra of L-His/Ag and D-His/Ag exhibit opposite signal characteristics, which further confirms their distinct chiral configurations. Notably, the CD spectra of L-His/Ag and D-His/Ag display prominent positive and negative peaks within the range of 350-600 nm, likely arising from chirality-induced asymmetric structures. This chiral feature significantly impacts the catalytic performance of the materials in subsequent experiments. Specifically, the CD intensity of L-His/Ag is more pronounced, suggesting a stronger chiral optical response. Therefore, in the following discussions, WS2 doped with L-His/Ag is primarily used as a representative sample to elucidate the effects of chirality on photocatalytic performance.
Figure 1. (A) Schematic illustration of the preparation of WS2 nanoflowers. (B) Preparation and loading of the chiral Ag nanocolloid. (C) CD and UV-Vis spectra of D/L/D,L-His/Ag.
The FESEM images displayed in Figure 2A and B demonstrate the uniform size and well-defined structure of the WS2 nanoflowers synthesized in this study. These nanoflowers exhibit an average particle size of
Figure 2. (A and B) SEM images of WS2. (C) TEM image of WS2@L-His/Ag. (D and E) HRTEM images of WS2@L-His/Ag. (F-J) WS2@L-His/Ag of S, Ag, and W STEM images and EDS elemental maps.
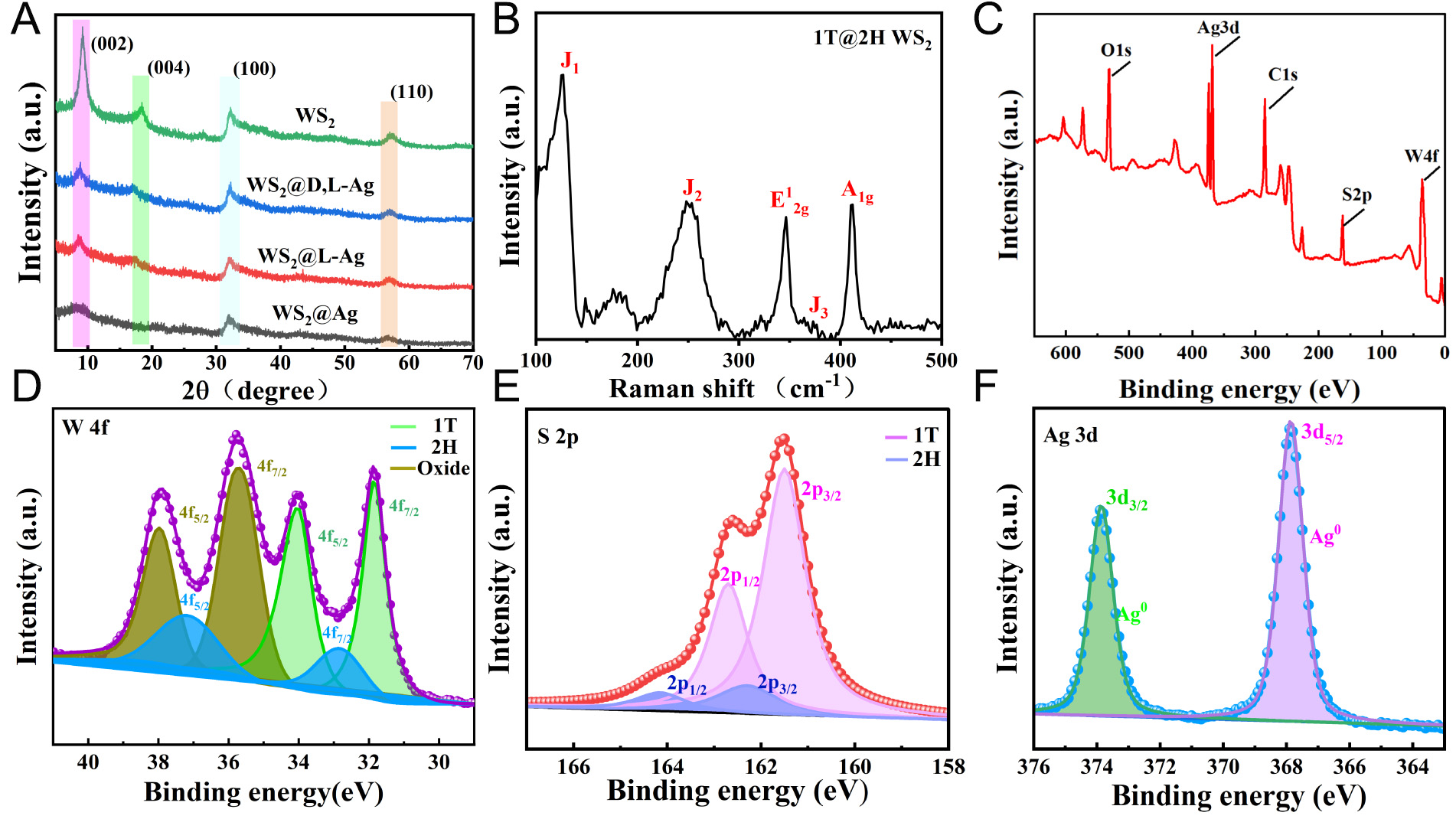
The X-ray diffraction (XRD) analysis, as shown by the black curve in Figure 3A, exhibits distinct peaks at 9.4°, 18.3°, 31.9°, and 57.3°. These peaks are associated with the (002) and (004) crystal planes of the 1T phase of WS2, as well as the (100) and (110) planes of the 2H phase of WS2, respectively. These findings confirm that the synthesized WS2 consists of a mixture of the 1T and 2H phases, denoted as 1T@2H WS2, which aligns with previous studies[23]. Upon loading WS2 with an equivalent amount of the (L/D,L) histidine nano-Ag colloidal solution, the intensity of the (002) diffraction peak of WS2 decreases. This reduction in intensity is likely due to the infiltration of chiral Ag nanoparticles into the pores of WS2, providing compelling evidence for the successful incorporation of chiral Ag nanoparticles onto WS2. Notably, the XRD patterns of WS2@L-His/Ag, WS2@D,L-His/Ag, and WS2@Ag do not display characteristic peaks of Ag, possibly because of the low loading content of Ag[24]. In the Raman spectra presented in Figure 3B, the prominent peaks at 337 and 419 cm-1 correspond to the in-plane E2g and out-of-plane A1g vibrational modes of 2H-WS2, respectively, while the J1, J2, and J3 peaks are attributed to 1T-WS2. These observations indicate the coexistence of the 1T and 2H phases in the WS2 nanosheets[25,26]. The Raman spectroscopic data further substantiate the presence of both 1T and 2H phases in the WS2 nanosheets. Collectively, the XRD and Raman analyses offer critical insights into the structural composition and crystalline phases of the synthesized WS2 and WS2@L-His/Ag composites.
Figure 3. (A) XRD patterns of WS2, WS2@D-His/Ag, WS2@L-His/Ag, WS2@D,L-His/Ag, and WS2@Ag. (B) Raman spectra of WS2. (C) XPS survey spectrum and high-resolution XPS spectra of WS2@L-Ag High-resolution XPS spectra of WS2@L-Ag (D) W 4f, (E) S 2p, and (F) Ag 3d.
To further investigate the elemental chemical states and compositions of the WS2@L-His/Ag composites, high-resolution X-ray photoelectron spectroscopy (XPS) measurements were performed. Figure 3C shows the survey XPS spectrum, which reveals that W, S, and Ag are the main elements. In addition, the O1s peak is detected because the WS2 nanosheets are easily oxidized in air. Figure 3D displays the high-resolution XPS spectra of the W 4f peaks in the WS2@L-His/Ag composite. The binding energies at 32.8 eV (W 4f7/2) and 37.2 eV (W 4f5/2) are characteristic of the 2H phase of WS2. Additionally, the binding energies at 36.7 eV
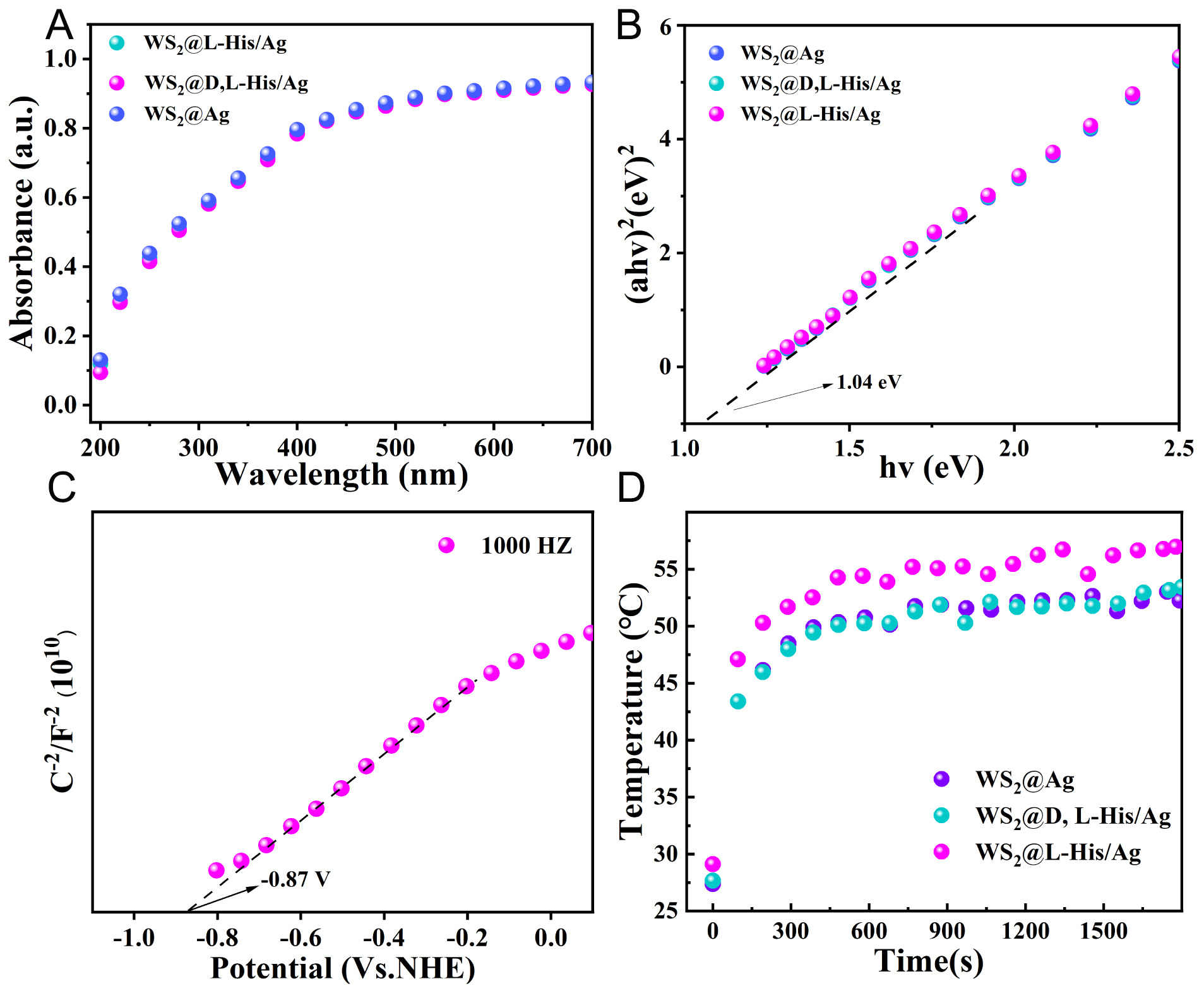
The optical properties of the composites were characterized using a UV-Vis absorption spectrophotometer, and the absorbance spectra of the samples were obtained. As shown in Figure 4A, the
Figure 4. (A and B) UV absorption spectra and band gap calculations of WS2@D,L/L-His/Ag and WS2@Ag, (C) WS2@L-His/Ag Mott-Schottky plots, (D) Surface temperature change, of the WS2@L-His/Ag, WS2@D,L-His/Ag, and WS2@Ag composites.
Photocatalytic degradation
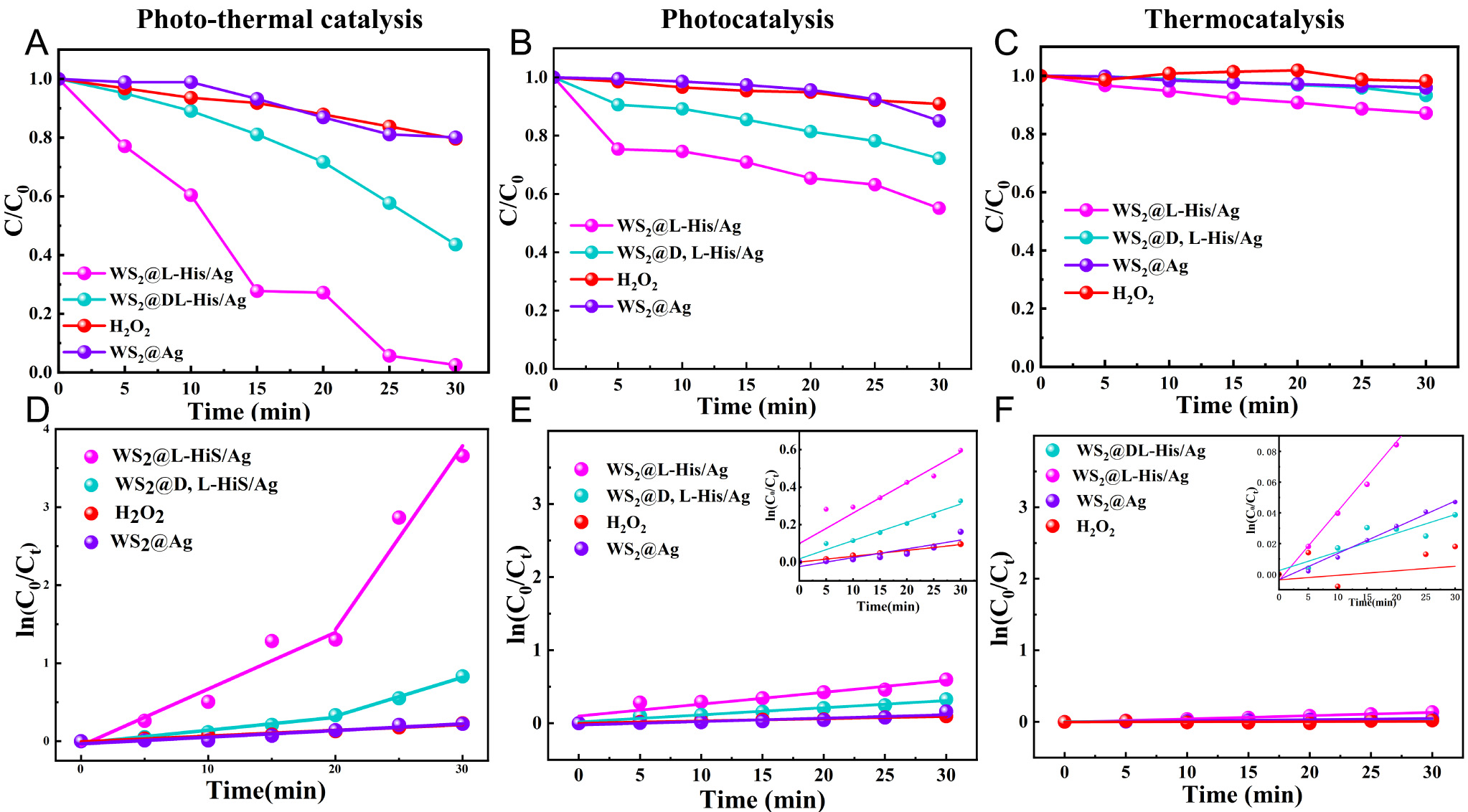
Figure 5 shows the degradation efficiency of RhB (C/C0) as a function of time (t). As illustrated in Figure 5A, the degradation rates of WS2@L-His/Ag, WS2@D,L-His/Ag, and WS2@Ag were 97.4%, 56.4%, and 19.9%, respectively. This indicates that the WS2@L-His/Ag composites exhibit significantly enhanced photocatalytic degradation performance compared to WS2@D,L-His/Ag, and WS2@Ag composites. Additionally, as shown in Supplementary Figure 3, no degradation of RhB was observed in the absence of a catalyst, which suggests that the chiral-induced spin selectivity (CISS) effect in WS2@L-His/Ag composites plays a critical role in promoting the photocatalytic degradation process[32-34]. WS2 serves as an excellent photothermal conversion medium and photocatalyst due to its ability to facilitate the separation and transfer of photogenerated carriers, which are key factors in the photocatalytic process. Owing to its narrow band gap, WS2 exhibits a broad light absorption range (as shown in Figure 4) and generates a pronounced photothermal effect. When photons with energy exceeding the band gap are absorbed, electron-hole pairs are generated above the band gap. These pairs can relax to the band edges via non-radiative processes, releasing excess energy as heat and increasing the system's temperature. The elevated temperature enhances carrier mobility, as higher temperatures typically result in faster carrier movement, thereby improving the photocatalytic activity of WS2 particles. Furthermore, Ag nanoparticles adsorbed onto WS2 surfaces exhibit surface plasmon resonance (SPR) effects within the visible region. The SPR effect not only increases the temperature but also enhances carrier mobility, further boosting the photocatalytic activity through photothermal effects.
Figure 5. Concentrations of the RhB dye as a function of time for the WS2@L-His/Ag, WS2@D,L-His/Ag, WS2@Ag, and H2O2 systems: (A) photothermal catalytic degradation, (B) photocatalytic degradation, and (C) thermocatalytic degradation. Graphs illustrating
The photocatalytic kinetics of WS2@L-His/Ag composites were investigated using the Langmuir-Hinshelwood first-order reaction model, expressed as ln(C0/Ct) = kt. In this equation, Ct and C0 denote the solution concentration at time t and the initial concentration under solar irradiation, respectively. It is evident that the WS2@L-His/Ag system exhibits the highest photothermal catalytic degradation rate. The enhanced degradation efficiency of WS2@chiral-His/Ag composites within 20 min can be attributed to their excellent photothermal properties, which enable the rapid degradation of residual RhB (as shown in
To further investigate the chiral-induced spin selectivity effect of WS2@L-His/Ag in the photocatalytic process, catalytic degradation experiments were conducted under pure light and pure heat conditions. For pure photocatalysis, a double-walled beaker reactor was placed within a cooling sleeve system with circulating water maintained at 25 °C to ensure a constant reaction temperature. To exclude potential thermal effects, pure thermal experiments were performed in darkness by heating the system to 32 °C using an external heating sleeve, while all other experimental steps remained identical. The results of these experiments are presented in Figure 5B, C, E, F. In Figure 5B and E, it is evident that the degradation efficiency under pure photocatalytic conditions is lower than that under photothermal conditions. However, the degradation trend remains consistent with the photothermal degradation results, where
In summary, the analysis of photocatalytic kinetics using the Langmuir-Hinshelwood modeling confirms that the WS2@L-His/Ag system exhibits the highest photocatalytic degradation rate. Furthermore, analogous results for the WS2@D-His/Ag system are illustrated in Supplementary Figure 5. To assess the stability of the catalyst, Supplementary Figure 6 displays the degradation rate of WS2@L-His/Ag after five catalytic degradation cycles. Additionally, XRD and SEM characterizations verify the maintenance of its structural integrity and morphological stability. Moreover, the degradation capability of the catalysts in complex systems was also validated, as shown in Supplementary Figure 7. The findings indicate that the WS2@L-His/Ag catalyst demonstrates excellent degradation performance even in mixed dye systems.
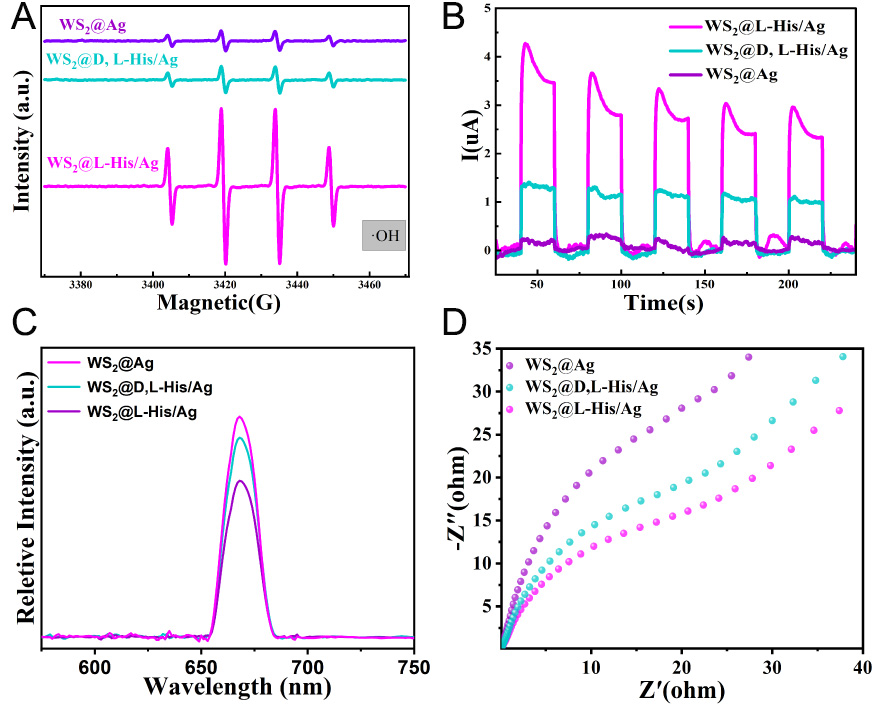
To investigate the photocatalytic reaction process and the regulatory mechanism of chiral molecules within the reaction system, we utilized EPR spectroscopy to detect the formation of active free radicals during degradation reactions. In these experiments, dimethyl pyridine N-oxide (DMPO) served as the spin trapping agent. Figure 6A illustrates the concentration of hydroxyl radicals in various systems after 5 min of the photothermal catalytic reaction. Notably, the concentration of hydroxyl radicals in each system aligns with the trend observed in Figure 5A: WS2@L-His/Ag > WS2@D,L-His/Ag > WS2@Ag. This consistency indicates that the incorporation of chiral molecules enhances the degradation performance by promoting the generation of hydroxyl radicals from hydrogen peroxide. Hydrogen peroxide is capable of producing highly reactive ·OH radicals in situ, which are pivotal for the oxidation of organic pollutants. The concentration of these hydroxyl radicals is a key determinant in improving degradation efficiency. The primary reactions responsible for the generation of ·OH radicals in the photocatalytic process are as follows:
Figure 6. (A) EPR spectra based on the H2O2 process, (B) Photocurrent responses of WS2@L-His/Ag, WS2@D,L-His/Ag, and WS2@Ag, (C) PL spectra of WS2@L-His/Ag, WS2@D,L-His/Ag, and WS2@Ag, (D) EIS spectra of WS2@L-His/Ag, WS2@D,L-His/Ag, and
To elucidate the factors contributing to the formation of more ·OH radicals, two primary reasons were identified: (1) an extended spectral range of light participating in photocatalytic reactions, and (2) enhanced separation and participation of electrons and holes in the reaction, which significantly reduces the recombination of electron-hole pairs within the system. To explore the latter aspect further, we measured the photocurrent response of the composites, as shown in Figure 6B. Notably, the incorporation of chiral molecules led to a marked increase in photocurrent density. Specifically, the L-His/Ag-loaded sample demonstrated the highest current density during the photoelectric reaction, consistent with the observed trend in photocatalytic degradation efficiency. This result indicates that the presence of chiral molecules enhances the involvement of electrons and holes in the catalytic process, likely due to the chiral-induced spin selectivity (CISS) effect. The spin-polarized electrons generated by this mechanism effectively suppress the recombination of electron-hole pairs, thereby promoting the generation of ·OH radicals and improving the overall catalytic performance.
To assess the carrier recombination efficiency by monitoring the hole recombination process, photoluminescence (PL) characterization was conducted on WS2@D,L-His/Ag, WS2@L-His/Ag, and
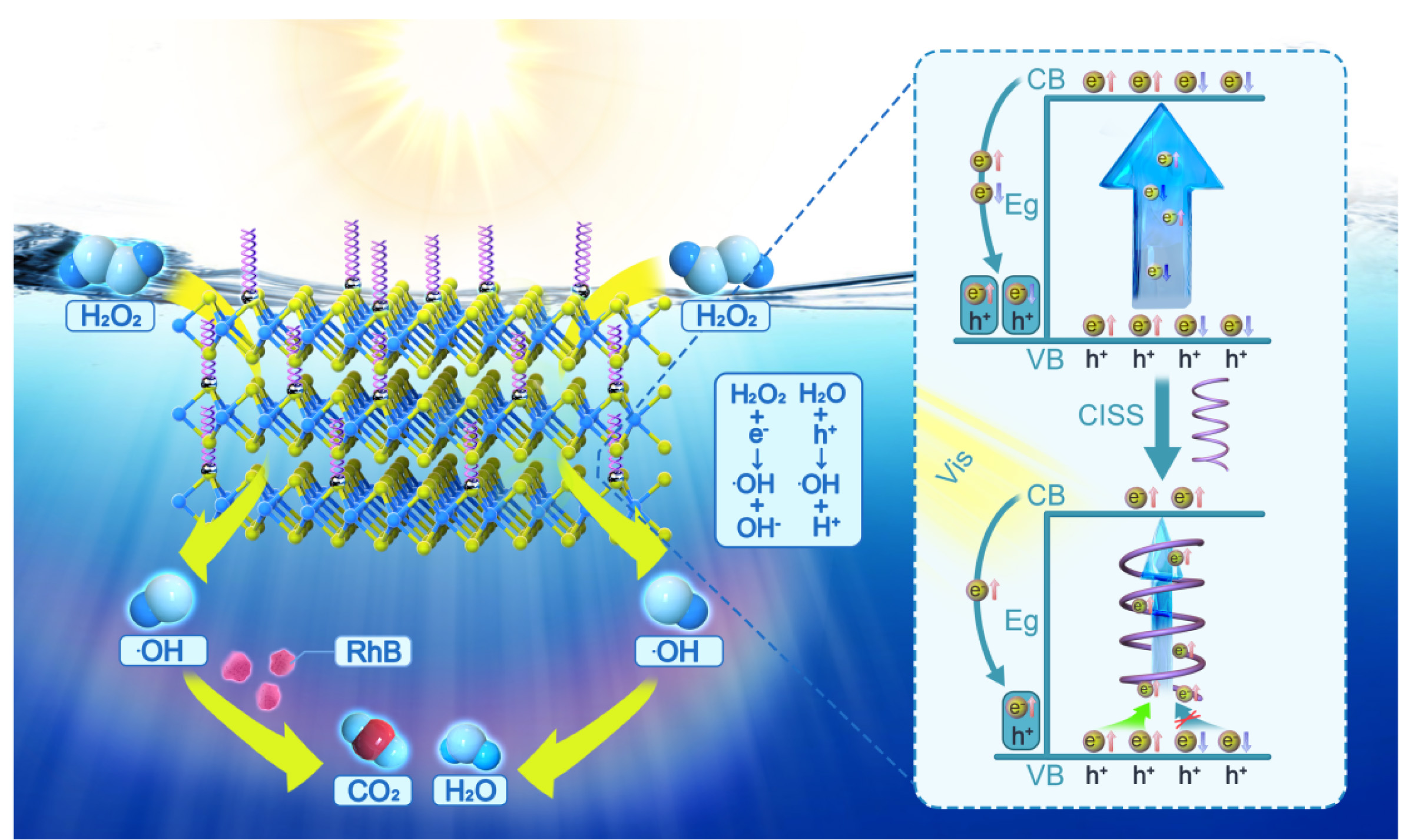
Figure 7 illustrates the mechanistic framework of the chiral-induced spin selectivity (CISS) effect that governs the photodegradation kinetics of organic pollutants. The proposed model elucidates a critical relationship between the spin polarization orientation of photogenerated holes and their spatial spin polarization gradients, where enhanced spatial spin polarization promotes coherent alignment of hole spin polarization vectors. In the WS2@L-His/Ag heterostructure with pronounced spatial spin polarization, spin-polarized holes exhibit spin-selective electron scavenging from hydroxide ions (OH-) via spin-momentum locking. Specifically, majority-spin holes (e.g., spin-down) predominantly capture electrons from OH- anions with parallel spin configurations. This spin-filtering mechanism generates hydroxyl radicals (·OH) with aligned spin states (e.g., spin-up), thereby establishing spin coherence within the radical ensemble. The spin alignment of ·OH radicals fundamentally determines their recombination dynamics through considerations of the Pauli exclusion principle. When ·OH radicals possess antiparallel spin configurations, their wavefunction symmetry allows efficient dimerization to form H2O2 via spin-allowed recombination, which depletes the effective concentration of ·OH radicals available for pollutant degradation. Conversely, parallel-spin-aligned ·OH radicals experience strong exchange interaction repulsion, suppressing their bimolecular recombination through spin-blockade effects. The resultant accumulation of spin-polarized ·OH radicals significantly enhances their oxidative capacity.
CONCLUSIONS
We have successfully synthesized and prepared a composite of tungsten sulfide embedded with chiral silver nanoparticles (WS2@L-His/Ag), which demonstrates a significant enhancement in the efficiency of photocatalytic degradation of organic pollutants. The key to this improvement lies in the incorporation of chiral molecules into the composite, which function as selective filters for light-excited transition electrons. These chiral molecules favor specific spin directions, effectively suppressing the recombination of electron-hole pairs. Consequently, the participating electrons facilitate the generation of hydroxyl radicals, leading to an overall improvement in photocatalytic performance. Additionally, the presence of silver nanoparticles on the WS2 surface induces localized surface plasmon resonance effects in the visible region, resulting in elevated temperatures and enhanced mobility of transition electrons, thereby further enhancing catalytic activity. The importance of the chiral-induced spin selectivity effect is highlighted in these findings, as it serves as the primary mechanism for improving the photocatalytic efficiency of the WS2@L-His/Ag composite. These results provide new insights into the design of advanced catalyst systems and offer promising strategies for effective environmental remediation.
DECLARATIONS
Acknowledgments
Thanks for the support from Fore Facilities in the State Key Laboratory of Marine Resource Utilization in South China Sea in this program.
Authors’ contributions
Writing - review & editing, visualization, supervision, resources, project administration: Wang, J.
Methodology, investigation, data curation: Liu, X.; Zheng, H.; Chu, D.; Pu, Y.
Visualization: Wang, D.
Resources: Liu, Z.
Availability of data and materials
The authors confirm that the data supporting the findings of this study are available within the article and its Supplementary Materials.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This research was funded by the National Natural Science Foundation of China (Grant Nos. 52161030).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Wang, H.; Li, X.; Zhao, X.; et al. A review on heterogeneous photocatalysis for environmental remediation: from semiconductors to modification strategies. Chinese. J. Catal. 2022, 43, 178-214.
2. Fujishima, A.; Zhang, X.; Tryk, D. TiO2 photocatalysis and related surface phenomena. Surf. Sci. Rep. 2008, 63, 515-82.
3. Liu, W.; Cao, L.; Cheng, W.; et al. Single-site active cobalt-based photocatalyst with a long carrier lifetime for spontaneous overall water splitting. Angew. Chem. Int. Ed. 2017, 56, 9312-7.
4. Wang, Y.; Vogel, A.; Sachs, M.; et al. Current understanding and challenges of solar-driven hydrogen generation using polymeric photocatalysts. Nat. Energy. 2019, 4, 746-60.
5. Niu, Z.; Yi, S.; Li, C.; et al. Supporting bimetallic sulfide on 3D TiO2 hollow shells to boost photocatalytic activity. Chem. Eng. J. 2020, 390, 124602.
6. Hu, H.; Choi, J. H. Synergistic effects of carbon vacancies in conjunction with phosphorus dopant across bilayer graphene for the enhanced hydrogen evolution reaction. ACS. Omega. 2024, 9, 16592-600.
7. Zhang, J.; Zhao, X.; Chen, L.; et al. Intrinsic mechanisms of morphological engineering and carbon doping for improved photocatalysis of 2D/2D carbon nitride van der waals heterojunction. Energy. Environ. Mater. 2023, 6, e12365.
8. Wang, Z.; Liu, S.; Zhao, X.; et al. Interfacial defect engineering triggered by single atom doping for highly efficient electrocatalytic nitrate reduction to ammonia. ACS. Mater. Lett. 2023, 5, 1018-26.
9. Wan, J.; Feng, C.; Zhang, H.; Wang, Y. Uv-induced stabilization of electron-deficient W single atoms for enhanced NO electroreduction. Chem. Eng. J. 2025, 510, 161833.
10. Fu, C. F.; Li, X.; Yang, J. A rationally designed two-dimensional MoSe2/Ti2CO2 heterojunction for photocatalytic overall water splitting: simultaneously suppressing electron-hole recombination and photocorrosion. Chem. Sci. 2021, 12, 2863-9.
11. Gao, W.; Peng, R.; Yang, Y.; et al. Electron spin polarization-enhanced photoinduced charge separation in ferromagnetic ZnFe2O4. ACS. Energy. Lett. 2021, 6, 2129-37.
12. Pan, L.; Ai, M.; Huang, C.; et al. Manipulating spin polarization of titanium dioxide for efficient photocatalysis. Nat. Commun. 2020, 11, 418.
13. Jnawali, G.; Xiang, Y.; Linser, S. M.; et al. Ultrafast photoinduced band splitting and carrier dynamics in chiral tellurium nanosheets. Nat. Commun. 2020, 11, 3991.
14. Naaman, R.; Paltiel, Y.; Waldeck, D. H. Chiral molecules and the electron spin. Nat. Rev. Chem. 2019, 3, 250-60.
15. Pu, Y.; Wang, T.; Lin, C.; et al. Enhancement of photocatalytic CO2 reduction in BiOBr through chirality-induced electron spin polarization regulation. Chem. Commun. 2025, 61, 2580-3.
16. Bian, Z.; Kato, K.; Ogoshi, T.; et al. Hybrid chiral MoS2 layers for spin-polarized charge transport and spin-dependent electrocatalytic applications. Adv. Sci. 2022, 9, e2201063.
17. Evers, F.; Aharony, A.; Bar-Gill, N.; et al. Theory of chirality induced spin selectivity: progress and challenges. Adv. Mater. 2022, 34, e2106629.
18. Lu, H.; Vardeny, Z. V.; Beard, M. C. Control of light, spin and charge with chiral metal halide semiconductors. Nat. Rev. Chem. 2022, 6, 470-85.
19. Mondal, P. C.; Roy, P.; Kim, D.; Fullerton, E. E.; Cohen, H.; Naaman, R. Photospintronics: magnetic field-controlled photoemission and light-controlled spin transport in hybrid chiral oligopeptide-nanoparticle structures. Nano. Lett. 2016, 16, 2806-11.
20. Mtangi, W.; Kiran, V.; Fontanesi, C.; Naaman, R. Role of the electron spin polarization in water splitting. J. Phys. Chem. Lett. 2015, 6, 4916-22.
21. Mtangi, W.; Tassinari, F.; Vankayala, K.; et al. Control of electrons' spin eliminates hydrogen peroxide formation during water splitting. J. Am. Chem. Soc. 2017, 139, 2794-8.
22. Zhang, W.; Gao, W.; Zhang, X.; Li, Z.; Lu, G. Surface spintronics enhanced photo-catalytic hydrogen evolution: mechanisms, strategies, challenges and future. Appl. Surf. Sci. 2018, 434, 643-68.
23. Ding, W.; Hu, L.; Liu, Q. C.; et al. Structure modulation induced enhancement of microwave absorption in WS2 nanosheets. Appl. Phys. Lett. 2018, 113, 243102.
24. Wu, S.; Cheng, P.; Han, J.; et al. Construction of two-dimensional Ag/WS2 hybrid membranes with self-cleaning ability by photocatalysis for efficient water filtration. J. Membr. Sci. 2022, 641, 119865.
25. Han, A.; Zhou, X.; Wang, X.; et al. One-step synthesis of single-site vanadium substitution in 1T-WS2 monolayers for enhanced hydrogen evolution catalysis. Nat. Commun. 2021, 12, 709.
26. Zhou, Y.; Ye, Q.; Shi, X.; et al. Regulating photocatalytic CO2 reduction selectivity via steering cascade multi-step charge transfer pathways in 1T/2H-WS2/TiO2 heterojuncitons. Chem. Eng. J. 2022, 447, 137485.
27. Pandey, R. P.; Rasool, K.; Madhavan, V. E.; Aïssa, B.; Gogotsi, Y.; Mahmoud, K. A. Ultrahigh-flux and fouling-resistant membranes based on layered silver/MXene (Ti3C2Tx) nanosheets. J. Mater. Chem. A. 2018, 6, 3522-33.
28. Grabchenko, M.; Mamontov, G.; Zaikovskii, V.; La, Parola. V.; Liotta, L.; Vodyankina, O. The role of metal-support interaction in Ag/CeO2 catalysts for CO and soot oxidation. Appl. Catal. B. Environ. 2020, 260, 118148.
29. Mani, N. P.; Sunil, K. S.; Tomy, A. M.; Sathyan, B.; Cyriac, J. Detection and screening of basic amino acids using the luminescence switching of a WS2 nanosheet-Ag2O nanoparticle composite. Sens. Diagn. 2022, 1, 485-95.
30. Fang, S.; Zhang, W.; Sun, K.; Hu, Y. H. Highly efficient thermo-photocatalytic degradation of tetracycline catalyzed by tungsten disulfide under visible light. Environ. Chem. Lett. 2023, 21, 1287-95.
31. Jia, P.; Han, Z.; Chen, J.; et al. Pt@WS2 Mott-Schottky heterojunction boosts light-driven active ion transport for enhanced ionic power harvesting. ACS. Nano. 2024, 18, 35729-37.
32. Kim, S.; Lim, Y. C.; Kim, R. M.; et al. A single chiral nanoparticle induced valley polarization enhancement. Small 2020, 16, e2003005.
33. Chen, P.; Wang, G.; Hao, C.; et al. Peptide-directed synthesis of chiral nano-bipyramids for controllable antibacterial application. Chem. Sci. 2022, 13, 10281-90.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data




















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.