Semaglutide in MASH with F2-F3 fibrosis: a holistic perspective on the ESSENCE phase 3 trial
Abstract
Metabolic dysfunction-associated steatohepatitis (MASH) with significant fibrosis (F2-F3) is a critical stage of metabolic dysfunction-associated steatotic liver disease (MASLD), during which intervention may avert transition to cirrhosis, hepatocellular carcinoma, and liver-related death. The phase 3 ESSENCE trial (NCT04822181) evaluated once-weekly subcutaneous semaglutide 2.4 mg vs. placebo over 240 weeks in adults with biopsy-confirmed MASH and F2-F3 fibrosis. A prespecified interim histologic assessment was performed at week 72 among the first 800 randomized participants. Semaglutide increased MASH resolution without worsening fibrosis and significantly increased the proportion of individuals achieving ≥ 1-stage fibrosis improvement compared with placebo, with a clinically meaningful number needed to treat. These histologic effects coincided with substantial weight reduction (~10%-11% vs. ~2% with placebo) and broad improvements in cardiometabolic risk profiles, including glycemia, blood pressure, lipid levels, and inflammatory biomarkers. Favorable shifts were also observed in non-invasive assessments, including the enhanced liver fibrosis score, vibration-controlled transient elastography, N-terminal type III Collagen Propeptide (PRO-C3), FibroScan-AST (FAST) score, and aminotransferases. Safety outcomes were consistent with the established semaglutide profile in type 2 diabetes and obesity. Here, we critically appraise ESSENCE from clinical and translational perspectives and consider the relative contributions of weight loss vs. potential weight-independent mechanisms. Semaglutide was also situated among emerging latestage therapies, including resmetirom, a thyroid hormone receptor β agonist with demonstrated antifibrotic and antisteatohepatitic activity. Finally, we outline the implications of adopting semaglutide as a disease-modifying agent in MASH with F2-F3 fibrosis, note the study limitations and remaining questions, and argue that combinatorial approaches targeting complementary metabolic and hepatocentric pathways may define the next phase of MASLD management.
Keywords
INTRODUCTION
Metabolic dysfunction-associated steatotic liver disease (MASLD) has become a leading cause of chronic liver disease worldwide, tightly linked to obesity, type 2 diabetes mellitus (T2DM), and other components of the metabolic syndrome[1]. Progression to metabolic dysfunction-associated steatohepatitis (MASH) with significant or advanced fibrosis (F2-F3) marks a threshold at which the risk of cirrhosis, hepatocellular carcinoma (HCC), and liver-related mortality rises sharply, and where targeted pharmacologic intervention is most likely to alter long-term prognosis[2].
Despite the burgeoning global burden, effective disease-modifying therapies for MASH with fibrosis have only recently begun to emerge[3]. Among these, glucagon-like peptide-1 receptor agonists (GLP1RAs) have attracted particular attention for their profound effects on body weight, glycemic control, and cardiovascular outcomes[4,5], which extend beyond metabolic effects[4], alongside emerging evidence of liver-specific benefits[5,6]. The phase 3 ESSENCE trial of semaglutide 2.4 mg once weekly in patients with biopsy-proven MASH and F2-F3 fibrosis represents a pivotal step toward integrating GLP1RA therapy into hepatology practice[7].
This commentary critically evaluates ESSENCE from the perspectives of hepatology research and clinical trial methodology, situates semaglutide within the broader MASLD therapeutic landscape, and explores mechanistic and translational implications for target-organ damage.
TRIAL DESIGN AND STUDY POPULATION
ESSENCE is a global, multicenter, randomized, double-blind, placebo-controlled phase 3 trial enrolling adults with histologically confirmed MASH and F2-F3 fibrosis, defined by central pathologist review according to standard scoring systems[7]. A total of 1,197 patients were randomized in a 2:1 ratio to receive semaglutide 2.4 mg subcutaneously once weekly or placebo for 240 weeks; the currently reported results derive from a prespecified interim analysis at week 72 in the first 800 randomized participants with paired liver biopsies[7].
The design aligns closely with contemporary U.S. Food and Drug Administration and European Medicines Agency guidance for MASH drug development, employing two co-primary histologic endpoints: (1) resolution of steatohepatitis without worsening of fibrosis stage; and (2) improvement in fibrosis by at least one stage without worsening of steatohepatitis.
Randomization was stratified by baseline fibrosis stage (F2 vs. F3) and T2DM status, recognizing the prognostic significance of these factors[1,2]. Baseline characteristics were typical of “metabolic MASH”, with a mean age of approximately 55 years and a mean body mass index (BMI) of approximately 35 kg/m2; more than half of the participants had T2DM, and approximately two-thirds exhibited F3 fibrosis. Non-invasive markers, including enhanced liver fibrosis (ELF) score and vibration-controlled transient elastography (VCTE), indicated a substantial fibrotic burden, consistent with an enriched high-risk population. The exclusion threshold for alcohol consumption was > 30 g/day for men and > 20 g/day for women.
From an external validity standpoint, the ESSENCE population is highly representative of the obese/diabetic MASH phenotype dominating hepatology clinics[1,2]. However, limited representation of “lean” MASH and certain ethnic groups (e.g., Black patients) constrains generalizability to those subpopulations, an issue shared by many MASH trials[1].
HISTOLOGIC EFFICACY AND CLINICAL INTERPRETABILITY
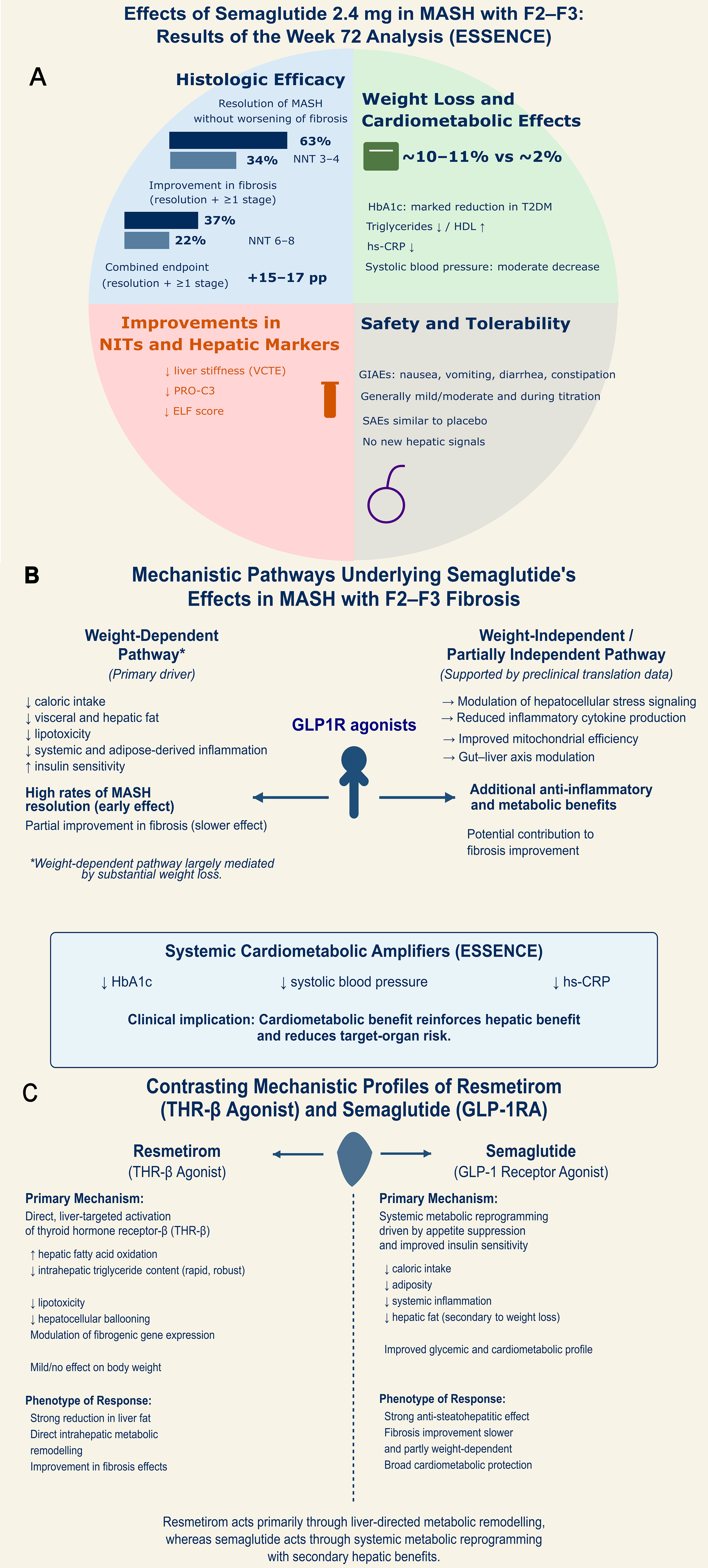
At week 72, semaglutide demonstrated robust histologic efficacy. In the intention-to-treat biopsy population, approximately two-thirds of the patients in the semaglutide arm achieved resolution of steatohepatitis without worsening of fibrosis compared with about one-third in the placebo arm. This corresponded to an absolute difference of ~25-30 percentage points and an estimated number needed to treat (NNT) of roughly 3-4[7]. Improvement in fibrosis by ≥ 1 stage, without worsening of steatohepatitis, occurred in approximately one-third of semaglutide-treated patients, compared with approximately one-fifth of those receiving placebo, corresponding to an absolute difference of 12-15 percentage points and an NNT of ~6-8. A sizeable fraction of patients receiving semaglutide also achieved the combined endpoint of MASH resolution plus ≥ 1-stage fibrosis improvement, with an absolute benefit of approximately 15-17 percentage points over placebo[7].
These results are striking when placed in context. Phase 2 data with subcutaneous semaglutide 0.4 mg daily in noncirrhotic MASH (F1-F3) demonstrated high rates of MASH resolution but less pronounced effects on fibrosis[8]. ESSENCE now extends and strengthens these findings by using a higher, obesity-approved dose of 2.4 mg weekly in a fibrosis-enriched F2-F3 population, showing both strong anti-steatohepatitic activity and significant antifibrotic effects over 72 weeks[7,8]. Observational and interventional data have established that improvement in fibrosis stage and resolution of steatohepatitis are associated with lower risks of hepatic decompensation, HCC, and mortality[1,2,9]. While these endpoints remain surrogates, the magnitude and consistency of the ESSENCE histologic signal strongly support semaglutide as a disease-modifying agent in this context.
Nonetheless, caution is warranted. Histologic changes over 72 weeks, although impressive, must ultimately be linked to long-term clinical outcomes. The ongoing 240-week follow-up in ESSENCE will be critical for determining whether semaglutide-driven histologic improvements translate into reductions in clinically meaningful events.
WEIGHT LOSS, CARDIOMETABOLIC EFFECTS, AND NON-INVASIVE MARKERS
Semaglutide 2.4 mg weekly produced substantial and sustained weight loss in ESSENCE, with mean reductions of approximately 10%-11% of baseline body weight vs. ~2% with placebo at 72 weeks, a pattern consistent with prior obesity trials such as STEP-1 (NCT03548935)[10]. This degree of weight loss alone would be expected to substantially improve hepatic steatosis, necroinflammation, and metabolic stress, given robust evidence linking ≥ 10% weight reduction to MASH resolution and fibrosis regression[11].
Beyond weight loss, semaglutide improved a broad range of cardiometabolic parameters in ESSENCE[7], consistent with prior data in T2DM and obesity[10,12]. These included: (1) significant reductions in glycated hemoglobin (HbA1c), particularly in patients with T2DM; (2) decreases in systolic blood pressure; (3) improvements in lipid profile (lower triglycerides, higher high-density lipoprotein [HDL] cholesterol); and (4) reductions in high-sensitivity C-reactive protein and other inflammatory markers.
Given that cardiovascular disease is a leading cause of death in MASLD/MASH[1,2], these parallel cardiometabolic benefits confer additional target-organ protection, further reinforcing the appeal of semaglutide in this high-risk population.
Non-invasive tests (NITs) in ESSENCE showed changes concordant with histologic improvements. Semaglutide reduced ELF score and VCTE-measured liver stiffness, and improved N-terminal type III collagen propeptide (PRO-C3) and the FibroScan-AST (FAST) score, markers associated with fibrotic activity and risk stratification in MASLD[7,13,14]. Liver enzymes, including alanine aminotransferase (ALT), aspartate aminotransferase (AST), and gamma-glutamyl transferase (GGT), also improved, indicating decreased hepatocellular injury and inflammation[7]. This triangulation between biopsy, NITs, and systemic cardiometabolic measures helps validate the biological plausibility of the treatment effect and supports the use of NITs in future biopsy-sparing strategies.
SAFETY AND TOLERABILITY
The safety profile of semaglutide in ESSENCE was consistent with its known profile from T2DM and obesity trials[10,12]. Gastrointestinal adverse events - principally nausea, vomiting, diarrhea, and constipation - were more frequent in the semaglutide arm but were generally mild to moderate and occurred primarily during dose escalation[7]. Notably, serious adverse events and discontinuations due to adverse events were similar between the semaglutide and placebo groups, and no new liver-specific safety signals emerged[7].
GLP1RAs have been associated with gallbladder-related events and rare pancreatitis in other settings[10,12], underscoring the need for vigilance in real-world hepatology practice, particularly among patients with advanced fibrosis and multiple comorbidities. However, the absence of hepatic decompensation or overt hepatotoxicity in the ESSENCE trial is reassuring and supports the use of semaglutide in patients with F2-F3 fibrosis, with appropriate monitoring. The ESSENCE findings are summarized in Figure 1A.
Figure 1. Mechanistic Basis and Clinical Efficacy of Semaglutide in MASH. (A) Clinical Efficacy, Safety, and Biomarker Response in the ESSENCE Trial (Week 72). The quadrant illustrates superior histologic efficacy in MASH resolution and fibrosis improvement compared with placebo, with favorable NNT values. Significant improvements in NITs, including ELF score, liver stiffness (VCTE), and PRO-C3; (B) Mechanistic Pathways Underlying Semaglutide’s Effects in MASH with F2-F3 Fibrosis. This panel illustrates a dual-pathway model for semaglutide hepatic benefits and reduction of overall target-organ risk in patients with cardiometabolic compromise: a weight-dependent pathway (primary driver), and a weight-independent or partially independent pathway, supported by reductions in HbA1c, systolic blood pressure, and hs-CRP; (C) Contrasting Mechanistic Profiles of Resmetirom and Semaglutide. Comparative overview of the primary pharmacological drivers for the two leading therapeutic classes in MASH: Resmetirom (a THR-β agonist) and semaglutide (a GLP1R agonist). MASH: Metabolic dysfunction-associated steatohepatitis; NNT: number needed to treat; NITs: non-invasive tests; ELF: enhanced liver fibrosis; VCTE: vibration-controlled transient elastography; PRO-C3: procollagen type III N-terminal peptide; HbA1c: glycated hemoglobin; hs-CRP: high-sensitivity C-reactive protein; THR-β: thyroid hormone receptor beta; GLP1R: glucagon-like peptide-1 receptor; F2-F3: fibrosis stage 2-3.
MECHANISTIC CONSIDERATIONS: WEIGHT-MEDIATED VS. WEIGHT-INDEPENDENT EFFECTS
A central mechanistic question is the extent to which semaglutide’s hepatic benefits are mediated by weight loss vs. weight-independent pathways [Figure 1B]. GLP1RAs reduce caloric intake and body weight, improve insulin sensitivity, and favorably modulate adipose tissue and systemic inflammation[10,12]. These changes are inherently hepatoprotective in MASLD/MASH[11]. At the same time, preclinical and translational studies suggest potential direct or partially weight-independent effects of GLP1RAs on hepatocellular metabolism, inflammation, and the gut-liver axis[4].
The available ESSENCE publication does not include individual participant data (IPD), multivariable models that adjust for weight loss, or mediation analyses to estimate effects independently of body weight or other metabolic variables[7]. Therefore, it remains unclear to what extent the robust histologic response is attributable specifically to semaglutide’s direct hepatic actions, rather than to its marked weight loss effect. It is noteworthy that semaglutide has no proven direct biological effect on the liver, as glucagon-like peptide-1 (GLP-1) receptors are not expressed in hepatic tissue (Protein Atlas, https://www.proteinatlas.org/ENSG00000112164-GLP1R)[15].
Prior phase 2 work in MASH (F1-F3) with lower-dose semaglutide demonstrated substantial MASH resolution but a more modest impact on fibrosis, suggesting that fibrosis regression may lag or be less tightly coupled to early weight loss[8]. Furthermore, in a trial involving patients with MASH-related compensated cirrhosis (F4), semaglutide improved weight and metabolic parameters but did not improve fibrosis stage or key hepatic clinical outcomes over the study period[16]. These data highlight the limited reversibility of established cirrhosis and suggest that metabolic improvement alone may be insufficient to remodel advanced fibrosis within typical trial durations.
Collectively, current evidence supports a model [Figure 1B] in which: (1) a large fraction of early MASH resolution in ESSENCE is likely explained by significant weight loss and associated metabolic reprogramming; and (2) fibrosis improvement may require both time and more targeted modulation of fibrogenic pathways and may be less directly proportional to short-term weight loss, particularly in advanced fibrosis.
Definitive clarification will require IPD-level modeling and causal mediation analyses, ideally integrating clinical, histologic, NIT, and omics data. Advanced proteomics reveals that semaglutide can normalize circulating protein profiles linked to MASH. Statistical modeling indicates that over one-third of these protein shifts drive disease resolution through biological pathways independent of weight reduction[17]. Regardless of the underlying mechanism and despite ongoing controversy about GLP1R in liver tissue, the clustering analysis using a cell type classifier (DeepCellSeek; deepcellseek.kiz.ac.cn)[18] suggested that circulating proteins significantly downregulated or upregulated by semaglutide may derive not only from pancreatic acinar cells but also hepatocytes, hepatic stellate cells, and Kupffer cells. Interestingly, similar proteomic profile changes were observed in STEP 1 and STEP 2, phase 3 trials evaluating the effects of semaglutide in participants with overweight/obesity with or without diabetes[19]. In the same cohort, semaglutide improved MASH-related SomaSignal profiles, which are aptamer-based proteomic profiles associated with MASH components[6].
COMPARISON WITH RESMETIROM AND THE CASE FOR COMBINATION STRATEGIES
Resmetirom, an orally active, liver-directed thyroid hormone receptor-β (THR-β) agonist, has recently shown positive phase 3 trial results in patients with MASH and F1-F3 fibrosis, improving both MASH resolution and fibrosis compared with placebo. This led to conditional regulatory approval in some jurisdictions[20]. Mechanistically, resmetirom enhances hepatic lipid oxidation, rapidly reduces liver fat content, and modulates intrahepatic metabolic and fibrogenic pathways, with relatively modest effects on body weight[20,21].
Semaglutide and resmetirom therefore occupy complementary mechanistic niches alongside other drugs, such as acetyl coenzyme A (acetyl-CoA) carboxylase inhibitors, peroxisome proliferator-activated receptor agonists, and fibroblast growth factor-21 analogs already in clinical trials[22]. Semaglutide acts primarily through the gut-pancreas-brain axis, driving substantial weight loss and systemic metabolic improvements, with secondary yet potent hepatic benefits. In contrast, resmetirom targets hepatic THR-β directly, improving intrahepatic lipid handling and fibrogenesis, with minor effects on systemic weight [Figure 1C].
From a systems biology and target-organ damage perspective, these findings provide a strong rationale for combination therapy. A GLP1RA, such as semaglutide, could provide potent upstream metabolic unloading (weight, insulin resistance, adipose inflammation), whereas resmetirom delivers focused hepatocentric modulation of intracellular lipid and fibrogenic pathways. Carefully designed trials assessing semaglutide-resmetirom combinations, integrated histologic, NIT, and molecular endpoints, could help define a new paradigm for multi-targeted metabolic and hepatic risk reduction in MASLD. It will be paramount to determine whether the combination exerts synergistic effects in reducing stage F2-F3 liver fibrosis and, more importantly, in promoting reversal of F4 liver fibrosis.
CLINICAL IMPLICATIONS AND FUTURE DIRECTIONS
For clinicians managing patients with MASH and F2-F3 fibrosis, ESSENCE positions semaglutide 2.4 mg weekly as an attractive option for patients with obesity and/or T2DM and high cardiometabolic risk[7]. In this prevalent phenotype, semaglutide may offer:
· High probability of MASH resolution within 72 weeks.
· Clinically meaningful likelihood of ≥ 1-stage fibrosis improvement.
· Substantial and sustained weight loss.
· Broad cardiometabolic risk factor modification.
· A safety profile consistent with extensive experience in diabetes and obesity.
Several gaps remain. Long-term data linking semaglutide-induced histologic changes to hard hepatic and cardiovascular outcomes are still needed. The efficacy and safety of semaglutide in lean or non-obese patients with MASLD/MASH and in underrepresented ethnic groups require further study. In F4 cirrhosis, available data suggest limited antifibrotic impact over typical trial horizons, although systemic benefits may still justify use in selected patients[16].
Beyond these clinical questions, ESSENCE invites deeper mechanistic inquiry, enabling formal mediation analyses to disentangle weight-dependent from weight-independent pathways. Systems-level analyses integrating transcriptomics, metabolomics, and microbiome data may complement these aims[23]. Such work would illuminate how interventions such as semaglutide rewire metabolic networks across the liver and extrahepatic organs. In this sense, omics-based analyses, particularly proteomic profiling, may help identify patient subgroups more likely to respond[24].
In conclusion, ESSENCE establishes semaglutide as a cornerstone therapy for MASH with F2-F3 fibrosis in metabolically high-risk patients, providing robust histologic improvement, substantial weight loss, and broad cardiometabolic benefit. The next frontier will be to incorporate semaglutide into rational combination strategies and to define, in mechanistic detail, how GLP1RAs modulate the complex interplay between systemic metabolism and target-organ damage in MASLD.
DECLARATIONS
Authors’ contributions
Made substantial contributions to the conception and design of the study and performed data analysis and interpretation: Pirola CJ, Sookoian S
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
None.
Conflicts of interest
Both authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Dulai PS, Singh S, Patel J, et al. Increased risk of mortality by fibrosis stage in nonalcoholic fatty liver disease: systematic review and meta-analysis. Hepatology. 2017;65:1557-65.
2. Younossi ZM, Koenig AB, Abdelatif D, Fazel Y, Henry L, Wymer M. Global epidemiology of nonalcoholic fatty liver disease-Meta-analytic assessment of prevalence, incidence, and outcomes. Hepatology. 2016;64:73-84.
3. Do A, Zahrawi F, Mehal WZ. Therapeutic landscape of metabolic dysfunction-associated steatohepatitis (MASH). Nat Rev Drug Discov. 2025;24:171-89.
4. Yabut JM, Drucker DJ. Glucagon-like peptide-1 receptor-based therapeutics for metabolic liver disease. Endocr Rev. 2023;44:14-32.
5. Banerjee M, Pal R, Pal S. Histological efficacy of anti-diabetic agents in MASH and the mediating role of weight loss: a network meta-analysis. Diabetes Obes Metab. 2026;28:287-95.
6. Schattenberg JM, Grønbæk H, Kliers I, et al. Proteomic signatures reflect effects of semaglutide treatment for MASH. JHEP Rep. 2025;7:101521.
7. Sanyal AJ, Newsome PN, Kliers I, et al.; ESSENCE Study Group. Phase 3 trial of semaglutide in metabolic dysfunction-associated steatohepatitis. N Engl J Med. 2025;392:2089-99.
8. Newsome PN, Buchholtz K, Cusi K, et al.; NN9931-4296 Investigators. A placebo-controlled trial of subcutaneous semaglutide in nonalcoholic steatohepatitis. N Engl J Med. 2021;384:1113-24.
9. Sanyal AJ, Friedman SL, McCullough AJ, Dimick-Santos L; American Association for the Study of Liver Diseases, United States Food and Drug Administration. Challenges and opportunities in drug and biomarker development for nonalcoholic steatohepatitis: findings and recommendations from an American Association for the Study of Liver Diseases-U.S. Food and Drug Administration Joint Workshop. Hepatology. 2015;61:1392-405.
10. Wilding JPH, Batterham RL, Calanna S, et al.; STEP 1 Study Group. Once-weekly semaglutide in adults with overweight or obesity. N Engl J Med. 2021;384:989-1002.
11. Vilar-Gomez E, Martinez-Perez Y, Calzadilla-Bertot L, et al. Weight loss through lifestyle modification significantly reduces features of nonalcoholic steatohepatitis. Gastroenterology. 2015;149:367-78.e5.
12. Marso SP, Bain SC, Consoli A, et al.; SUSTAIN-6 Investigators. Semaglutide and cardiovascular outcomes in patients with type 2 diabetes. N Engl J Med. 2016;375:1834-44.
13. Newsome PN, Sasso M, Deeks JJ, et al. FibroScan-AST (FAST) score for the non-invasive identification of patients with non-alcoholic steatohepatitis with significant activity and fibrosis: a prospective derivation and global validation study. Lancet Gastroenterol Hepatol. 2020;5:362-73.
14. Rosenberg WM, Voelker M, Thiel R, et al.; European Liver Fibrosis Group. Serum markers detect the presence of liver fibrosis: a cohort study. Gastroenterology. 2004;127:1704-13.
15. Petta S, Kim K, Targher G, et al. Focus on semaglutide 2.4 mg/week for the treatment of metabolic dysfunction-associated steatohepatitis. Liver Int. 2025;45:e70407.
16. Loomba R, Abdelmalek MF, Armstrong MJ, et al.; NN9931-4492 investigators. Semaglutide 2·4 mg once weekly in patients with non-alcoholic steatohepatitis-related cirrhosis: a randomised, placebo-controlled phase 2 trial. Lancet Gastroenterol Hepatol. 2023;8:511-22.
17. Jara M, Norlin J, Kjær MS, et al. Modulation of metabolic, inflammatory and fibrotic pathways by semaglutide in metabolic dysfunction-associated steatohepatitis. Nat Med. 2025;31:3128-40.
18. Xiao T, Hua D, Wang Y, Lu X, Zhang C. Benchmarking large language models for cell typing in single-cell RNA-Seq. Brief Bioinform. 2025;26:bbaf677.
19. Maretty L, Gill D, Simonsen L, et al. Proteomic changes upon treatment with semaglutide in individuals with obesity. Nat Med. 2025;31:267-77.
20. Harrison SA, Bedossa P, Guy CD, et al.; MAESTRO-NASH Investigators. A phase 3, randomized, controlled trial of resmetirom in NASH with liver fibrosis. N Engl J Med. 2024;390:497-509.
21. Harrison SA, Bashir MR, Guy CD, et al. Resmetirom (MGL-3196) for the treatment of non-alcoholic steatohepatitis: a multicentre, randomised, double-blind, placebo-controlled, phase 2 trial. Lancet. 2019;394:2012-24.
22. Zhou XD, Fan QY, Byrne CD, et al. Combination therapies for metabolic dysfunction-associated steatohepatitis: challenges and opportunities. Gut. 2026;75:815-25.
23. Pirola CJ, Sookoian S. Panomics in metabolic dysfunction-associated steatotic liver disease: unravelling the drivers of disease heterogeneity. Clin Mol Hepatol. 2026;32:156-69.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data


















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.