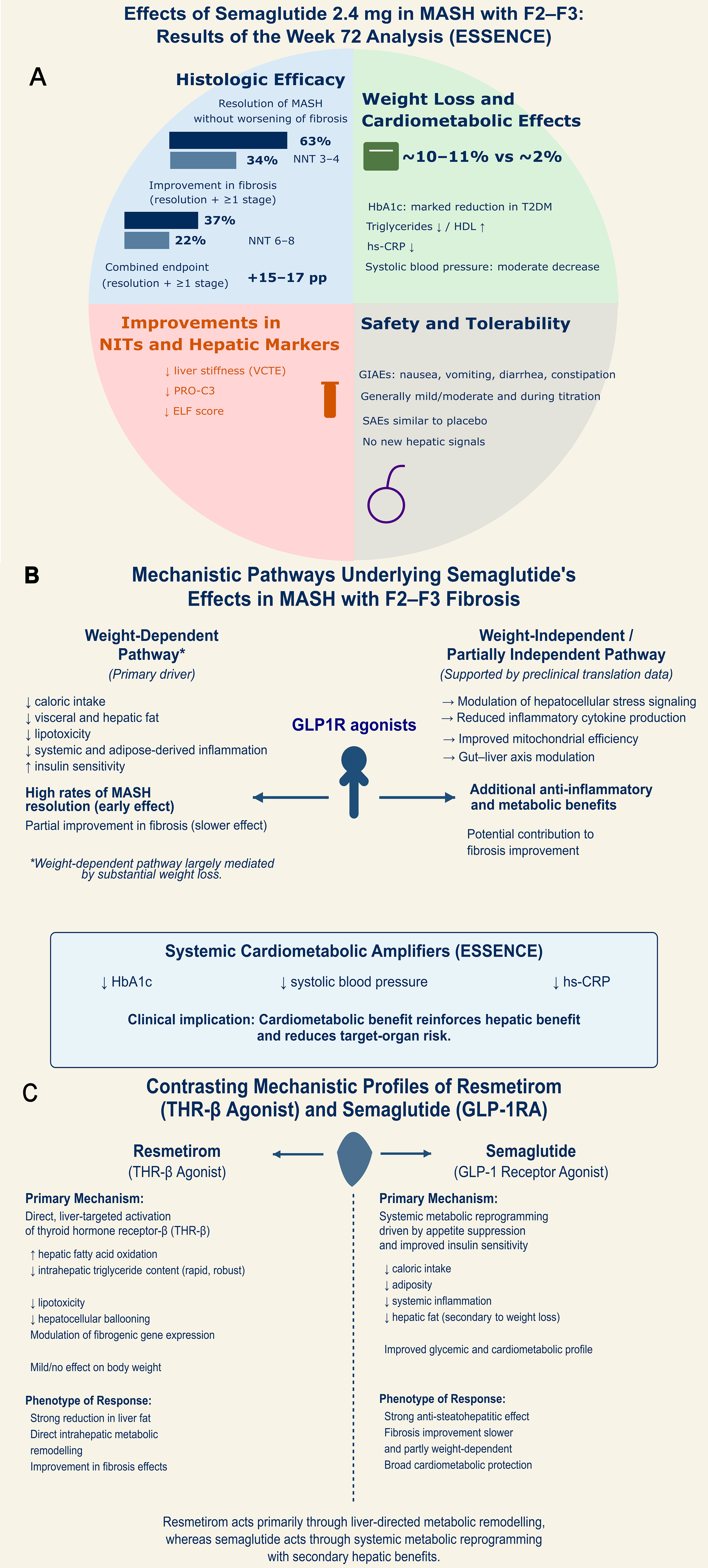
fig1
Figure 1. Mechanistic Basis and Clinical Efficacy of Semaglutide in MASH. (A) Clinical Efficacy, Safety, and Biomarker Response in the ESSENCE Trial (Week 72). The quadrant illustrates superior histologic efficacy in MASH resolution and fibrosis improvement compared with placebo, with favorable NNT values. Significant improvements in NITs, including ELF score, liver stiffness (VCTE), and PRO-C3; (B) Mechanistic Pathways Underlying Semaglutide’s Effects in MASH with F2-F3 Fibrosis. This panel illustrates a dual-pathway model for semaglutide hepatic benefits and reduction of overall target-organ risk in patients with cardiometabolic compromise: a weight-dependent pathway (primary driver), and a weight-independent or partially independent pathway, supported by reductions in HbA1c, systolic blood pressure, and hs-CRP; (C) Contrasting Mechanistic Profiles of Resmetirom and Semaglutide. Comparative overview of the primary pharmacological drivers for the two leading therapeutic classes in MASH: Resmetirom (a THR-β agonist) and semaglutide (a GLP1R agonist). MASH: Metabolic dysfunction-associated steatohepatitis; NNT: number needed to treat; NITs: non-invasive tests; ELF: enhanced liver fibrosis; VCTE: vibration-controlled transient elastography; PRO-C3: procollagen type III N-terminal peptide; HbA1c: glycated hemoglobin; hs-CRP: high-sensitivity C-reactive protein; THR-β: thyroid hormone receptor beta; GLP1R: glucagon-like peptide-1 receptor; F2-F3: fibrosis stage 2-3.