Enhanced DC-bias stability and reliability in BaTiO3 Ceramics via B-site Ca doping induced long-range order disruption
Abstract
With the ongoing miniaturization of multilayer ceramic capacitors (MLCCs), there is an increasing demand for dielectric materials that simultaneously exhibit high dielectric constant, excellent DC-bias stability, and high reliability. To address this challenge, B-site Ca doping was employed to regulate the polar structure of
Keywords
INTRODUCTION
Multilayer ceramic capacitors (MLCCs) are essential passive components in modern energy systems, widely employed in 5G/6G communications, smart devices, and new energy vehicles, with their performance largely dependent on the properties of dielectric materials. BaTiO3 (BT) has emerged as the preferred dielectric material system due to its high dielectric constant and low dielectric loss in high-capacitance MLCCs. Traditional research has predominantly focused on broadening its temperature stability. However, the ongoing trend toward miniaturization and thinner dielectric layers in MLCCs has significantly increased the operating electric field across the dielectric, placing stringent demands on the material: not only should a high dielectric constant be maintained, but excellent DC-bias stability and long-term reliability should also be ensured.
In this context, Ca-doped BT ceramics have attracted increasing attention owing to their potential in tuning dielectric performance. To date, research has mainly focused on A-site Ca-doping, which has been reported to improve Curie temperature[1,2], increase insulation resistance[3], and enhance reliability[4,5] compared to undoped BT. Regarding reliability enhancement, Sakabe et al. demonstrated that in (Ba1-xCax)mTiO3 (m > 1) systems, the lattice contraction induced by the smaller A-site Ca2+ ions, coupled with local lattice distortion at the Ti site caused by B-site Ca2+ occupation, significantly suppresses oxygen vacancy migration and enhances lifetime by more than tenfold[6]. Moreover, improved insulation resistance under reducing atmospheres has also been linked to Ca2+ substituting Ti4+, which reduces electronic conductivity and promotes oxide ion conduction[7]. Interestingly, even in Ba0.97Ca0.03TiO3-based ceramics, trace amounts of Ca occupying B-sites have been detected and associated with the formation of defect dipoles
The DC-bias stability is another important property for MLCC applications. BT ceramics exhibit a drop in dielectric constant with an increase in electric fields, primarily due to the suppression of anharmonic vibrations of Ti4+ within the Ti-O octahedra, as well as inhibited domain wall motion and polarization switching[10,11]. While these mechanisms are key to high spontaneous polarization under zero field, they also lead to poor DC-bias performance. To resolve the trade-off between high polarization and DC-bias stability, various approaches have been investigated, including tuning the tetragonality (c/a ratio) to reduce anharmonicity[12], decreasing the volume fraction of core in core-shell structures[13], modifying shell layers via doping[14], refining grain size[15], and introducing amorphous phases[16] to downscale domain size. However, these modulation strategies are often accompanied by a decrease in dielectric constant. Relaxor ferroelectrics, composed of micro-domains and/or polar nanoregions (PNRs), have emerged as promising candidates for balancing dielectric constant and DC-bias stability, offering clear advantages over conventional ferroelectric ceramics[17]. In BT-based systems, B-site doping has been shown to disrupt
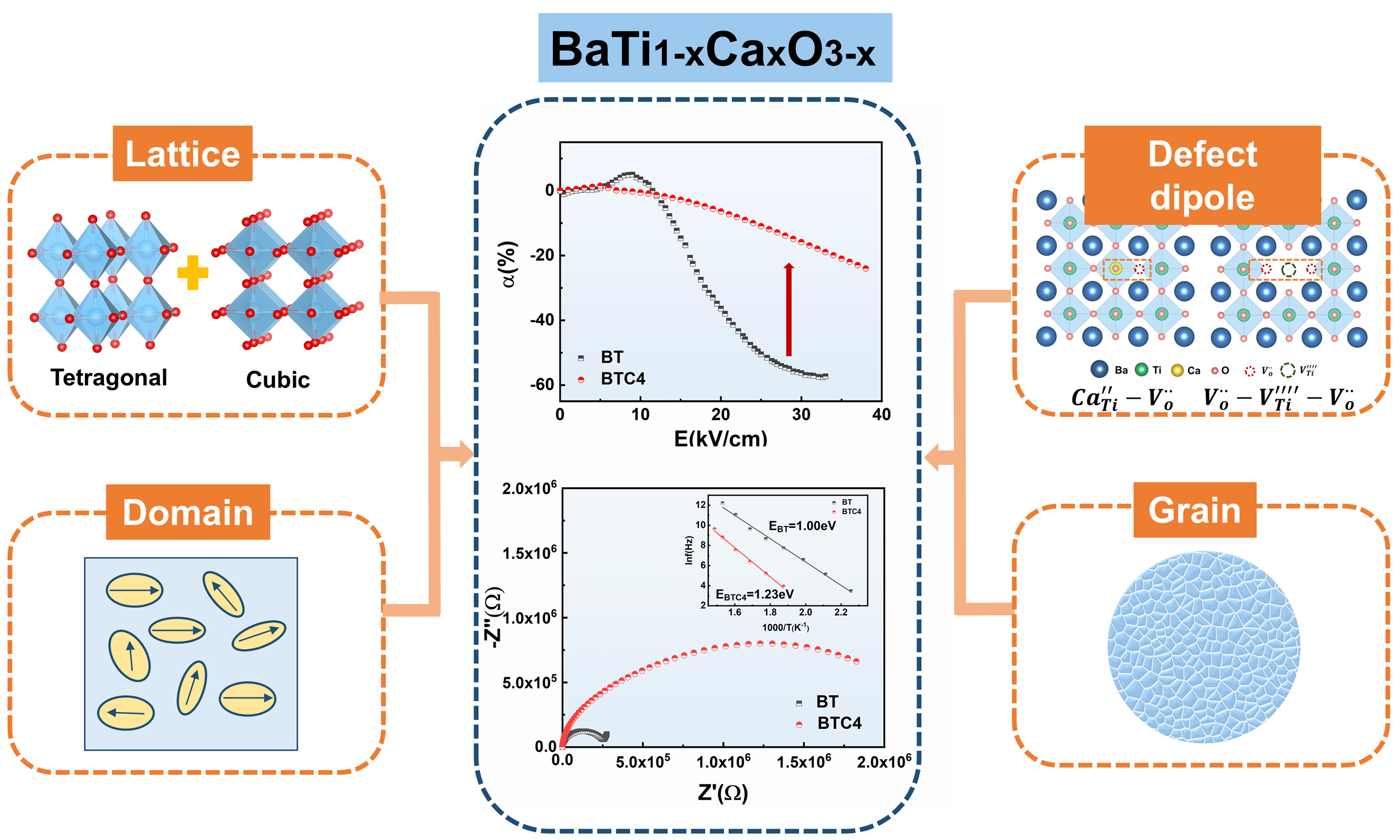
In this work, we systematically investigate the effects of B-site Ca doping on the structure and performance of BT-based ceramics, as shown in Figure 1. The results demonstrate that Ca substitution at the B-site facilitates the formation of tetragonal PNRs embedded within a non-polar cubic matrix, achieving a balance of high dielectric constant and excellent DC-bias stability. The B-site Ca doping induces local structural distortions and defect dipoles, which effectively trap oxygen vacancies and suppress their migration, improving reliability and strong non-reducibility. By establishing a direct correlation between ionic occupation and dielectric behavior, this study provides fundamental insights into the structure-property relationships of BT-based ceramics. The results suggest that B-site Ca-doped BT ceramics show significant potential for high-capacitance, high-reliability MLCC applications.
MATERIALS AND METHODS
Sample preparation
The Ba100-xCaxTiO3 (BCT) and BaTi100-xCaxO3 (BTC) (x = 2, 4, 6) ceramics were fabricated by a conventional solid-state reaction method and were named as BCxT and BTCx, respectively. Pure BT ceramic was also prepared for comparison. The raw materials, BaCO3 (99.8%, Aladdin), TiO2 (99.0%, Aladdin), and CaCO3 (99.99%, Aladdin) were weighed according to the design stoichiometric composition and ball-milled with zirconia balls and ethanol for 6 h. The powders were dried and then calcined at 1,100 °C (BCT) and 1,200 °C (BTC) in air for 2 h [Supplementary Figure 1], followed by a second ball-milling process for 6 h in ethanol. After drying, 6 wt% PVA was added as a binder, and the powders were granulated and then uniaxially pressed into 13 mm pellets at 200 MPa. All samples were sintered at 1,325-1,375 °C for 2 h in either air or
Structural characterization
Crystalline phase formation was identified via X-ray powder diffraction (XRD; D8 Advance, Bruker) with a Cu Kα source at room temperature. High-resolution synchrotron powder XRD and synchrotron X-ray pair distribution function (PDF) patterns were collected at BL44B2 beamline of SPring-8 (Hyogo, Japan) at room temperature, employing a focused monochromatic beam at 50.00 keV [λ = 0.490030(1) Å], which was precisely calibrated through Le Bail refinement of LaB6 standard data. Synchrotron diffraction data were corrected for anomalous scattering effects before structural analysis, and Rietveld refinements were performed using the JANA2006 crystallographic software suite. The X-ray absorption spectra (XAS), including X-ray absorption near-edge structure (XANES) and extended X-ray absorption fine structure (EXAFS) at Ca K-edge, were collected at the Singapore Synchrotron Light Source center. The Ba K-edge EXAFS data were measured at room temperature in transmission mode using Beamline NW10A of Photon Factory Advanced Ring (PF-AR), High Energy Accelerator Research Organization (KEK). X-rays were varied using the fixed-exit Si (111) double-crystal monochromator, with higher-order harmonic X-rays removed using a rhodium-coated harmonic-rejection mirror. A bent cylindrical mirror was used to focus the incident light. All the XANES spectra were measured in the transmission mode. All the spectra were normalized using the Athena software[19]. Thermal etching was performed on the cross-section of ceramic samples for grain size analysis using scanning electron microscopy (SEM; SU8220, Hitachi). Transmission electron microscope (TEM) samples were prepared using focused ion beam (FIB) milling (Helios Nanolab G3 UC, FEI) for microstructure observation. Atomic-resolution high-angle annular dark-field scanning transmission electron microscopy (HADDF-STEM) images were obtained using a
Electrical properties characterization
Temperature-dependent dielectric properties were measured using an impedance analyzer (E4990a, Keysight) with a temperature-controlling system (DMS-2000, Partulab) over the temperature range of
where εr(0) and εr(E) are the dielectric constant under zero bias field and applied bias field E, respectively. Polarization electric field (P-E) loops were measured at 1 kHz using a ferroelectric tester (TF Analyzer 2000E, aixACCT). The DC breakdown voltage was tested using a high-voltage breakdown tester
RESULTS AND DISCUSSION
Electrical performance of BTC ceramics
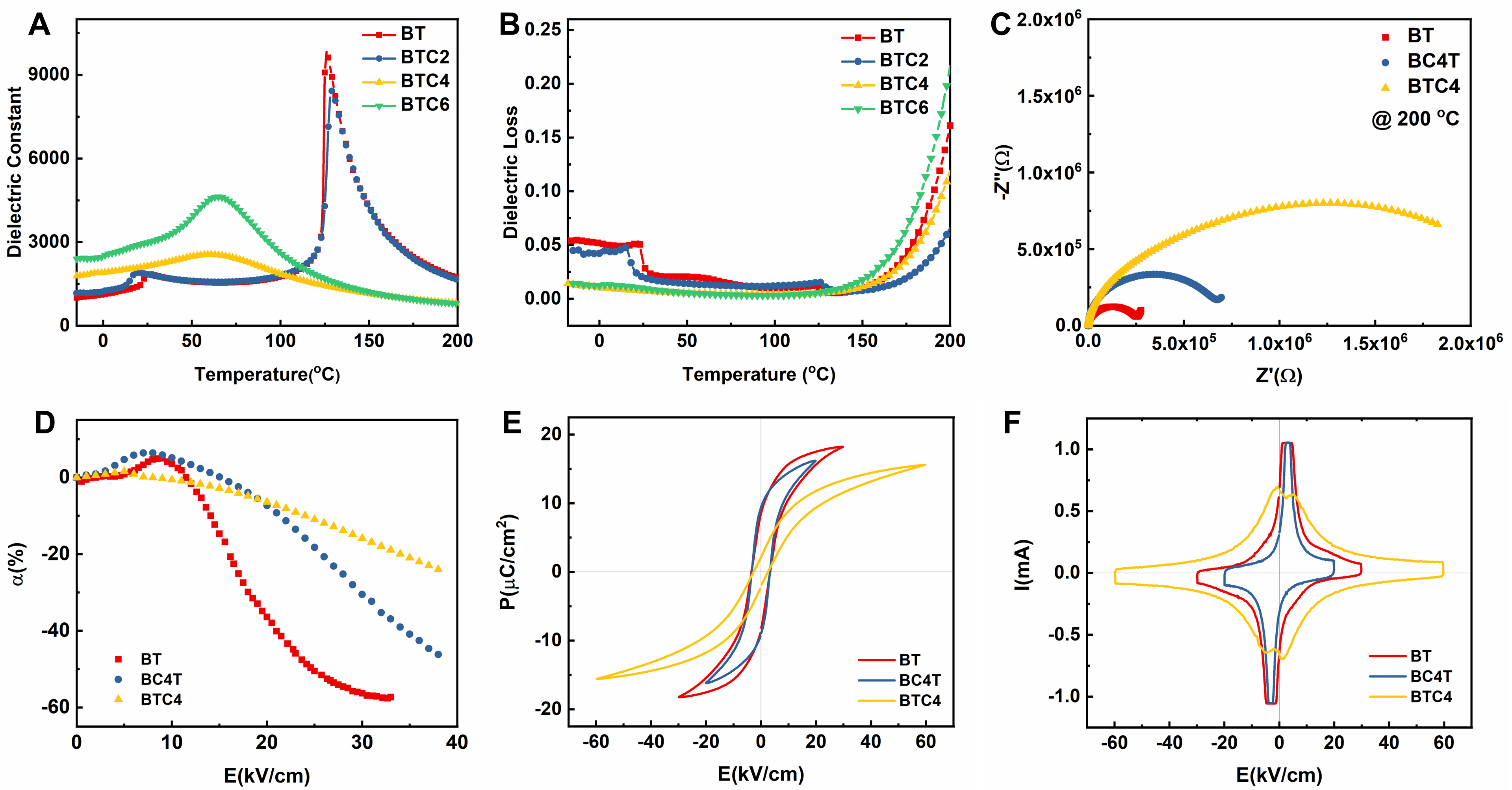
The effect of Ca doping on the temperature-dependent phase transition behavior of BT strongly depends on the substitution site [Figure 2A and B, Supplementary Figure 2]. For BCT ceramics, the Tc increases slightly from 129 °C (BT) to 134 °C as the Ca concentration increases, while the Tc of BTC ceramics decreases sharply to 60 °C (BTC4), accompanied by a significant broadening of the Curie peak[8]. The
Figure 2. Temperature dependence of (A) dielectric constant and (B) dielectric loss of BTC ceramics measured at 1 kHz; (C) Complex impedance spectra of BT, BC4T, and BTC4 ceramics measured at 200 °C; (D) The bias-field-dependent DC-bias stability coefficient (α) of BT, BC4T, and BTC4 ceramics; (E) P-E hysteresis loops and (F) corresponding I-E curves for BT, BC4T, and BTC4 ceramics. BTC: BaTi100-xCaxO3; BT: BaTiO3.
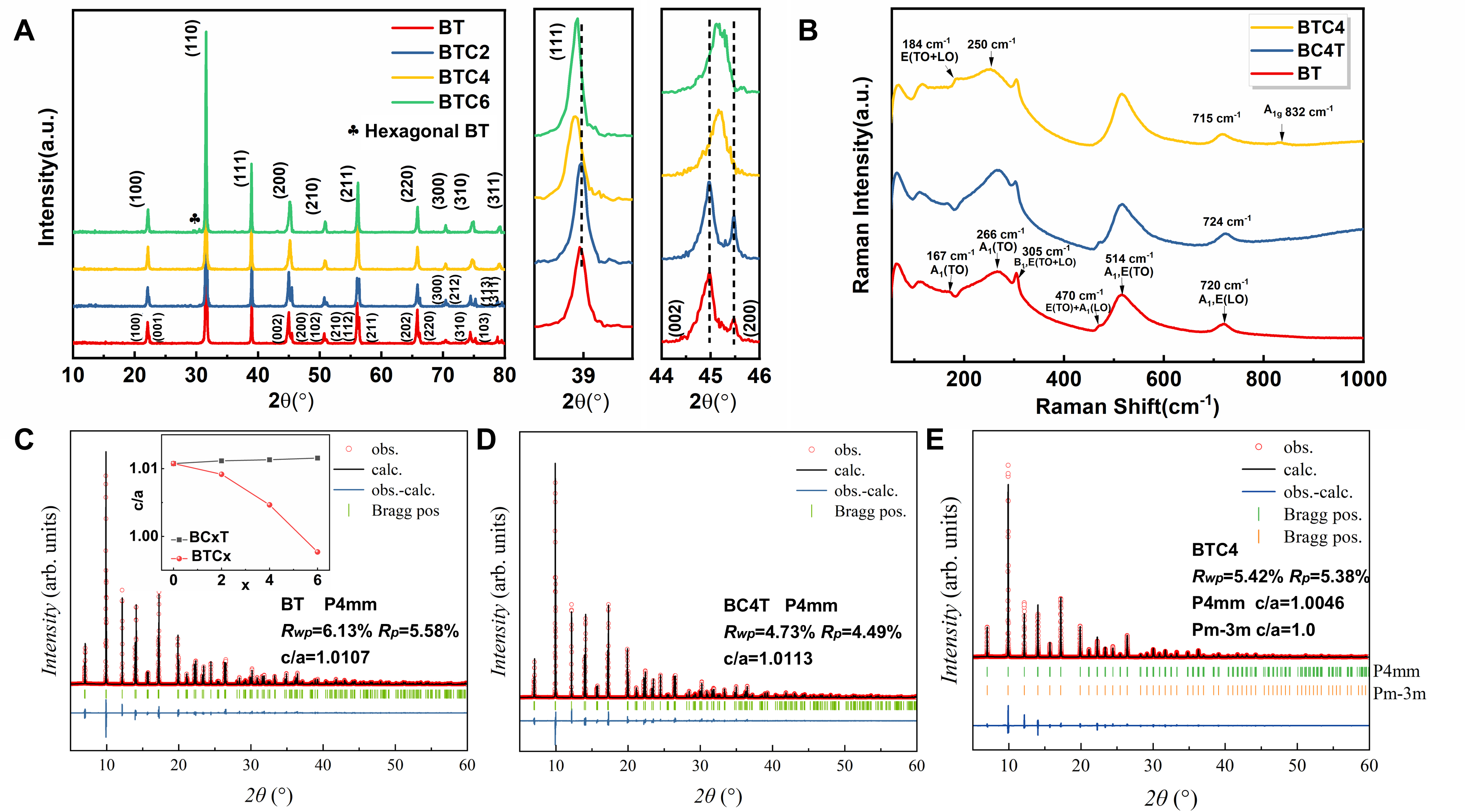
Figure 3. (A) XRD patterns of BTC ceramics, with enlarged views of (111), (002), and (200) diffraction peaks; (B) Raman spectra of BT, BC4T, and BTC4 ceramics measured at room temperature. Synchrotron powder XRD patterns of (C) BT, (D) BC4T, and (E) BTC4 ceramics, with accompanying Rietveld refinement results. A blue trace in the residual plot highlights the deviations between the measured and computed values. The positions of Bragg peaks are marked by vertical green and orange indicators, serving as reference points for crystallographic analysis. The primary diffraction peaks of all samples align well with theoretical Bragg positions (vertical ticks), confirming phase integrity. The refined profiles exhibit excellent agreement with peak positions and relative intensities, with high reliability factors. The illustration in Figure 3C shows the variation of the c/a ratio with Ca doping content for the BCT and BTC ceramics, as calculated from Rietveld refinement results. For the BTC ceramics, the c/a ratio was obtained by averaging the c/a values of the tetragonal and cubic phases. BTC: BaTi100-xCaxO3; XRD: X-ray powder diffraction; BT: BaTiO3; BCT: Ba100-xCaxTiO3.
The polarization behavior of BTC4 ceramic
To understand the coexistence of a high dielectric constant and superior DC-bias stability in BTC4 ceramic, it is essential to investigate the impact of B-site Ca substitution on the microstructure and clarify its influence on polarization mechanisms. In the BTC system, increasing Ca content leads to a gradual shift of the (111) diffraction peak to lower angles and the merging of the (200) and (002) peaks, as shown in Figure 3A. These changes indicate a progressive expansion of the unit cell and a reduction in tetragonality, reflecting a transition toward a pseudo-cubic structure. This structural evolution is primarily attributed to the substitution of Ti4+ (0.60 Å) by larger Ca2+ ions (0.99 Å)[6-9], which introduces significant local lattice distortion due to the large size mismatch. Such a high mismatch limits the solid solubility of Ca2+ at B-sites (x~0.04), and excess doping leads to the formation of the hexagonal BT phase in BTC6[8]. In contrast, in the BCT system, where Ca2+ substitutes at the A-site (Ba2+: 1.61 Å vs. Ca2+: 1.34 Å), leading to unit cell shrinkage and enhanced tetragonal stability[1,8]. This is evidenced by the shift of the (111) peak to higher angles and the persistent splitting of (200)/(002) peaks, as illustrated in Supplementary Figure 6. These structural evolutions are further validated by Raman spectra [Figure 3B]. The disappearance of the 167 cm-1 and
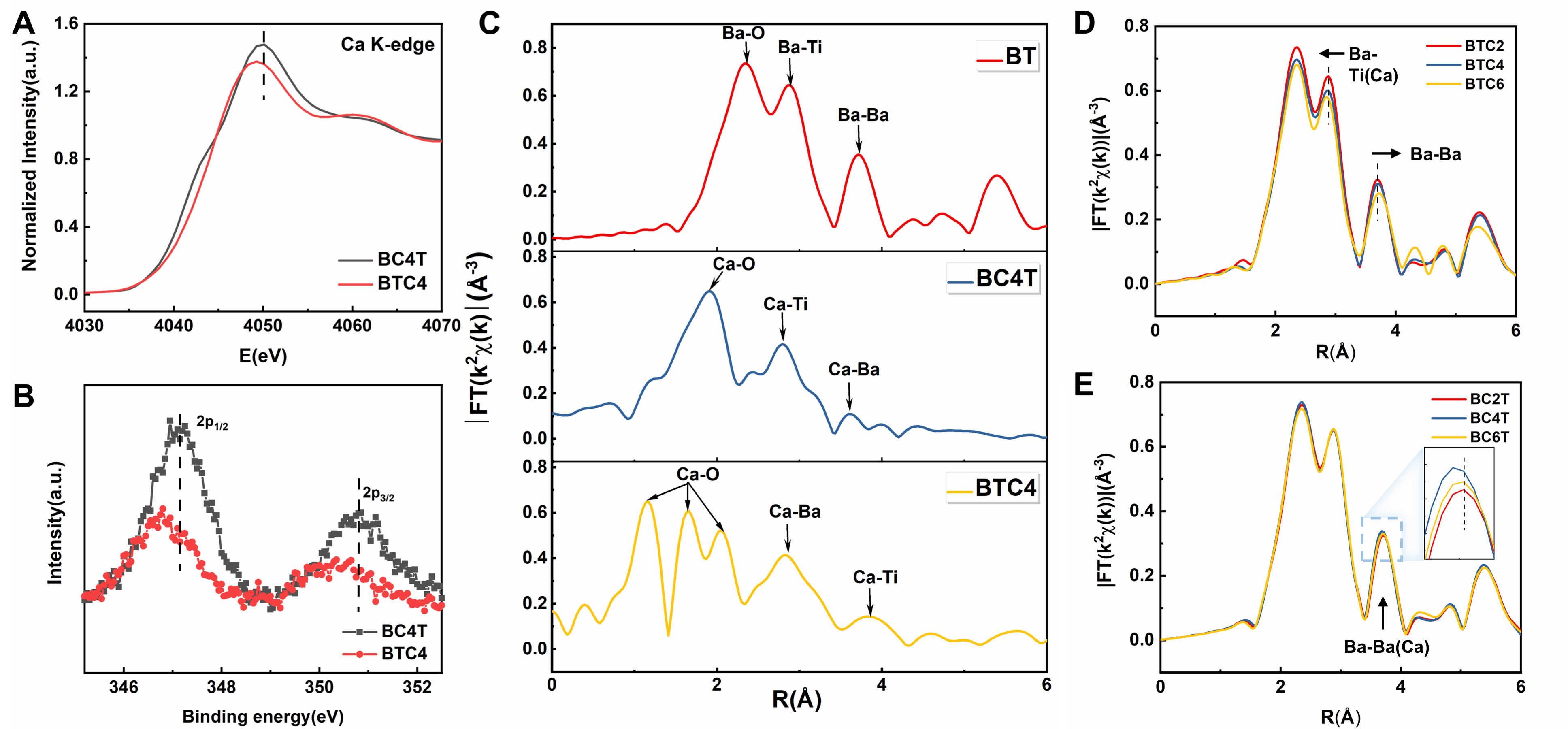
Since the atomic-scale Energy-dispersive X-ray spectroscopy (EDS) results [Supplementary Figure 9] could not determine the substitution sites of Ca atoms in the BTC4 sample, XANES, EXAFS, and XPS analyses were conducted to comprehensively clarify the local structure. As shown in Figure 4A, the Ca K-edge absorption edge in BTC4 shifts to lower energy with a decrease in white line intensity compared to BC4T. This indicates that B-site Ca substitution induces hybridization between Ca 3d and O 2p orbitals via the pseudo-Jahn-Teller effect (PJTE)[31,32], as white line intensity typically correlates with the density of unoccupied states. In Figure 4B, the Ca 2p orbital in BTC4 shifts toward lower binding energy, confirming partial electron transfer from O 2p to Ca 3d. In Figure 4C, the EXAFS spectrum of BC4T exhibits features similar to those of BT, with the first three peaks corresponding to Ca-O (~1.9 Å), Ca-Ti (~2.8 Å), and Ca-Ba (~3.6 Å) bonds. In contrast, BTC4 ceramic shows three split peaks of the Ca-O bond, indicating that Ca not only replaces the B-site but also undergoes displacement along the [001] direction. The PJTE effect typically leads to displacements along the [111][31,32], but Coulomb interactions caused by oxygen vacancies dominate and surpass the PJTE effect, resulting in Ca displacement along the [001] in BTC4 ceramic. This local structural distortion explains the presence of the P4mm phase in BTC4. It is well known that the ferroelectric phase of BT originates from the correlated alignment of local polar Ti displacements[33]. The presence of B-site Ca substitution and oxygen vacancies disrupts the -Ti-O-Ti- chain, weakening local Ti displacement correlations and promoting pseudo-cubic phase formation[8]. In Figure 4D, the Ba-Ba bond length increases while the Ba-Ti bond length decreases as the Ca doping content increases in BTC ceramics. This trend is attributed to the substitution of Ca for Ti, which leads to an increase in the lattice parameter a and a decrease in c. Furthermore, the reduction in both Ba-O and Ba-Ba bonds with increasing Ca content is consistent with the decreased unit cell volume caused by the substitution of Ca for Ba in BCT ceramics [Figure 4E][4]. These observations align well with the fitted lattice parameters presented in Supplementary Table 1, supporting the structural evolution induced by Ca doping.
Figure 4. (A) XANES spectra of the Ca K-edge in BC4T and BTC4 ceramics; (B) XPS spectra of Ca 2p in BC4T and BTC4 ceramics, which were obtained by subtracting the XPS intensity of BT from those of BC4T and BTC4, respectively; (C) Fourier transform (FT) of k-space EXAFS spectra for Ba in BT ceramic, and Ca in BC4T and BTC4 ceramics; FT of k-space EXAFS spectra for the Ba element in (D) BTC ceramics and (E) BCT ceramics. XANES: X-ray absorption near-edge structure; XPS: X-ray photoelectron spectroscopy; BT: BaTiO3; EXAFS: extended X-ray absorption fine structure; BTC: BaTi100-xCaxO3; BCT: Ba100-xCaxTiO3.
TEM analysis was performed to investigate the polar structure of BTC4 ceramics. As shown in Figure 5A, no obvious macro-domains are observed, but an incommensurate superlattice is detected by the diffraction pattern and the local strain field images shown in Figure 5B and C, likely induced by regular local lattice distortions associated with Ca substitution[34]. HADDF-STEM was acquired along the [001] zone axis, and the results revealed the presence of PNRs in BTC4. The regions in Figure 5D where polarization vectors are aligned along the {001} crystal plane family exhibit tetragonal symmetry. The relatively high polarization intensity in these regions, as shown in Figure 5E, further confirms the presence of a locally tetragonal structure. In contrast, areas with minimal polarization displacement can be attributed to a pseudo-cubic phase. The substitution of Ti by Ca disrupts the long-range order of Ti displacements, preventing the formation of ferroelectric domains and instead leading to the development of PNRs. Grain refinement also benefits the production of PNRs, ensuring the high dielectric constant in BTC4 [Supplementary Figure 10]. PNRs exhibit short-range order with random orientations, resulting in a weak polarization response under an electric field, which contributes to excellent DC-bias stability in BTC4. The c/a ratio mapping [Figure 5F] further reveals that highly polarized PNRs are not isolated but embedded and interconnected within the non-polar matrix. The bridging between PNRs can facilitate polarization rotation and reduce
Figure 5. (A) TEM image of BTC4 ceramic; (B) Enlarged view of (A), with the inset showing the corresponding diffraction pattern; (C) Localized strain field images of (B) calculated by the Geometric phase analysis (GPA) method. The two-dimensional strain field is characterized by normal strain components exx and eyy, which describe lattice elongation or compression along the X and Y directions, respectively. The shear strain component exy represents in-plane lattice distortion due to shear deformation, while the rotation component Rxy quantifies local rigid-body lattice rotation. High-angle annular dark-field scanning transmission electron microscopy (HADDF-STEM) images of BTC4 ceramics; (D) the polarization vector, (E) the polarization strength of the B site, and (F) the c/a ratio. Polarization vectors were defined as the displacement between the B-site cations and the center of the four nearest neighboring A-site cations, with polarization strength corresponding to the absolute magnitude of these vectors. TEM: Transmission electron microscope; BTC: BaTi100-xCaxO3.
The enhanced insulation in BTC4 ceramic
The high impedance, high activation energy, high breakdown strength, and extended lifetime
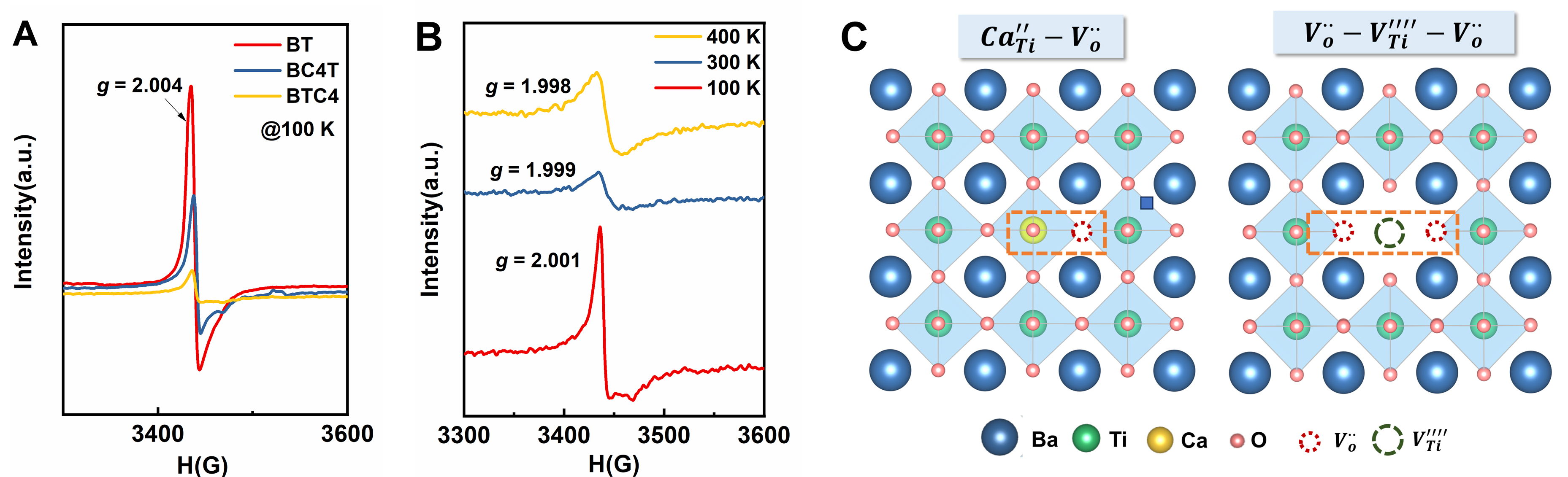
While Ca substitution at the Ba site is isovalent, Ca substituting for the Ti atom is aliovalent, leading to the formation of oxygen vacancies to preserve charge neutrality. This is corroborated by XPS analysis, which reveals a higher oxygen vacancy concentration in BTC4 ceramics [Supplementary Figure 12]. However, the higher activation energy [Supplementary Figure 3] and the mismatch between Z” and M” [Supplementary Figure 4] suggest that oxygen vacancies are localized. The EPR spectroscopy was employed to probe the defect structures. Figure 6A shows that all the BT, BC4T, and BTC4 ceramics exhibit a distinct EPR
The non-reducibility in BTC4 ceramic
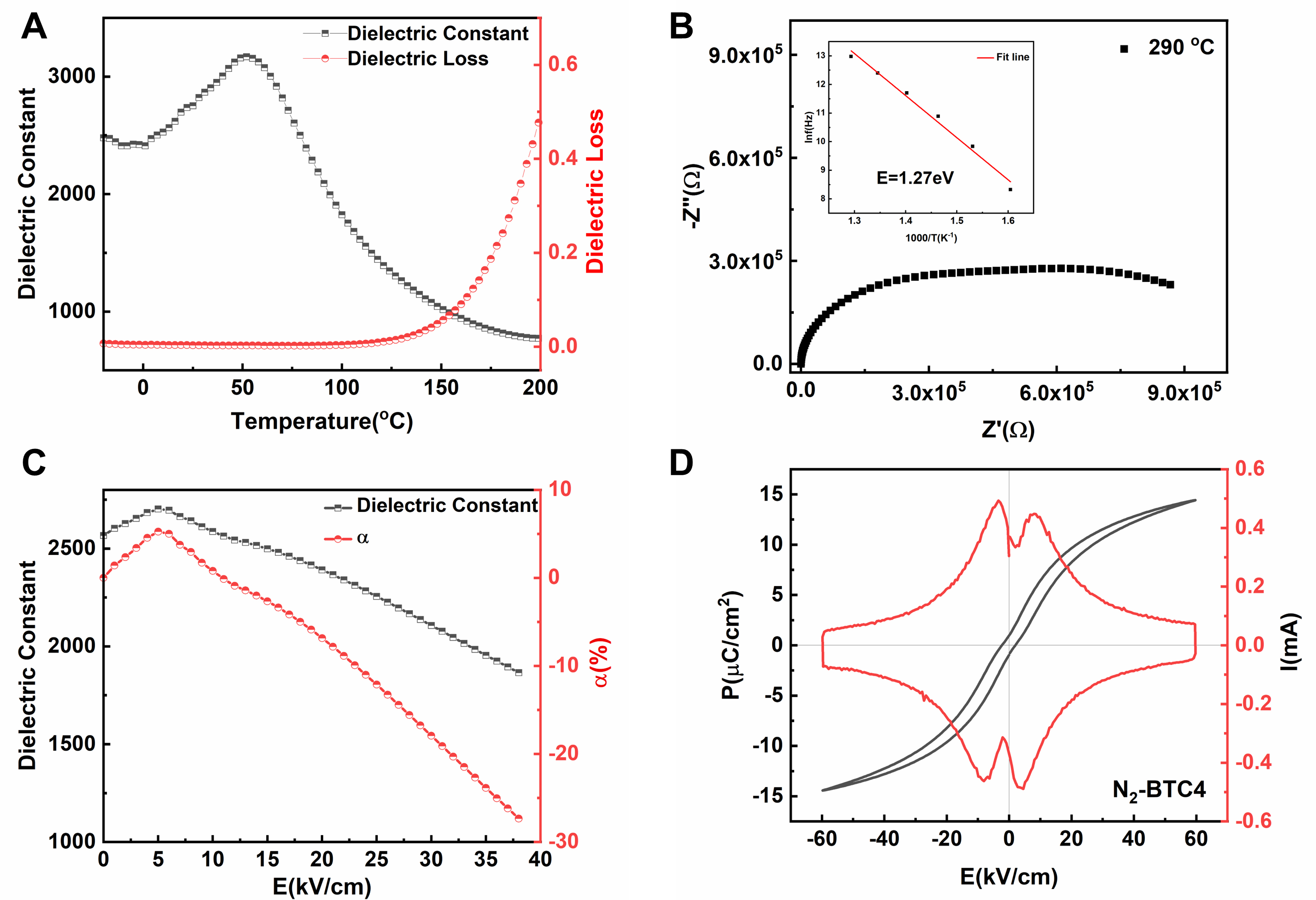
Non-reducibility is a crucial property of dielectric materials for MLCCs. Experimental results show that the BTC4 ceramics sintered in N2/H2 atmosphere (N2-BTC4) exhibit outstanding resistance to reduction. As shown in Figure 7A, the temperature-dependent dielectric properties of N2-BTC4 closely resemble those of BTC4, with the Tc shifting to approximately 50 °C and no significant dielectric loss peak. Notably, N2-BTC4 exhibits an impedance several orders of magnitude higher than that of N2-BT and N2-BC4T ceramics, indicating its superior insulation performance [Supplementary Figure 13]. Although N2-BTC4 theoretically has a higher oxygen vacancy concentration than BTC4, its calculated activation energy (1.27 eV) is higher than that of BTC4 [Figure 7B]. This suggests that oxygen vacancies are more strongly localized in N2-BTC4, likely forming a greater number of
Figure 7. (A) Temperature dependence of dielectric constant and dielectric loss at 1 kHz for N2-BTC4 ceramic; (B) Complex impedance spectra of N2-BTC4 ceramic, with the inset showing the Arrhenius plot of ln(f) versus 1000/T; (C) Dielectric constant and DC-bias stability coefficient as a function of the bias-field for N2-BTC4 ceramic; (D) P-E hysteresis loops and the corresponding I-E curves measured at 60 kV/cm for N2-BTC4 ceramic.
CONCLUSIONS
This study demonstrates that the pseudo-cubic structure of BTC ceramics consists of coexisting tetragonal and cubic phases, forming a unique microstructure in which tetragonal PNRs are embedded and interconnected within a non-polar cubic matrix. Such a configuration originates primarily from the disruption of the -Ti-O-Ti- chains caused by Ca substitution at the Ti site. The formation of PNRs and the reduction in the c/a ratio collectively contribute to the improved DC-bias stability. Moreover, the presence of tetragonal PNRs is essential for maintaining a high dielectric constant, thereby enabling a desirable balance between dielectric constant and DC-bias stability. Specifically, the reduced c/a ratio correlates with a decreased Tc, while the embedding PNRs lead to broadened dielectric peaks. In addition, oxygen vacancies in BTC ceramics are trapped by immobile cation defects (
DECLARATIONS
Acknowledgments
The authors would like to thank the synchrotron beamline RIKEN BL44B2 (proposal Nos. 2023B1295, 2023B1376, and 2024A1235) at SPring-8 for the beamtime allocation. Dr. Kenichi Kato is greatly acknowledged for his support during synchrotron experiments at BL44B2.
Authors’ contributions
Made substantial contributions to the conception and design of the study: He, Z.; Liu, Z.
Performed data acquisition: He, Z.; Chen, X.
Provided technical and material support: Chen, X.; Zhang, J; Fu, Z.
Performed data analysis and interpretation: Meng, X.; Fan, D.; Zhang, F.
Revised the language: Ma, M.; Li, Y.; Liu, Z.
Availability of data and materials
The original contributions presented in this study are included in the article/Supplementary Materials. Further inquiries can be directed to the corresponding author.
Financial support and sponsorship
This work was supported by the National Key R&D Program of China (No. 2021YFB3800604).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Levin, I.; Krayzman, V.; Woicik, J. C. Local-structure origins of the sustained Curie temperature in (Ba,Ca)TiO3 ferroelectrics. Appl. Phys. Lett. 2013, 102, 162906.
2. Fu, D.; Itoh, M.; Koshihara, S. Y.; Kosugi, T.; Tsuneyuki, S. Anomalous phase diagram of ferroelectric (Ba,Ca)TiO3 single crystals with giant electromechanical response. Phys. Rev. Lett. 2008, 100, 227601.
3. Hsiang, H.; Yang, Y.; Huang, C.; Yang, K. Dielectric properties of BaTiO3 and Ba0.95Ca0.05TiO3 sintered in a reducing atmosphere. Ceram. Int. 2023, 49, 28751-7.
4. Yoon, S.; Kang, S.; Kwon, S.; Hur, K. Resistance degradation behavior of Ca-doped BaTiO3. J. Mater. Res. 2010, 25, 2135-42.[DOI:10.1557/JMR.2010.0278.
5. Huang, X.; Zhang, L.; Wang, P.; et al. Excellent permittivity-temperature stability and reliability performance of ultra-thin Ba0.97Ca0.03TiO3 -based MLCCs. J. Asian. Ceram. Soc. 2023, 11, 146-58.
6. Sakabe, Y.; Wada, N.; Hiramatsu, T.; Tonogaki, T. Dielectric properties of fine-grained BaTiO3 ceramics doped with CaO. Jpn. J. Appl. Phys. 2002, 41, 6922-5.
7. Sakabe, Y.; Takagi, H. Nonreducible mechanism of {(Ba1- xCax)O}mTiO2 (m >1) ceramics. Jpn. J. Appl. Phys. 2002, 41, 6461-5.
8. Zhang, L.; Thakur, O. P.; Feteira, A.; et al. Comment on the use of calcium as a dopant in X8R BaTiO3-based ceramics. Applied. Physics. Letters. 2007, 90, 142914.
9. Lee, S. J.; Park, I. J.; Han, Y. H. Relaxation behavior of oxygen vacancy complexes in Ba(Ti1-xCax)O3-x. Mater. Chem. Phys. 2010, 121, 161-4.
10. Johnson, K. M. Variation of dielectric constant with voltage in ferroelectrics and its application to parametric devices. J. Appl. Phys. 1962, 33, 2826-31.
11. Hennings, D.; Schnell, A.; Simon, G. Diffuse ferroelectric phase transitions in Ba(Ti1-yZry)O3 ceramics. J. Am. Ceram. Soc. 1982, 65, 539-44.
12. Yoon, S.; Kim, M.; Kim, D. Influence of tetragonality (c/a) on dielectric nonlinearity and direct current (dc) bias characteristics of (1-x)BaTiO3-xBi0.5Na0.5TiO3 ceramics. J. Appl. Phys. 2017, 122, 154103.
13. Wen, H.; Wang, X.; Chen, R.; Li, L. Modeling of dielectric behaviors of multilayer ceramic capacitors under a direct current bias field. J. Am. Ceram. Soc. 2006, 89, 550-6.
14. Zhang, W.; Jiang, Y.; Xiao, M.; et al. High DC-bias stability and reliability in BaTiO3-based multilayer ceramic capacitors: the role of the core-shell structure and the electrode. ACS. Appl. Mater. Interfaces. 2024, 16, 1158-69.
15. Zhu, C.; Cai, Z.; Guo, L.; Li, L.; Wang, X. Grain size engineered high-performance nanograined BaTiO3 -based ceramics: experimental and numerical prediction. J. Am. Ceram. Soc. 2021, 104, 273-83.
16. Yuan, Q.; Yao, F.; Cheng, S.; et al. Bioinspired hierarchically structured all-inorganic nanocomposites with significantly improved capacitive performance. Adv. Funct. Mater. 2020, 30, 2000191.
17. Yang, L.; Kong, X.; Cheng, Z.; Zhang, S. Ultra-high energy storage performance with mitigated polarization saturation in lead-free relaxors. J. Mater. Chem. A. 2019, 7, 8573-80.
18. Zhu, M.; Li, S.; Zhang, H.; et al. Diffused phase transition boosted dye degradation with Ba (ZrxTi1-x)O3 solid solutions through piezoelectric effect. Nano. Energy. 2021, 89, 106474.
19. Ravel, B.; Newville, M. Athena, artemis, hephaestus: data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron. Radiat. 2005, 12, 537-41.
20. Kong, L. B.; Li, S.; Zhang, T. S.; et al. Electrically tunable dielectric materials and strategies to improve their performances. Prog. Mater. Sci. 2010, 55, 840-93.
21. Krayzman, V.; Levin, I.; Woicik, J. C.; Bridges, F.; Nelson, E. J.; Sinclair, D. C. Ca K-edge X-ray absorption fine structure in BaTiO3-CaTiO3 solid solutions. J. Appl. Phys. 2013, 113, 044106.
22. Yang, Y.; Hao, H.; Zhang, L.; et al. Structure, electrical and dielectric properties of Ca substituted BaTiO3 ceramics. Ceram. Int. 2018, 44, 11109-15.
23. Hao, X. A review on the dielectric materials for high energy-storage application. J. Adv. Dielect. 2013, 03, 1330001.
24. He, Z.; Cao, M.; Tao, Y.; et al. Anomalous dielectric nonlinearity in niobium and aluminum Co-doped SrTiO3 ceramics with giant permittivity and low dielectric loss. J. Phys. Chem. C. 2019, 123, 18142-9.
25. Randall, C. A.; Fan, Z.; Reaney, I.; Chen, L.; Trolier-mckinstry, S. Antiferroelectrics: history, fundamentals, crystal chemistry, crystal structures, size effects, and applications. J. Am. Ceram. Soc. 2021, 104, 3775-810.
26. Zhang, L.; Ben, L.; Thakur, O. P.; et al. Ferroelectric aging and recoverable electrostrain in BaTi0.98Ca0.02O2.98 ceramics. J. Am. Ceram. Soc. 2008, 91, 3101-4.
27. Dobal, P. S.; Dixit, A.; Katiyar, R. S.; Yu, Z.; Guo, R.; Bhalla, A. S. Micro-Raman scattering and dielectric investigations of phase transition behavior in the BaTiO3-BaZrO3 system. J. Appl. Phys. 2001, 89, 8085-91.
28. Perry, C. H.; Hall, D. B. Temperature dependence of the raman spectrum of BaTiO3. Phys. Rev. Lett. 1965, 15, 700-2.
29. Zheng, H.; Csete de Györgyfalva, G. D. C.; Quimby, R.; et al. Raman spectroscopy of B-site order-disorder in CaTiO3-based microwave ceramics. J. Eur. Ceram. Soc. 2003, 23, 2653-9.
30. Siny, I.; Tao, R.; Katiyar, R.; Guo, R.; Bhalla, A. Raman spectroscopy of Mg-Ta order-disorder in BaMg13Ta23O3. J. Phys. Chem. Solids. 1998, 59, 181-95.
31. Bersuker, I. B. Pseudo-Jahn-teller effect-a two-state paradigm in formation, deformation, and transformation of molecular systems and solids. Chem. Rev. 2013, 113, 1351-90.
32. Bersuker, I. B. Pseudo Jahn-Teller origin of perovskite multiferroics, magnetic-ferroelectric crossover, and magnetoelectric effects: the d0-d10 problem. Phys. Rev. Lett. 2012, 108, 137202.
33. Asokan, K.; Jan, J. C.; Chiou, J. W.; et al. Electronic structures of Ba1-xCaxTiO3 studied by X-ray absorption spectroscopy and theoretical calculation. J. Phys. Condens. Matter. 2001, 13, 11087-95.
34. Li, C.; Hong, J. S.; Huang, Y. H.; et al. Pinched P-E hysteresis loops in Ba4Sm2Fe0.5Ti3Nb6.5O30 ceramic with tungsten bronze structure. Appl. Phys. Lett. 2019, 115, 082901.
35. Takenaka, H.; Grinberg, I.; Liu, S.; Rappe, A. M. Slush-like polar structures in single-crystal relaxors. Nature 2017, 546, 391-5.
36. Zhang, M.; Lan, S.; Yang, B. B.; et al. Ultrahigh energy storage in high-entropy ceramic capacitors with polymorphic relaxor phase. Science 2024, 384, 185-9.
37. Wei, X.; Yao, X. Nonlinear dielectric properties of barium strontium titanate ceramics. Mater. Sci. Eng. B. 2003, 99, 74-8.
38. Dunbar, T. D.; Warren, W. L.; Tuttle, B. A.; Randall, C. A.; Tsur, Y. Electron paramagnetic resonance investigations of lanthanide-doped barium titanate: dopant site occupancy. J. Phys. Chem. B. 2004, 108, 908-17.
39. Kolodiazhnyi, T.; Petric, A. Analysis of point defects in polycrystalline BaTiO3 by electron paramagnetic resonance. J. Phys. Chem. Solids. 2003, 64, 953-60.
40. Ward, R. E.; Freeman, C. L.; Dean, J. S.; Sinclair, D. C.; Harding, J. H. Using metadynamics to obtain the free energy landscape for cation diffusion in functional ceramics: dopant distribution control in rare earth-doped BaTiO3. Adv. Funct. Mater. 2020, 30, 1905077.
41. Erhart, P.; Albe, K. Modeling the electrical conductivity in BaTiO3 on the basis of first-principles calculations. J. Appl. Phys. 2008, 104, 044315.
42. Ren, X. Large electric-field-induced strain in ferroelectric crystals by point-defect-mediated reversible domain switching. Nat. Mater. 2004, 3, 91-4.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data





















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.