Bidirectional associations between coronary artery disease and epilepsy: cross-sectional and prospective analyses from the UK biobank
Abstract
Aim: This study aimed to evaluate the bidirectional association between coronary artery disease (CAD) and epilepsy and to examine the modifying effect of genetic susceptibility.
Methods: Using data from the UK Biobank, we conducted cross-sectional and longitudinal analyses. In the cross-sectional analysis (n = 502,359), logistic regression estimated odds ratios for the association between CAD and epilepsy. In Cohort 1 (n = 496,921 without baseline epilepsy), stratified Cox models assessed the risk of incident epilepsy by CAD status. In Cohort 2 (n = 475,130 without baseline CAD), the risk of incident CAD by epilepsy status was similarly evaluated. Polygenic risk scores for CAD and epilepsy were incorporated to assess genetic modification.
Results: The median baseline age was 58.0 years, and 45.6% were male. Over a median follow-up of 13.8 years, 3,590 participants developed epilepsy and 39,223 developed CAD. CAD was significantly associated with epilepsy both cross-sectionally and longitudinally (hazard ratio (HR) 1.32; 95% confidence interval (CI): 1.15-1.51; P < 0.001). Conversely, epilepsy increased CAD risk (HR 1.31; 95%CI: 1.12-1.54; P < 0.001). These associations were consistent across age, sex, and body mass index (BMI) strata. CAD predicted epilepsy only in participants with low genetic risk, while epilepsy predicted CAD mainly in those with high genetic risk.
Conclusions: Our findings indicate a bidirectional association between CAD and epilepsy. Additionally, this association exhibits heterogeneity across subgroups and may be influenced by genetic susceptibility. These results underscore the need for further studies to elucidate the underlying mechanisms and clinical implications.
Keywords
INTRODUCTION
Coronary artery disease (CAD) is the leading cause of morbidity and mortality globally[1-3]. According to the Global Burden of Disease (GBD) study, CAD was diagnosed in an estimated 197 million individuals worldwide in 2019, causing 9.14 million deaths and accounting for 49.2% of all cardiovascular disease (CVD)-related fatalities[1,4,5]. While the established risk factors for CAD have been extensively studied, the potential contributions of nervous system disorders—particularly epilepsy—have not been fully elucidated[6-11].
Epilepsy is a prevalent neurological disorder, affecting more than 70 million individuals globally[12-17]. Beyond its effects on physical health, epilepsy also imposes significant psychological, social, and economic burdens[18-23]. Moreover, studies suggest that epilepsy may elevate the risk of CAD in affected individuals by promoting the release of catecholamines and glucocorticoids, while CAD-related hypoperfusion or myocardial injury may exacerbate the frequency and severity of seizures, creating a vicious cycle[24-34].
Despite these insights, the interplay between epilepsy and CAD remains underexplored. To date, only two small-scale cross-sectional studies in the United States have demonstrated a significantly higher prevalence of heart disease in individuals with epilepsy, as well as an increased prevalence of epilepsy in patients with heart disease[35,36]. Additionally, several European studies have investigated the cardiovascular risks and mortality associated with epilepsy, indicating that individuals with epilepsy have a higher prevalence of CVD and increased cardiovascular mortality compared to those without epilepsy[37-39]. These studies suggest that epilepsy may be a risk factor for CVD. Nevertheless, no studies have specifically examined the role of CAD in the onset of epilepsy, nor have they explored the potential contribution of epilepsy to the development of CAD. Moreover, the possible influence of genetic susceptibility on the relationship between these two conditions remains unexplored.
In this study, we conducted a comprehensive analysis using data from the UK Biobank to investigate the bidirectional relationship between CAD and epilepsy. First, we assessed the cross-sectional associations between CAD and epilepsy. Subsequently, we performed prospective cohort analyses to evaluate the association between baseline CAD and subsequent epilepsy and the association between baseline epilepsy and subsequent CAD. We further explored subgroup differences based on demographic and clinical characteristics and investigated the moderating effect of genetic predisposition. This research not only advances our understanding of the shared pathophysiological mechanisms of CAD and epilepsy but also holds promise for informing integrated management strategies and personalized treatment approaches.
MATERIAL AND METHODS
Data source and study design
The UK Biobank is a large population-based prospective study that recruited over 500,000 participants aged 40-69 years in the UK between 2006 and 2010[40]. The researchers gathered information on the participants' sociodemographic characteristics, lifestyle choices, medical history, and physical measurements at recruitment. Ethical approval for the study was obtained from the National Health Service (NHS) North West Research Ethics Committee, and all participants signed an informed consent form. The study was conducted under UK Biobank application number 77195. More comprehensive information is available at https://www.ukbiobank.ac.uk/.
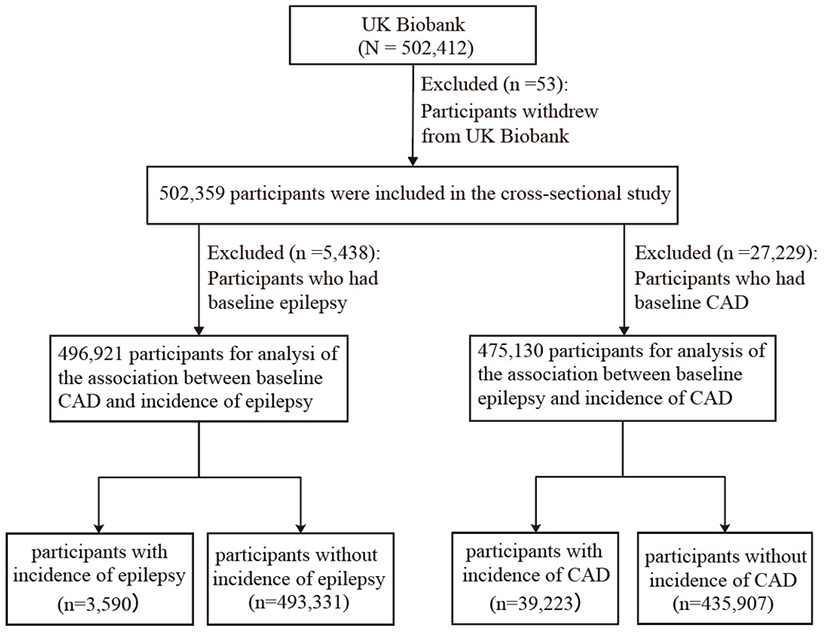
In the present study, we synthesized evidence from one cross-sectional study and two prospective cohort studies. First, among 502,412 participants in the UK Biobank, we excluded 53 participants who withdrew informed consent, leaving 502,359 participants included in the cross-sectional analysis to explore the associations between CAD and epilepsy. Subsequently, we conducted two prospective cohort studies. For Cohort 1, we excluded 5,438 participants with a diagnosis of epilepsy at baseline, leaving 496,921 participants to investigate the association between baseline CAD and the risk of developing epilepsy. For Cohort 2, we excluded 27,229 participants with CAD at baseline, leaving 475,130 participants to examine the relationship between epilepsy and the incidence of new-onset CAD. The complete participant selection process is illustrated in Figure 1.
Ascertainment of CAD and epilepsy
Cases of CAD and epilepsy were identified from health-related records of the International Classification of Diseases, 10th Edition (ICD-10), coded from self-reported information, primary care data, death registration records, and hospitalization data. The diagnostic codes for CAD are ICD-10 I20-I25, and those for epilepsy are ICD-10 G40 and G41. Detailed ICD-10 codes used to define CAD and epilepsy are provided in Supplementary Table 1. Follow-up commenced upon participants' enrollment and persisted until the date of diagnosis, loss of follow-up, mortality, or conclusion of the study (January 2, 2023)—whichever came first.
Data collection
We collected covariates based on a variety of data sources, including medical history, physical measurements, touch-screen questionnaires, and laboratory indicators. These covariates included age (continuous), sex (male/female) and Body mass index (BMI, continuous). Ethnicity was categorized as White, Asian/Asian British, Black/Black British, Chinese, Mixed, or Other. Smoking status (current/previous/never) and alcohol consumption (current/previous/never) were recorded. Physical activity level was classified as high, moderate, or low. Education level included college/university degree; A/AS levels (Advanced/Advanced Subsidiary levels); and NVQ (National Vocational Qualification). Other education categories included HND (Higher National Diploma); HNC (Higher National Certificate); other professional qualifications; O levels/GCSEs (General Certificate of Secondary Education) or equivalent; or none. Townsend deprivation index was included as a continuous variable. This area-based measure incorporates unemployment, lack of car ownership, non-home ownership, and household overcrowding. Higher scores indicate greater material deprivation. Laboratory indicators (all continuous) included total triglycerides, total cholesterol, HDL-C (high-density lipoprotein cholesterol), LDL-C (low-density lipoprotein cholesterol), GLU (glucose), and CRP (C-reactive protein). Clinical indicators (both continuous) were systolic blood pressure (SBP) and diastolic blood pressure (DBP). We also considered comorbidities and therapeutic medications relevant to CAD and epilepsy. These included hypertension, hyperlipidemia, diabetes, depression, cerebrovascular disease, hyperthyroidism, central nervous system infections, lipid-lowering drugs, antihypertensive medications, and insulin.
Polygenic risk score
The genotypic data, quality control procedures, polygenic risk score (PRS) construction, and evaluation for the UK Biobank have been described in previous reports[41]. In Cohort 1, we excluded participants who failed genetic data quality control (N = 14,999); in Cohort 2, we excluded those with missing genetic data
Statistical analyses
Baseline characteristics of participants were presented as medians with interquartile ranges (IQRs) for continuous variables and as frequencies and percentages for categorical variables. The difference between groups was tested by the Mann-Whitney U test. Categorical variables were compared using the chi-square test or Fisher's exact test. The overall missing rates for the covariates used in this study are presented in Supplementary Table 2. Missing data were imputed using the MICE package in R through the chained equations method. The imputed datasets were used in the logistic and Cox regression models described below.
In the cross-sectional analyses, logistic regression was used to evaluate the associations between CAD and epilepsy, with results expressed as odds ratios (ORs) and 95% confidence intervals (CIs). In the cohort analyses, stratified Cox proportional hazards regression was used to evaluate the relationship between baseline CAD and subsequent epilepsy, as well as between epilepsy and new-onset CAD. The results were expressed as hazard ratios (HRs) and 95% CIs. The proportional hazards assumption for the stratified Cox model was tested using the Schoenfeld residuals method and was confirmed to be satisfied. Both the logistic regression models and stratified Cox proportional hazards models employed three adjustment models. Model 1 adjusted for age, sex, and ethnicity. Model 2 further adjusted for BMI, smoking status, alcohol consumption, physical activity, education level, and the Townsend Deprivation Index. Building upon Model 2, Model 3 additionally adjusted for SBP, laboratory test measures (total triglycerides, total cholesterol, HDL-C, LDL-C, GLU, and CRP), and comorbidity/medication history. Comorbidities included hypertension, hyperlipidemia, diabetes mellitus, depression, cerebrovascular disease, hyperthyroidism, CNS infections (Cohort 1 only), and CVD family history (Cohort 2 only). Therapeutic medications included lipid-lowering drugs, antihypertensive medications, and insulin.
To investigate potential variations in the bidirectional relationship between CAD and epilepsy across different subgroups, we conducted stratified analyses based on the following factors: age (≤ 60 years vs.
Several sensitivity analyses were conducted to test the robustness of the main findings of this study: (1) Participants without major covariates in the cross-sectional and cohort studies were excluded, and the main analysis was repeated; (2) Participants in the longitudinal cohort with follow-up durations of less than two years were excluded, and the main analysis was repeated; (3) The proportional risk model was extended using a competing risk model, with death from other causes treated as a competing risk, to evaluate the stability of the findings; (4) A mixed-effects model was used in the longitudinal cohort to adjust for potential effects of the UK Biobank Assessment Centre, and the main analysis was repeated; (5) Participants with baseline cerebrovascular disease and head trauma were excluded, and the analysis was repeated in the longitudinal cohort. Furthermore, E-value analysis was employed to assess the robustness of the associations between CAD and new-onset epilepsy, as well as between epilepsy and new-onset CAD, in order to address potential unmeasured confounding factors[48,49].
All statistical analyses were performed by R-4.0.0 (R Foundation for Statistical Computing, Vienna, Austria). P values <0.05 were considered statistically significant.
RESULTS
Baseline characteristics of individuals in the cross-sectional study
Table 1 presents the baseline characteristics of 502,359 participants from the cross-sectional study, including 27,229 (5.42%) with CAD and 5,438 (1.08%) with epilepsy. The median age of all participants was 58.0 years (IQR, 50.0, 63.0), with 45.6% being male, 94.59% of White ethnicity, and a median BMI of 26.74 kg/m2 (IQR, 24.14, 29.91). Compared with the non-CAD and non-epilepsy groups, participants in the CAD and epilepsy groups had higher BMI, lower DBP, higher CRP, higher smoking frequency, lower drinking frequency, lower exercise frequency, lower education level, and higher Townsend deprivation index (all P < 0.001). These groups also had a higher prevalence of baseline comorbidities (all P < 0.001). Furthermore, they were more likely to use lipid-lowering drugs, blood pressure medications, and insulin (all P < 0.001). Notably, the prevalence of epilepsy was significantly higher in the CAD group than in the non-CAD group (1.78% vs. 1.04%, P < 0.001). Similarly, the prevalence of CAD was higher in individuals with epilepsy compared to those without epilepsy (8.92% vs. 5.38%, P < 0.001).
Baseline characteristics of 502,359 participants stratified by CAD and epilepsy in the Cross-Sectional Study
| Characteristics | Overall | CAD | Non-CAD | P-value | Epilepsy | Non-epilepsy | P-value |
| Participants, n (%) | 502,359 | 27,229(5.4%) | 475,130(94.6%) | 5,438(1.1%) | 496,921(98.9%) | ||
| Age (year, median (IQR)) | 58.00 [50.00, 63.00] | 63.00 [59.00, 66.00] | 57.00 [50.00, 63.00] | < 0.001 | 57.00 [49.00, 63.00] | 58.00 [50.00, 63.00] | < 0.001 |
| Sex, n (%) | |||||||
| Male | 229,065 (45.60) | 18,673 (68.58) | 210,392 (44.28) | < 0.001 | 2,701 (49.67) | 226,364 (45.55) | < 0.001 |
| Female | 273,294 (54.40) | 8,556 (31.42) | 264,738 (55.72) | < 0.001 | 2,737 (50.33) | 270,557 (54.45) | < 0.001 |
| BMI (kg/m2, median (IQR)) | 26.74 [24.14, 29.91] | 28.73 [25.98, 32.14] | 26.63 [24.06, 29.76] | < 0.001 | 27.18 [24.22, 30.74] | 26.74 [24.14, 29.90] | < 0.001 |
| DBP (mmHg, median (IQR)) | 82.00 [75.00, 89.00] | 79.00 [72.50, 86.00] | 82.00 [75.50, 89.00] | < 0.001 | 81.50 [74.50, 88.50] | 82.00 [75.00, 89.00] | 0.006 |
| SBP (mmHg, median (IQR)) | 136.00 [124.50, 149.50] | 137.50 [125.50, 150.00] | 136.00 [124.50, 149.50] | < 0.001 | 135.00 [123.00, 148.00] | 136.00 [124.50, 149.50] | < 0.001 |
| GLU (mmol/L, median (IQR)) | 4.93 [4.60, 5.32] | 5.09 [4.69, 5.65] | 4.92 [4.60, 5.30] | < 0.001 | 4.89 [4.55, 5.28] | 4.93 [4.60, 5.32] | < 0.001 |
| CRP (mg/L, median (IQR)) | 1.33 [0.66, 2.76] | 1.59 [0.78, 3.35] | 1.32 [0.65, 2.73] | < 0.001 | 1.77 [0.81, 3.81] | 1.33 [0.65, 2.75] | < 0.001 |
| Smoking, n (%) | < 0.001 | < 0.001 | |||||
| Current | 52,960 (10.60) | 3,400 (12.61) | 49,560 (10.49) | 792 (14.69) | 52,168 (10.56) | ||
| Previous | 173,002 (34.64) | 13,221 (49.04) | 159,781 (33.82) | 1,787 (33.14) | 171,215 (34.66) | ||
| Never | 273,447 (54.75) | 10,341 (38.35) | 263,106 (55.69) | 2,813 (52.17) | 270,634 (54.78) | ||
| Alcohol, n (%) | < 0.001 | < 0.001 | |||||
| Current | 460,236 (91.92) | 23,628 (87.16) | 436,608 (92.19) | 4,448 (82.42) | 455,788 (92.02) | ||
| Previous | 18,091 (3.61) | 1,775 (6.55) | 16,316 (3.45) | 544 (10.08) | 17,547 (3.54) | ||
| Never | 22,378 (4.47) | 1,705 (6.29) | 20,673 (4.37) | 405 (7.50) | 21,973 (4.44) | ||
| Ethnic, n (%) | < 0.001 | < 0.001 | |||||
| White | 472,564 (94.59) | 25,432 (94.06) | 447,132 (94.62) | 5,231 (96.82) | 467,333 (94.57) | ||
| Asian or Asian British | 9,878 (1.98) | 889 (3.29) | 8,989 (1.90) | 73 (1.35) | 9,805 (1.98) | ||
| Black or Black British | 8,058 (1.61) | 330 (1.22) | 7,728 (1.64) | 47 (0.87) | 8,011 (1.62) | ||
| Chinese | 1,573 (0.31) | 36 (0.13) | 1,537 (0.33) | 7 (0.13) | 1,566 (0.32) | ||
| Mixed | 2,953 (0.59) | 110 (0.41) | 2,843 (0.60) | 21 (0.39) | 2,932 (0.59) | ||
| Other ethnic group | 4,555 (0.91) | 240 (0.89) | 4,315 (0.91) | 24 (0.44) | 4,531 (0.92) | ||
| Physical activities, n (%) | < 0.001 | < 0.001 | |||||
| High | 162,093 (40.29) | 7,619 (36.17) | 154,474 (40.52) | 1,469 (36.50) | 160,624 (40.33) | ||
| Moderate | 163,984 (40.77) | 8,393 (39.84) | 155,591 (40.82) | 1,574 (39.11) | 162,410 (40.78) | ||
| Low | 76,189 (18.94) | 5,054 (23.99) | 71,135 (18.66) | 982 (24.40) | 75,207 (18.88) | ||
| Education, n (%) | < 0.001 | < 0.001 | |||||
| College or university degree | 161,100 (32.73) | 5,369 (20.28) | 155,731 (33.44) | 1,443 (27.28) | 159,657 (32.79) | ||
| A/AS levels, NVQ, HND, HNC, other professional qualifications, and equivalent | 113,821 (23.12) | 6,257 (23.64) | 107,564 (23.09) | 1,158 (21.89) | 112,663 (23.14) | ||
| O levels/GCSEs or CSEs or equivalent | 132,052 (26.83) | 5,867 (22.17) | 126,185 (27.09) | 1,414 (26.73) | 130,638 (26.83) | ||
| None of the above | 85,254 (17.32) | 8,976 (33.91) | 76,278 (16.38) | 1,274 (24.09) | 83,980 (17.25) | ||
| Townsend deprivation index (median (IQR)) | -2.14 [-3.64, 0.55] | -1.51 [-3.32, 1.79] | -2.17 [-3.66, 0.48] | < 0.001 | -1.30 [-3.19, 1.95] | -2.14 [-3.65, 0.53] | < 0.001 |
| Total Triglycerides (mmol/L, median (IQR)) | 1.48 [1.05, 2.15] | 1.67 [1.17, 2.38] | 1.47 [1.04, 2.13] | < 0.001 | 1.44 [1.02, 2.13] | 1.48 [1.05, 2.15] | 0.005 |
| Total Cholesterol (mmol/L, median (IQR)) | 5.65 [4.91, 6.42] | 4.52 [3.91, 5.24] | 5.70 [4.98, 6.46] | < 0.001 | 5.64 [4.87, 6.47] | 5.65 [4.91, 6.42] | 0.684 |
| HDL Cholesterol (mmol/L, median (IQR)) | 1.40 [1.17, 1.67] | 1.18 [1.00, 1.41] | 1.41 [1.18, 1.69] | < 0.001 | 1.45 [1.19, 1.77] | 1.40 [1.17, 1.67] | < 0.001 |
| LDL Cholesterol (mmol/L, median (IQR)) | 3.52 [2.94, 4.12] | 2.70 [2.26, 3.24] | 3.56 [3.00, 4.15] | < 0.001 | 3.45 [2.88, 4.09] | 3.52 [2.94, 4.12] | 0.019 |
| Comorbidity/Medication History, n (%) | |||||||
| Hypertension | 134,477 (26.77) | 17,460 (64.12) | 117,017 (24.63) | < 0.001 | 1,595 (29.33) | 132,882 (26.74) | < 0.001 |
| Hyperthyroidism | 5,439 (1.08) | 399 (1.47) | 5,040 (1.06) | < 0.001 | 66 (1.21) | 5,373 (1.08) | 0.383 |
| Diabetes | 26,261 (5.23) | 5,080 (18.66) | 21,181 (4.46) | < 0.001 | 354 (6.51) | 25,907 (5.21) | < 0.001 |
| Depression | 40,916 (8.14) | 2,864 (10.52) | 38,052 (8.01) | < 0.001 | 766 (14.09) | 40,150 (8.08) | < 0.001 |
| Cerebrovascular disease | 8,936 (1.78) | 1,964 (7.21) | 6,972 (1.47) | < 0.001 | 584 (10.74) | 8,352 (1.68) | < 0.001 |
| Hypertriglyceridemia | 75,147 (14.96) | 16,432 (60.35) | 58,715 (12.36) | < 0.001 | 1,032 (18.98) | 74,115 (14.91) | < 0.001 |
| Central nervous system inflammation | 3,111 (0.62) | 210 (0.77) | 2,901 (0.61) | 0.001 | 141 (2.59) | 2,970 (0.60) | < 0.001 |
| CVD family history | 91,332 (18.74) | 7,398 (28.17) | 83,934 (18.21) | < 0.001 | 966 (18.53) | 90,366 (18.75) | 0.711 |
| Cholesterol-lowering drug | 98,303 (19.57) | 21,666 (79.57) | 76,637 (16.13) | < 0.001 | 1,281 (23.56) | 97,022 (19.52) | < 0.001 |
| Antihypertensive drug | 113,365 (22.57) | 18,333 (67.33) | 95,032 (20.00) | < 0.001 | 1,276 (23.46) | 112,089 (22.56) | 0.115 |
| Insulin | 5,902 (1.17) | 1,266 (4.65) | 4,636 (0.98) | < 0.001 | 97 (1.78) | 5,805 (1.17) | < 0.001 |
| Epilepsy | 5,438 (1.08) | 485 (1.78) | 4,953 (1.04) | < 0.001 | / | / | / |
| CAD | 27,229 (5.42) | / | / | / | 485 (8.92) | 26,744 (5.38) | < 0.001 |
Association of CAD and epilepsy in the cross-sectional analysis
The risks associated with the presence of epilepsy in individuals with CAD and the presence of CAD in individuals with epilepsy are presented in Tables 2 and 3. Compared with individuals without CAD, those with CAD had a significantly higher risk of epilepsy (multivariable-adjusted OR, 1.25; 95%CI: 1.10-1.43,
Association of coronary artery disease with epilepsy in cross-sectional analysis
| Groups | Model 1 | Model 2 | Model 3 | |||
| OR (95%CI) | P-value | OR (95%CI) | P-value | OR (95%CI) | P-value | |
| Non-CAD | 1 (reference) | - | 1 (reference) | - | 1 (reference) | - |
| CAD | 1.79 (1.62-1.97) | < 0.001 | 1.42 (1.26-1.60) | < 0.001 | 1.25 (1.10-1.43) | < 0.001 |
Association of epilepsy with coronary artery disease in cross-sectional analysis
| Groups | Model 1 | Model 2 | Model 3 | |||
| OR (95%CI) | P-value | OR (95%CI) | P-value | OR (95%CI) | P-value | |
| Non-Epilepsy | 1 (reference) | - | 1 (reference) | - | 1 (reference) | - |
| Epilepsy | 1.79 (1.62-1.97) | < 0.001 | 1.47 (1.28-1.68) | < 0.001 | 1.45 (1.29-1.62) | < 0.001 |
Baseline characteristics of individuals in the prospective cohort study
We subsequently conducted two longitudinal cohort analyses to examine the temporal relationship between CAD and epilepsy. In Cohort 1, we investigated the association between baseline CAD and subsequent epilepsy, using the non-CAD group as the control. In Cohort 2, we explored the relationship between baseline epilepsy and new-onset CAD, with the non-epilepsy group as the control.
In Cohort 1, baseline characteristics of 496,921 participants are presented in Supplementary Table 3, including 26,744 (5.4%) with CAD and 470,177 (94.6%) without CAD. The median age was 58.0 years (IQR, 50.0-63.0), and 226,364 (45.55%) of the participants were male. Compared to those without baseline CAD, individuals with baseline CAD were more likely to be older, male, have a higher BMI, smoke frequently, be less educated, engage in less physical activity, and have higher baseline SBP (all P < 0.001). They also had a higher Townsend deprivation index, elevated total triglycerides, and lower total cholesterol, HDL-C, and LDL-C levels (all P < 0.001). Additionally, they used more lipid-lowering medications, antihypertensive drugs, insulin, and exhibited a higher prevalence of comorbidities (all P < 0.001).
In Cohort 2, the baseline information of 475,130 participants involved in the analysis of the associations between epilepsy and the incidence of CAD is shown in Supplementary Table 4, including 4,953 (1.0%) with epilepsy and 470,177 (99.0%) without epilepsy. The median age was 57.0 years (IQR, 50.0-63.0), with 44.3% of participants being male. Compared with participants without epilepsy, those with epilepsy were more likely to be younger, male, have a higher BMI, smoke more frequently, be of white ethnicity, have lower educational attainment, be less physically active, and have higher Townsend deprivation index scores (all
Association between baseline CAD and the development of epilepsy in the longitudinal cohort 1
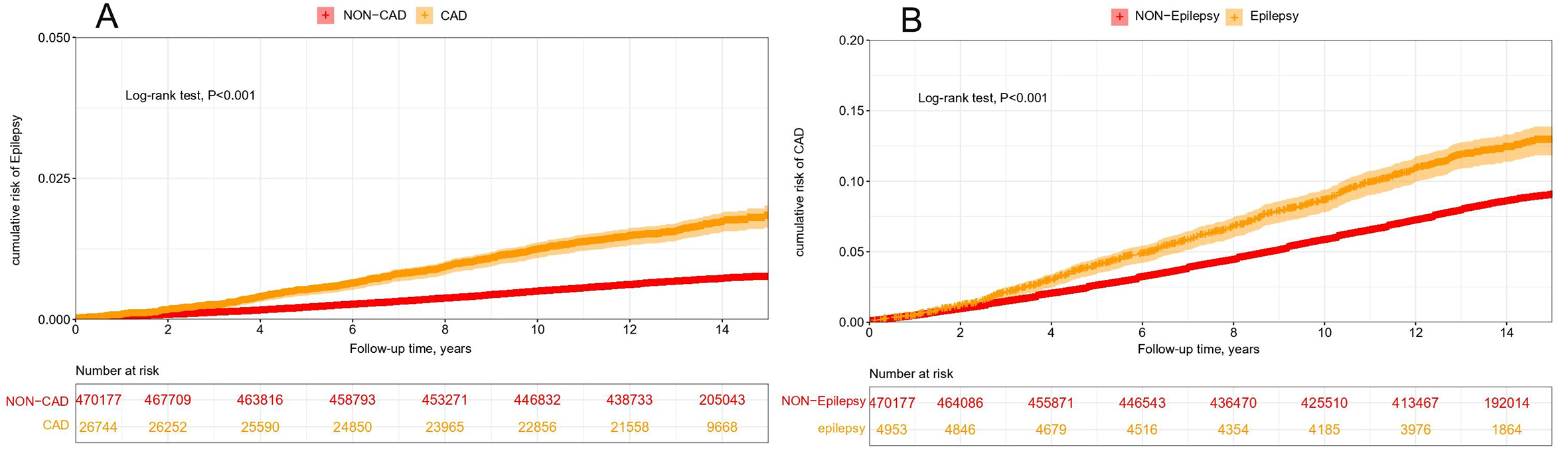
As shown in Supplementary Table 3, during a median follow-up of 13.82 years, a total of 3,590 (0.72%) out of 496,921 participants developed new-onset epilepsy. Among these, 3,183 (0.68%) cases occurred in the non-CAD group, while 407 (1.52%) cases were observed in the CAD group. The Kaplan-Meier curve demonstrates a significantly higher cumulative incidence of epilepsy in the CAD group compared to the non-CAD group (log-rank P < 0.001, Figure 2A). Subsequently, we assessed the association between CAD and new-onset epilepsy [Table 4]. After adjusting for all potential confounders, the HR for the CAD group was 1.32 (95%CI: 1.15-1.51, P < 0.001).
Figure 2. Cumulative risk of epilepsy in patients with CAD and cumulative risk of CAD in patients with epilepsy. (A) Cumulative risk of epilepsy in CAD participants and non-CAD participants. (B) Cumulative risk of CAD in epilepsy and non-epilepsy participants. CAD: coronary artery disease.
Association between baseline CAD and the incidence of epilepsy in the prospective cohort analysis
| Groups | No. of participants | Events/person-years | Model 1 | Model 2 | Model 3 | |||
| HR (95%CI) | P-value | HR (95%CI) | P-value | HR (95%CI) | P-value | |||
| Non-CAD | 470,177 | 3,183/6,721,643 | 1 (reference) | - | 1 (reference) | - | 1 (reference) | - |
| CAD | 26,744 | 407/354,194 | 1.86 (1.67-2.06) | < 0.001 | 1.60 (1.42-1.82) | < 0.001 | 1.32 (1.15-1.51) | < 0.001 |
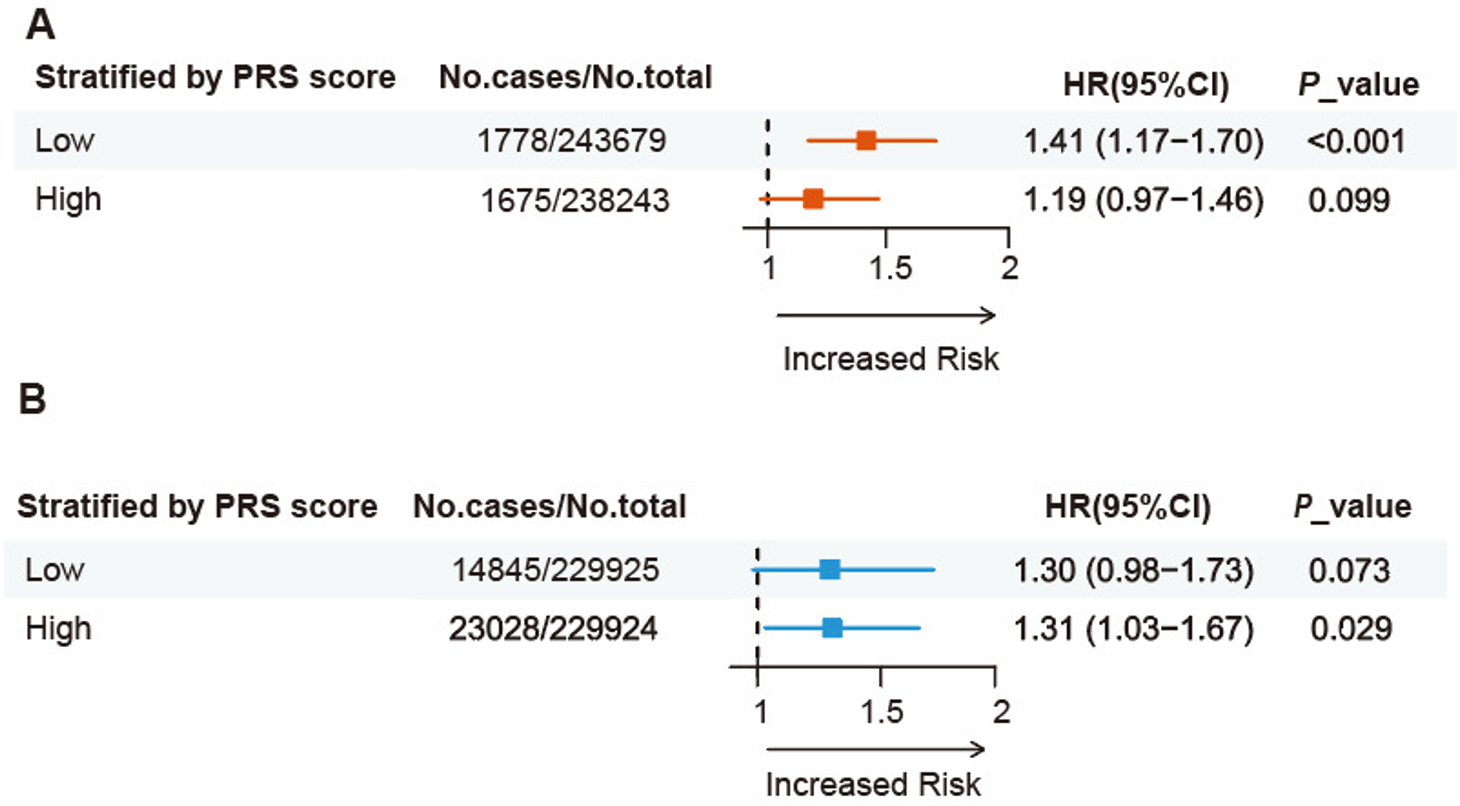
Furthermore, we divided participants into low and high genetic risk groups based on their median epilepsy PRS score and conducted a subgroup analysis [Figure 3A]. Interestingly, in the multivariate adjusted Cox regression model (n = 481,922), we found that the association between CAD and new-onset epilepsy was significant only among participants with low genetic susceptibility to epilepsy (HR, 1.41, 95%CI: 1.17-1.70,
Figure 3. Genetic susceptibility-adjusted bidirectional analysis of epilepsy and CAD in the prospective cohort. (A) Association between CAD and incident epilepsy, stratified by or adjusted for genetic susceptibility to epilepsy. (B) Association between epilepsy and incident CAD, stratified by or adjusted for genetic susceptibility to CAD.
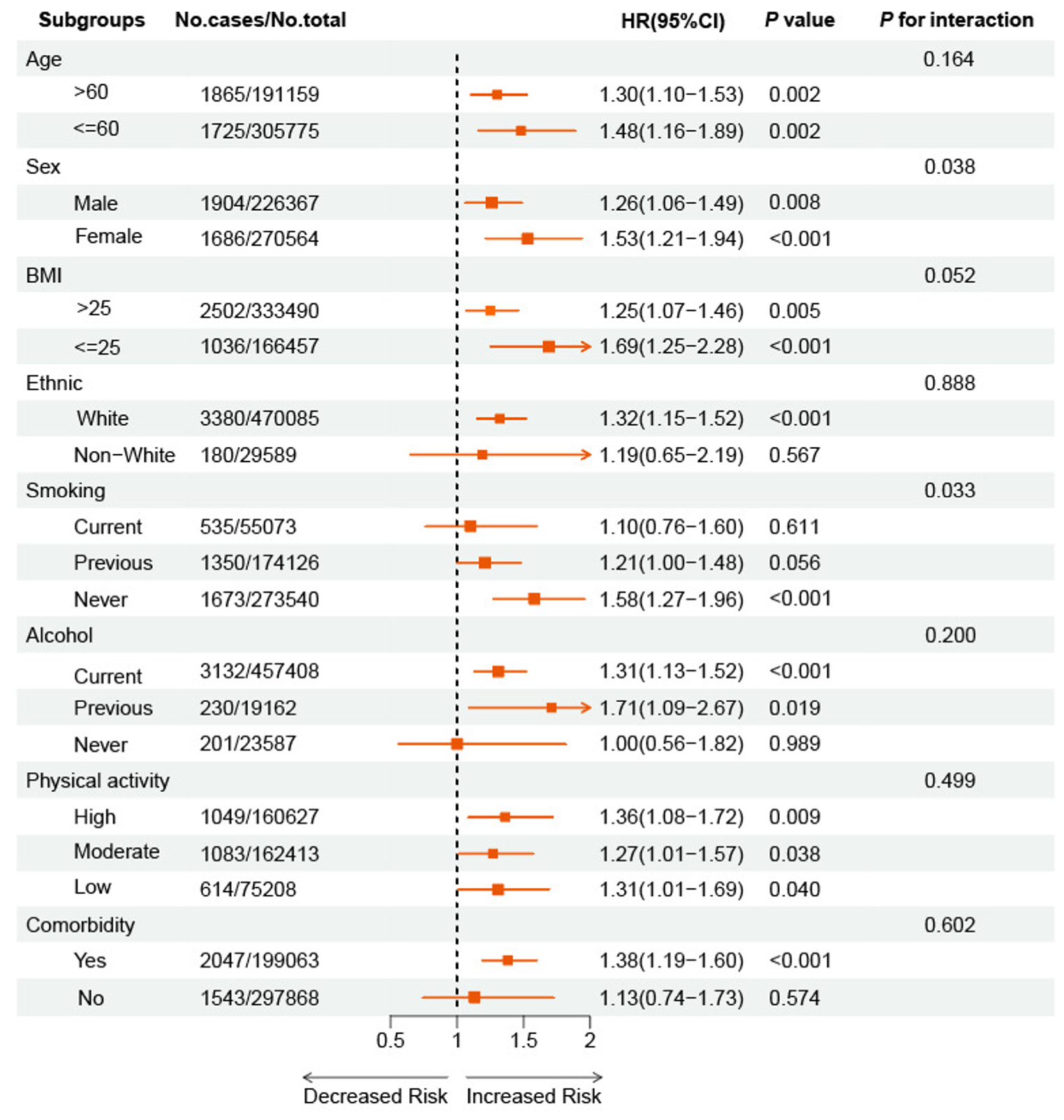
Finally, we conducted subgroup analyses based on age, sex, BMI, ethnicity, smoking status, alcohol consumption, physical activity, and comorbidities [Figure 4]. Notably, we found that the association remained significant regardless of age, sex, obesity status, physical activity levels, and among White participants, as well as in individuals with comorbidities. Additionally, we found an interaction between sex, smoking status, and the effect of CAD on epilepsy (P for interaction < 0.05).
Figure 4. Relationship between CAD and the incidence of epilepsy, stratified by covariates. Multivariable stratified Cox proportional hazards regression models were used to estimate hazard ratios (HRs) and 95% confidence intervals (CIs) for the association between CAD and incident epilepsy across different subgroups. Model adjusted age, sex, BMI, ethnicity, alcohol drinking status, smoking status, physical activity, education, Townsend deprivation index, total cholesterol, LDL cholesterol, HDL cholesterol, total triglycerides, glucose, systolic blood pressure, C-reactive protein, and comorbidity/medication history. (Omit the corresponding grouping variable from the relevant subgroup analysis (e.g., do not adjust for the sex factor in the sex subgroup)). CAD: Coronary artery disease; BMI, body mass index; LDL: low-density lipoprotein; HDL: high-density lipoprotein.
Association between baseline epilepsy and the development of CAD in the longitudinal cohort 2
In analyzing the risk of incident CAD associated with epilepsy, we followed a total of 475,130 participants for a median of 13.73 years. During this period, 576 (11.6%) epilepsy participants and 38,647 (8.3%) non-epilepsy individuals developed CAD [Supplementary Table 4]. Notably, the prevalence of all CAD-related outcomes and death was significantly higher in participants with epilepsy than in those without
Association between baseline epilepsy and the incidence of cad in the prospective cohort analysis
| Groups | No. of participants | Events/person-years | Model 1 | Model 2 | Model 3 | |||
| HR (95%CI) | P-value | HR (95%CI) | P-value | HR (95%CI) | P-value | |||
| Non-Epilepsy | 470,177 | 38,647/6,499,446 | 1 (reference) | - | 1 (reference) | - | 1 (reference) | - |
| Epilepsy | 4,953 | 576/65,143 | 1.52 (1.40-1.65) | < 0.001 | 1.28 (1.12-1.47) | < 0.001 | 1.31 (1.12-1.54) | < 0.001 |
Moreover, subgroup analysis stratified by the median PRS (n = 459,849) for CAD revealed that although the association between epilepsy and new-onset CAD was significant only in participants with high genetic risk, there was no significant difference in risk between low genetic risk and high genetic risk participants. The HRs were 1.30 (95%CI: 0.98-1.73,
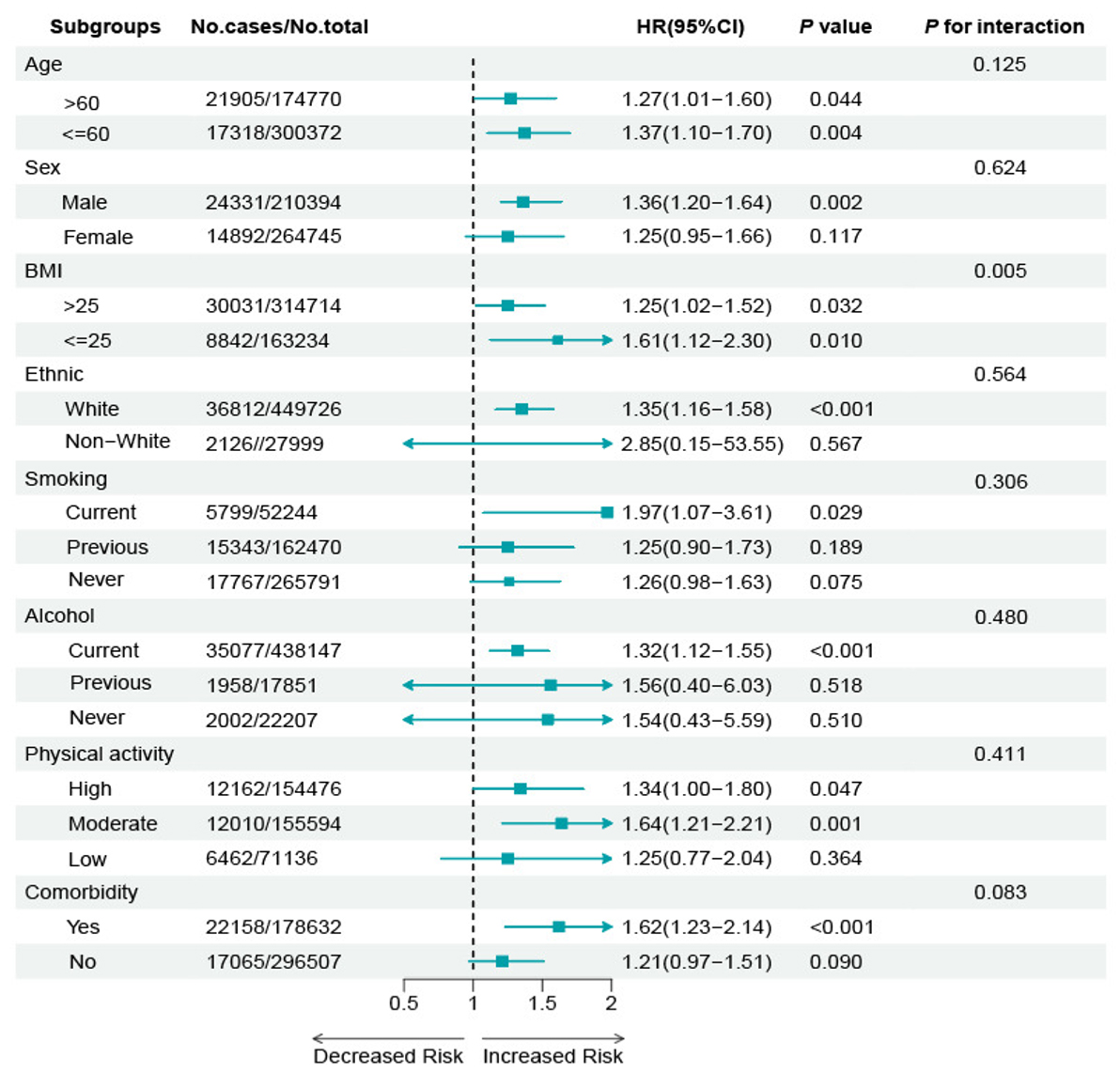
Furthermore, we conducted subgroup analyses based on age, sex, BMI, ethnicity, smoking status, alcohol consumption, physical activity, and comorbidities [Figure 5]. We found that the association between epilepsy and new-onset CAD remained significant regardless of age, BMI, and among male participants, white participants, current smokers, current drinkers, and those with comorbidities. Additionally, we identified an interaction between BMI and the effect of epilepsy on CAD (P for interaction < 0.05).
Figure 5. Relationship between epilepsy and the incidence of CAD, stratified by covariates. Multivariable stratified Cox proportional hazards regression models were used to estimate hazard ratios (HRs) and 95% confidence intervals (CIs) for the association between epilepsy and incident CAD across different subgroups. Model adjusted age, sex, BMI, ethnicity, alcohol drinking status, smoking status, physical activity, education, Townsend deprivation index, total cholesterol, LDL cholesterol, HDL cholesterol, total triglycerides, glucose, systolic blood pressure, C-reactive protein, and comorbidity/medication history. (Omit the corresponding grouping variable from the relevant subgroup analysis (e.g., do not adjust for the sex factor in the sex subgroup)). CAD: Coronary artery disease; BMI: body mass index; LDL: low-density lipoprotein; HDL: high-density lipoprotein.
Sensitivity analysis
Several additional sensitivity analyses were performed to evaluate the robustness of our findings, with results detailed in Supplementary Tables 7-11. These analyses included excluding participants with a follow-up duration of less than two years, restricting the primary analysis to participants with complete major covariate data, applying Fine-Gray competing risk models, using a mixed-effects model to adjust for institutional factors, and repeating the analysis in the longitudinal cohort after excluding participants with baseline cerebrovascular disease and head trauma. The results of all sensitivity analyses were generally consistent with the main conclusion. In addition, we conducted E-value analyses for both cohorts. The E-values of the point estimates for the two cohorts were 1.97 and 1.95, respectively. This indicates that unmeasured confounding factors need to have a moderate to strong association with the exposure factor and the outcome, and this association should not be affected by the measured covariates, in order to fully explain the observed association. Therefore, it is unlikely that the main findings can be solely explained by unmeasured confounding factors.
DISCUSSION
This large cross-sectional and prospective cohort study provides evidence of a bidirectional association between CAD and epilepsy. Cross-sectional analyses demonstrated a clear association between CAD and epilepsy. In longitudinal analyses, individuals with CAD had a higher risk of new-onset epilepsy than those without CAD, while individuals with epilepsy were more likely to develop incident CAD than those without epilepsy. Furthermore, the association between CAD and new-onset epilepsy was significant only in participants with low genetic susceptibility, whereas the association between epilepsy and new-onset CAD was significant only in those with high genetic susceptibility. These findings were consistent across subgroups stratified by age, sex, and BMI. Overall, the results deepen our understanding of the mechanisms underlying the bidirectional relationship between these two diseases and may inform more integrated preventive and therapeutic strategies in clinical practice.
Our study on the bidirectional relationship between CAD and epilepsy extends previous research findings. Cross-sectional studies in the U.S. population have shown that 21% of adults with epilepsy also reported a history of heart disease, 9% higher than the 12% observed in adults without epilepsy. Additionally, adults with a history of heart disease were nearly twice as likely to report a history of epilepsy compared with those without heart disease[35]. In addition to cross-sectional evidence on the association between epilepsy and CAD, our study also considered the temporal sequence of the two conditions. We used two longitudinal cohorts to examine the relationship between baseline CAD and incident epilepsy, as well as between baseline epilepsy and incident CAD. Previous cohort studies from the United States and the United Kingdom have reported findings consistent with ours, showing a higher prevalence of CVD among individuals with epilepsy and a greater incidence of heart disease during follow-up[37-39]. Building on these findings, we further demonstrated that individuals with CAD had a higher risk of new-onset epilepsy than those without CAD, and individuals with epilepsy had a higher risk of incident CAD than those without epilepsy. Additionally, we investigated the potential influence of genetic susceptibility on this association. Overall, our study extends the existing literature and supports a bidirectional association between CAD and epilepsy, highlighting the need for a holistic approach to management, including integrated strategies for health promotion, prevention, diagnosis, treatment, and prognosis to reduce the burden of these coexisting conditions.
Our study also explored the relationship between CAD and epilepsy across different subgroups. We found that the association remained statistically significant regardless of age or BMI level. This may be attributed to shared risk factors and the presence of brain-heart interactions, which are observed across different age and BMI groups[50]. In the sex-stratified analysis, the association between baseline epilepsy and incident CAD was statistically significant in males but not in females. This difference may be related to sex-specific baseline cardiovascular risk. Males generally have a higher risk of CAD at a younger age, which may amplify the observable effect of epilepsy on CAD. In contrast, premenopausal females are protected by estrogen, which lowers their cardiovascular risk and may mask the influence of epilepsy[51]. Furthermore, we observed that the bidirectional association between epilepsy and CAD was significant among individuals with comorbidities, but not among those without. This may be due to the role of comorbidities as 'amplifiers' of risk or as mediating factors that enhance the interaction between epilepsy and CAD through mechanisms such as chronic inflammation, endothelial dysfunction, and pharmacological interactions[52,53].
Interestingly, the association between baseline CAD and incident epilepsy was statistically significant only among individuals with low genetic susceptibility. One possible explanation is that individuals with a high genetic risk for epilepsy may possess more responsive neuroprotective feedback mechanisms. For example, enhanced regulatory functions of calcium channel-related genes may attenuate the impact of exogenous risk factors such as CAD on brain function, thereby masking their pro-epileptogenic effects[54]. However, we cannot exclude the possibility that unmeasured factors such as longer epilepsy duration or poorer seizure control in the high-risk group may have influenced the results, as epilepsy-related cardiac injury may be a cumulative process[55]. In contrast, the effect of baseline epilepsy on incident CAD was significant only among individuals with high genetic susceptibility. Existing studies have indicated that epilepsy and CAD may share common risk factors and partially overlapping genetic risk loci, such as those associated with chronic inflammation, oxidative stress, and ion channel dysfunction[56-59]. However, to date, no large-scale GWAS have systematically examined the shared genetic variants between these two conditions. Future studies are needed to investigate this potential genetic relationship.
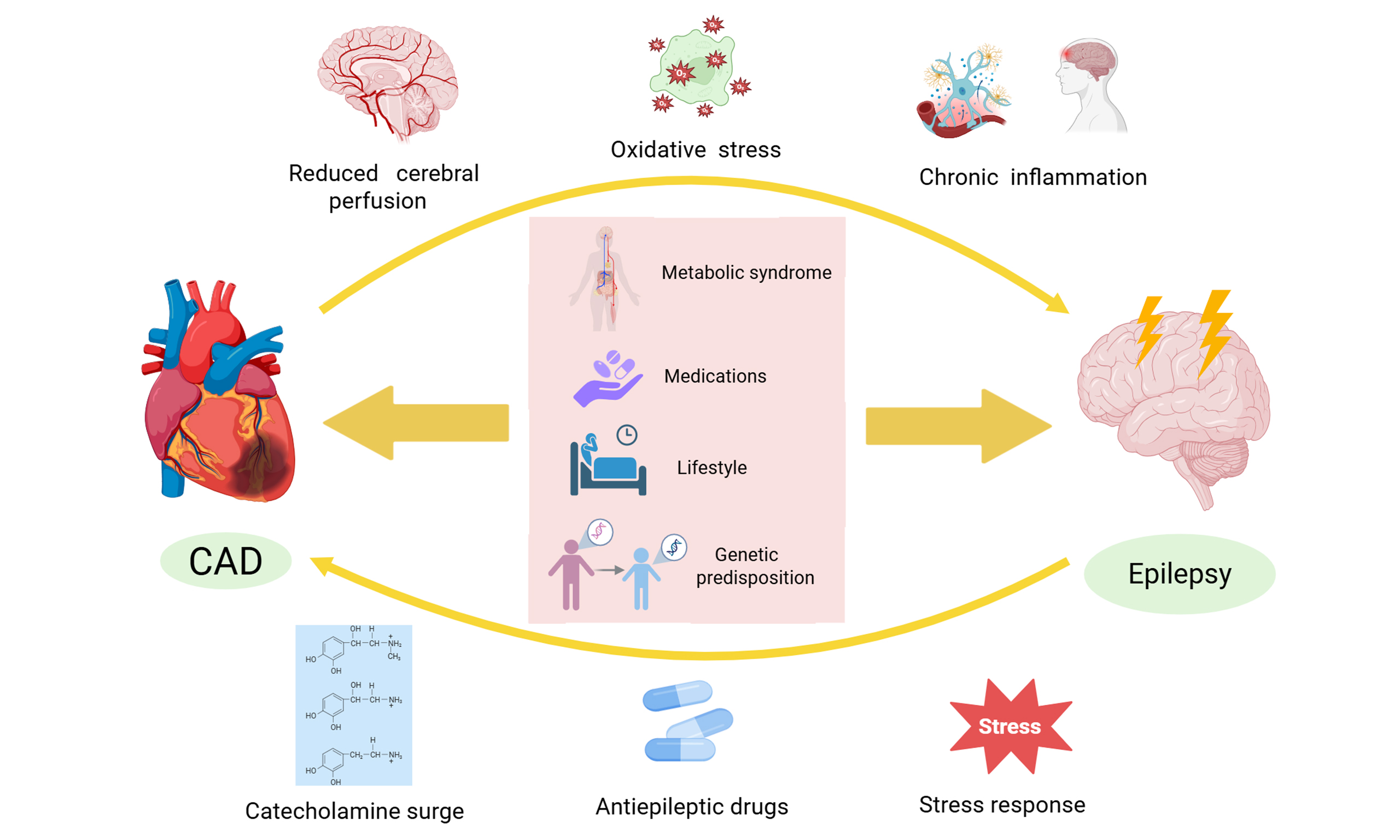
One of the mechanisms explaining the bidirectional relationship between CAD and epilepsy may be the presence of common risk factors. These include metabolic syndrome, medications, lifestyle, genetic predisposition, and other factors[19,31-33,56-58]. Moreover, epilepsy patients may be at higher risk for CAD due to several interrelated mechanisms. One hypothesis is that seizure-induced catecholamine release causes repetitive myocardial injury ('epileptic heart'), resulting in chronic myocardial and coronary artery damage, which promotes myocardial fibrosis, accelerated atherosclerosis, and cardiac dysfunction[60-62]. Additionally, certain antiepileptic drugs (AEDs) have been shown to adversely affect lipid metabolism and blood glucose levels, thereby increasing the risk of CAD[63-65]. Furthermore, seizures often trigger a stress response with elevated glucocorticoids, which may promote atherosclerosis by impairing endothelial function and reducing vasodilation, thereby increasing CAD risk[66]. Apart from the abovementioned effects between CAD and epilepsy, patients with CAD may have an increased risk of epilepsy due to several interrelated factors. CAD often reduces systemic blood flow, leading to cerebral ischemia and subsequent hypoxia. These conditions can alter brain electrophysiology, increasing susceptibility to epileptic activity[67,68]. Moreover, CAD is frequently accompanied by chronic inflammation and oxidative stress, both of which can contribute to neuronal damage and disrupt normal electrical activity in the brain, further elevating the risk of epilepsy[69].
Our study has several limitations. First, the observed association between epilepsy and CAD is based on evidence from cross-sectional and cohort studies, which precludes the establishment of a causal relationship. Second, epilepsy diagnoses in the UK Biobank relied on hospital records and self-report, which may be subject to misclassification, particularly with conditions that mimic epilepsy, such as syncope. Importantly, this inaccuracy is unlikely to be related to CAD status, meaning it would only weaken our results. Thus, the real associations between CAD and epilepsy may be stronger than we found. Third, although our observational analysis adjusted for a wide range of potential confounders, residual confounding cannot be entirely excluded. In particular, adjustment for the potential effects of antiseizure medications on CAD risk was not possible due to missing data. Fourth, as most participants in the UK Biobank were of UK descent, our findings may not be generalizable to other ethnic populations. Fifth, stroke is an important potential confounder in the CADepilepsy relationship, as it shares vascular risk factors with CAD and poststroke seizures may lead to an epilepsy diagnosis. To address this, we performed sensitivity analyses excluding participants with baseline stroke. Although the associations were attenuated, they remained directionally consistent, suggesting that stroke alone is unlikely to fully explain the observed associations. Sixth, due to data limitations, we lacked detailed clinical information on epilepsy (subtype, duration, seizure control, seizure frequency) and CAD severity, as well as more precise measures of body composition. These factors may modify cardiovascular risk and influence observed associations. For example, cumulative cardiac injury in chronic epilepsy may differ from new-onset cases, and CAD severity spectrum could affect prognosis. Future studies with richer clinical phenotyping and body composition data are needed to address these questions.
Conclusion
This study is the first to systematically reveal the bidirectional temporal relationship between CAD and epilepsy in a large-scale population, while also exploring the heterogeneity of this association across different subpopulations and its potential modification by genetic susceptibility. Our findings suggest that for these two highly comorbid conditions, interdisciplinary collaboration and integrated disease management should be strengthened to enable more effective health interventions and improved clinical outcomes.
DECLARATIONS
Acknowledgments
We sincerely thank the participants and staff of the UK Biobank for their invaluable contributions to this research. The graphical abstract was created with BioRender.com (Created in BioRender. Zhang, L. (2026)
Authors’ contributions
Conceptualization, methodology, investigation, visualization, writing - original draft, writing - review & editing, supervision: Zhang L
Conceptualization, Investigation, Data curation, Visualization, Writing - original draft, Writing - review & editing: Liu W
Methodology, data curation, validation: Xue Y, Wang L, Gong C
Supervision, project administration, funding acquisition: She ZG
Conceptualization, supervision, methodology, resources, writing - review & editing, funding acquisition, project administration: Li H
Availability of data and materials
The data supporting the findings of this study are available from the UK Biobank (https://www.ukbiobank.ac.uk/); however, restrictions apply to their availability, as they were used under license for the current study and are therefore not publicly available. The data are available from the authors upon reasonable request and with permission from the UK Biobank. Researchers can apply for access by submitting an application through the UK Biobank Access Management System (https://www.ukbiobank.ac.uk/enable-your-research/apply-for-access).
AI and AI-assisted tools Statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Science Foundation of China (82270390, JCRCFZ-2022-006), the Hubei Province Innovation Platform Construction Project (20204201117303072238), and the Jiangxi Province Thousand Talents Project (jxsq2023101002).
Conflicts of interest
Zhigang She is an Associate Editor of The Journal of Cardiovascular Aging. Zhigang She was not involved in any steps of the editorial process, notably including reviewer selection, manuscript handling, or decision-making, while the other authors have declared that they have no conflicts of interest.
Ethical approval and consent to participate
Ethical approval for the UK Biobank study was obtained from the North West Multi-centre Research Ethics Committee (MREC) (reference number: 21/NW/0157). This work was conducted under UK Biobank application number 77195. All participants provided written informed consent prior to participation. The research was conducted in accordance with the ethical standards of the 1964 Declaration of Helsinki and its later amendments.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Bauersachs R, Zeymer U, Brière JB, Marre C, Bowrin K, Huelsebeck M. Burden of coronary artery disease and peripheral artery disease: a literature review. Cardiovasc Ther. 2019;2019:8295054.
2. Leong DP, Joseph PG, McKee M, et al. Reducing the global burden of cardiovascular disease, part 2: prevention and treatment of cardiovascular disease. Circ Res. 2017;121:695-710.
3. Townsend N, Kazakiewicz D, Lucy Wright F, et al. Epidemiology of cardiovascular disease in Europe. Nat Rev Cardiol. 2022;19:133-43.
4. Soppert J, Lehrke M, Marx N, Jankowski J, Noels H. Lipoproteins and lipids in cardiovascular disease: from mechanistic insights to therapeutic targeting. Adv Drug Delivery Rev. 2020;159:4-33.
5. Wang W, Hu M, Liu H, et al. Global burden of disease study 2019 suggests that metabolic risk factors are the leading drivers of the burden of ischemic heart disease. Cell Metab. 2021;33:1943-56.e2.
6. Adams LA, Anstee QM, Tilg H, Targher G. Non-alcoholic fatty liver disease and its relationship with cardiovascular disease and other extrahepatic diseases. Gut. 2017;66:1138-53.
7. Fowkes R, Byrne M, Sinclair H, Tang E, Kunadian V. Coronary artery disease in patients with dementia. Coron Artery Dis. 2016;27:511-20.
8. Stakos DA, Stamatelopoulos K, Bampatsias D, et al. The Alzheimer's disease amyloid-beta hypothesis in cardiovascular aging and disease: JACC focus seminar. J Am Coll Cardiol. 2020;75:952-67.
9. Huang X, Wang Z, Lei F, et al. Association of urban environments with Atherosclerotic cardiovascular disease: A prospective cohort study in the UK Biobank. Environ Int. 2024;193:109110.
10. Sun T, Yang J, Lei F, et al. Artificial sweeteners and risk of incident cardiovascular disease and mortality: evidence from UK Biobank. Cardiovasc Diabetol. 2024;23:233.
11. Targher G, Byrne CD, Tilg H. NAFLD and increased risk of cardiovascular disease: clinical associations, pathophysiological mechanisms and pharmacological implications. Gut. 2020;69:1691-705.
12. Epilepsy Collaborators. Global, regional, and national burden of epilepsy, 1990-2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet Public Health. 2025;10:e203-27.
13. 2021 Nervous System Disorders Collaborators. Global, regional, and national burden of disorders affecting the nervous system, 1990-2021: a systematic analysis for the Global Burden of Disease Study 2021. Lancet Neurol. 2024;23:344-81.
14. Ding D, Zhou D, Sander JW, Wang W, Li S, Hong Z. Epilepsy in China: major progress in the past two decades. Lancet Neurol. 2021;20:316-26.
15. Fiest KM, Sauro KM, Wiebe S, et al. Prevalence and incidence of epilepsy: a systematic review and meta-analysis of international studies. Neurology. 2017;88:296-303.
16. De Luca R, Arrè V, Nardone S, et al. Gastrointestinal microbiota and inflammasomes interplay in health and disease: a gut feeling. Gut. 2025;75:161-75.
17. Symonds JD, Elliott KS, Shetty J, et al. Early childhood epilepsies: epidemiology, classification, aetiology, and socio-economic determinants. Brain. 2021;144:2879-91.
18. Singh G, Sander JW. The global burden of epilepsy report: Implications for low- and middle-income countries. Epilepsy Behav. 2020;105:106949.
19. Stefanidou M, Himali JJ, Devinsky O, et al. Vascular risk factors as predictors of epilepsy in older age: The Framingham Heart Study. Epilepsia. 2022;63:237-43.
20. Devinsky O, Spruill T, Thurman D, Friedman D. Recognizing and preventing epilepsy-related mortality: a call for action. Neurology. 2016;86:779-86.
21. Ellis CA, Petrovski S, Berkovic SF. Epilepsy genetics: clinical impacts and biological insights. Lancet Neurol. 2020;19:93-100.
22. Thurman DJ, Logroscino G, Beghi E, et al. The burden of premature mortality of epilepsy in high-income countries: A systematic review from the Mortality Task Force of the International League Against Epilepsy. Epilepsia. 2017;58:17-26.
23. Mayer J, Fawzy AM, Bisson A, et al. Epilepsy and the risk of adverse cardiovascular events: A nationwide cohort study. Eur J Neurol. 2024;31:e16116.
24. Fialho GL. Epilepsy and the heart: can 'brain arrhythmia' lead to cardiac arrhythmias? Back to the basics. Eur Heart J. 2024;45:853-4.
26. Husein N, Josephson CB, Keezer MR. Understanding cardiovascular disease in older adults with epilepsy. Epilepsia. 2021;62:2060-71.
27. Nyberg J, Aberg MA, Torén K, Nilsson M, Ben-Menachem E, Kuhn HG. Cardiovascular fitness and later risk of epilepsy: a Swedish population-based cohort study. Neurology. 2013;81:1051-7.
28. Neri S, Gasparini S, Pascarella A, et al. Epilepsy in cerebrovascular diseases: a narrative review. Curr Neuropharmacol. 2023;21:1634-45.
29. Hesdorffer DC, Tomson T, Benn E, et al. Combined analysis of risk factors for SUDEP. Epilepsia. 2011;52:1150-9.
30. Xu K, Gao X, Xia G, et al. Rapid gut dysbiosis induced by stroke exacerbates brain infarction in turn. Gut. 2021;70:1486-94.
31. Ioannou P, Foster DL, Sander JW, et al. The burden of epilepsy and unmet need in people with focal seizures. Brain Behav. 2022;12:e2589.
32. Terrone G, Salamone A, Vezzani A. Inflammation and epilepsy: preclinical findings and potential clinical translation. Curr Pharm Des. 2017;23:5569-76.
33. Simats A, Sager HB, Liesz A. Heart-brain axis in health and disease: role of innate and adaptive immunity. Cardiovasc Res. 2025;120:2325-35.
34. Wannamaker BB, Wilson DA, Malek AM, Selassie AW. Stroke after adult-onset epilepsy: a population-based retrospective cohort study. Epilepsy Behav. 2015;43:93-9.
35. Zack M, Luncheon C. Adults with an epilepsy history, notably those 45-64 years old or at the lowest income levels, more often report heart disease than adults without an epilepsy history. Epilepsy Behav. 2018;86:208-10.
36. Terman SW, Aubert CE, Hill CE, Skvarce J, Burke JF, Mintzer S. Cardiovascular disease risk, awareness, and treatment in people with epilepsy. Epilepsy Behav. 2021;117:107878.
37. Shah RA, Chahal CAA, Ranjha S, et al. Cardiovascular disease burden, mortality, and sudden death risk in epilepsy: a UK biobank study. Can J Cardiol. 2024;40:688-95.
38. Annegers JF, Hauser WA, Shirts SB. Heart disease mortality and morbidity in patients with epilepsy. Epilepsia. 1984;25:699-704.
39. Bucci T, Mbizvo GK, Rivera-Caravaca JM, et al. Epilepsy-heart syndrome: incidence and clinical outcomes of cardiac complications in patients with epilepsy. Curr Probl Cardiol. 2023;48:101868.
40. Sudlow C, Gallacher J, Allen N, et al. UK biobank: an open access resource for identifying the causes of a wide range of complex diseases of middle and old age. PLoS Med. 2015;12:e1001779.
41. Bycroft C, Freeman C, Petkova D, et al. The UK Biobank resource with deep phenotyping and genomic data. Nature. 2018;562:203-9.
42. Thompson DJ, Wells D, Selzam S, et al. A systematic evaluation of the performance and properties of the UK biobank polygenic risk score (PRS) release. PLoS ONE. 2024;19:e0307270.
43. International League Against Epilepsy Consortium on Complex Epilepsies. Electronic address: epilepsy-austin@unimelb.edu.au. Genetic determinants of common epilepsies: a meta-analysis of genome-wide association studies. Lancet Neurol. 2014;13:893-903.
44. International League Against Epilepsy Consortium on Complex Epilepsies. GWAS meta-analysis of over 29,000 people with epilepsy identifies 26 risk loci and subtype-specific genetic architecture. Nat Genet. 2023;55:1471-82.
45. Heyne HO, Pajuste FD, Wanner J, et al. Polygenic risk scores as a marker for epilepsy risk across lifetime and after unspecified seizure events. Nat Commun. 2024;15:6277.
46. Klarin D, Natarajan P. Clinical utility of polygenic risk scores for coronary artery disease. Nat Rev Cardiol. 2022;19:291-301.
48. Mathur MB, Ding P, Riddell CA, VanderWeele TJ. Web site and R package for computing E-values. Epidemiology. 2018;29:e45-7.
49. VanderWeele TJ, Ding P. Sensitivity analysis in observational research: introducing the E-value. Ann Intern Med. 2017;167:268-74.
50. Costagliola G, Orsini A, Coll M, Brugada R, Parisi P, Striano P. The brain-heart interaction in epilepsy: implications for diagnosis, therapy, and SUDEP prevention. Ann Clin Transl Neurol. 2021;8:1557-68.
53. Rana A, Musto AE. The role of inflammation in the development of epilepsy. J Neuroinflammation. 2018;15:144.
54. International League Against Epilepsy Consortium on Complex Epilepsies. Genome-wide mega-analysis identifies 16 loci and highlights diverse biological mechanisms in the common epilepsies. Nat Commun. 2018;9:5269.
55. Sivathamboo S, Liu Z, Sutherland F, et al. Serious cardiac arrhythmias detected by subcutaneous long-term cardiac monitors in patients with drug-resistant epilepsy. Neurology. 2022;98:e1923-32.
56. Hall SA, Lesniewski LA. Targeting vascular senescence in cardiovascular disease with aging. J Cardiovasc Aging. 2024;4:16.
57. Muthu S, Tran Z, Thilagavathi J, et al. Aging triggers mitochondrial, endoplasmic reticulum, and metabolic stress responses in the heart. J Cardiovasc Aging. 2025;5:4.
58. Sun Q, Karwi QG, Wong N, Lopaschuk GD. Advances in myocardial energy metabolism: metabolic remodelling in heart failure and beyond. Cardiovasc Res. 2024;120:1996-2016.
59. Dichgans M, Malik R, König IR, et al. Shared genetic susceptibility to ischemic stroke and coronary artery disease: a genome-wide analysis of common variants. Stroke. 2014;45:24-36.
60. Verrier RL, Pang TD, Nearing BD, Schachter SC. Epileptic heart: a clinical syndromic approach. Epilepsia. 2021;62:1780-9.
62. Wang J, Huang P, Yu Q, et al. Epilepsy and long-term risk of arrhythmias. Eur Heart J. 2023;44:3374-82.
63. Chuang YC, Chuang HY, Lin TK, et al. Effects of long-term antiepileptic drug monotherapy on vascular risk factors and atherosclerosis. Epilepsia. 2012;53:120-8.
64. Beghi E, Bizzi A, Codegoni AM, Trevisan D, Torri W. Valproate, carnitine metabolism, and biochemical indicators of liver function. Collaborative Group for the Study of Epilepsy. Epilepsia. 1990;31:346-52.
65. Li J, Shlobin NA, Thijs RD, et al. Antiseizure medications and cardiovascular events in older people with epilepsy in the canadian longitudinal study on aging. JAMA Neurol. 2024;81:1178-86.
66. MacLeod C, Hadoke PWF, Nixon M. Glucocorticoids: fuelling the fire of atherosclerosis or therapeutic extinguishers? Int J Mol Sci. 2021:22.
67. Xu Y, Fan Q. Relationship between chronic hypoxia and seizure susceptibility. CNS Neurosci Ther. 2022;28:1689-705.
68. Grigg-Damberger M, Foldvary-Schaefer N. Hypoxia not AHI in adults with sleep apnea midlife markedly increases risk of late-onset epilepsy-Carosella CM et al Sleep apnea, hypoxia, and late-onset epilepsy: the Atherosclerosis Risk in Communities study SLEEP-2023-0175.R1. Sleep. 2024;47:zsad252.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.