fig17
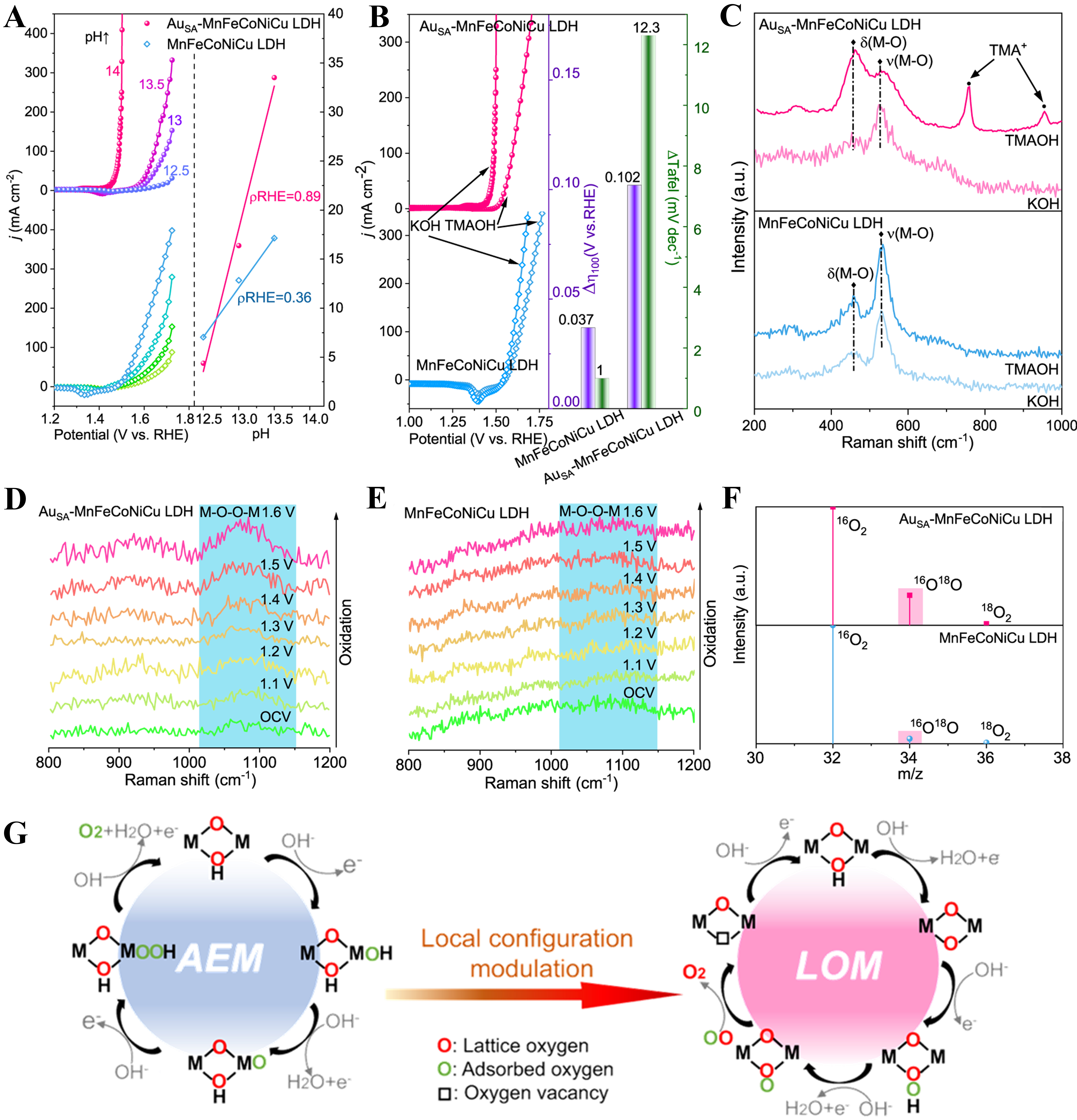
Figure 17. (A) LSV curves in KOH and log j vs. pH plot at 1.45 V vs. RHE for proton reaction order; (B) Polarization curves of AuSA-MnFeCoNiCu LDH and MnFeCoNiCu LDH in 1.0 M KOH/TMAOH, with corresponding Δη100 and Tafel slope variations; (C) Raman spectra of AuSA-MnFeCoNiCu LDH and MnFeCoNiCu LDH after 30 min electrolysis at 1.45 V vs. RHE in 1.0 M KOH/TMAOH; (D) In situ Raman spectra of MnFeCoNiCu LDH; (E) In situ Raman spectra of AuSA-MnFeCoNiCu LDH; (F) 18O isotope-labeling mass spectra; (G) Adsorbate evolution mechanism and lattice oxygen oxidation mechanism on MnFeCoNiCuOOH. (A-G) Reprinted with permission from Ref.[131]. Copyright 2023, Springer Nature. AEM: Adsorbate evolution mechanism; LOM: lattice oxygen mechanism; LSV: linear sweep voltammetry; SA: single atom; RHE: reversible hydrogen electrode; Δη100: refers to the overpotential at a current density of 100 mA cm-2.