Challenges, strategies and prospects in interfaces between Li metal anode and polyether-based solid-state electrolytes
Abstract
The high fluidity and flammability of conventional organic liquid electrolyte pose critical safety risk and lead to premature cell failure in commercial lithium-ion batteries. Solid polymer electrolytes, particularly polyether-based systems such as polyethylene oxide, are promising alternatives to liquid electrolytes, enabling safer, high-energy-density lithium metal batteries. However, polyether-based all-solid-state batteries (ASSBs), like liquid systems, face significant interfacial challenges - including chemical instability, poor contact, and slow ionic transport - that reduce overall performance. This review systematically surveys the evolution and categories of polyether-based electrolytes and provides a comprehensive analysis of interfacial phenomenon that undermines their application potential in ASSBs. We highlight recent advances in interfacial engineering strategies, including the optimization of polyether electrolyte, the design of in situ/ex situ solid state electrolyte interphases layers, and the development of composite Li metal anode. Finally, we outline promising directions and opportunities for addressing the interfacial bottlenecks to accelerate the practical realization of high-performance polyether-based solid state Li metal batteries.
Keywords
INTRODUCTION
Since their commercialization in 1991 by Sony Corporation, lithium-ion batteries have demonstrated remarkable advantages in fields such as portable electronic devices and smart vehicles, profoundly impacting human daily life. However, due to the fluidity and flammability of organic liquid electrolytes, conventional lithium-ion batteries suffer from potential safety hazards such as thermal runaway, electrolyte leakage and explosions[1,2]. Conventional lithium-ion batteries are approaching theoretical energy-density limits, making them insufficient to meet the growing demand for next-generation energy-storage systems with higher energy and power densities. The development of energy dense, high safety lithium-based energy storage systems is imminent.
The use of solid-state electrolytes (SSEs) instead of traditional liquid electrolytes in lithium battery systems is considered an ideal strategy to address current safety issues. The SSEs can effectively avoid the electrolyte leakage and combustion caused by safety issues since they are non-volatile, non-flammable and inherently resistant to leakage[3,4]. More importantly, SSEs are regarded as highly compatible with high-voltage cathodes and lithium metal anodes, facilitating the development of next-generation energy-dense systems such as Li-S and Li-O2 batteries. Employing lithium metal as the anode is essential for achieving high energy density of cell due to its exceptional specific capacity (3,860 mAh·g-1) and extremely low electrochemical potential (-3.04 V versus SHE)[5,6]. Although metallic lithium was historically employed before graphite anode in lithium-ion batteries, its practical employment has been hindered by the severe dendrite growth on the lithium metal in the liquid electrolyte[7].
SSEs have long been regarded as the most viable solution for avoiding the dendrite formation in high energy-density batteries with lithium metal anodes. Generally, SSEs can be classified into oxide ceramic electrolytes, sulfide electrolytes, and polymer electrolytes[8]. In Figure 1A-C, representative garnet-type oxide, argyrodite-type sulfide, and polyether-based polymer electrolytes are compared in terms of their key characteristics. The oxide and sulfide electrolytes exhibit high ionic conductivity, whereas polymer electrolytes demonstrate advantages in flexibility, cost-effectiveness, processability, and environmental stability. Owing to their intrinsic mechanical compliance, solid polymer electrolytes (SPEs) enable improved interfacial contact and reduced mechanical mismatch during cycling. In addition, their solution- or melt-based processing allows simple fabrication at low cost. These integrated merits make polyether-based SPEs, particularly those based on polyethylene oxide (PEO), highly attractive for next-generation flexible and high-safety lithium metal batteries. In polyether-based SPEs, the ether groups coordinate with lithium ions to facilitate salt dissociation, while the flexible polymer chains support efficient lithium-ion transport. Based on the type of polyether matrix, SPEs can be categorized into PEO, polyethylene glycol (PEG), and poly(1,3-dioxolane) [P(DOL)] [Figure 2A].
Figure 1. Radar charts illustrating the key properties of three representative solid-state electrolyte systems: (A) Garnet-type oxide electrolyte; (B) Argyrodite-type sulfide electrolyte; (C) Polyether-based polymer electrolyte.
Figure 2. (A) Chemical structures of PEO, poly(poly(ethylene glycol) methyl ether methacrylate) [P(PEGMA), a PEG-derived polyether] and P(DOL); (B) Key interfacial challenges of polyether electrolytes against Li metal anodes and corresponding modification strategies. PEO: Polyethylene oxide; P(PEGMA): poly[poly(ethylene glycol) methyl ether methacrylate]; PEG: polyethylene glycol; P(DOL): poly(1,3-dioxolane); SPE: solid polymer electrolyte; SEI: state electrolyte interphases; 3D: three-dimensional.
Despite early optimism, there is growing evidence that Li dendrites can still form and propagate in polyether SPE-based cells during cycling even under limited current densities. The critical current density (CCD), defined as the maximum current density that can be sustained without short circuit, is generally limited to approximately 0.2-0.5 mA·cm-2 under commonly reported laboratory conditions [Table 1]. Furthermore, the short circuit in all-solid-state batteries (ASSBs) occurs even faster than that in liquid electrolyte systems, contradicting early expectations. Considering the complex chemical and physical properties at the electrode-electrolyte interface, the mechanism of dendrite growth in polyether-based SPEs is far more intricate than initially expected. These interfacial failures are not governed by a single factor but arise from the intrinsic coupling among chemical reactivity, mechanical instability, and ion transport limitations[26].
Summary of reported CCD values of polyether-based SPEs and corresponding testing parameters
| SPEs | Thickness (µm) | Temperature (°C) | Salt type | Areal capacity (mAh·cm-2) | CCD (mA·cm-2) | References |
| PEGDMA-IL-LiTFSI | 70 | 30 | LiTFSI | 0.5 | 0.5 | [9] |
| HBPC-SEs | / | 60 | LiTFSI | 0.25 | 0.4 | [10] |
| M-S-PEGDA | 50 | 25 | LiTFSI | 0.25 | 0.5 | [11] |
| PM-PEO | 78 | 65 | LiTFSI | 0.3 | 0.3 | [12] |
| PEGDA-TA-P(VDF-HFP) | 50 | 25 | LiTFSI | 0.2 | 0.4 | [13] |
| CA-PEGMEA-SN | 40 | 25 | LiTFSI | 0.5 | 0.5 | [14] |
| PEGMA-LAGP-Li | 8.5 | 30 | LiTFSI | 0.5 | 0.5 | [15] |
| PEO/LiTFSI | / | 60 | LiTFSI | 0.45 | 0.2 | [16] |
| PEO/Zr-MOF | 143 | 60 | LiTFSI | 0.43 | 0.43 | [17] |
| PEO-UIO66 | 100 | 60 | LiTFSI | 0.3 | 0.3 | [18] |
| PEO-3D-LLZAO | 160 | 60 | LiTFSI | 0.4 | 0.4 | [19] |
| PEO/SN/MOF | 100 | 60 | LiTFSI | 0.4 | 0.4 | [20] |
| PAN-PEO-3D-LLZAO | 150 | 60 | LiTFSI | 0.44 | 0.44 | [21] |
| PEO/BaTiO3 @PAN/MWCNT | 70 | 25 | LiClO4 | 0.5 | 0.5 | [22] |
| 3D Zeo/PEO | / | 25 | LiTFSI | 0.21 | 0.42 | [23] |
| PDOL | / | 30 | LiTFSI | 0.25 | 0.5 | [24] |
| P-PETEA-MOF | 100-150 | 25 | LiTFSI | 0.5 | 0.5 | [25] |
Although several recent reviews have summarized the Li metal-polymer electrolyte interface from material or strategy-oriented perspectives, a mechanism-centered analysis specifically focused on polyether systems remains scarce[27,28]. In particular, the fundamental interfacial challenges and corresponding regulation strategies related to chemical reactivity, mechanical instability, and ion-transport limitations have not been systematically organized. Therefore, a problem-oriented and integrative analysis is urgently required.
In this review, the development of polyether-based SPEs, the anode-electrolyte interface issues in ASSBs, the corresponding Li dendrite suppression strategies are systematically discussed and summarized. We first introduce the evolution and classification of polyether electrolytes. We then examine the major interfacial issues in polyether SPE-based ASSBs including chemical instability, mechanical stability, and sluggish ion transport at the Li/SPE interface. In addition, we briefly discuss the techno-economic aspects of polyether-based SPEs to assess their practical deployment potential. Finally, we underscore recent advances in interfacial engineering, including polyether structure optimization, in situ and ex-situ artificial solid state electrolyte interphases (SEI) design, and the development of composite Li metal anodes [Figure 2B]. We conclude by outlining future research directions to guide the rational design of polyether electrolytes for advanced solid-state lithium batteries.
POLYETHER ELECTROLYTE
Polyether-based SPEs generally consist of lithium salts dissolved in an ether-based polymer matrix, which serves both as a host for the salts and as a medium for Li+ conduction[29]. Ion transport occurs through coordination between Li+ ions and ether oxygen lone pairs, involving migration along polymer chain segments and hopping between coordinating sites facilitated by local chain dynamics[30]. At room temperature, the high crystallinity of many polyether matrices restricts chain mobility, resulting in slow Li+ transport. Lithium salt type and concentration affect both salt dissociation and polymer crystallinity, thereby influencing ionic conductivity. In addition to optimizing the lithium salt, functional additives such as plasticizers, ionic liquids, or inorganic fillers are commonly employed to enhance chain mobility and improve Li+ transport[31]. Given the diversity of polyether matrices and tuning strategies, the following subsections review the main polyether electrolytes, including PEO, PEG derivatives, and P(DOL), focusing on their structural features, ion transport mechanisms, and the effects of additives on ionic transport properties.
PEO-based electrolyte
PEO offers several advantages as an electrolyte matrix, including good membrane-forming capability, lightweight nature and high lithium salt solubility. The conductive behavior of alkali metal ions in PEO was first found by Wright et al.[32] in 1973. Armand et al.[33] subsequently proposed the application of PEO-based polymer electrolytes in lithium-ion batteries, opening a new direction for polymer electrolyte research. However, PEO tends to crystallize due to its regular -CH2-CH2-O- structure. This promotes ordered chain packing and lamellar crystal formation [Figure 3A and B][34,35]. High-molecular-weight PEO remains largely crystalline at ambient temperature, severely restricting segmental chain mobility and leading to low ionic conductivity (10-8-10-7 S·cm-1) [Figure 3C][35,39], ultimately compromising the rate capability and energy output of ASSBs. Consequently, considerable research has focused on suppressing crystallinity and enhancing room-temperature transport performance in PEO-based SPEs.
Figure 3. (A) Chemical structure of PEO; (B) Scheme of semi-crystalline PEO; Figure 3B is reprinted with permission from Ref. [34], Copyright © 2015 Royal Society of Chemistry.; (C) Ionic conductivities of PEO electrolyte with various Li salts. Figure 3C is reprinted with permission from Ref. [35], Copyright © 2020 Wiley; (D) Scheme for the synthesis of P(PEGDE). Figure 3D is reprinted with permission from Ref. [36], Copyright © 2019 American Chemical Society; (E) The optical images of (α) PEGDE solution and (β) crosslinked solid electrolyte pure P(PEGDE); (F) The temperature dependence of ionic conductivity for the composite P(PEGDE) polymer electrolytes. Figure 3E and F is reprinted with permission from Ref. [37], Copyright © 2017 Wiley; (G) Reaction mechanism illustrating how LiDFOB initiates polymerization of DOL; (H) Ionic conductivity of the poly-DOL electrolyte versus temperature. Figure 3G and H is reprinted with permission from Ref. [38], Copyright © 2020 Royal Society of Chemistry. PEO: Polyethylene oxide; P(PEGDE): poly[poly(ethylene glycol) diglycidyl ether]; PEGDE: poly(ethylene glycol) diglycidyl ether; LiDFOB: lithium difluoro(oxalato)borate; DOL: 1,3-dioxolane.
In PEO-based SPEs, plasticizers such as succinonitrile or ionic additives (e.g., phosphoric acid and sulfonated polysulfone) are often introduced to weaken intermolecular interactions between PEO chains, thereby enhancing segmental mobility and ionic conductivity[40,41]. Nevertheless, the electrochemical stability of PEO in Li metal battery systems remains largely underexplored. Most attention has focused on the degradation of PEO on the high-voltage cathode side[42-45], relatively few investigations have concentrated on the formation and stability of the SEI layer on the Li metal anode side.
PEG-derived electrolytes: P(PEGMA), P(PEGDA), and P(PEGDE)
Poly(ethylene glycol) methyl ether methacrylate (PEGMA) and poly(ethylene glycol) diglycidyl ether (PEGDE) are PEG derivatives featuring polymerizable groups, can form amorphous, crosslinked networks through chemical reactions, providing a solution to the crystallization issue of high-molecular-weight PEO. Their tunable crosslinked architectures render them particularly suitable for use as solid or gel electrolytes in lithium metal batteries. The tunable crosslinking degree offers additional flexibility to tailor mechanical stability and electrochemical performance of the electrolyte matrix.
PEGMA contains terminal vinyl groups that undergo free radical polymerization to form a chemically crosslinked gel-like polymer network [P(PEGMA)]. Nava et al.[46] prepared amorphous P(PEGMA) electrolyte via free-radical polymerization of the PEGMA monomer with LiPF6 incorporated. Unlike the strong interaction forces to solvate Li + ions in the PEO crystalline region, the amorphous morphology of P(PEGMA) forms weak coordination shells with Li + ions, enabling the enhanced ionic conductivity
Poly(ethylene glycol) diacrylate (PEGDA) contains two terminal acrylate groups and is most often polymerized via ultraviolet (UV)-induced photopolymerization. The resulting crosslinked network exhibits low crystallinity, enhanced mechanical robustness, and favorable film-forming characteristics. Zhang et al.[48] developed a PEGDA-based gel polymer electrolyte (GPE) by in-situ UV curing of PEGDA in the presence of LiPF6 and ethylene carbonate (EC)/diethyl carbonate (DEC). The resulting GPE showed high ionic conductivity (~10-3 S·cm-1) due to Li + transport within the swollen gel matrix, but associated with low mechanical strength. To overcome this drawback, Yuan et al.[49] designed an ultrathin (16 μm) composite polymer electrolyte (PE-CPE) by UV polymerizing PEGDA with succinonitrile (SN) and LiTFSI within a polyethylene (PE) separator framework. This PE-CPE achieved high room-temperature conductivity (1.11 × 10-4 S·cm-1) and excellent mechanical strength (6.37 MPa). Solid-state LiCoO2|Li cells using this electrolyte delivered 145.3 mAh·g-1 at 0.2 C and retained 76.1% capacity after 385 cycles, demonstrating the viability of PEGDA-based in-situ cured electrolytes for high-efficiency, flexible all-solid-state cells.
In contrast to PEGMA and PEGDA, PEGDE undergoes cationic ring-opening polymerization, typically initiated by Lewis acid catalysts such as lithium salts[36]. The resulting P(PEGDE) networks are chemically stable and amorphous, combining moderate mechanical flexibility with enhanced ion transport [Figure 3D and E]. For example, Cui et al.[37] reported that a crosslinked PEGDE-based electrolyte prepared by lithium salt LiDFOB-induced cationic polymerization of PEGDE. This crosslinked framework inhibits crystallization, resulting in an amorphous phase and excellent ionic conductivity of 8.9 × 10-5 S·cm-1 at ambient temperature [Figure 3F]. Similarly, Nair et al.[36] also synthesized a cross-linked PEGDE-based electrolyte using LiBF4 salt to initiate the polymerization reaction, demonstrating excellent cycling performance in LiFePO4-based lithium metal batteries. However, in most cases, the crosslinked structure also restricts the free movement of the polymer chains, leading to a limited increase in ionic conductivity.
P(DOL)-based electrolyte
1,3-Dioxolane (DOL) is commonly utilized as a solvent or co-solvent in liquid electrolytes due to its excellent Li + transport capabilities. Liu et al.[38] and Ma et al.[50] found that DOL can form a amorphous polymer matrix for the polyether electrolyte via ring-opening polymerization induced by the PF5 Lewis acid [Figure 3G], resulting in high ionic conductivity (> 10-5 S·cm-1 at 25 ℃) [Figure 3H]. Zhao et al.[51] demonstrated that the in situ formed P(DOL) electrolyte initiated by Al(OTf)3 salt performs well in Li-S and Li-LiFePO4 batteries. Owing to its smaller molecular structure, DOL exhibits enhanced dielectric properties and improved fluidity compared with PEGMA and PEGDE. Consequently, the DOL monomer can sufficiently penetrate the pores of the electrode, enabling the intimate electrode-electrolyte contact and the formation of well-connected ion-conductive pathways inside the electrode following in situ polymerization.
To further enhance the performance of P(DOL)-based electrolytes, various functional components are often incorporated to improve both ionic and mechanical properties. Salt additives, such as Sn(OTf)2 and Al(OTf)3, together with inert fillers (e.g., SnF2, AlF3), primarily facilitate interfacial Li+ transport and can promote the formation of Li-Al and Li-Sn alloy species at the interface, thereby improving interfacial stability[52]. Meanwhile, supporting skeletons, such as Li6.4La3Zr1.4Ta0.6O12 layers or polyimide nanofiber films, act as robust structural frameworks that reinforce the electrolyte and suppress deformation during cycling[52]. Despite these improvements, P(DOL)-SPE still suffers from limited thermal stability. It tends to decompose at 110 °C or even at the lower temperature of 40 °C under vacuum[53]. Such decomposition not only leads to gas evolution and swelling of pouch cells but can also disrupt the electrode-electrolyte interface and promote uneven Li deposition. To address these issues and enable safer solid-state cells with P(DOL)-based electrolytes, the formation of a high-performance SEI layer is necessary to guide Li deposition and mitigate thermal degradation.
Environmental impact of polyether-based SPEs
Besides electrochemical performance, the environmental impact of polyether-based SPEs is another crucial consideration. Life cycle assessment (LCA) data are primarily available for PEO/LiTFSI, as reported by Larrabide et al.[54], which exhibits the lowest global warming potential (GWP) among representative SPEs, at approximately 0.37 kg CO2-equivalent per gram of PEO electrolyte. The polymer matrix itself contributes less than 0.6% of the total impact, indicating that LiTFSI and solvent usage dominate the environmental burden. Sensitivity analyses suggest that environmental impacts can be further reduced through solvent recovery, the use of less fluorinated lithium salts, and improved energy efficiency during fabrication. Despite its favorable environmental profile, PEO/LiTFSI suffers from relatively low room-temperature ionic conductivity, necessitating further optimization. Alternative designs, such as block copolymers (e.g., PCL-PPC-PCL/LiTFSI), may offer a better balance between electrochemical performance and reduced environmental footprint. Environmental assessments for PEG-derived and P(DOL)-based SPEs remain limited[55], highlighting the need for further investigation of these emerging systems.
ANODE/ELECTROLYTE INTERFACIAL ISSUES
The interface between the lithium metal anode and polyether-based SPEs is governed by coupled chemical reactions, mechanical stress, and Li+ transport. These factors collectively determine battery lifetime and energy density. During cycling, the high reactivity of lithium can partially decompose the polyether electrolyte, forming a SEI with heterogeneous composition. Variations in SEI composition and structure, combined with its limited Li+ conductivity, lead to localized ionic fluxes, promoting uneven lithium deposition and dendrite formation[56,57]. Mechanical stress and volumetric changes associated with lithium plating and stripping further disrupt interfacial contact, causing cracks or local SEI failure, which in turn exacerbates uneven ion distribution and dendrite formation[58]. The interplay among chemical, mechanical, and ionic transport processes establishes a persistent feedback loop. Understanding this coupled mechanism is crucial for analyzing interfacial chemical and mechanical instabilities, as well as the limitations of ion transport.
Chemical instability
Due to the strong chemical reactivity of lithium, pronounced parasitic reactions are typically observed at the Li/PEO interface[59-62]. In 1985, Fauteux first identified the presence of the high-resistance passivating film on a lithium electrode coupled with PEO-based SPE using AC impedance methods[63]. Moreover, Fauteux and Croce et al.[64,65] demonstrated that the passivating film progressively thickened, leading to a steady increase in interfacial impedance that eventually dominated the overall cell resistance. Furthermore, as the critical information, the composition of the passivating film was further investigated. Lisowska-Oleksiak[66] and Le Granvalet-Mancini et al.[67] demonstrated that Li/PEO-LiX SEI consisted of lithium alkoxides Li-O-R and Li-O-R-O-Li. Notably, around the same period, Kong et al.[68] used the SEI to describe the passivating film and reported that the interphase formed at the Li/PEO-LiN(CF3SO2)2 interface is approximately 12 nm thick, primarily consisting of Li2O, along with minor components such as LiF, Li2CO3 and LiOH. Building on these earlier studies, recent work by Elena E. Ushakova’s group and Sheng’s group[69,70] further validated the SEI composition on lithium metal anodes in PEO-based SPE. Advanced in-situ X-ray spectroscopy (XPS) and transmission electron microscopy (TEM) techniques revealed the presence of lithium alkoxides (ROLi), Li2O, LiOH, Li2CO3, and LiF species [Figure 4A-C].
Figure 4. (A) Recognized crystalline grains of Li metal, Li2O, Li2CO3, and LiOH in interface; (B) The corresponding FFT of (A), red circle: Li, 2.48 Å, yellow circle: Li2O, 2.66 Å, blue circle: Li2CO3, 3.02 Å, white circle: LiOH, 4.35 Å; (C) Schematic diagram for the identified mosaic structure of interface. Figure 4A-C is reprinted with permission from Ref. [70], Copyright © 2020 Wiley. FFT: Fast Fourier transform; SPE: solid polymer electrolyte.
Mechanical instability
Although SPEs show better interfacial adhesion than oxide and sulfide electrolytes, their viscoelastic nature and limited mechanical compliance still cause stress accumulation during cycling. Repeated lithium stripping and plating induce significant volume fluctuations of the Li metal anode[58]. In this process, the mechanical loads are shared between the SEI and the surrounding polyether electrolyte, with the SEI serving as the main mechanical buffer at the Li/electrolyte interface.
This coupled mechanical response is essential for preserving interfacial integrity. The elastic properties of individual SEI components differ significantly [Table 2]. Nanoscale mechanical measurements, including atomic force microscopy (AFM), reveal that crystalline LiF and Li2O components have high Young’s moduli but low Pugh’s ratios (B/G < 1.75)[74], indicating their brittleness and susceptibility to crack formation under interfacial stress. In contrast, amorphous Li2CO3 component exhibits a higher B/G ratio (> 1.75), allowing better plastic deformation and stress accommodation, although its lower stiffness may limit dendrite suppression[75]. Besides, depth-resolved characterizations such as X-ray photoelectron spectroscopy (XPS) depth profiling and time-of-flight secondary ion mass spectrometry (TOF-SIMS) further reveal pronounced compositional heterogeneity across the SEI thickness[76,77]. Such heterogeneous distribution of stiff-brittle (LiF, Li2O) and relatively ductile (Li2CO3) domains results in non-uniform stress and localized cracking during cycling, which accelerates dendrite growth and capacity decay of the cell.
Elastic properties and Pugh’s ratio (B/G) of major SEI components with different microstructures
| SEI components | Microstructure | Young’s modulus | Bulk modulus (B) | Shear modulus (G) | B/G |
| LiF | Crystalline | 135.3 | 89.4 | 54.2 | 1.65 |
| Amorphous | 58.1 | 45.5 | 21.1 | 2.16 | |
| Li2CO3 | Crystalline | 54.8 | 50.5 | 22.2 | 2.27 |
| Amorphous | 36.2 | 43.1 | 13.3 | 3.24 | |
| Li2O | Crystalline | 108 | ~57 | 45.6 | 1.25 |
Sluggish ionic transport
Beyond mechanical and chemical instabilities, Li+ transport across the anode-electrolyte interface often limits the overall reaction kinetics of the cell. This interfacial process therefore governs rate capability and Li stripping/plating behavior in full cells. In recent decades, extensive research has focused on enhancing the ionic conductivity of PEO SPEs to levels comparable with those of commercial liquid electrolytes. However, the interphase components resulting from polyether decomposition exhibit retarded ionic conductivity
STRATEGIES OF OPTIMIZING SOLID ELECTROLYTE INTERPHASE
Improvement of polymer electrolyte
Adopting polyether electrolytes with enhanced elastic property
A major challenge for polyether-based electrolytes is their poor adaptability to the dynamic volume changes of lithium metal anodes, which often leads to interfacial instability during cycling. To address this, researchers have incorporated elastic networks into polyether matrices, aiming to enhance mechanical resilience and maintain stable electrode-electrolyte contact.
For instance, Cui et al.[83] designed an elastic Li-ion conductor with dual crosslinks for a polyether-based electrolyte, consisting of poly (propylene oxide) (PPO) elastomer, SiO2 nanoparticles, LiTFSI, and propylene carbonate (PC). The integration of dynamic amide hydrogen bonds from Jeffamine precursors within the chemically crosslinked PPO matrix imparts high mechanical resilience to the SPE (0.32 MJ·m-3). Using this elastic electrolyte, the LiFePO4|Li cell shows excellent cycling stability, with a capacity retention rate of 94% after 100 cycles. Similarly, Guo et al.[84] reported a highly stretchable and elastic polyther-based electrolyte by in situ incorporating Li salts and PEO chains into an elastomeric poly(urethane-urea) matrix (PU-EO12/LiTFSI/TEG41%). In this electrolyte, the reversible noncovalent crosslinking and permanent covalent crosslinking formed between poly(urethane-urea) and PEO chains impart the SPE with pronounced elastic recovery behavior and mechanical resilience. The Li|Li symmetric cell using this electrolyte demonstrated extended operational resilience with a low polarization of 80 mV over 2,200 h at 0.1 mA·cm-2, attributed to the formation of a self-adaptive, continuous ion transport interface [Figure 5A-C]. Later, Zheng et al.[86] synthesized an elastic polyurethane (PU) polymer electrolyte matrix via polyaddition of poly(ethylene glycol) (PEG) and isocyanate. The PU chains were crosslinked into a polymer network via hydrogen bonding (N-H···O), resulting in a breaking elongation of up to 3,000%. This elasticity allowed the electrolyte to maintain intimate electrode contact during cycling, delivering stable performance in LFP|Li cells over 700 cycles at 0.5 C.
Figure 5. (A) Schematic illustration of the elastomeric polymer network of the PU electrolyte; (B) Schematic illustration of the self-adaptive electrolyte-electrode interface; (C) Photographs showing the adhesion of the PU-EO12/LiTFSI/TEG41% electrolyte to LiFePO4 cathode material. Figure 5A-C is reprinted with permission from Ref. [84], Copyright © 2021 Royal Society of Chemistry; (D) The chemical structure of electrolyte; (E) Long-term cycling performance of Li/Li symmetric cells using PCPE-550 and PEO-SPE at
More recently, a novel 3D interconnected elastic polymer electrolyte (PCEE) was designed based on PEGDA incorporating a plastic crystalline succinonitrile (SN) phase[87]. The in situ polymerized PCEE effectively accommodated volume changes during cycling, enabling a LiFePO4|PCEE|Li full cell to maintain a discharge capacity of 93 mAh·g-1 at 1 C over 1,000 cycles, with minimal capacity fading (only 0.005% per cycle). In a further advancement, Ren et al.[85] constructed a supramolecularly crosslinked polyether electrolyte (PCPE) by incorporating a tri-armed boron-centered oligomer (MBO) into a PEO/LiTFSI matrix [Figure 5D]. The flexible methoxy-terminated polyethylene glycol (MPEG) arms and dynamic Lewis acid-base interactions formed a mechanically robust and elastic polymer network, which improved structural adaptability at the Li interface. As a result, the electrolyte exhibited an enhanced elastic modulus of 1.84 MPa, enabling dendrite-free cycling for over 2,600 h and stable performance in Li symmetric cells [Figure 5E].
Providing polyether electrolytes with high mechanical strength
High-modulus SPE membranes act as physical obstacles to lithium dendrite propagation. For polyether-based electrolytes, stiffness enhancement strategies include blending or copolymerizing with rigid polymer segments, designing cross-linked polyether chains, or adding inorganic fillers and mechanical scaffolds.
Blending/copolymerization with rigid polymers. Generally, PEO segments function as the ionic conductor, while the rigid block contributes mechanical strength. Li et al.[88] blended PEO with high-strength poly(m-phenylene isophthalamide) (PMIA) in a suitable ratio, significantly enhancing the Young’s modulus of pristine PEO SPE from 0.98 to 37.07 MPa. The mechanically robust PMIA matrix effectively suppressed lithium dendrite growth, endowing the PMIA|PEO SPE-based Li|Li symmetric cell with extended cycling stability of 468 h at 0.1 mA·cm-2. However, the simple blending of these two segments leads to poor miscibility and macro-phase separation, resulting in uneven components distribution within the membrane.
To overcome this issue, covalent linkage between polyether segments and rigid blocks in copolymers has been explored to improve compatibility and mechanical integrity[89-91]. Zhang et al.[92] fabricated a PEO-b-poly(trimethyl-N-{[2-(dimethylamino)ethyl methacrylate]-7-propyl}-ammonium bis(trifluoromethanesulfonyl)imide) (PEO-b-PDM-dTFSI) copolymer. Fortunately, the covalent linking of polyether and rigid segments in copolymers can significantly enhance the intrinsic compatibility between these components. The presence of the rigid PDM-dTFSI block endows the PEO-b-PDM-dTFSI/LiTFSI SPE with high mechanical strength (104 Pa) even at 100 ℃, which is approximately 10,000 times greater than that of the PEO/LiTFSI electrolyte [Figure 6A and B]. Furthermore, the tendency for phase separation was nearly eliminated by adjusting the block ratio of PDM-dTFSI and PEO, as well as the content of LiTFSI salts. The Li|Li symmetric cell utilizing this copolymer SPE demonstrates long-term stability over 1,000 h at
Figure 6. (A) Schematic illustration of the fabrication of SPEs; (B) Storage moduli (G’) of the PEO114-b-PDMn-dTFSI/LiTFSI electrolytes with different doping ratios upon heating when n = 15. Figure 6A and B is reprinted with permission from Ref. [92], Copyright © 2022 Multidisciplinary Digital Publishing Institute; (C) Fabrication and polymerization pathways of PDOL and F-PDOL electrolytes. Figure 6C is reprinted with permission from Ref. [93], Copyright © 2025 Wiley. SPEs: Solid polymer electrolytes; PEO: polyethylene oxide; PEO-b-PDM-dTFSI: PEO-b-poly(trimethyl-N-{[2-(dimethylamino)ethyl methacrylate]-7-propyl}-ammonium bis(trifluoromethanesulfonyl) imide); LiTFSI: lithium bis(trifluoromethanesulfonyl)imide; PDOL: poly(1,3-dioxolane); F-PDOL: F-contained PDOL; DOL: 1,3-dioxolane; OFBO: 2,2’-(2,2,3,3,4,4,5,5-Octafluorohexane-1,6-diyl) bis (oxirane).
Designing the cross-linked polyether chain structures. Constructing a polyether macromolecule network via cross-linking of liner polymer chains can improve both the mechanical robustness and thermal resistance of polyether. For instance, Cui et al.[94] developed a crosslinked poly(1,3-dioxolane) (PSiDOL) electrolyte by introducing a multifunctional siloxane-based crosslinker to construct a robust polyether network. The resulting structure significantly enhanced the tensile strength of the membrane (25.7 MPa) and promoted the development of a durable SEI layer enriched with LixSiOy and LiF. Owing to its reinforced mechanical integrity, the PSiDOL electrolyte exhibited a high CCD of 0.5 mA·cm-2 and enabled dendrite-free cycling for over 4,000 h in Li|Li symmetric cells at 0.2 mA·cm-2. Similarly, Tang et al.[91] developed cross-linked polymer electrolytes (CNPE) based on PEO and poly(propylene oxide) (PPO) oligomers using surface-modified SiO2 nanoparticles as multifunctional cross-linkers. The stable cross-linked network endowed the membrane with enhanced structural stability, allowing the Li|Li symmetric cell to sustain lithium plating/stripping at a high current density of 2.4 mA·cm-2 for up to 1,700 h.
To further strengthen the mechanical robustness and interfacial stability of polyether-based SPEs, Sun et al.[95] proposed an innovative dual-crosslinked network design. This strategy combines a UV-induced self-polymerized network formed by trimethylolpropane triacrylate (TMPTA), which rapidly constructs a uniform and rigid crosslinked skeleton, with a secondary, randomly crosslinked network derived from PEO and triethylene glycol dimethacrylate (TEGDME). This structural feature not only enhances the homogeneity of the membrane but also significantly reinforces its mechanical strength, achieving a tensile stress of up to 1.91 MPa. Benefiting from the improved structural integrity and interfacial compatibility, LFP|Li cells employing this SPE can retain 99.6% of their initial capacity with nearly 100% coulombic efficiency over 240 cycles.
Compositing with inorganic nanofillers. The inorganic fillers play a very crucial role in affecting the mechanical property of polyether-based SPE. Based on their dimensional properties, they can be divided into four types: particles (0D), wire-like fillers (1D), two-dimensional (2D) sheet-like fillers and three-dimensional (3D) network-like fillers.
0D oxide ceramic nanoparticles, such as SiO2[96], Al2O3[97], ZrO2[98], TiO2[99] and Li6.4La3Zr1.4Ta0.6O12 (LLZTO)[100], are commonly employed as mechanical strengthening fillers in PEO-based SPEs due to their high modulus. For example, when 6.85% fumed SiO2 was added into the PEO-LiClO4 SPE [Figure 7A], the tensile strength can be increased by 2.1 times (1.9 MPa)[101]. The symmetric cell with the SiO2-containing SPE exhibits stable plating/stripping behavior for 380 h, significantly longer than the cycling life (210 h) of the symmetric cell with pristine PEO SPE. However, these 0D fillers tend to cluster within the polymer host due to their large interfacial energy, limiting their ability to form uniform, continuous reinforcing network.
Figure 7. (A) Physical and structural diagrams of the PEO-LiClO4 and PEO-LiClO4-Fumed-SiO2 SPE. Figure 7A is reprinted with permission from Ref. [101], Copyright © 2021 Royal Society of Chemistry; (B) Scanning electron microscopy (SEM) images of vertically aligned 1D LLTO nano-arrays; (C) Cross-sectional morphology of the PEO/LLTO nano-arrays with top view sample inset. Figure 7B and C is reprinted with permission from Ref. [102] Copyright © 2025 Royal Society of Chemistry; (D) Schematic diagram of the preparing process for PEO/2D MOFs CSE; (E) Stress-strain curves of SPEs; (F) Voltage profiles of PEO/2D MOF electrolyte-based Li/Li symmetric cells. Figure 7D-F is reprinted with permission from Ref. [103], Copyright © 2023 Wiley. PEO: Polyethylene oxide; SPE: solid polymer electrolyte; 1D: wire-like fillers; LLTO: Li0.5La0.5TiO3; 2D: two-dimensional; MOF: metal-organic framework; CSE: composite solid electrolyte; FS: fumed silica.
1D nanostructures can provide more continuous load transfer and mechanical reinforcement compared to 0D particles. As depicted in Figure 7B and C, Li et al.[102] synthesized vertically aligned 1D Li0.5La0.5TiO3 (LLTO) nano-arrays via a sol-gel template method, which were embedded into a PEO matrix to construct a flexible composite polymer electrolyte (VLSPE). Compared to the 0D LLTO nanoparticle/PEO electrolyte with a mechanical strength of 5.4 MPa, the aligned 1D LLTO nanowire/PEO electrolyte exhibited a significantly higher strength of 7.5 MPa[102]. This substantial enhancement in mechanical robustness effectively stabilized the interface with the lithium anode throughout prolonged cycling, enabling Li|Li symmetric cells to function reliably for over 800 h. Similarly, Wen et al.[104] fabricated 1D HAP nanowires embedded in PEO electrolyte, where the HAP nanowires establish a 3D network. Due to the continuous inorganic phase, the PEO-HAP composite solid polymer electrolyte (SPE) achieves a Young’s modulus of
Compared to 0D and 1D nanofillers, 2D nanoflakes exhibit more effective modification effects due to their higher surface-to-volume ratio[106]. Shim et al.[107] incorporated 2D boron nitride nanosheets (BNs) into PEO-LiTFSI matrix. Remarkably, the tensile strength and Young’s modulus of PEO SPE increased from 0.85 and 0.72 MPa to 2.62 and 2.91 MPa, respectively, upon the addition of BN. As a result, lithium dendrite propagation is significantly restrained. In addition, the interactions between PEO/LiTFSI chains and BN nanosheets lead to the formation of multiple interfacial sites, promoting efficient ion conduction. The symmetric cell with PEO/BN SPE demonstrates stable operation for 1,200 h without short circuit at
Incorporating the scaffold supports. The 3D framework can more effectively remove clustering fillers and provide mechanical support for SPE membrane[108,109]. Introducing solid-state electrolytes into porous frameworks can substantially improve the mechanical stability of SPEs. For example, Li et al.[109] demonstrated a composite electrolyte by filling PEO SPE in a 3D Ga-doping LLZO (Ga-LLZO) garnet framework [Figure 8A-C]. The interconnected 3D garnet architecture, prepared via sacrificial template method, endows the composite electrolytes with a high tensile strength of 21.8 MPa. The 3D garnet composite SPE in Li|Li symmetric configuration shows a long cycling life of 360 h at 0.4 mA·cm-2 with the reduced dendrite growth. Additionally, Mu et al.[111] introduced a vertically aligned Li1.3Al0.3Ti1.7(PO4)3 (LATP) scaffold into a PEGDA-based polymer matrix to construct a composite SPE. The aligned ceramic framework markedly reinforced the structural stability of the electrolyte and provided fast Li+ transport channels along the vertical direction, thereby reducing interfacial resistance and improving electrode-electrolyte contact. As a result, the Li|Li symmetric cell exhibited excellent cycling stability for over 4,000 h at 0.1 mA·cm-2 and
Figure 8. (A) Synthesis procedure of 3D garnet/PEO composite electrolyte; (B) SEM image of the 3D garnet framework; (C) SEM image of 3D garnet composite electrolyte. Figure 8A-C is reprinted with permission from Ref. [109], Copyright © 2019 American Chemical Society; (D) Cross-section SEM images of PEPL electrolyte; (E) Voltage profiles of Li plating/stripping cycles at 0.1 mA·cm-2. Figure 8D and E is reprinted with permission from Ref. [110], Copyright © 2019 Wiley. 3D: Three-dimensional; PEO: polyethylene oxide; SEM: scanning electron microscopy; PEPL: polyethylene-based polymer electrolyte; LLZO: lithium lanthanum zirconium oxide; PPL: polyethylene-based polymer lithium electrolyte; PL: polymer electrolyte.
Some polymer scaffolds, such as commercial polyolefin separators[110], porous nanocellulose membrane[112,113], and polyacrylonitrile nanofibers film[114,115] are flexible, mechanically strong, and lightweight, and are also used as mechanical support for SPEs. For example, Wu et al.[110] infiltrated PEO/LiTFSI polymer electrolyte into a 5 µm thick polyethylene (PE) separator to develop a high-strength polymer electrolyte (PEPL). The robust yet flexible PE separator provides structural reinforcement to the soft PEO electrolyte, leading to a more than 1000-fold increase in Young’s modulus (445 vs. 0.2 MPa). Meanwhile, the PEPL electrolyte exhibits significantly lower thickness (7.5 µm) compared to pure PEO SPE (120 µm), which shortens the Li + diffusion time and distance between cathode and anode. Due to its sufficient mechanical strength[110], the ultrathin PEPL electrolyte supports stable lithium electrode operation in a symmetric cell for 1,500 h at
Tung et al.[116] incorporated a heat-resistant Kevlar-derived aramid (ANF) nanofibers membrane into PEO to increase the dendrite-suppression modulus of the electrolyte. The ultimate Young’s modulus of the PEO/ANF membrane was measured as 4.95 GPa, which is higher than that of lithium (4.91 GPa). Crucially, the membranes feature pore sizes smaller than the typical dendrite growth regions, thereby hindering lithium dendrite intrusion into the SPE. Besides, the Celgard PE melted completely after being stored at 200 °C for
Therefore, composite 3D frameworks composed of mechanically flexible polymer materials and ionically conductive ceramics were introduced as skeletons to overcome the above-mentioned issues of inorganic and polymer scaffolds. For example, Zhang et al.[114] reported a composite solid polymer electrolyte (PPL) constructed by electrospinning a 3D framework of polyacrylonitrile (PAN) and LLZTO, followed by infiltration with PEO and LiTFSI. The introduction of the continuous PAN/LLZTO network substantially enhanced the mechanical performance of the electrolyte, with the tensile strength reaching 9.47 MPa, approximately 11 times that of the pristine PEO membrane. This mechanically reinforced structure effectively mitigated lithium dendrite growth, enabling the Li|Li symmetric cell to cycle stably for over
Based on the above, for 3D fillers suitable for bendable solid-state energy storage batteries, it is essential to construct a flexible and strong 3D framework with continuous, aligned, and precisely controlled structures. Therefore, establishing novel design strategies is essential for composite 3D architectures, which should combine high flexibility, robust mechanical strength, and structural resilience.
In summary, electrolytes with enhanced mechanical rigidity can restrain dendritic lithium propagation. However, it is worth noting that electrolyte with high mechanical strength may compromise interfacial contact with the electrodes, potentially enlarging the interfacial resistance and lowering the electrochemical performance. Hence, an appropriate balance between mechanical strength and flexibility is necessary to suppress lithium dendrites while maintaining low interfacial resistance.
Endowing polyether electrolytes with self-healing property
The electrolyte breakage caused by lithium dendrite growth or significant volume changes in the anode impairs Li+ transport at the interface, leading to battery capacity attenuation or even short circuits. To address these problems, polyether SPEs with self-repair capabilities have been developed by incorporating dynamic covalent bonds and supramolecular interactions into polymer matrices. These dynamic networks allow autonomous repair of interfacial damage and enhance their tolerance to mechanical deformation[118-121].
For instance, Pei et al.[122] reported a poly(ether-urethane)-based SPE (PTMG-HDI-BHDS) with intrinsic self-healing capability by introducing dynamic disulfide bonds and hydrogen bonding into the polymer network [Figure 9A]. The resulting dual-network architecture enabled spontaneous self-healing at room temperature [Figure 9B] and maintained close interfacial contact with the Li anode, delivering outstanding cycling stability over 6,000 h at 0.2 mA·cm-2 in symmetric Li|Li cells. Similarly, Chen et al.[124] implemented a comparable strategy by combining disulfide bonds and hydrogen bonding within a polyether framework, which significantly improved both interfacial compatibility and electrochemical reversibility, further confirming the general applicability of this approach.
Figure 9. (A) Chemical structures of self-healing SPE; (B) The interfacial self-healing process of PTMG-HDI-BHDS SPE. Figure 9A and B is reprinted with permission from Ref. [122], Copyright © 2024 Springer Nature; (C) Schematic diagram of the self-healing mechanism; (D) Schematic representation of Li dendrite evolution in a self-healing electrolyte, which is suppressed by the electrolyte. Figure 9C and D is reprinted with permission from Ref. [123], Copyright © 2022 Wiley. SPE: Solid polymer electrolyte; PTMG-HDI-BHDS: poly(ether-urethane)-based SPE.
Building upon this, Li et al.[123] developed a self-healing polymer electrolyte by in situ copolymerizing 2-[3-(6-methyl-4-oxo-1,4-dihydropyrimidin-2-yl)ureido]ethyl methacrylate (UPyMA) and ethylene glycol methyl ether acrylate (EGMEA), constructing a dynamic network via supramolecular interactions between UPy moieties [Figure 9C]. This supramolecular structure enables the polymer matrix to autonomously repair cracks and voids within 2 h at room temperature, thereby maintaining interfacial integrity against lithium dendrite growth and electrode volume fluctuations [Figure 9D]. Moreover, the self-healing polymer electrolyte maintains stable Li plating/stripping behavior in symmetric Li|Li cells for over 650 h at
Recently, a novel self-healing PEO-based SPE was prepared through the condensation polymerization of poly(ethylene glycol) diamine with 1,3,5-triformylbenzene[125]. Through dynamically cross-linked imine bonds, the healed electrolyte regained stretchability up to five times its initial length after 24 h of self-healing. As a result, Li|Li symmetrical cells could maintain a stable cycle for 1,200 h at 0.1 mA·cm-2.
In summary, the incorporation of dynamic covalent and non-covalent interactions into polyether-based SPE imparts intrinsic self-healing ability, allowing autonomous repair of bulk and interfacial damage during cycling. This self-healing behavior not only improves the mechanical durability of the electrolyte, but also ensures stable interfacial contact with lithium metal, thereby suppressing dendrite growth and enhancing long-term cycling performance.
Construction of interfacial layer
In situ creation of SEI
Polyether electrolytes are thermodynamically unstable against Li metal and tend to undergo reductive decomposition under practical operating conditions. This decomposition gives rise to SEI formation comprising Li2CO3 and Li2O precipitate on Li metal surface. However, this in situ SEI is often mechanically weak and ionically insulating, promoting filamentary lithium growth and increasing interfacial resistance. Therefore, how to construct a robust and fast-ion conducting SEI by adjusting the components on the Li metal anode is strongly considered. Accordingly, representative in situ SEI engineering strategies are discussed in this section.
(1) LiF-rich SEI with high mechanical strength
Although LiF is intrinsically brittle, its high Young’s modulus (135.3 GPa) enables it to mechanically suppress the growth of lithium dendrites[71,126]. Consequently, extensive studies have demonstrated that increasing the LiF content in the SEI leads to significantly improved anode performance. A common approach is to tailor the polyether electrolyte composition, such as the choice of lithium salt or additive, to promote the formation of LiF-rich SEI. For instance, Armand’s team found that SEI layers formed with LiFSI-added PEO (PEO-LiFSI) electrolytes contained more LiF content than those formed with LiTFSI-added PEO (PEO-LiTFSI) electrolytes[127]. This is due to the fact that S-F bonds in FSI- of LiFSI salts are more easily reduced than the C-F bonds in TFSI- of LiTFSI salts. The increased LiF component is essential for achieving a mechanically and electrochemically stable SEI layer, leading to superior cycling performance in cells with PEO-LiFSI electrolytes compared to those with PEO-LiTFSI electrolytes [Figure 10A and B]. Besides, Wang et al.[128] introduced lithium trifluoro (perfluoro-tert-butyloxyl) borate {Li[(CF3)3COBF3], LiTFPFB} into PEO-based SPE. The fluoroalkoxyl groups of TFPFB- anion in the salt also participates in the formation of SEI, thereby enriching it with LiF [Figure 10C]. As a result, the resulting Li|Li symmetric cell exhibits a long cycling life of 600 h at 0.25 mA·cm-2, compared to only 220 h for the cell without the LiTFPFB-based interfacial layer [Figure 10D].
Figure 10. (A) XPS spectra of Li deposits formed on Cu in LiX/DME electrolytes; (B) Depth-dependent atomic concentrations (Li, S, F, N) of the Li deposits. Figure 10A and B is reprinted with permission from Ref. [127], Copyright © 2018 American Chemical Society; (C) Multilayer design of the salt-differentiated SPE; (D) Voltage profiles of Li/Li symmetric cells with the salt-differentiated SPE at
Similarly, inorganic additives such as lithium nitrate (LiNO3) have also been shown to promote LiF formation in solid polymer electrolytes. Zhang et al.[129] introduced 5 wt% LiNO3 into a PEO/LLZTO (PLLE) solid-state electrolyte and observed a significantly enhanced interfacial stability. During cycling, the presence of LiNO3 facilitates the decomposition of LiTFSI, thereby increasing the LiF content within the SEI and effectively mitigating undesired reactions at the PEO/Li interface. As a result, the modified PLLE electrolyte exhibits a higher CCD of 1.0 mA·cm-2, and the corresponding LFP|Li cell retains 128.5 mAh·g-1 (84.5% of the maximum capacity) after 200 cycles, in contrast to the rapid performance degradation observed in the LiNO3-free system.
Fluorine-containing organic additives also play a critical role in promoting the formation of LiF-rich interface. Fluoroethylene carbonate (FEC), known for forming stable SEI in liquid systems, provides similar benefits in solid-state systems. For example, Li et al.[130] incorporated FEC additive into PEO-Li0.35La0.55TiO3 (PEO-LLTO) electrolyte, where its coordination with Li+ endowed more freely mobile Li+ ions migrate to locally damaged regions during cycling, forming a dynamic, LiF-rich, self-repairing interfacial layer. This design enabled stable lithium deposition/stripping behavior at 0.2 mA·cm-2 for 800 h. Besides, FEC was also introduced into a DOL-based polymer electrolyte, where it synergized with perfluorodecanoic acid (PFDA) to construct a dual-layered SEI[131]. The inner LiF-rich layer, derived from the decomposition of FEC and LiTFSI, provided mechanical robustness to resist dendritic lithium penetration, while the outer organic layer accommodated volume fluctuations. While FEC is beneficial to promote the formation of a LiF-rich SEI and stabilize the Li/electrolyte interface, its liquid nature and flammability may compromise the mechanical robustness and flame retardancy of the composite solid electrolyte, necessitating careful consideration before practical applications.
(2) Li3N-rich SEI with high ionic conductivity
The presence of Li3N in the SEI has been widely demonstrated to significantly enhance interfacial ion transport due to its high ionic conductivity (~1.2 × 10-4 S·cm-1)[132]. To this end, various strategies have been explored to in situ construct Li3N-rich interphases at the Li metal interface a phenomenon confirmed by the majority of studies. For example, Hu et al.[133] developed a sandwich-structured PEO-TiN/PEO-LiYF4/ PEO-TiN polymer electrolyte [Figure 11A]. The incorporation of TiN additives facilitates the formation of Li3N interface components via in situ chemical reactions with the Li metal anode. This enhancement effectively promotes Li + transfer across the Li/electrolyte interface, thereby mitigating Li dendrites growth, as depicted in Figure 11B. As a result, LFP cell employing this electrolyte showed a high-capacity retention rate of 74.18% after 300 cycles. In another study, Li et al.[135] developed an in situ Li3N-forming interface by introducing 2,2’-azobisisobutyronitrile (AIBN) as a nitrogen-containing additive. The Li3N interface layer effectively improves the reversibility of the Li deposition/stripping, and the corresponding Li|Li symmetric cell exhibits the superior cycling stability at 0.2 mA·cm-2, with a cycle life at least four times that of PEO electrolytes without the AIBN additive.
Figure 11. (A) Optical image of the PEO-TiN/PEO-LiYF4/PEO-TiN membrane; (B) Schematic illustration of the cyclic evolution between sandwich-CSE and Li metal and the suppression of Li dendrite processes in a lithium symmetric cell. Figure 11A and B is reprinted with permission from Ref. [133], Copyright © 2023 American Chemical Society; (C) Schematic of species distribution in the CSEs-1%Li2S6 membrane and at the lithium metal anode interface; (D and E) TOF-SIMS depth profiles and 3D reconstruction of the sputtered volume of Li2S-, Li2S2- species. Figure 11C-E is reprinted with permission from Ref. [134], Copyright © 2021 Wiley. PEO: Polyethylene oxide; CSE: composite solid electrolyte; TOF-SIMS: time-of-flight secondary ion mass spectrometry; LiTFSI: lithium bis(trifluoromethanesulfonyl) imide; LLZTO: Li6.4La3Zr1.4Ta0.6O12.
In addition to Li3N, other inorganic species with high ionic conductivity - such as Li3P and Li2S - have been extensively investigated for the construction of fast ion-conducting interphases aimed at enhancing interfacial lithium-ion transport. Furthermore, the Li3P and Li2S components exhibit high ionic conductivity and are utilized to enhance the lithium ion diffusion rate within the SEI[136,137]. For example, a thin Li3P layer is constructed in situ at the Li/electrolyte interface by introducing black phosphorus (BP) additive into a crosslinked polymer electrolyte[137]. The Li|Li symmetric cell with this Li3P layer demonstrates low interfacial resistance and stable cycling performance at 0.3 mA·cm-2. Fang et al.[134] incorporated Li2S6 into a PEO-based polymer electrolyte, enabling the in situ generation of an ultrathin Li2S/Li2S2 layer upon contact with lithium metal [Figure 11C-E]. This interphase facilitates interfacial Li+ transport and mitigates dendritic lithium propagation. According to R. A. Huggins, the ionic conductivity of Li2S could reach up to 10-5 S·cm-1[138]. Therefore, the Li|Li symmetric cell with the Li2S6-containing electrolyte delivers excellent cyclability and a high CCD of 0.9 mA·cm-2. Furthermore, lithium polysulfidophosphate (LPS) was used as an additive in a PEO-based SPE to form a highly Li ion conductive and stable Li3PS4/Li2S/LiF layer[80]. The Li|Li cell incorporating LPS into the SPE demonstrates an interfacial resistance as low as 10 Ω·cm2, enabling stable operation for 3,475 h at 0.2 mA·cm-2. Moreover, an all-solid-state LFP|Li cell maintains a high discharge capacity of 127.6 mAh·g-1 after 1,000 cycles at 1 C.
(3) Li alloy-rich SEI with high Li ion diffusion rate
Recently, alloy phases have been constructed on Li metal surfaces to facilitate rapid Li+ diffusion. Therefore, some inorganic components such as SbF3, In2O3, AgF, SnF2 and CoF2 have been introduced into polyether-based electrolytes, which react with Li metal to form Li-rich alloy phase with high lithium ion diffusion rate, thus optimizing the lithium deposition behavior by accelerating Li + transport at the eletrode-eletrolyte interface[139-143]. For example, SbF3 is used as PEO electrolyte additive to in-situ generate a Li-Sb alloy-rich SEI layer on the Li metal anode[141]. The lithophilic Li-Sb component promotes Li + migration at the interface, resulting in improved CCD and an extended cycle life (980 h) for Li|Li symmetric cell at
Chen et al.[144] further demonstrated the generation of Li-In alloy-rich SEI in a gel polymer electrolyte (GPE), synthesized via in situ polymerization of DOL and dimethoxyethane (DME) initiated by indium trifluoromethanesulfonate [In(OTF)3]. The generated Li-In alloy (e.g., Li3In2, Li13In3) and LiF-rich SEI effectively reduced the energy barrier for Li+ deposition, enabling stable symmetric cell cycling over 4,000 h. Likewise, Lu et al.[145] developed a PDOL-based GPE polymerizing DOL initiated by gallium trifluoromethanesulfonate [Ga(OTF)3]. The Ga3 + ions not only initiated polymerization but also reacted with Li metal to form a hybrid SEI comprising LiF, Li2O, and Li-Ga alloys. DFT calculations revealed that the Li-Ga alloy exhibited strong Li+ adsorption (-2.05 eV), promoting uniform Li deposition. Consequently, Li|Li symmetric cells exhibited stable cycling for 1,600 h at 0.2 mA·cm-2, and LiFePO4|Li cells maintained 90.2% capacity after 200 cycles at 1 C.
Besides, other metal-based additives such as bismuthine, Al(EtO)3 and zinc bis(2-ethylhexanoate) were also introduced to in situ form LixBi, LiAl and LiZn alloy interface species[146-148], which accelerate Li+ conduction and suppress parasitic reactions at the Li/electrolyte interface.
Based on the quantitative comparison summarized in Table 3, several trends can be discerned. LiF-containing SEI systems generally exhibit moderate to high critical current densities (typically
Comparative summary of in situ SEI engineering strategies in polyether-based ASSBs: interfacial characteristics and realistic symmetric-cell performance metrics
| SPEs | Dominant SEI componens | Critical current density (mA·cm-2) | Interfacial resistance (Ω) | Cycle life of Li|Li cells | Operating current density (mA·cm-2) | Areal Capacity (mAh·cm-2) | Over- potential (mV) | Operating Temperature (°C) | References |
| PEO-PbrILs + LiTFSI | LiF | 1.1 | 300 | 1,400 h | 0.2 | 0.2 | 12.2 | 60 | [149] |
| PEO-POF | 0.5 | ~380 | > 1,000 h | 0.1 | 0.1 | ~250 | 60 | [150] | |
| PEO + TiO2(B) | 1.6 | ~250 | 1,000 h | 0.2 | 0.1 | ~100 | 50 | [151] | |
| PEGDA/o-LLZO | 2 | ~150 | 1,0000 h | 0.1 | 0.1 | ~100 | 60 | [152] | |
| P@PBA | 0.5 | ~25 | > 3,500 h | 0.3 | 0.3 | 86.1 | 60 | [153] | |
| P(PEGMEA) + TMP + LiNO3 | Li3N | / | 17.67 | 1,500 h | 0.2 | 0.4 | 52 | 25 | [154] |
| PEO + LiNO3 + LiTFSI | 1 | ~50 | 240 h | 0.4 | 0.4 | 75 | 60 | [129] | |
| PEO-LiTFSI-LiFSI-LiNO3 | / | ~60 | 1,200 h | 0.1 | 0.1 | ~60 | 60 | [155] | |
| PEO + LiTFSI + Li10Si0.3PS6.7C17.8 | Li2S | 0.54 | 50 | 1,400 h | 0.18 | 0.09 | 90 | 55 | [156] |
| PEO + LPS + LiTFSI | 1 | ~30 | > 3,000 h | 0.2 | 0.4 | ~100 | 60 | [80] | |
| CPMEA LiTFSI + phosphorus powder | Li3P | 0.7 | 19 | 350 h | 0.1-0.7 | 0.1 | 80 | 60 | [136] |
| PEO + LiTFSI + SnO2&In2O3 | LiSn + LiIn | 0.7 | ~95 | 1000 h | 0.2 | 0.2 | 47 | 60 | [157] |
| P(DME) + LiFSI + LiNO3 + SnI2 | Sn@LixSn + Li3N | / | ~45 | > 800 h | 1 | 1 | ~50 | 75 | [158] |
| PEGDE + LiDFOB Al(EtO)3 + liquid electrolyte | LiAl | 4.9 | ~60 | 3,000 h | 0.1 | 0.1 | ~20 | 30 | [148] |
| P(DOL) + DME + In(OTF)3 | Li3In2 + Li13In3 + LiF | / | ~8.4 | 4,000 h | 0.2 | 0.2 | ~10 | 25 | [144] |
| PEO + LiTFSI + In2O3 | LiIn | / | ~200 | > 1,200 h | 0.1 | 0.1 | 27 | 60 | [142] |
| PEO + LLZTO + SbF3 | LiSb + LiF | 1.5 | ~80 | 980 h | 0.2 | 0.2 | ~50 | 60 | [141] |
| PEO + LLZTO + CoF2 | LiCo + LiF | 1.3 | 55.6 | 1,200 h | 0.2 | 0.2 | 76 | 60 | [140] |
| PEO + LiTFSI + AgF | LiAg + LiF | 1 | ~120 | 1,800 h | 0.2 | 0.2 | ~300 | 60 | [139] |
In addition, several application-related trends can be identified. Most stable cycling demonstrations are conducted at elevated temperatures (50-60 °C), reflecting the temperature dependence of polyether-based electrolytes, whereas only a limited number of systems exhibit stable operation near room temperature (25-30 °C). Moreover, extended lifetimes in symmetric cells are typically achieved under relatively mild testing conditions (≤ 0.2 mA·cm-2 and ≤ 0.2-0.4 mAh·cm-2). In contrast, reports demonstrating stable cycling at higher current densities and areal capacities (≥ 1 mA·cm-2 and ≥ 1 mAh·cm-2) remain scarce.
Ex situ constructing coating
Prior to battery assembly, lithium metal is often chemically pre-treated to form an ex situ interface layer, which can effectively suppress undesirable reactions between the polyether electrolyte and lithium anode. Such an interface layer can also mitigate interfacial dendrite growth by enhancing its mechanical strength, improving the uniformity of Li-ion flux, or reducing the charge transfer resistance across the interface. Up to now, inorganic, polymer, and polymer/inorganic composites serve as common options for ex situ coating.
(1) Ex situ inorganic coating with high mechanical strength
Constructing artificial SEI layer using inorganic material has been revealed as a viable route for forming stable interface, which can isolate the electrolyte from the Li anode, and physically suppress the dendrite growth.
For instance, Fan et al.[159] fabricated a 5 nm-thick Al2O3 layer on PEO SPE surface via atomic layer deposition technique to mechanically inhibit the evolution of lithium dendrites. As a result, the Li|Li symmetric cell with Al2O3 coated PEO can cycle stably for 500 h at 0.6 mA·cm-2, whereas the cell with bare PEO SPE cycled only for 250 h due to mechanical failure of the relatively weak electrolyte. In a similar approach, Shi et al.[160] developed an ultrathin (~5 nm) atomic layer deposition (ALD)-deposited Al2O3 layer on a PEO-TiO2 CSE, which served not only as a mechanically robust interlayer but also chemically stabilized the electrode-electrolyte interface. Consequently, the Li|Li symmetric cell with CSE/Al2O3 delivered an impressive ultralong cycling life over 1,200 h at 0.1 mA·cm-2, significantly outperforming the uncoated CSE (390 h) and bare PEO electrolyte (134 h). However, a potential limitation of such metal oxide coatings lies in their intrinsically poor lithium-ion conductivity[161], which may impede efficient Li+ transport across the interface and increase interfacial resistance - thereby compromising long-term electrochemical performance if not properly engineered.
(2) Ex situ inorganic coating with fast ionic transport
To overcome the limitations of metal oxides, certain inorganic compounds that can react with Li metal to form Li-ion conductive species (e.g., Li3N) have been ex situ constructed as interface layers, thereby simultaneously enhancing Li+ transport kinetics and mechanical stability at the electrolyte-anode interface. For example, a Mg3N2 layer was employed to decorate the PEO SPE, which could in situ transform the ionic conductor Li3N and the benign electronic conductor Mg metal after in contact with Li foil [Figure 12A][162]. This interface layer could not only mechanically suppress the dendrite growth, but also could alleviate the dendrite formation by homogenizing the distribution of
Figure 12. (A) Schematic illustration of the cycling behavior in PEO and PEO-Mg3N2 electrolytes. Figure 12A is reprinted with permission from Ref. [162], Copyright © 2020 Wiley; (B and C) Digital images of pristine and Zn-coated Li foils; (D and E) Cross-sectional SEM and EDS mapping of the Zn-coated Li anode; (F) The cycling stability of Zn-coated Li at 0.1 mA·cm-2. Figure 12B-F is reprinted with permission from Ref. [163], Copyright © 2021 Wiley. PEO: Polyethylene oxide; SEM: scanning electron microscopy; EDS: energy dispersive X-ray spectroscopy.
In addition to inorganic non-metallic interface components (Li3N, Li2S), Li-rich alloy components can also be generated by constructing ex-situ interface layer, thereby regulating the ions transport behavior at electrolyte-anode interface. For example, Deng et al.[163] prepared a LiZn alloy interface layer via an alloy interaction between the pre-sputtered Zn thin film and Li foil [Figure 12B-E]. Owing to the high lithium-ion diffusion coefficient of the LiZn alloy (4.7 × 10-8 cm2·s-1), PEO-based symmetric cells employing LiZn-protected lithium anodes exhibited stable cycling over 900 h at 0.1 mA·cm-2 [Figure 12F]. Similarly, a dense Li-Sn alloy layer was formed by pre-sputtering a thin Sn film onto the Li metal surface, which subsequently reacted in situ with lithium[164]. Compared to the LiZn alloy, the Li-Sn alloy offers an even higher Li+ diffusion coefficient (~6 × 10-8 cm2·s-1), resulting in an extended symmetric cell lifespan of 2,400 h at 0.1 mA·cm-2. The corresponding LiFePO4|Li full cell retained 85% of its initial capacity after 500 cycles.
Most recently, Liu et al.[165] introduced a uniform and ultra-thin (~35 nm) Ag nano interlayer onto the surface of PEO/succinonitrile(SN)-based electrolytes via magnetron sputtering, which spontaneously reacted with Li metal to form a Li-Ag alloy interphase. The resulting AgLi-rich interlayer enhanced interfacial wettability and provided fast ion transport channels. Moreover, The AgLi interphase also suppressed the side reactions between SN and lithium metal, contributing to improved interface chemical stability. Benefiting from the alloy-rich and stable interfacial structure, the Ag-modified symmetric cells exhibited prolonged cycling stability over 1,700 h at 0.05 mA·cm-2. In addition, ex situ coating of Al, Pt and ZnO layers has also been employed to generate Li-Al, Li-Pt and Li-Zn alloy interphases in situ during cycling[166,167]. These alloy layers further accelerate interfacial Li+ transport and improve electrochemical performance. However, although these alloy interlayers effectively facilitate Li+ transport, their intrinsic electronic conductivity may compromise interface performance by inducing undesired lithium plating above the alloy layer or triggering electrolyte decomposition (e.g., PEO degradation), thereby posing challenges to long-term cycling stability.
To address the challenges posed by the electronic conductivity of pure alloy layers, Wang et al.[168] constructed a LiF/Li3Sb composite layer in situ by spraying the SbF3-contained dimethoxyethane solution on fresh Li foil. Herein, the LiF component formed simultaneously with the Li3Sb alloy bestows insulating qualities to the interface layer, preventing reduction of the Li + on the surface. Due to the improved mechanical stability and ionic diffusion rate at the interface, the SbF3-modified Li (SbF3@Li)|SbF3@Li cell with PEO-Li6.4La3Zr1.4Ta0.6O12 electrolyte can cycle stably for more than 300 h at 0.2 mA·cm-2, while maintaining a stable interfacial structure between the electrolyte and SbF3@Li. These results highlight that the suitable inorganic materials as ex situ coatings are essential for preventing the decomposition of polyether electrolytes as well as the formation and growth of dendrites.
(3) Ex situ polymer coating for improving interfacial contact
In addition to inorganic coatings, polymer materials that are chemically stable with lithium metal, capable of accommodating anode volume changes during cycling, or able to react with lithium to form lithium-ion conductors, have been explored as surface coatings to enhance the interfacial stability between the anode and solid electrolytes.
For example, Cui’s group introduced an adaptive layer (ABL) consisting of PEO and low molecular-weight polypropylene carbonate (PPC) to ensure interface stability [Figure 13A-E][169]. In the presence of the viscoelastic layer, the interface resistance of the Li|LiFePO4 cell only increased by 20% after 150 cycles due to the stabilized interface contact, which is much lower than the increase (117%) in the resistance of cell without this layer [Figure 13F]. Similarly, Wang et al.[171] proposed an asymmetric multilayered solid composite electrolyte (MSCE) design consisting of a PEO buffer layer, a PEO-LLZTO composite middle layer, and a PPC coating on the anode side. The PPC layer serves as a self-sacrificial component that reacts with lithium to form Li alkoxides and alkyl carbonates (e.g., ROCO2Li, ROLi), thereby improving interfacial wettability and reducing resistance. Benefiting from this adaptive architecture, the MSCE enabled excellent interfacial contact and dendrite suppression, achieving a high ionic conductivity of 3.21 × 10-4 S·cm-1 at 30 °C. Consequently, LiFePO4|MSCE|Li cells exhibited 94.5% capacity retention after 150 cycles at 0.1 C, while symmetric Li|MSCE|Li cells maintained stable cycling for 500 h at 0.4 mA·cm-2.
Figure 13. (A-C) Photographs of fully dried PEO SPE, ABL, and PPC SPE; (D) Cross-sectional SEM images of Li|Li symmetric cells without ABL layer after cycling; (E) Cross-sectional SEM images of Li|Li symmetric cells with ABL layer after cycling; (F) Nyquist plots of full cell before and after cycling at 50 °C. Figure 13A-F is reprinted with permission from Ref. [169], Copyright © 2019 American Chemical Society; (G) Illustration of Tween-grafted lithium metal; Figure 13G is reprinted with permission from Ref. [170], Copyright © 2018 American Chemical Society. PEO: Polyethylene oxide; SPE: solid polymer electrolyte; ABL: adaptive layer; PPC: polypropylene carbonate; SEM: scanning electron microscopy; W/O: without; CPE: constant phase element.
Moreover, Liu et al.[170] constructed a polymer interfacial layer by grafting tween-20 with sequential oxyethylene groups on lithium metal anode (TG-Li) to facilitate Li+ conduction and stabilize the interface [Figure 13G]. As a result, the cycling stability of LFP|TG-Li cell is much better than the LFP|Li cell, delivering a reversible capacity of 132.5 mAh·g-1 after 200 cycles at 1 C. Moreover, the F-rich poly(vinylidene fluoride) (PVDF) polymer applied as a functional coating on PEO SPE can react with lithium, resulting in the in situ formation of a LiF-enriched interface layer[172]. Besides, after constructing PVDF skin, the tensile strength of PEO electrolyte also increased nearly ten times, which is beneficial to mechanically inhabiting the Li dendrite growth. The Li|Li symmetric cell shows a stable long-term cycling performance over 1,000 h at 0.1 mA·cm-2 and maintains stable Li/PEO interface resistance.
In addition, polymer interface layers have also been integrated into asymmetric polymer electrolyte architectures to further improve interfacial contact with lithium metal. Recent studies demonstrated that the construction of bilayer or quasi-double-layer electrolytes with tailored interfacial chemistries on the anode side can effectively stabilize the Li surface, enhance ionic conductivity, and suppress dendrite formation, thereby enabling long-term cycling stability even under high-voltage or high-rate conditions[173,174].
(4) Ex situ polymer/inorganic coating with excellent comprehensive performance
Recently, by combining the complementary advantages of polymer and inorganic materials, composites have become a better choice for artificial layers. For instance, Wang’s group constructed a poly(vinylidene-co-hexafluoropropylene)/lithium nitrate (PVDF-HFP/LiNO3, PFH/LN) composite layer on Li anode surface for PEO-LLZTO electrolyte[175]. In the artificial layer, PVDF-HFP can physically suppress Li dendrites penetration and thus maintain the structural integrity of interface layer due to its high mechanical strength and moderate flexibility; the LiNO3 additives could react with Li to form highly ionic conductive Li3N components, thus inducing the uniform Li deposition at the interface. With the PFN/LN artificial layer, the cycle life (1,800 h) of Li|Li symmetric cell is much longer than that (96 h) of the cell with bare Li anode at 0.2 mA·cm-2. Similarly, the PVDF-HFP/CuF2 composite could also be employed as Li metal interface layer[176]. The in-situ reaction of CuF2 with Li metal anode generates LiF and Cu components, which improves the ion transport rate and t interfacial mechanical robustness. Applied in the Li|Li symmetric cell with PEO-based electrolyte, the PVDF-HFP/CuF2 modified Li anode demonstrates extended cycle life (1,000 h) than the bare Li at 0.4 mA·cm-2. Furtherly, the LFP full cell with the modified Li anode shows excellent cycle stability (0.032% capacity fading rate per cycle) at 1 C. Consequently, the synergistic effect of polymer and inorganic components could efficiently regulate the Li plating/stripping behavior to illustrate excellent interface stability.
Composite Li metal anode
Designing 3D composite anode to decrease interface mechanical deformation
Since the volume change of the Li anode during cycling is infinite due to its hostless nature, it would cause SEI damage and discontinuous electrolyte-electrode contact. Therefore, developing 3D Li composite anode is quite effective on maintaining the mechanical stability, as the 3D structure can accommodate the huge volume change of Li metal, thereby reducing the interface mechanical deformation.
Due to the hostless nature of lithium metal, the anode undergoes significant and virtually unlimited volume changes during repeated deposition/dissolution, which leads to continuous damage of the SEI and loss of contact at the electrolyte-electrode interface. To address this issue, the design of 3D lithium composite anodes has emerged as a promising strategy for enhancing mechanical stability. The porous architecture of 3D hosts can effectively buffer the drastic volume fluctuations of Li metal, thereby mitigating interfacial mechanical stress and deformation.
To stabilize the interface between lithium metal and polyether-based solid electrolytes, various 3D anode architectures have been explored. For instance, Chen et al.[177] fabricated a 3D Li anode via infusing molten Li into Ni foam disk for dendrite-free ASSBs [Figure 14A-F]. The volume change alleviated by 3D Li extends the cycle life to 600 h at 0.5 mA·cm-2 for the symmetric cell with poly(ethylene carbonate)-based SPE, whereas the cell with Li foil can only cycle for 315 h. Later, Ye et al.[178] prepared a 3D-structured lithium/copper mesh composite anode for PEO-based solid-state batteries [Figure 14G-I], which endows the LFP full cell with the enhanced cycle stability with high interfacial stability.
Figure 14. (A and B) The illustration of Li deposition behaviors of Li metal cell with bare Li foil and 3D Li metal anode; (C and D) Digital images of Ni foam and Li-Ni composite anode; (E) SEM image of Ni foam; (F) SEM image of Li-Ni composite anode. Figure 14A-F is reprinted with permission from Ref. [177], Copyright © 2019 Wiley; (G) EDS mapping images of LG composite anode; (H) Photograph of copper mesh; (I) Photograph of lithium/copper mesh. Figure 14G-I is reprinted with permission from Ref. [178], Copyright © 2021 American Chemical Society. 3D: Three-dimensional; SEM: scanning electron microscopy; EDS: energy dispersive X-ray spectroscopy; PEO: polyethylene oxide.
However, in contrast to liquid electrolyte batteries, where the electrolyte readily penetrates a 3D network, incorporating such 3D anode structures in all-solid-state batteries to sustain ionic conduction remains challenging. Therefore, Zhu et al.[179] put forward a 3D composite anode (LG), which possesses a vertical alternating array structure. This structure not only provides continuous lithium-ion transport channels into the composite anode, but also accommodates the cyclic expansion and contraction of lithium during repeated cycling. The symmetric cells based on this 3D composite anode can operate stably for 800 cycles at a large capacity of 5 mAh·cm-2, and the corresponding solid-state cell exhibits 93.26% capacity retention rate after 830 cycles at 5 C. Besides, Liu et al.[180] incorporated the 3D composite Li anode [metallic Li in layered reduced graphene oxide layers (Li-rGO)] with the pre-loaded flowable PEG ion-conducting interphase into the ASSB system. The 3D rGO host could compensate for the Li metal volume changes, and the penetrated PEG components could enhance the intrinsic ionic diffusion capacity of anode. As a result, the cycle life of symmetric cell with Li-rGO anode could be improved by nearly seven times compared to that of the cell a flat Li foil at 0.5 mA·cm-2. Furthermore, Zhu et al.[181] constructed lithium ion diffusion pathways by incorporating Li-Al alloy with strong lithium affinity and in situ-formed Li3N within the 3D lithium/rGO composite anode (LAG), effectively suppressing dendrite growth and enabling impressive electrochemical performance (capacity retention of 86.3% after 200 cycles for the solid state LFP|LAG cell).
As a complementary approach, Park et al.[182] explored the use of 3D porous hosts combined with viscous polymer electrolytes to stabilize lithium deposition. The viscous nature of the poly(ethylene glycol) dimethyl ether (PEGDME)-based polymer electrolyte allowed it to effectively infiltrate the 3D host structure, forming continuous ion transport pathways and accommodating lithium volume changes during cycling. This design promoted uniform Li deposition and suppressed short-circuiting even at high areal capacities (5 mAh·cm-2). In addition, Liu et al.[183] developed an integrated 3D lithium-boron (LiB) fiber anode combined with polymer electrolyte to construct dual ion/electron transport pathways. The electrolyte effectively infiltrated the porous LiB scaffold, forming continuous ion-conduction channels while maintaining mechanical flexibility. The 3D structure enabled more uniform lithium deposition and reduced interfacial stress, while the in-situ formed stable SEI further enhanced interfacial stability. These results highlight that 3D composite anodes can effectively buffer volume changes and improve the structural integrity of the lithium-polyether electrolyte interface.
Employing Li-alloy anode to improve interface chemical stability
Li can be mixed with Si, Ge, Mg, In, Zn, Si, Al, etc. to form Li alloys. Compared with Li metal, Li-alloy anodes exhibit slightly higher operating potentials, which can reduce parasitic reactions with SPEs and enhance interfacial chemical stability. Furthermore, many Li alloys possess higher Li diffusion coefficient, which can promote uniform Li distribution during cycling, mitigating the formation of lithium dendrites.
For example, LiAl alloys are often used as anode materials for liquid batteries because of their good air stability, high Li diffusion coefficient (6.0 × 10-10 Ω-1·cm-1) and relatively high redox potential of 0.3 V versus Li/Li+ [Figure 15A][186-188]. Until 2001, Bang et al.[189] and coworkers first investigated the cyclability of the LiAl alloy anode material in the LiAl|PEO30-LiTFSI|V2O5 solid state cells. Due to the low lithium activity in the LiAl alloy, the parasitic reaction on the anode significantly reduced. As a result, the PEO-based ASSBs with Li-Al anodes maintained a coulombic efficiency close to 100%. Later, Pan et al.[184] also applied Li-Al alloy as the anode material in an all-solid-state Li-S battery. The symmetric cell with Li0.8Al alloy anode shows steady operation for over 2,500 h at 0.5 mA·cm-2, further implying the excellent anode-electrolyte compatibility [Figure 15B]. And they concluded that the volume change (74.95%) is modest during the biphasic lithiation reaction of Al, which further promotes interfacial mechanical stability and anode structural stability. Besides, the Li10GeP2S12 electrolyte exhibits excellent stability upon contact with Li0.8Al anode because of the smaller potential difference between sulfides and alloy than that between sulfides and Li metal. However, when the
Figure 15. (A) Schematic of the practical stability window of the Li10GeP2S12 electrolyte and the chemical potential of different electrodes; (B) Galvanostatic Li plating/stripping profiles of the Li-LGPS-Li cell at 0.5 mA·cm-2. Figure 15A and B is reprinted with permission from Ref. [184], Copyright © 2022 American Association for the Advancement of Science; (C) Schematics of the two fabricated electrodes; (D and E) TOF-SIMS analysis of 7Li+ in the modified electrode after charging to 2.7 V (D) and 3.7 V (E). Figure 15C-E is reprinted with permission from Ref. [185], Copyright © 2023 Wiley. LGPS: Li10GeP2S12; TOF-SIMS: time-of-flight secondary ion mass spectrometry; HOMO: highest occupied molecular orbital; LUMO: lowest unoccupied molecular orbital; SE: solid electrolyte.
Consequently, Yan et al.[192] synthesized a hard-carbon-stabilized LiSi alloy anode (LiSH46), in which the hard-carbon can effectively buffer the volume change of Si during the lithiation process. The thickness of anode only increases from 44.7 to 46.4 μm after activation at 0.1 C and further to 47.1 μm after ten cycles at
Polyether SPE/high-voltage cathode interface: stability and optimization
Interfacial degradation mechanisms of polyether SPE at high-voltage cathodes
While the previous sections have primarily addressed the Li metal anode/polyether-based SPE interface, the stability of polyether electrolytes at high-voltage cathodes is equally critical for practical full-cell operation. Unlike the anode interface, where dendrite growth dominates, the cathode interface is primarily challenged by the oxidative decomposition of polyether-based electrolytes under high potentials.
Polyether-based polymers, such as PEO, PEG-derivates and P(DOL), are intrinsically susceptible to oxidation. Ether oxygen units can be oxidized at ~4-4.5 V versus Li/Li+, forming radicals or carbocations that generate resistive products, including lithium alkoxides, carbonates, and other oxygen-containing species, which hinder Li+ transport[194,195]. This oxidative degradation is further exacerbated in contact with high-voltage layered cathodes, such as NCM811, LiNi0.6Co0.2Mn0.2O2 (NCM622), and LiCoO2, which accelerate polymer decomposition and lower the practical oxidative onset below ~4 V versus Li/Li+[42]. Factors contributing to degradation include the oxidative activity of partially delithiated Ni- or Co-rich cathodes, conductive carbon additives, high electrode surface area, and reactive species such as HTFSI generated during electrolyte oxidation, which can further propagate polymer chain degradation. Collectively, these reactions form a resistive cathode-electrolyte interphase, causing voltage polarization and capacity decay, which underscores the dominant role of interfacial chemistry and reaction kinetics in determining electrolyte stability.
Building on this understanding, researchers have developed the following strategies to mitigate interfacial instability at high-voltage cathodes, which can be broadly categorized into cathode surface coatings and polyether electrolyte/interphase engineering.
Strategies for stabilizing the polyether SPE/high-voltage cathode interface
(1) High-voltage cathode surface coatings
Surface coatings on high-voltage cathodes (e.g., NCM811, NCM622, LiCoO2) function as protective barriers that prevent direct contact between the polyether-based SPE and the active cathode material, thereby mitigating oxidative degradation and interfacial side reactions while preserving Li+ transport pathways. For example, Dai et al.[196] applied a lithium borate (Li2O-B2O3) coating that effectively isolated the cathode from PEO, suppressing parasitic reactions and facilitating ionic transport. Similarly, Chen et al.[197] introduced a nanoscale LATP coating onto NCM811, which increased the interfacial stability of the cathode up to 4.31 V vs. Li/Li+ and suppressed parasitic reactions with the PVDF/PEO-LLZTO composite electrolyte. Beyond inorganic coatings, Li et al.[198] constructed a high-voltage, high-conductivity interfacial layer of polyfluoroalkyl acrylate (PTFEMA) directly on the NCM811 cathode via in situ solvent-free electropolymerization. This layer expanded the electrochemical oxidation window of PEO SPE from 4.3 to 5.1 V, resulting in enhanced cycling stability of NCM811|PEO|Li cells, with an initial discharge capacity of 179.8 mAh·g-1 retained at 119.7 mAh·g-1 after 50 cycles at 0.1 C.
(2) Polyether electrolyte and interphase engineering
Parallel efforts have focused on tailoring the polyether-based electrolyte itself to enhance interfacial stability with high-voltage cathodes, mainly through three strategies: (i) Additive engineering. Chen et al.[199] incorporated a multifunctional additive (e.g., tris(pentafluorophenyl)borane) into PEO, which reduced polymer crystallinity, enhanced ionic conductivity, increased the Li+ transference number via anion trapping, and optimized SEI/ cathode-electrolyte interphase (CEI) formation, thereby enabling stable operation above 4.6 V. Similarly, Kim et al.[200] introduced a functional sulfate additive into a thermally cross-linked PEO-based gel polymer electrolyte, forming a stable interfacial layer that effectively suppressed oxidative decomposition and side reactions on Ni-rich cathodes; (ii) In situ interphase construction. Ye et al.[201] constructed a CEI on NCM811 via pre-reaction with trace liquid electrolyte, which isolated PEO from direct contact with the cathode and suppressed its catalytic decomposition; (iii) Double-layer polymer electrolyte design. The bilayer design proposed by Arrese-Igor et al.[202], comprising a high-voltage-tolerant poly(propylene carbonate)(PPC) on the cathode side and PEO in the separator, not only ensures suitable energy-level alignment but also promotes stable chemical interactions with both the high-voltage cathode and the lithium-metal anode. This architecture enabled LiNixMnyCozO2|Li cells to deliver 160 mAh·g-1 at
Collectively, these strategies, including targeted cathode coatings, additive engineering, in situ interface formation, and double-layer polymer design, constitute a coherent framework for stabilizing high-voltage cathode-polyether SPE interfaces, enhancing ionic transport, and improving cycling performance in solid-state lithium metal batteries.
TECHNO-ECONOMIC CONSIDERATIONS FOR PRACTICAL DEPLOYMENT
Beyond interface chemistry and electrochemical stability, the practical deployment of polyether-based solid-state batteries must be assessed from a techno-economic perspective. Although detailed cost modeling requires proprietary industrial data, a semi-quantitative assessment can be conducted by examining raw material costs, manufacturing requirements, infrastructure demand, stack-pressure dependency, and scalability constraints.
Raw material cost and supply considerations
Raw material cost constitutes a key component of electrolyte economics. Polyether-based SPEs rely on industrially mature polymer matrices such as PEO, P(DOL), and PEG derivatives, which are produced at large scale. Bulk polymer matrices are typically priced at approximately $5-15 kg-1, depending on molecular weight and purity. In these systems, the primary cost contribution often arises from battery-grade lithium salts (e.g., LiTFSI), whereas the polymer backbone itself benefits from established supply chains and relatively low base cost.
In contrast, oxide and sulfide solid electrolytes require high-purity ceramic or sulfide precursors. Garnet-type oxides depend on materials such as ZrO2, La2O3, and dopants including Ta or Nb, with high-purity ceramic precursors potentially reaching hundreds of dollars per kilogram[203]. Argyrodite-type sulfide systems rely on Li2S, phosphorus compounds, and in some cases critical elements such as Ge, placing material costs typically in the tens to hundreds of dollars per kilogram range and introducing additional supply considerations[204].
Although electrolyte material cost alone does not determine overall cell cost, the relatively low base-polymer price and mature supply infrastructure provide polyether systems with a structural advantage in upstream material economics and scalability.
Manufacturing energy consumption and infrastructure requirements
Processing requirements strongly influence capital expenditure and operational costs in solid-state battery manufacturing. Garnet-type oxides typically require high-temperature sintering (> 1,000 °C), precision ceramic processing, and high-pressure lamination to ensure densification and interfacial contact[205]. These steps substantially increase energy consumption and equipment investment. Argyrodite-type sulfides avoid extreme sintering temperatures but necessitate ultra-dry (< ppm H2O) environments, inert-atmosphere handling, and strict environmental control during synthesis and cell assembly, thereby elevating facility cost and operational complexity[206].
Polyether-based SPEs can be prepared via solution casting, extrusion, roll-to-roll coating, or in situ polymerization at moderate temperatures. The absence of high-temperature densification and ultra-dry processing reduces infrastructure requirements and energy consumption[207]. Moreover, polymer-processing techniques are inherently compatible with existing lithium-ion battery coating lines, which may reduce industrial transition barriers and facilitate scale-up.
Stack pressure and system-level economic impact
In addition to material and processing factors, the stack pressure applied during cell assembly plays a direct role in module-level costs and mechanical design. Oxide SSEs, such as Garnet-type systems, owing to their rigid and brittle nature, typically require sustained multi-megapascal pressure to maintain stable electrode-electrolyte contact. This necessitates structural reinforcement, increasing packaging mass and mechanical complexity while potentially reducing gravimetric and volumetric energy density. Sulfide SSEs exhibit improved deformability and better initial contact; however, moderate pressure is still generally required to suppress interfacial void formation, particularly in lithium metal systems. Although lower than that of oxides, this requirement remains a practical constraint for large-format cells.
In contrast, polyether-based SPEs possess intrinsic mechanical compliance and conformal interfacial contact, enabling stable operation without high stack pressure or complex interfacial engineering[208]. Lower pressure requirements can simplify module architecture, reduce structural reinforcement demands, and decrease balance-of-system costs. This advantage becomes increasingly significant as cell dimensions scale up.
Scalability and manufacturing yield
Scalability depends not only on material cost but also on mechanical robustness and manufacturing yield. Oxide SSEs are intrinsically brittle and susceptible to cracking during handling, lamination, and scale-up, which may reduce production yield and increase defect-related losses. Sulfide SSE materials, although mechanically softer, impose strict environmental controls that constrain processing flexibility and elevate operational complexity. In contrast, polyether-based electrolytes exhibit intrinsic mechanical flexibility, thin-film processability, and compatibility with continuous roll-to-roll manufacturing, while enabling improved interfacial conformity with electrodes[209]. These characteristics collectively suggest the potential for higher production yield and reduced defect-related waste during large-scale fabrication.
Techno-economic assessment of solid-state electrolytes
A structured comparison of polymer, oxide, and sulfide SSEs from a techno-economic perspective is summarized in Table 4.
Comparative evaluation of manufacturing complexity and industrial scalability among polyether-based, oxide, and sulfide SSEs
| Evaluation metric | Polyether-based SSE | Oxide SSE (e.g., garnet-type) | Sulfide SSE (e.g., argyrodite-type) |
| Raw material complexity | Low-moderate | High (multi-element ceramics) | Moderate-high (sensitive sulfide chemistry) |
| Precursor characteristics | Commodity polymer matrix; lithium salt dominant | Multi-element ceramic precursors; dopants required | Moisture-sensitive precursors; controlled synthesis |
| High-temperature sintering | Not required | Required (> 1,000 °C) | Not required |
| Ultra-dry processing | Not mandatory | Not mandatory | Strictly required |
| Roll-to-roll compatibility | Excellent | Limited | Moderate |
| Required stack pressure | Low-moderate | Moderate-high | Moderate-high |
| Manufacturing maturity | Mature polymer industry | Developing | Early industrialization stage |
| Process energy intensity | Low-moderate (no high-T densification) | High (high-T sintering) | Moderate (dry-room control) |
Overall, the above analysis indicates that polyether-based SPEs exhibit a structurally favorable techno-economic profile, particularly in terms of raw material accessibility, processing simplicity, infrastructure compatibility, and reduced stack-pressure dependency. In contrast, oxide electrolytes, while offering strong intrinsic stability, impose substantial energy, equipment, and mechanical design burdens due to high-temperature sintering and sustained pressure requirements. Sulfide systems occupy an intermediate position, benefiting from improved interfacial contact but constrained by strict environmental control and moderate pressure demand.
CONCLUSION AND OUTLOOK
Polyether-based electrolytes are promising candidates for high-energy all-solid-state lithium batteries due to their favorable safety and stability. This review summarizes key interfacial challenges affecting cycling performance, including chemical and mechanical instability and limited Li+ transport. It also presents strategies to stabilize the Li metal anode, including polymer electrolyte modification, interface layers, and composite anodes. Practical deployment also depends on factors such as material availability, processing requirements, and scalability. Significant challenges remain to realize practical polyether-based all-solid-state batteries [Figure 16].
Figure 16. Schematic illustration of significant challenges and strategies to achieve excellent ASSBs. ASSBs: All-solid-state batteries; SPE: solid polymer electrolyte; SEI: state electrolyte interphase; 3D: three-dimensional.
(1) Development of gradient, multilayered polyether electrolytes
Gradient multilayered polyether electrolytes with functional anode contact coatings should be developed to comprehensively improve the interfacial issues of ASSBs. As discussed above, electrolyte layers with excellent elasticity can ensure intimate electrode contact, mechanically robust layers may help suppress the propagation of lithium dendrites, and self-healing layers offer the potential to repair damage caused by dendrite formation. The integration of these advantages can be achieved through construction of multi-layered electrolytes. Besides, the ex situ constructed SEI layer could precisely design interface components and improve the interface chemical stability by physically separating the electrolyte from the anode. Consequently, designing multilayered polyether electrolytes with anode contact coating is considered to simultaneously optimize the interface mechanical/chemical stability and ionic transport at the interface by designing different layers with various functions.
(2) Endowing 3D Li-alloy anode with high strength and balanced ionic/electronic conductivity
As mentioned above, the use of a 3D scaffold structure can effectively limit volumetric changes in the lithium metal anode, enhancing interfacial mechanical stability. Therefore, lithium-rich alloys, which exhibit good chemical stability with the electrolyte, can further improve interfacial mechanical stability through the large specific surface area of the 3D scaffold and the domain-limiting effect.
As for 3D electrode, the effective Li + /e- pathways ensure the continuity of the electrochemical reaction, which could promote the Li deposition within the 3D framework rather than on the anode surface. Besides, the mechanical strength of the 3D framework should also be increased to prevent the irregular morphologies of lithium deposition (e.g., dendrites, dead Li) from damaging the electrode structure.
(3) Achieving appropriate cyclability with practical operating current density (≈ 2 mA·cm-2)
Although there have been numerous reports improving the cycling stability of polyether-based Li|Li symmetric cell with the single optimization strategy, the operating current density is lower than the practical operating current density (~2 mA·cm-2). Therefore, a single approach cannot resolve all issues of electrode/electrolyte interfaces. The combination of modification strategies, such as constructing an artificial SEI, creating a 3D composite lithium metal anode, utilizing elastic polyether SPE, and developing alloy anode, should be considered.
(4) Better understanding of the SEI film in polyether electrolyte based all-solid-state batteries
A more clear understanding of the SEI films helps us design better interfacial modification strategies. More in-depth discussion and theoretical calculation of the interface reaction will provide guidance for building a more stable interface. Besides, advanced nondestructive methods such as solid-state nuclear magnetic resonance (NMR), in situ TEM, TOF-SIMS, neutron scattering, and operando X-ray are crucial for real-time probing of interfacial composition and structure and for clarifying lithium-ion transport mechanisms, providing theoretical insight into solid-solid interfaces.
(5) Challenges in large-scale application and industrial translation
Beyond laboratory optimization, polyether-based ASSBs face significant practical hurdles for mass production and industrial translation. Fabricating thin and uniform solid electrolyte membranes with sufficient mechanical strength is crucial; however, large-area production often leads to thickness variations, surface defects, or pores, which can impede ion transport, reduce battery performance, and accelerate degradation. Cost control is also a major concern, as laboratory approaches frequently rely on high-purity materials, specialized solvents, or energy-intensive processes that may be impractical for industrial deployment. In addition, the membranes must be compatible with electrodes and assembly processes, ensuring intimate contact, preserving structural integrity during fabrication, and adapting to winding, stacking, or dry-coating assembly techniques. These challenges collectively limit the smooth transition of polyether-based ASSBs from lab-scale research to industrial application.
To address these issues, industry-relevant strategies such as cathode-supported architectures, solution processing for large-area membranes, and bipolar stacking with internal series connections can help bridge the gap between laboratory and commercial production. Implementing these approaches can enhance uniform ion transport, maintain mechanical robustness, and enable scalable fabrication, providing a practical pathway toward the large-scale application of polyether-based ASSBs.
(6) AI-assisted interface development
While polyether-based electrolytes offer a promising pathway toward safer, high-energy ASSBs, their practical deployment remains limited by interfacial instability. Future progress requires a shift from empirical optimization to rational, data-driven interface engineering. In this regard, artificial intelligence (AI) and machine learning (ML) provide powerful tools to decode the complex composition-structure-interface-performance relationships and accelerate materials development [Figure 17].
Figure 17. AI/ML framework for stable interfaces in polyether-based ASSBs. AI: Artificial intelligence; ML: machine learning; ASSBs: all-solid-state batteries; XRD: X-ray diffraction; XPS: X-ray spectroscopy; SEI: state electrolyte interphase.
Firstly, AI-assisted high-throughput screening can efficiently explore the vast chemical space of polyether matrices, lithium salts, additives, and fillers, enabling the prediction of optimal multicomponent formulations. For example, Kim et al.[210] combined ML with DFT to screen over 10,000 dual-doped LLZO compositions, identifying candidates that achieved a room-temperature ionic conductivity of 8.7 × 10-4 S·cm-1 in polymer composite electrolytes. Such approaches move beyond trial-and-error and support the rational design of multifunctional interlayers for dendrite suppression and interphase stabilization.
Secondly, ML can guide interphase chemistry and atomic-scale interface design by predicting SEI/CEI compositions, reaction pathways, and interface energetics. Physics-informed ML frameworks, such as InterOptimus[211], enable rapid evaluation of interface stability with DFT-level accuracy at substantially reduced computational cost, offering atomic-level insights for designing stable lithium/electrolyte interfaces.
Finally, data-driven models can elucidate interfacial failure mechanisms and define quantitative safety thresholds. By analyzing multimodal operando and post-mortem data, ML enables early identification of interface degradation and dendrite initiation, while physics-informed models incorporating electrochemical, thermal, and mechanical coupling can predict cell lifetime and operational limits. Together, these advances highlight the transformative role of AI/ML in accelerating the development of stable and safe polyether-based solid-state batteries.
In spite of the persistent challenges, polyether-based electrolytes remain a highly promising avenue for achieving high-energy-density and safe lithium metal batteries. To translate this promise into reality, future research must deepen the fundamental exploration of interfacial deterioration mechanisms while pioneering more rational design strategies. Moving beyond traditional empirical approaches, the integration of innovative interfacial engineering and advanced characterization with transformative computational intelligence, ranging from AI-accelerated material discovery, ML-guided manufacturing optimization, and data-driven failure-prediction frameworks, will establish a systematic and efficient development roadmap. This integrated, data-centric paradigm is essential for evolving polyether electrolytes from promising candidates into reliable, high-performance components of next-generation energy storage systems.
DECLARATIONS
Authors’ contributions
Writing-original draft: Zhang, M.
Writing-review and editing: Zhang, Y.; Zheng, Y.; Zhou, Z.; Wang, Z. (Zhiqiao Wang)
Collecting and analyzing: Shao, A.; Li, X.; Wang, H.; Liu, J.; Li, C.; Zhu, J.; Liu, F.
Supervision: Wang, Z. (Zhaohui Wang); Ma, Y.
Funding acquisition: Zhang, M.; Wang, Z. (Zhaohui Wang); Ma, Y.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
The authors acknowledge the financial support of the National Natural Science Foundation of China (Nos. 22479046, 52173229 and 52373229), the Natural Science Foundation of Shaanxi (Nos. 2023-JC-JQ-15 and 2019KJXX-099), the Key Research and Development Projects of Shaanxi Province (No. 2019ZDLGY04-05), Young Talent Fund of Association for Science and Technology in Shaanxi, China (No. 20250451), Scientific Research Program Funded by Education Department of Shaanxi Provincial Government (No. 24JK0503), Fund of Hubei Key Laboratory of Energy Storage and Power Battery (Hubei University of Automotive Technology) (No. ZDK22025A03), the Shaanxi Innovation Capacity Support Program (Science and Technology Innovation Team, No. 2025RS-CXTD-027), the Youth Innovation Team of Shaanxi Universities Project (No. 24JP085) and the Fundamental Research Funds for the Central Universities (No. 3102019JC005).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Etacheri, V.; Marom, R.; Elazari, R.; Salitra, G.; Aurbach, D. Challenges in the development of advanced Li-ion batteries: a review. Energy. Environ. Sci. 2011, 4, 3243.
2. Liu, K.; Liu, Y.; Lin, D.; Pei, A.; Cui, Y. Materials for lithium-ion battery safety. Sci. Adv. 2018, 4, eaas9820.
4. Manthiram, A.; Yu, X.; Wang, S. Lithium battery chemistries enabled by solid-state electrolytes. Nat. Rev. Mater. 2017, 2, 16103.
5. Cheng, X. B.; Zhang, R.; Zhao, C. Z.; Zhang, Q. Toward safe lithium metal anode in rechargeable batteries: a review. Chem. Rev. 2017, 117, 10403-73.
6. Xu, W.; Wang, J.; Ding, F.; et al. Lithium metal anodes for rechargeable batteries. Energy. Environ. Sci. 2014, 7, 513-37.
7. Lin, D.; Liu, Y.; Cui, Y. Reviving the lithium metal anode for high-energy batteries. Nat. Nanotechnol. 2017, 12, 194-206.
8. Han, X.; Gong, Y.; Fu, K. K.; et al. Negating interfacial impedance in garnet-based solid-state Li metal batteries. Nat. Mater. 2017, 16, 572-9.
9. Wang, M.; Zhang, H.; Li, Y.; Liu, R.; Yang, H. A three-dimensional co-continuous network structure polymer electrolyte with efficient ion transport channels enabling ultralong-life all solid-state lithium metal batteries. J. Energy. Chem. 2024, 94, 635-45.
10. Wang, C.; Zhang, X.; Sun, X.; Zhang, Y.; Wang, Q.; Sun, J. Aliphatic hyperbranched polycarbonates solid polymer electrolytes with high Li-ion transference number for lithium metal batteries. Macromol. Rapid. Commun. 2024, 45, e2300645.
11. Wang, H.; Wang, Q.; Cao, X.; et al. Thiol-branched solid polymer electrolyte featuring high strength, toughness, and lithium ionic conductivity for lithium-metal batteries. Adv. Mater. 2020, 32, e2001259.
12. Fan, Y.; Zhu, M.; Wang, H.; et al. Polyamine-mediated proton/TFSI- dual capture enables high-voltage PEO-based all-solid-state Li batteries. Adv. Mater. 2026, 38, e20538.
13. Zuo, X.; Cheng, Y.; Xu, L.; et al. A novel thioctic acid-functionalized hybrid network for solid-state batteries. Energy. Storage. Mater. 2022, 46, 570-6.
14. Gong, Y.; Wang, C.; Xin, M.; et al. Ultra-thin and high-voltage-stable Bi-phasic solid polymer electrolytes for high-energy-density Li metal batteries. Nano. Energy. 2024, 119, 109054.
15. Zhang, K.; Wu, F.; Wang, X.; et al. 8.5 µm-thick flexible-rigid hybrid solid-electrolyte/lithium integration for air-stable and interface-compatible all-solid-state lithium metal batteries. Adv. Energy. Mater. 2022, 12, 2200368.
16. Ma, Y.; Wan, J.; Yang, Y.; et al. Scalable, ultrathin, and high-temperature-resistant solid polymer electrolytes for energy-dense lithium metal batteries. Adv. Energy. Mater. 2022, 12, 2103720.
17. Li, J.; Yang, R.; Jiang, A.; et al. Ultra-stable all-solid-state lithium metal batteries facilitated by in-situ LiF-rich single-ion conductor composite polymer electrolytes. Chem. Eng. J. 2025, 514, 162820.
18. Zhang, C.; Zhang, S.; Zhang, Y.; et al. Regulating Lewis acid-base interaction in poly (ethylene oxide)-based electrolyte to enhance the cycling stability of solid-state lithium metal batteries. Small. Struct. 2023, 5, 2300301.
19. Zhang, Q.; Yang, R.; Li, C.; et al. In-situ coupling construction of interface bridge to enhance electrochemical stability of all solid-state lithium metal batteries. J. Energy. Chem. 2024, 89, 18-26.
20. Wei, L.; Xu, X.; Xi, K.; et al. Polydopamine-induced metal-organic framework network-enhanced high-performance composite solid-state electrolytes for dendrite-free lithium metal batteries. ACS. Appl. Mater. Interfaces. 2024, 16, 878-88.
21. Zhang, Q.; Yang, R.; Li, C.; et al. Composite solid electrolytes with cation assisted effect to enhance the electrochemical performance of all solid-state lithium metal batteries. Ceram. Int. 2025, 51, 12738-47.
22. Annu; Arifeen, W. U.; Abideen, Z. U.; Ko, T. J.; Shin, D. K. Evaluation of BaTiO3 enhanced bioinspired modified-chitosan integrated polymer electrolyte membrane for stable Li-metal batteries. J. Energy. Storage. 2026, 141, 119233.
23. Luo, Z.; Cui, Y.; Zhang, Z.; Li, M.; Yu, J. A solid composite electrolyte based on three-dimensional structured zeolite networks for high-performance solid-state lithium metal batteries. Chem. Sci. 2026, 17, 3533-43.
24. Yang, X.; Sun, C.; Li, R.; et al. Percolating dynamic ion-conduction network in composite polymer electrolyte for high-performance lithium metal batteries. Chem. Eng. J. 2025, 520, 165754.
25. Zhou, J.; Wang, X.; Fu, J.; et al. A 3D cross-linked metal-organic framework (MOF)-derived polymer electrolyte for dendrite-free solid-state lithium-ion batteries. Small 2024, 20, e2309317.
26. Wang, Z.; Du, Z.; Liu, Y.; et al. Metal-organic frameworks and their derivatives for optimizing lithium metal anodes. eScience 2024, 4, 100189.
27. Wu, X.; Zhang, W.; Qu, H.; et al. An oriented design of a π-conjugated polymer framework for high-performance solid-state lithium batteries. Energy. Environ. Sci. 2025, 18, 1835-46.
28. Majeed, M. K.; Hussain, A.; Hussain, G.; et al. Interfacial engineering of polymer solid-state lithium battery electrolytes and li-metal anode: current status and future directions. Small 2024, 20, e2406357.
29. Chattopadhyay, J.; Pathak, T. S.; Santos, D. M. F. Applications of polymer electrolytes in lithium-ion batteries: a review. Polymers 2023, 15, 3907.
30. Yang, H.; Wu, N. Ionic conductivity and ion transport mechanisms of solid-state lithium-ion battery electrolytes: a review. Energy. Sci. Eng. 2022, 10, 1643-71.
31. Guchok, O.; Ardel, G.; Keidar, T. D.; et al. Understanding of the ion transport in blended TPU-PEO polymer electrolytes. J. Solid. State. Electrochem. 2025, 6323.
32. Fenton, D.; Parker, J. M.; Wright, P. Complexes of alkali metal ions with poly(ethylene oxide). Polymer 1973, 14, 589.
34. Xue, Z.; He, D.; Xie, X. Poly(ethylene oxide)-based electrolytes for lithium-ion batteries. J. Mater. Chem. A. 2015, 3, 19218-53.
35. Xu, S.; Sun, Z.; Sun, C.; et al. Homogeneous and fast ion conduction of PEO-based solid-state electrolyte at low temperature. Adv. Funct. Mater. 2020, 30, 2007172.
36. Nair, J. R.; Shaji, I.; Ehteshami, N.; et al. Solid polymer electrolytes for lithium metal battery via thermally induced cationic ring-opening polymerization (CROP) with an insight into the reaction mechanism. Chem. Mater. 2019, 31, 3118-33.
37. Cui, Y.; Liang, X.; Chai, J.; et al. High performance solid polymer electrolytes for rechargeable batteries: a self-catalyzed strategy toward facile synthesis. Adv. Sci. 2017, 4, 1700174.
38. Liu, Q.; Cai, B.; Li, S.; et al. Long-cycling and safe lithium metal batteries enabled by the synergetic strategy of ex situ anodic pretreatment and an in-built gel polymer electrolyte. J. Mater. Chem. A. 2020, 8, 7197-204.
39. Zhao, Y.; Tao, R.; Fujinami, T. Enhancement of ionic conductivity of PEO-LiTFSI electrolyte upon incorporation of plasticizing lithium borate. Electrochim. Acta. 2006, 51, 6451-5.
40. Ai, C.; Shu, Y.; Zhao, Z.; Guo, H.; Chen, S.; Yi, Q. Advances in the application of ionic liquids in PEO-based lithium-ion solid-state electrolytes: from the perspective of fillers. J. Mater. Chem. A. 2025, 13, 13632-56.
41. Rawat, S.; Singh, P. K.; Pandey, S. P.; Singh, R. C. Review on comparative study of solid polymer electrolyte polyethylene oxide (PEO) doped with ionic liquid. Macromol. Symp. 2024, 413, 2300050.
42. Cabañero Martínez, M. A.; Boaretto, N.; Naylor, A. J.; et al. Are polymer-based electrolytes ready for high-voltage lithium battery applications? An overview of degradation mechanisms and battery performance. Adv. Energy. Mater. 2022, 12, 2201264.
43. Qiu, J.; Liu, X.; Chen, R.; et al. Enabling stable cycling of 4.2 V high-voltage all-solid-state batteries with PEO-based solid electrolyte. Adv. Funct. Mater. 2020, 30, 1909392.
44. Li, Z.; Zhang, H.; Sun, X.; Yang, Y. Mitigating interfacial instability in polymer electrolyte-based solid-state lithium metal batteries with 4 V cathodes. ACS. Energy. Lett. 2020, 5, 3244-53.
45. Lu, J.; Zhou, J.; Chen, R.; et al. 4.2 V poly(ethylene oxide)-based all-solid-state lithium batteries with superior cycle and safety performance. Energy. Storage. Mater. 2020, 32, 191-8.
46. Nava, D.; Guzmán, G.; Vazquez-arenas, J.; Cardoso, J.; Gomez, B.; Gonzalez, I. An experimental and theoretical correlation to account for the effect of LiPF6 concentration on the ionic conductivity of poly(poly (ethylene glycol) methacrylate). Solid. State. Ionics. 2016, 290, 98-107.
47. Wang, S.; Zhang, L.; Zeng, Q.; Liu, X.; Lai, W.; Zhang, L. Cellulose microcrystals with brush-like architectures as flexible all-solid-state polymer electrolyte for lithium-ion battery. ACS. Sustain. Chem. Eng. 2020, 8, 3200-7.
48. Zhang, S.; Xia, X.; Xie, D.; et al. Facile interfacial modification via in-situ ultraviolet solidified gel polymer electrolyte for high-performance solid-state lithium ion batteries. J. Power. Sources. 2019, 409, 31-7.
49. Yuan, B.; Zhao, B.; Wang, Q.; et al. A thin composite polymer electrolyte with high room-temperature conductivity enables mass production for solid-state lithium-metal batteries. Energy. Storage. Mater. 2022, 47, 288-96.
50. Ma, Q.; Yue, J.; Fan, M.; et al. Formulating the electrolyte towards high-energy and safe rechargeable lithium-metal batteries. Angew. Chem. Int. Ed. Engl. 2021, 60, 16554-60.
51. Zhao, Q.; Liu, X.; Stalin, S.; Khan, K.; Archer, L. A. Solid-state polymer electrolytes with in-built fast interfacial transport for secondary lithium batteries. Nat. Energy. 2019, 4, 365-73.
52. Yang, H.; Jing, M.; Wang, L.; Xu, H.; Yan, X.; He, X. PDOL-based solid electrolyte toward practical application: opportunities and challenges. Nanomicro. Lett. 2024, 16, 127.
53. Li, W.; Gao, J.; Tian, H.; et al. SnF2-catalyzed formation of polymerized dioxolane as solid electrolyte and its thermal decomposition behavior. Angew. Chem. Int. Ed. Engl. 2022, 61, e202114805.
54. Larrabide, A.; Rey, I.; Lizundia, E. Environmental impact assessment of solid polymer electrolytes for solid-state lithium batteries. Adv. Energy. and. Sustain. Res. 2022, 3, 2200079.
55. Mandade, P.; Weil, M.; Baumann, M.; Wei, Z. Environmental life cycle assessment of emerging solid-state batteries: a review. Chem. Eng. J. Adv. 2023, 13, 100439.
56. Klinsmann, M.; Hildebrand, F. E.; Ganser, M.; Mcmeeking, R. M. Dendritic cracking in solid electrolytes driven by lithium insertion. J. Power. Sources. 2019, 442, 227226.
57. Lee, S.; Kim, J.; Lee, J.; Byeon, J. Evaluation of cracking damage in electrode materials of a LMO/Al-Lix lithium-ion battery through analysis of acoustic emission signals. J. Mater. Res. Technol. 2023, 24, 5235-49.
58. Chen, S.; Wei, X.; Zhang, G.; et al. Mechanical strain signal based early warning for failure of different prismatic lithium-ion batteries. J. Power. Sources. 2023, 580, 233397.
59. Mirsakiyeva, A.; Ebadi, M.; Araujo, C. M.; Brandell, D.; Broqvist, P.; Kullgren, J. Initial steps in PEO decomposition on a Li metal electrode. J. Phys. Chem. C. 2019, 123, 22851-7.
60. Ebadi, M.; Marchiori, C.; Mindemark, J.; Brandell, D.; Araujo, C. M. Assessing structure and stability of polymer/lithium-metal interfaces from first-principles calculations. J. Mater. Chem. A. 2019, 7, 8394-404.
61. Ebadi, M.; Costa, L. T.; Araujo, C. M.; Brandell, D. Modelling the polymer electrolyte/Li-metal interface by molecular dynamics simulations. Electrochim. Acta. 2017, 234, 43-51.
62. Zhou, Q.; Ma, J.; Dong, S.; Li, X.; Cui, G. Intermolecular chemistry in solid polymer electrolytes for high-energy-density lithium batteries. Adv. Mater. 2019, 31, e1902029.
63. Fauteux, D. Formation of a passivating film at the lithium-PEO-LiCF3SO3 interface. Solid. State. Ionics. 1985, 17, 133-8.
64. Fauteux, D. Lithium electrode/PEO-based polymer electrolyte interface behavior between 60° and 120 °C. J. Electrochem. Soc. 2019, 135, 2231-7.
65. Croce, F.; Scrosati, B. Interfacial phenomena in polymer-electrolyte cells: lithium passivation and cycleability. J. Power. Sources. 1993, 43, 9-19.
66. Lisowska-oleksiak, A. The interface between lithium and poly(ethylene-oxide). Solid. State. Ionics. 1999, 119, 205-9.
67. Le Granvalet-mancini, M. Characterization of the passivation layer at the polymer electrolyte/lithium electrode interface. Solid. State. Ionics. 2000, 135, 283-90.
68. Kong, F.; Mclarnon, F. Spectroscopic ellipsometry of lithium/polymer electrolyte interfaces. J. Power. Sources. 2000, 89, 180-9.
69. Ushakova, E. E.; Frolov, A.; Reveguk, A. A.; Usachov, D. Y.; Itkis, D. M.; Yashina, L. V. Solid electrolyte interface formation between lithium and PEO-based electrolyte. Appl. Surf. Sci. 2022, 589, 153014.
70. Sheng, O.; Zheng, J.; Ju, Z.; et al. In situ construction of a LiF-enriched interface for stable all-solid-state batteries and its origin revealed by cryo-TEM. Adv. Mater. 2020, 32, e2000223.
71. Shin, H.; Park, J.; Han, S.; Sastry, A. M.; Lu, W. Component-/structure-dependent elasticity of solid electrolyte interphase layer in Li-ion batteries: experimental and computational studies. J. Power. Sources. 2015, 277, 169-79.
72. Wang, C.; Lin, R.; He, Y.; et al. Tension-induced cavitation in Li-metal stripping. Adv. Mater. 2023, 35, e2209091.
73. Ye, H.; Gui, S.; Wang, Z.; et al. In situ measurements of the mechanical properties of electrochemically deposited Li2CO3 and Li2O nanorods. ACS. Appl. Mater. Interfaces. 2021, 13, 44479-87.
74. Pugh, S. XCII. Relations between the elastic moduli and the plastic properties of polycrystalline pure metals. London,. Edinburgh,. Dublin. Philos. Mag. J. Sci. 2009, 45, 823-43.
75. Li, J. L.; Wang, Y. N.; Sun, S. Y.; et al. Understanding and regulating the mechanical stability of solid electrolyte interphase in batteries. Adv. Energy. Mater. 2024, 15, 2403845.
76. Li, J.; Hu, H.; Fang, W.; et al. Impact of fluorine-based lithium salts on SEI for all-solid-state PEO-based lithium metal batteries. Adv. Funct. Mater. 2023, 33, 2303718.
77. Zhang, J.; Li, S.; Wang, X.; et al. Construction of stable Li2O-rich solid electrolyte interphase for practical PEO-based Li-metal batteries. Adv. Energy. Mater. 2023, 14, 2302587.
78. Wu, H.; Jia, H.; Wang, C.; Zhang, J. G.; Xu, W. Recent progress in understanding solid electrolyte interphase on lithium metal anodes. Adv. Energy. Mater. 2020, 11, 2003092.
79. Pan, J.; Cheng, Y.; Qi, Y. General method to predict voltage-dependent ionic conduction in a solid electrolyte coating on electrodes. Phys. Rev. B. 2015, 91, 134116.
80. Xu, J.; Li, J.; Li, Y.; et al. Long-life lithium-metal all-solid-state batteries and stable li plating enabled by in situ formation of Li3PS4 in the SEI layer. Adv. Mater. 2022, 34, e2203281.
81. Cheng, Z.; Zhao, W.; Wang, Q.; et al. Beneficial redox activity of halide solid electrolytes empowering high-performance anodes in all-solid-state batteries. Nat. Mater. 2025, 24, 1763-72.
82. Wang, Z. Y.; Zhao, C. Z.; Yao, N.; et al. The regulation of solid electrolyte interphase on composite lithium anodes in solid-state batteries. Angew. Chem. Int. Ed. Engl. 2025, 64, e202414524.
83. Lopez, J.; Sun, Y.; Mackanic, D. G.; et al. A dual-crosslinking design for resilient lithium-ion conductors. Adv. Mater. 2018, 30, e1804142.
84. Guo, Y.; Qu, X.; Hu, Z.; Zhu, J.; Niu, W.; Liu, X. Highly elastic and mechanically robust polymer electrolytes with high ionic conductivity and adhesiveness for high-performance lithium metal batteries. J. Mater. Chem. A. 2021, 9, 13597-607.
85. Ren, Y.; Chen, S.; Odziomek, M.; et al. Mixing functionality in polymer electrolytes: a new horizon for achieving high-performance all-solid-state lithium metal batteries. Angew. Chem. Int. Ed. Engl. 2025, 64, e202422169.
86. Zheng, T.; Cui, X.; Chu, Y.; Li, H.; Pan, Q. Ultrahigh elastic polymer electrolytes for solid-state lithium batteries with robust interfaces. ACS. Appl. Mater. Interfaces. 2022, 14, 5932-9.
87. Lee, M. J.; Han, J.; Lee, K.; et al. Elastomeric electrolytes for high-energy solid-state lithium batteries. Nature 2022, 601, 217-22.
88. Liu, L.; Mo, J.; Li, J.; et al. Comprehensively-modified polymer electrolyte membranes with multifunctional PMIA for highly-stable all-solid-state lithium-ion batteries. J. Energy. Chem. 2020, 48, 334-43.
89. Huang, C.; Zhang, L. Miscibility of poly(vinylidene fluoride) and atactic poly(methyl methacrylate). J. Appl. Polym. Sci. 2004, 92, 1-5.
90. Xiao, Q.; Wang, X.; Li, W.; Li, Z.; Zhang, T.; Zhang, H. Macroporous polymer electrolytes based on PVDF/PEO-b-PMMA block copolymer blends for rechargeable lithium ion battery. J. Membr. Sci. 2009, 334, 117-22.
91. Tang, S.; Lan, Q.; Xu, L.; et al. A novel cross-linked nanocomposite solid-state electrolyte with super flexibility and performance for lithium metal battery. Nano. Energy. 2020, 71, 104600.
92. Zhang, Z. K.; Ding, S. P.; Ye, Z.; Xia, D. L.; Xu, J. T. PEO-based block copolymer electrolytes containing double conductive phases with improved mechanical and electrochemical properties. Mater. 2022, 15, 7930.
93. Duan, S.; Zhang, L.; Zheng, Y.; et al. “Rigid exterior, soft interior” design enables high-voltage polyether electrolytes for quasi-solid-state batteries. Angew. Chem. Int. Ed. Engl. 2025, 64, e202502728.
94. Cui, J.; Du, Y.; Zhao, L.; et al. Thermal stable poly-dioxolane based electrolytes via a robust crosslinked network for dendrite-free solid-state Li-metal batteries. Chem. Eng. J. 2023, 461, 141973.
95. Fu, F.; Lu, W.; Zheng, Y.; et al. Regulating lithium deposition via bifunctional regular-random cross-linking network solid polymer electrolyte for Li metal batteries. J. Power. Sources. 2021, 484, 229186.
96. Liu, S.; Imanishi, N.; Zhang, T.; et al. Effect of nano-silica filler in polymer electrolyte on Li dendrite formation in Li/poly(ethylene oxide)-Li(CF3SO2)2N/Li. J. Power. Sources. 2010, 195, 6847-53.
97. Chung, S.; Wang, Y.; Persi, L.; et al. Enhancement of ion transport in polymer electrolytes by addition of nanoscale inorganic oxides. J. Power. Sources. 2001, 97-98, 644-8.
98. Dam, T.; Tripathy, S. N.; Paluch, M.; Jena, S. S.; Pradhan, D. K. Investigations of relaxation dynamics and observation of nearly constant loss phenomena in PEO20-LiCF3SO3-ZrO2 based polymer nano-composite electrolyte. Electrochim. Acta. 2016, 202, 147-56.
99. Polu, A. R.; Rhee, H. Effect of TiO2 nanoparticles on structural, thermal, mechanical and ionic conductivity studies of PEO12-LiTDI solid polymer electrolyte. J. Ind. Eng. Chem. 2016, 37, 347-53.
100. Chen, L.; Li, Y.; Li, S.; Fan, L.; Nan, C.; Goodenough, J. B. PEO/garnet composite electrolytes for solid-state lithium batteries: from “ceramic-in-polymer” to “polymer-in-ceramic”. Nano. Energy. 2018, 46, 176-84.
101. Lei, R.; Yang, Y.; Yu, C.; Xu, Y.; Li, Y.; Li, J. A facile preparation of PEO-LiClO4-fumed SiO2 composite solid-state electrolyte with improved electrochemical performance for lithium-metal batteries. Sustain. Energy. Fuels. 2021, 5, 1538-47.
102. Li, S.; Wang, Y.; Anguita, J.; Yang, K.; Silva, S. R. P. Constructing vertically aligned Li + transport pathways in a flexible solid polymer composite electrolyte by a soft template approach. Nanoscale 2025, 17, 6833-40.
103. Xu, L.; Xiao, X.; Tu, H.; et al. Engineering functionalized 2D metal-organic frameworks nanosheets with fast Li + conduction for advanced solid Li batteries. Adv. Mater. 2023, 35, e2303193.
104. Wen, J.; Zhang, R.; Zhao, Q.; et al. Hydroxyapatite nanowire-reinforced poly(ethylene oxide)-based polymer solid electrolyte for application in high-temperature lithium batteries. ACS. Appl. Mater. Interfaces. 2020, 12, 54637-43.
105. Yu, J.; Wang, C.; Li, S.; Liu, N.; Zhu, J.; Lu, Z. Li + -containing, continuous silica nanofibers for high Li + conductivity in composite polymer electrolyte. Small 2019, 15, e1902729.
106. Hu, L.; Tang, Z.; Zhang, Z. New composite polymer electrolyte comprising mesoporous lithium aluminate nanosheets and PEO/LiClO4. J. Power. Sources. 2007, 166, 226-32.
107. Meng, X.; Zhang, D.; Mo, J.; et al. Room-temperature solid-state metallic lithium batteries based on high-content boron nitride nanosheet-modified polymer electrolytes. Appl. Surf. Sci. 2024, 648, 158962.
108. Li, R.; Guo, S.; Yu, L.; et al. Morphosynthesis of 3D macroporous garnet frameworks and perfusion of polymer-stabilized lithium salts for flexible solid-state hybrid electrolytes. Adv. Mater. Interfaces. 2019, 6, 1900200.
109. Li, Z.; Sha, W. X.; Guo, X. Three-dimensional garnet framework-reinforced solid composite electrolytes with high lithium-ion conductivity and excellent stability. ACS. Appl. Mater. Interfaces. 2019, 11, 26920-7.
110. Wu, J.; Rao, Z.; Cheng, Z.; Yuan, L.; Li, Z.; Huang, Y. Ultrathin, flexible polymer electrolyte for cost-effective fabrication of all-solid-state lithium metal batteries. Adv. Energy. Mater. 2019, 9, 1902767.
111. Mu, Y.; Yang, L.; Feng, Y.; et al. Achieving superior critical current density and rate performance in solid-state lithium batteries via vertically aligned LATP arrays. Nano. Energy. 2025, 138, 110885.
112. Zhou, K.; Zhang, M.; Zhang, X.; et al. A cellulose reinforced polymer composite electrolyte for the wide-temperature-range solid lithium batteries. Chem. Eng. J. 2023, 464, 142537.
113. Wang, T.; Zhang, M.; Zhou, K.; et al. A hetero-layered, mechanically reinforced, ultra-lightweight composite polymer electrolyte for wide-temperature-range, solid-state sodium batteries. Adv. Funct. Mater. 2023, 33, 2215117.
114. Zhang, Z.; YingHuang; Zhang, G.; Chao, L. Three-dimensional fiber network reinforced polymer electrolyte for dendrite-free all-solid-state lithium metal batteries. Energy. Storage. Mater. 2021, 41, 631-41.
115. Shi, P.; Ma, J.; Huang, Y.; et al. A thin and high-strength composite polymer solid-state electrolyte with a highly efficient and uniform ion-transport network. J. Mater. Chem. A. 2021, 9, 14344-51.
116. Tung, S. O.; Ho, S.; Yang, M.; Zhang, R.; Kotov, N. A. A dendrite-suppressing composite ion conductor from aramid nanofibres. Nat. Commun. 2015, 6, 6152.
117. Liu, Y.; Xu, H.; Wang, T.; Chen, M. PAN@UiO66 nanofibers with fast Li + transfer and outstanding mechanical performance for composite solid polymer electrolytes. ACS. Appl. Energy. Mater. 2025, 8, 3793-801.
118. Cao, Y.; Morrissey, T. G.; Acome, E.; et al. A transparent, self-healing, highly stretchable ionic conductor. Adv. Mater. 2017, 29.
119. Munaoka, T.; Yan, X.; Lopez, J.; et al. Ionically conductive self-healing binder for low cost Si microparticles anodes in Li-ion batteries. Adv. Energy. Mater. 2018, 8, 1703138.
120. Jaumaux, P.; Liu, Q.; Zhou, D.; et al. Deep-eutectic-solvent-based self-healing polymer electrolyte for safe and long-life lithium-metal batteries. Angew. Chem. Int. Ed. Engl. 2020, 59, 9134-42.
121. Cordier, P.; Tournilhac, F.; Soulié-Ziakovic, C.; Leibler, L. Self-healing and thermoreversible rubber from supramolecular assembly. Nature 2008, 451, 977-80.
122. Pei, F.; Wu, L.; Zhang, Y.; et al. Interfacial self-healing polymer electrolytes for long-cycle solid-state lithium-sulfur batteries. Nat. Commun. 2024, 15, 351.
123. Li, Z.; Fu, J.; Zheng, S.; Li, D.; Guo, X. Self-healing polymer electrolyte for dendrite-free Li metal batteries with ultra-high-voltage Ni-rich layered cathodes. Small 2022, 18, e2200891.
124. Chen, K.; Sun, Y.; Zhang, X.; Liu, J.; Xie, H. A self-healing and nonflammable cross-linked network polymer electrolyte with the combination of hydrogen bonds and dynamic disulfide bonds for lithium metal batteries. Energy. Environ. Mater. 2023, 6, e12568.
125. Zhang, L.; Zhang, P.; Chang, C.; Guo, W.; Guo, Z. H.; Pu, X. Self-healing solid polymer electrolyte for room-temperature solid-state lithium metal batteries. ACS. Appl. Mater. Interfaces. 2021, 13, 46794-802.
126. Li, Z.; Wang, L.; Huang, X.; He, X. Unveiling the mystery of LiF within solid electrolyte interphase in lithium batteries. Small 2024, 20, e2305429.
127. Eshetu, G. G.; Judez, X.; Li, C.; et al. Ultrahigh performance all solid-state lithium sulfur batteries: salt anion’s chemistry-induced anomalous synergistic effect. J. Am. Chem. Soc. 2018, 140, 9921-33.
128. Wang, C.; Wang, T.; Wang, L.; et al. Differentiated lithium salt design for multilayered PEO electrolyte enables a high-voltage solid-state lithium metal battery. Adv. Sci. 2019, 6, 1901036.
129. Zhang, Z.; Wang, J.; Zhang, S.; et al. Stable all-solid-state lithium metal batteries with Li3N-LiF-enriched interface induced by lithium nitrate addition. Energy. Storage. Mater. 2021, 43, 229-37.
130. Li, H.; Liu, W.; Yang, X.; et al. Fluoroethylene carbonate-Li-ion enabling composite solid-state electrolyte and lithium metal interface self-healing for dendrite-free lithium deposition. Chem. Eng. J. 2021, 408, 127254.
131. Li, X.; Liu, Q.; Wang, Y.; et al. In situ polymerized solid-state electrolyte enabling inorganic-organic dual-layered SEI film for stable lithium metal batteries. Small 2025, 21, e2411104.
132. Ionic conductivity and mechanical properties of the solid electrolyte interphase in lithium metal batteries. Energy. Mater. 2023.
133. Hu, L.; Gao, X.; Li, Z.; et al. Layered polymer stacking for stable interfaces and dendrite growth inhibition in all-solid-state lithium batteries. ACS. Appl. Mater. Interfaces. 2023, 15, 38485-95.
134. Fang, R.; Xu, B.; Grundish, N. S.; et al. Li2S6-integrated PEO-based polymer electrolytes for all-solid-state lithium-metal batteries. Angew. Chem. Int. Ed. Engl. 2021, 60, 17701-6.
135. Li, H.; Li, L.; Zheng, J.; et al. Thermal decomposition assisted construction of nano-Li3N sites interface layer enabling homogeneous Li deposition. ChemSusChem 2023, 16, e202202220.
136. Suyama, M.; Yubuchi, S.; Deguchi, M.; Sakuda, A.; Tatsumisago, M.; Hayashi, A. Importance of Li-metal/sulfide electrolyte interphase ionic conductivity in suppressing short-circuiting of all-solid-state Li-metal batteries. J. Electrochem. Soc. 2021, 168, 060542.
137. Wu, N.; Li, Y.; Dolocan, A.; et al. In situ formation of Li3P layer enables fast Li + conduction across Li/solid polymer electrolyte interface. Adv. Funct. Mater. 2020, 30, 2000831.
138. Huggins, R. A. Recent results on lithium ion conductors. In International Symposium on Solid Ionic and Ionic-Electronic Conductors; Elsevier, 1977; pp 773-81.
139. Ding, J.; Mou, J.; Lin, S.; et al. In situ construction of a Li-Ag&LiF interface enables stable cycling of all-solid-state lithium-metal batteries. J. Colloid. Interface. Sci. 2025, 686, 1000-8.
140. Li, H.; Jiu, H.; Zhang, L.; et al. CoF2-modified PEO-LLZTO composite polymer electrolyte to optimize Li + transport pathway. J. Electroanal. Chem. 2025, 984, 119035.
141. Wang, A.; Yi, M.; Chang, S.; et al. Regulating Li deposition behavior at anodic interface induced by SbF3 electrolyte additive in all-solid-state Li metal batteries. Chem. Eng. J. 2023, 474, 145593.
142. Zhang, X.; Zhang, H.; Geng, Y.; et al. A multifunctional nano filler for solid polymer electrolyte toward stable cycling for lithium-metal anodes in lithium-sulfur batteries. Chem. Eng. J. 2022, 444, 136328.
143. Wu, K.; Li, A.; Tan, J.; et al. SnF2-catalyzed lithiophilic-lithiophobic gradient interface for high-rate PEO-based all-solid-state batteries. Angew. Chem. Int. Ed. Engl. 2024, 63, e202410347.
144. Chen, X.; Chu, F.; Li, D.; Si, M.; Liu, M.; Wu, F. In situ polymerized fluorine-free ether gel polymer electrolyte with stable interface for high-voltage lithium metal batteries. Adv. Funct. Mater. 2024, 35, 2421965.
145. Lu, B.; Hu, L.; Zhang, W.; et al. Li-Ga alloy-contained hybrid solid electrolyte interphase induced by in situ polymerization for high-performance lithium metal batteries. ACS. Appl. Mater. Interfaces. 2024, 16, 55314-24.
146. Xu, L.; Li, J.; Xiang, Y.; et al. Few-layer bismuthene enabled solid-state Li batteries. Energy. Storage. Mater. 2022, 52, 655-63.
147. Zeng, Z.; Liu, G.; Jiang, Z.; Peng, L.; Xie, J. Zinc bis(2-ethylhexanoate), a homogeneous and bifunctional additive, to improve conductivity and lithium deposition for poly (ethylene oxide) based all-solid-state lithium metal battery. J. Power. Sources. 2020, 451, 227730.
148. Ou, C.; Ye, S.; Li, Z.; et al. The intermolecular interaction enables ordered ion transport in quasi-solid-state electrolyte for ultra-long life lithium-metal battery. Energy. Storage. Mater. 2024, 67, 103277.
149. Liu, J.; Mao, R.; Wang, T.; Liu, X.; Qu, Y.; Zhang, B. In situ construction of rich-LiF/LiBr SEI by ion-supported porous polymer strategy for high interfacial stability all-solid-state lithium metal batteries. Adv. Funct. Mater. 2025, 36, e23301.
150. Liang, W.; Zhou, X.; Zhang, B.; et al. The versatile establishment of charge storage in polymer solid electrolyte with enhanced charge transfer for LiF-rich SEI generation in lithium metal batteries. Angew. Chem. Int. Ed. Engl. 2024, 63, e202320149.
151. Li, J.; Cai, Y.; Zhang, F.; et al. Exceptional interfacial conduction and LiF interphase for ultralong life PEO-based all-solid-state batteries. Nano. Energy. 2023, 118, 108985.
152. Mu, Y.; Chu, Y.; Shi, Y.; et al. Constructing robust LiF-enriched interfaces in high-voltage solid-state lithium batteries utilizing tailored oriented ceramic fiber electrolytes. Adv. Energy. Mater. 2024, 14, 2400725.
153. Zheng, Y.; Yang, N.; Duan, S.; et al. Dual-enhanced charge transfer through prelithiation strategy in polymer electrolyte enables robust LiF-rich SEI for ultralong-life all-solid-state batteries. Adv. Funct. Mater. 2025, 36, e11011.
154. Zhang, X.; Jia, M.; Zhang, Q.; et al. LiNO3 and TMP enabled high voltage room-temperature solid-state lithium metal battery. Chem. Eng. J. 2022, 448, 137743.
155. Liu, H.; Liu, X.; Zhang, N.; et al. Functional ternary salt construction enabling an in-situ Li3N/LiF-enriched interface for ultra-stable all-solid-state lithium metal batteries. J. Energy. Chem. 2025, 101, 68-75.
156. Su, Y.; Zhang, X.; Du, C.; et al. An all-solid-state battery based on sulfide and PEO composite electrolyte. Small 2022, 18, e2202069.
157. Chen, X.; Wang, Z.; Si, M.; et al. Reinforced anti-oxidative degradation and interface stabilization in bimetal oxide filler-based PEO electrolytes for lithium metal batteries. Adv. Funct. Mater. 2025, 35, 2504306.
158. Zhao, Z.; Joe, H. J.; Desalegn, B. Z.; Kim, S. K.; Wang, D.; Seo, J. G. Homogeneous Li flux and mechanical stable solid-electrolyte interphase enabled by different solvation chemistry for lithium metal batteries. Adv. Funct. Mater. 2025, 35, e08653.
159. Fan, Z.; Ding, B.; Zhang, T. Solid/solid interfacial architecturing of solid polymer electrolyte-based all-solid-state lithium-sulfur batteries by atomic layer deposition. Small 2019, 15, e1903952.
160. Shi, Y.; Fan, Z.; Ding, B.; et al. Atomic-scale Al2O3 modified PEO-based composite polymer electrolyte for durable solid-state Li-S batteries. J. Electroanal. Chem. 2021, 881, 114916.
161. Wang, L.; Zhang, L.; Wang, Q.; et al. Long lifespan lithium metal anodes enabled by Al2O3 sputter coating. Energy. Storage. Mater. 2018, 10, 16-23.
162. Yan, M.; Liang, J. Y.; Zuo, T. T.; et al. Stabilizing polymer-lithium interface in a rechargeable solid battery. Adv. Funct. Mater. 2019, 30, 1908047.
163. Deng, J.; Wang, Y.; Qu, S.; et al. Fast Li + transport of Li-Zn alloy protective layer enabling excellent electrochemical performance of Li metal anode. Batteries. Supercaps. 2020, 4, 140-5.
164. Zhang, Y.; Wang, H.; Yang, Y.; et al. Polyacrylonitrile fibers network reinforced polymer electrolyte with Li-Sn alloy layer protected Li anode toward ultra-long cycle lifespan for room-temperature solid-state batteries. Chem. Eng. J. 2023, 461, 141993.
165. Liu, Y.; Yang, T.; Fang, R.; et al. Ultra-homogeneous dense Ag nano layer enables long lifespan solid-state lithium metal batteries. J. Energy. Chem. 2024, 96, 110-9.
166. Zhang, S.; Sun, Q.; Hou, G.; et al. Boosting fast interfacial Li + transport in solid-state Li metal batteries via ultrathin Al buffer layer. Nano. Res. 2022, 16, 6825-32.
167. Liu, M.; Wang, C.; Cheng, Z.; et al. Controlling the lithium-metal growth to enable low-lithium-metal-excess all-solid-state lithium-metal batteries. ACS. Mater. Lett. 2020, 2, 665-70.
168. Wang, A.; Li, J.; Yi, M.; et al. Stable all-solid-state lithium metal batteries enabled by ultrathin LiF/Li3Sb hybrid interface layer. Energy. Storage. Mater. 2022, 49, 246-54.
169. Yang, H.; Zhang, Y.; Tennenbaum, M. J.; et al. Polypropylene carbonate-based adaptive buffer layer for stable interfaces of solid polymer lithium metal batteries. ACS. Appl. Mater. Interfaces. 2019, 11, 27906-12.
170. Liu, J.; Qian, T.; Wang, M.; Zhou, J.; Xu, N.; Yan, C. Use of tween polymer to enhance the compatibility of the Li/electrolyte interface for the high-performance and high-safety quasi-solid-state lithium-sulfur battery. Nano. Lett. 2018, 18, 4598-605.
171. Wang, Z.; Tan, J.; Cui, J.; et al. A novel asymmetrical multilayered composite electrolyte for high-performance ambient-temperature all-solid-state lithium batteries. J. Mater. Chem. A. 2024, 12, 4231-9.
172. Ye, Q.; Liang, H.; Wang, S.; et al. Fabricating a PVDF skin for PEO-based SPE to stabilize the interface both at cathode and anode for Li-ion batteries. J. Energy. Chem. 2022, 70, 356-62.
173. Chai, Y.; Ning, D.; Zhou, D.; et al. Construction of flexible asymmetric composite polymer electrolytes for high-voltage lithium metal batteries with superior performance. Nano. Energy. 2024, 130, 110160.
174. Pan, J.; Zhang, Y.; Wang, J.; et al. A quasi-double-layer solid electrolyte with adjustable interphases enabling high-voltage solid-state batteries. Adv. Mater. 2022, 34, e2107183.
175. Wang, J.; Zhang, Z.; Ying, H.; et al. An effective artificial layer boosting high-performance all-solid-state lithium batteries with high coulombic efficiency. J. Materiomics. 2022, 8, 257-65.
176. Wang, J.; Zhang, Z.; Ying, H.; Han, G.; Han, W. In-situ formation of LiF-rich composite interlayer for dendrite-free all-solid-state lithium batteries. Chem. Eng. J. 2021, 411, 128534.
177. Chen, L.; Li, W.; Fan, L. Z.; Nan, C. W.; Zhang, Q. Intercalated electrolyte with high transference number for dendrite-free solid-state lithium batteries. Adv. Funct. Mater. 2019, 29, 1901047.
178. Ye, Y.; Deng, Z.; Gao, L.; et al. Lithium-rich anti-perovskite Li2OHBr-based polymer electrolytes enabling an improved interfacial stability with a three-dimensional-structured lithium metal anode in all-solid-state batteries. ACS. Appl. Mater. Interfaces. 2021, 13, 28108-17.
179. Zhu, Y.; Han, Y.; Guo, Q.; et al. Lithium- gel polymer electrolyte composite anode with large electrolyte-lithium interface for solid-state battery. Electrochim. Acta. 2021, 394, 139123.
180. Liu, Y.; Lin, D.; Jin, Y.; et al. Transforming from planar to three-dimensional lithium with flowable interphase for solid lithium metal batteries. Sci. Adv. 2017, 3, eaao0713.
181. Zhu, J.; Cai, D.; Li, J.; et al. In-situ generated Li3N/Li-Al alloy in reduced graphene oxide framework optimizing ultra-thin lithium metal electrode for solid-state batteries. Energy. Storage. Mater. 2022, 49, 546-54.
182. Park, B.; Oh, C.; Yu, S.; et al. Coupling of 3D porous hosts for Li metal battery anodes with viscous polymer electrolytes. J. Electrochem. Soc. 2022, 169, 010511.
183. Liu, Z.; Fu, X.; Li, Z.; et al. Integrated anode with 3D electron/ion conductive network for stable lithium metal batteries. Energy. Storage. Mater. 2024, 66, 103201.
184. Pan, H.; Zhang, M.; Cheng, Z.; et al. Carbon-free and binder-free Li-Al alloy anode enabling an all-solid-state Li-S battery with high energy and stability. Sci. Adv. 2022, 8, eabn4372.
185. Kim, M.; Kim, M. J.; Oh, Y. S.; Kang, S.; Shin, T. H.; Lim, H. T. Design strategies of Li-Si alloy anode for mitigating chemo-mechanical degradation in sulfide-based all-solid-state batteries. Adv. Sci. 2023, 10, e2301381.
186. Abraham, K. Recent developments in secondary lithium battery technology. J. Power. Sources. 1985, 14, 179-91.
187. Huggins, R. Alternative materials for negative electrodes in lithium systems. Solid. State. Ionics. 2002, 152-153, 61-8.
188. Lee, J.; Jeong, S. H.; Nam, J. S.; et al. Toward thin and stable anodes for practical lithium metal batteries: A review, strategies, and perspectives. EcoMat 2023, 5, e12416.
189. Bang, H. J.; Kim, S.; Prakash, J. Electrochemical investigations of lithium-aluminum alloy anode in Li/polymer cells. J. Power. Sources. 2001, 92, 45-9.
190. Zhang, Y.; Chen, R.; Wang, S.; et al. Free-standing sulfide/polymer composite solid electrolyte membranes with high conductance for all-solid-state lithium batteries. Energy. Storage. Mater. 2020, 25, 145-53.
191. Hänsel, C.; Singh, B.; Kiwic, D.; Canepa, P.; Kundu, D. Favorable interfacial chemomechanics enables stable cycling of high-Li-content Li-In/Sn anodes in sulfide electrolyte-based solid-state batteries. Chem. Mater. 2021, 33, 6029-40.
192. Yan, W.; Mu, Z.; Wang, Z.; et al. Hard-carbon-stabilized Li-Si anodes for high-performance all-solid-state Li-ion batteries. Nat. Energy. 2023, 8, 800-13.
193. Huang, Y.; Shao, B.; Han, F. Li alloy anodes for high-rate and high-areal-capacity solid-state batteries. J. Mater. Chem. A. 2022, 10, 12350-8.
194. Chen, L.; Venkatram, S.; Kim, C.; Batra, R.; Chandrasekaran, A.; Ramprasad, R. Electrochemical stability window of polymeric electrolytes. Chem. Mater. 2019, 31, 4598-604.
195. Pandian, S.; Adiga, S.; Tagade, P.; Hariharan, K.; Mayya, K.; Lee, Y. Electrochemical stability of ether based salt-in-polymer based electrolytes: computational investigation of the effect of substitution and the type of salt. J. Power. Sources. 2018, 393, 204-10.
196. Dai, Y.; Hou, Z.; Luo, G.; et al. Improving the interfacial stability of ultrahigh-nickel cathodes with PEO-based electrolytes by targeted chemical reactions. Chem. Sci. 2024, 15, 12964-72.
197. Chen, X.; Li, Y.; Lu, Y.; et al. LATP-coated LiNi0.8Co0.1Mn0.1O2 cathode with compatible interface with ultrathin PVDF-reinforced PEO-LLZTO electrolyte for stable solid-state lithium batteries. J. Materiomics. 2024, 10, 682-93.
198. Li, Q.; Zhang, X.; Peng, J.; et al. Engineering a high-voltage durable cathode/electrolyte interface for all-solid-state lithium metal batteries via in situ electropolymerization. ACS. Appl. Mater. Interfaces. 2022, 14, 21018-27.
199. Cheng, Z.; Xiang, J.; Yuan, L.; et al. Multifunctional additive enables a “5H” PEO solid electrolyte for high-performance lithium metal batteries. ACS. Appl. Mater. Interfaces. 2024, 16, 21924-31.
200. Kim, S. H.; Park, N.; Bo Lee, W.; Park, J. H. Functional sulfate additive-derived interfacial layer for enhanced electrochemical stability of PEO-based polymer electrolytes. Small 2024, 20, e2309160.
201. Ye, M.; Chen, J.; Deng, H.; et al. In-situ electrochemical passivation for constructing high-voltage PEO-based solid-state lithium battery. Chem. Eng. J. 2024, 488, 151108.
202. Arrese-igor, M.; Martinez-ibañez, M.; Pavlenko, E.; et al. Toward high-voltage solid-state Li-metal batteries with double-layer polymer electrolytes. ACS. Energy. Lett. 2022, 7, 1473-80.
203. Schreiber, A.; Rosen, M.; Waetzig, K.; et al. Oxide ceramic electrolytes for all-solid-state lithium batteries - cost-cutting cell design and environmental impact. Green. Chem. 2023, 25, 399-414.
204. Wu, Y.; Zhang, Z.; Zhang, Q.; et al. Industrialization challenges for sulfide-based all solid state battery. eTransportation 2024, 22, 100371.
205. Kong, W.; Jiang, Z.; Liu, Y.; et al. Stabilizing Li1.3Al0.3Ti1.7(PO4)3|Li metal anode interface in solid-state batteries by Kevlar aramid nanofiber-based protective coating. Adv. Funct. Mater. 2023, 33, 2306748.
206. Wang, S.; Lou, C.; Wu, X.; et al. Large-scale manufacturing sulfide superionic conductor for advancing all-solid-state batteries. Matter 2025, 8, 102135.
207. Lee, W.; Lee, J.; Yu, T.; et al. Advanced parametrization for the production of high-energy solid-state lithium pouch cells containing polymer electrolytes. Nat. Commun. 2024, 15, 5860.
208. Song, Z.; Chen, F.; Martinez-Ibañez, M.; et al. A reflection on polymer electrolytes for solid-state lithium metal batteries. Nat. Commun. 2023, 14, 4884.
209. Cywar, R. M.; Rorrer, N. A.; Hoyt, C. B.; Beckham, G. T.; Chen, E. Y. Bio-based polymers with performance-advantaged properties. Nat. Rev. Mater. 2021, 7, 83-103.
210. Kim, J.; Sun, J.; Kim, J.; et al. Machine learning-driven discovery of innovative hybrid solid electrolytes for high-performance all-solid-state batteries. Chem. Eng. J. 2025, 511, 161926.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data



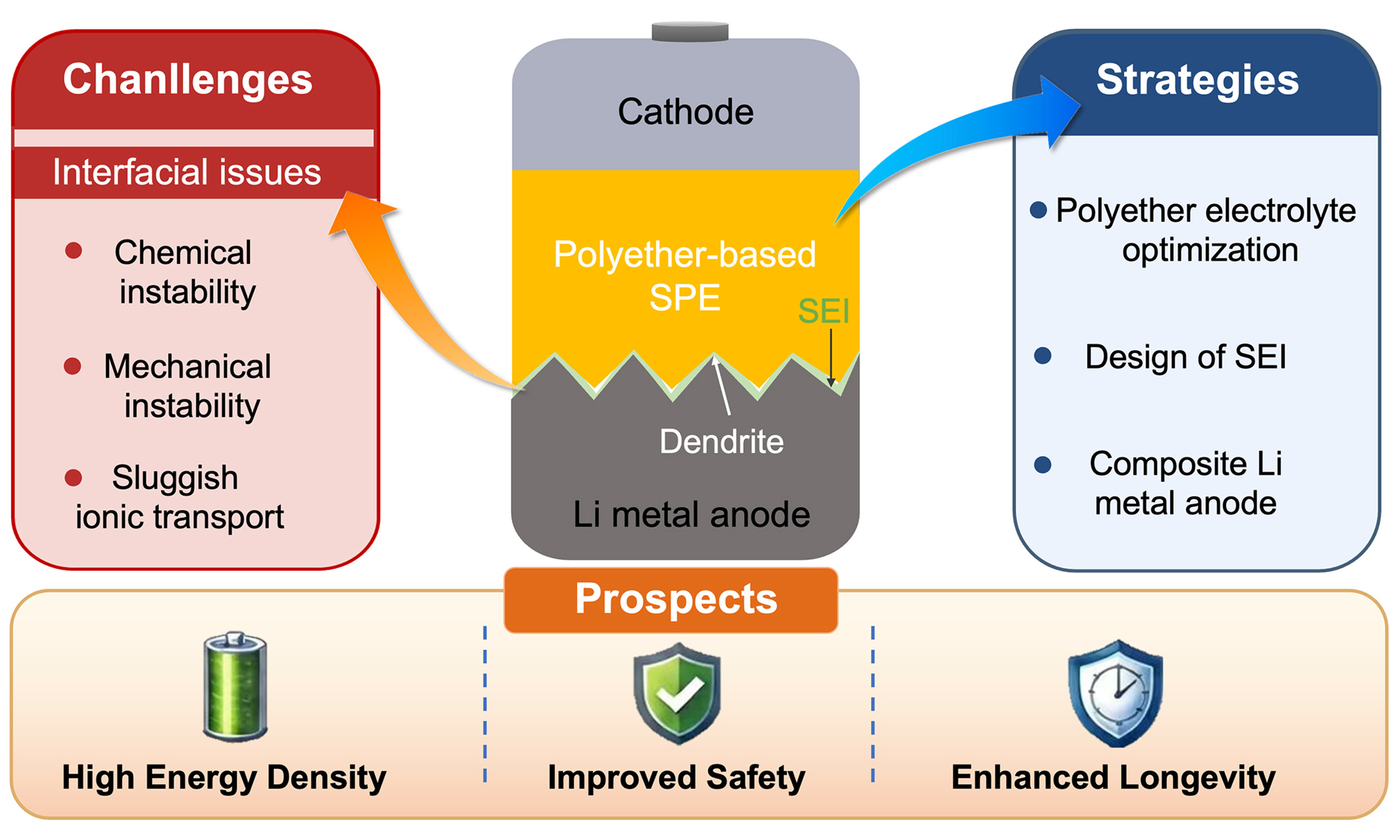
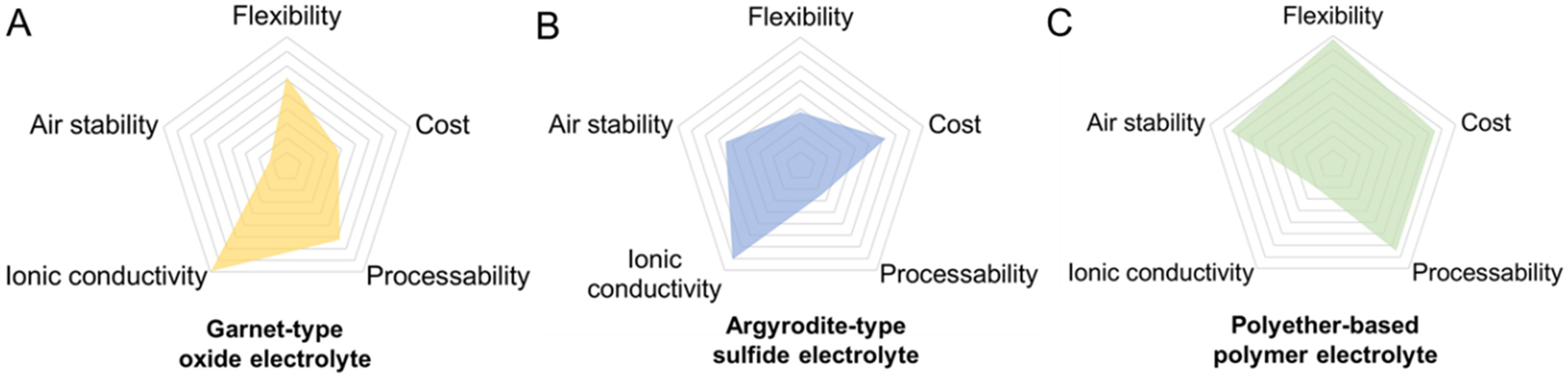
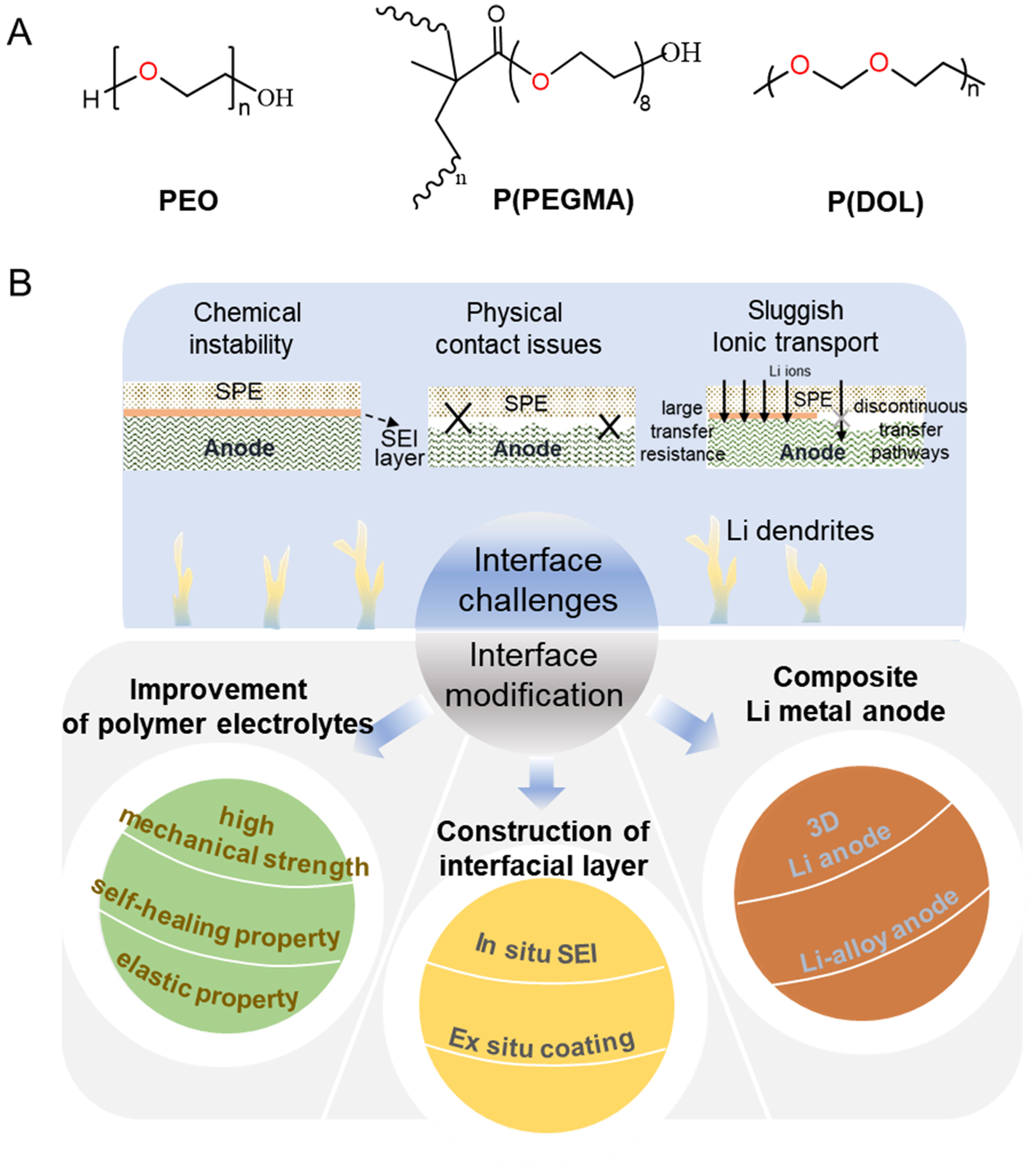
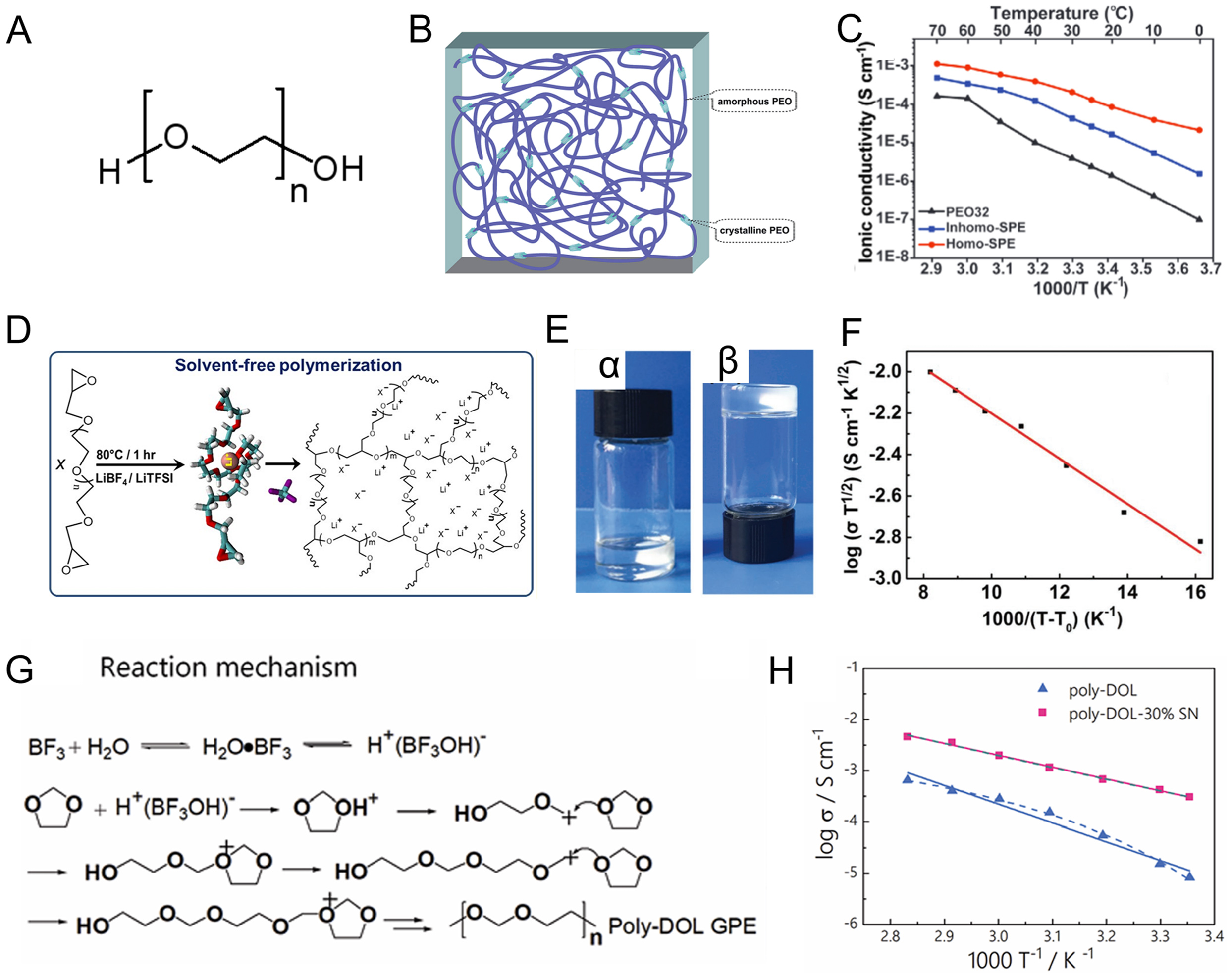
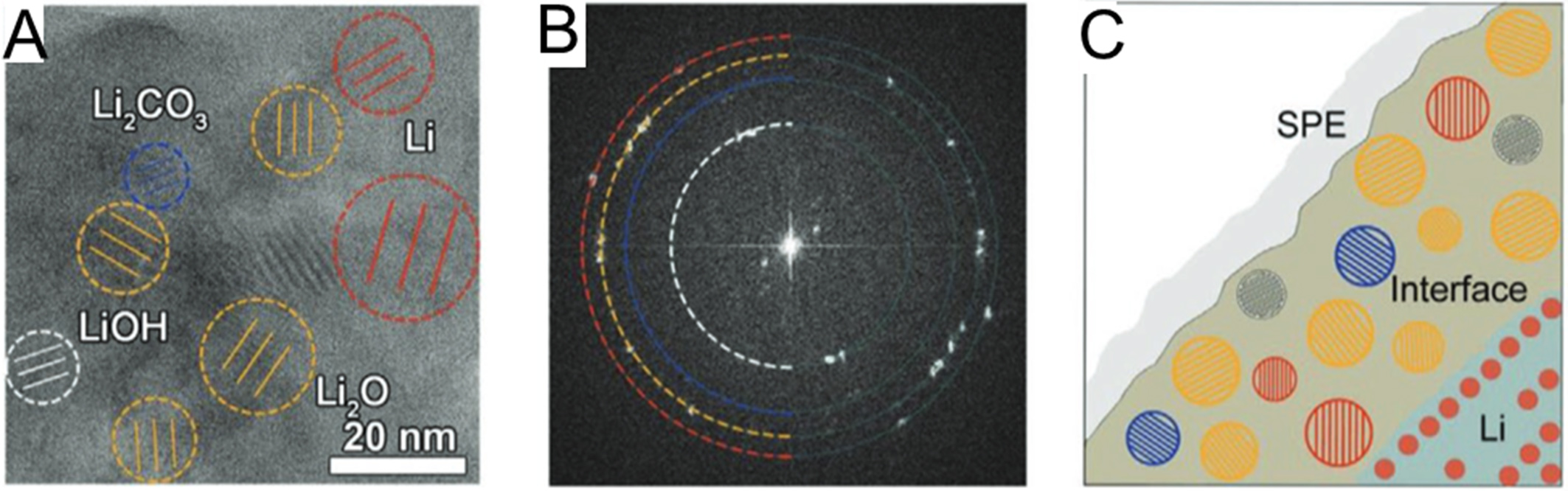
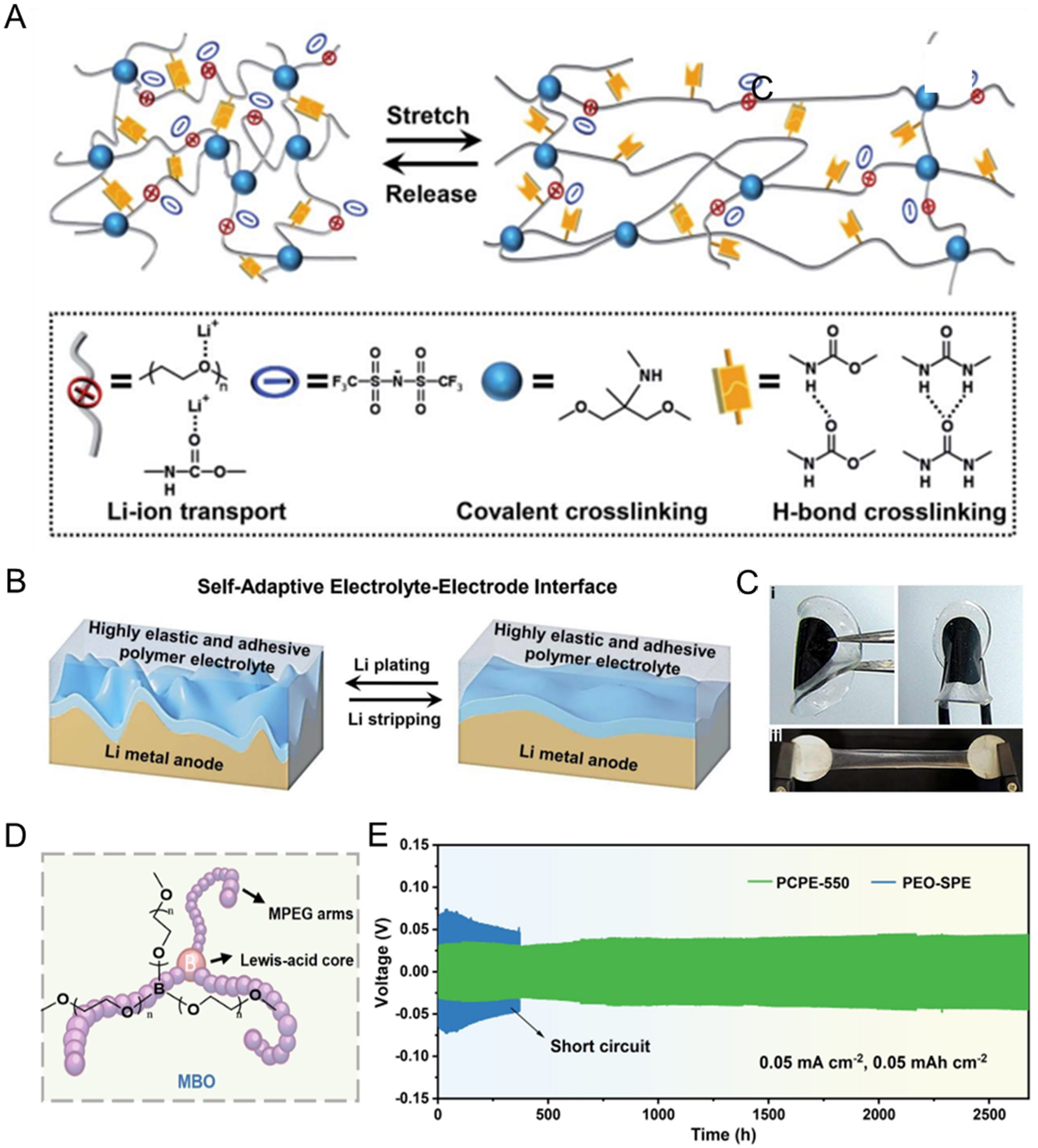
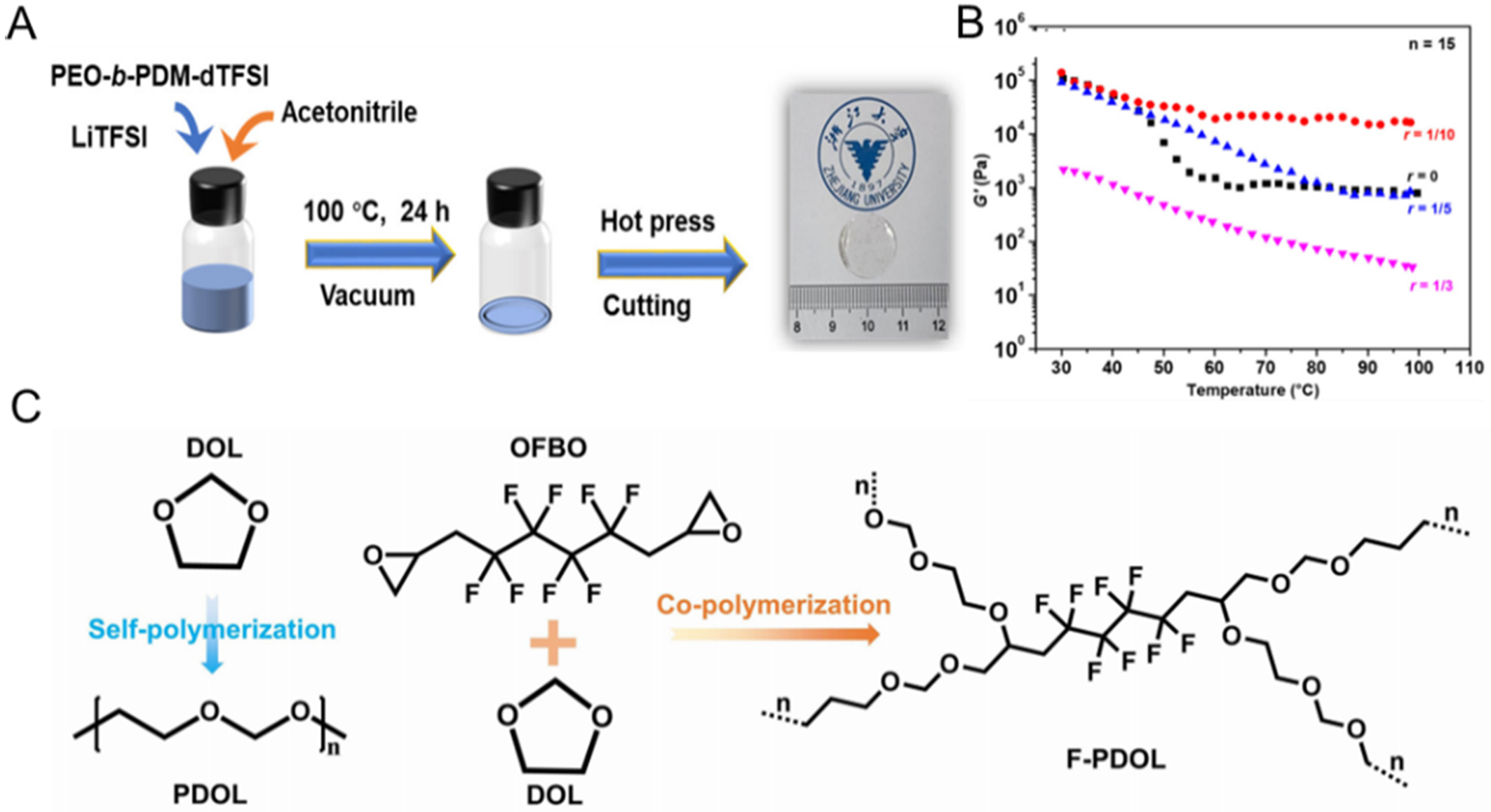
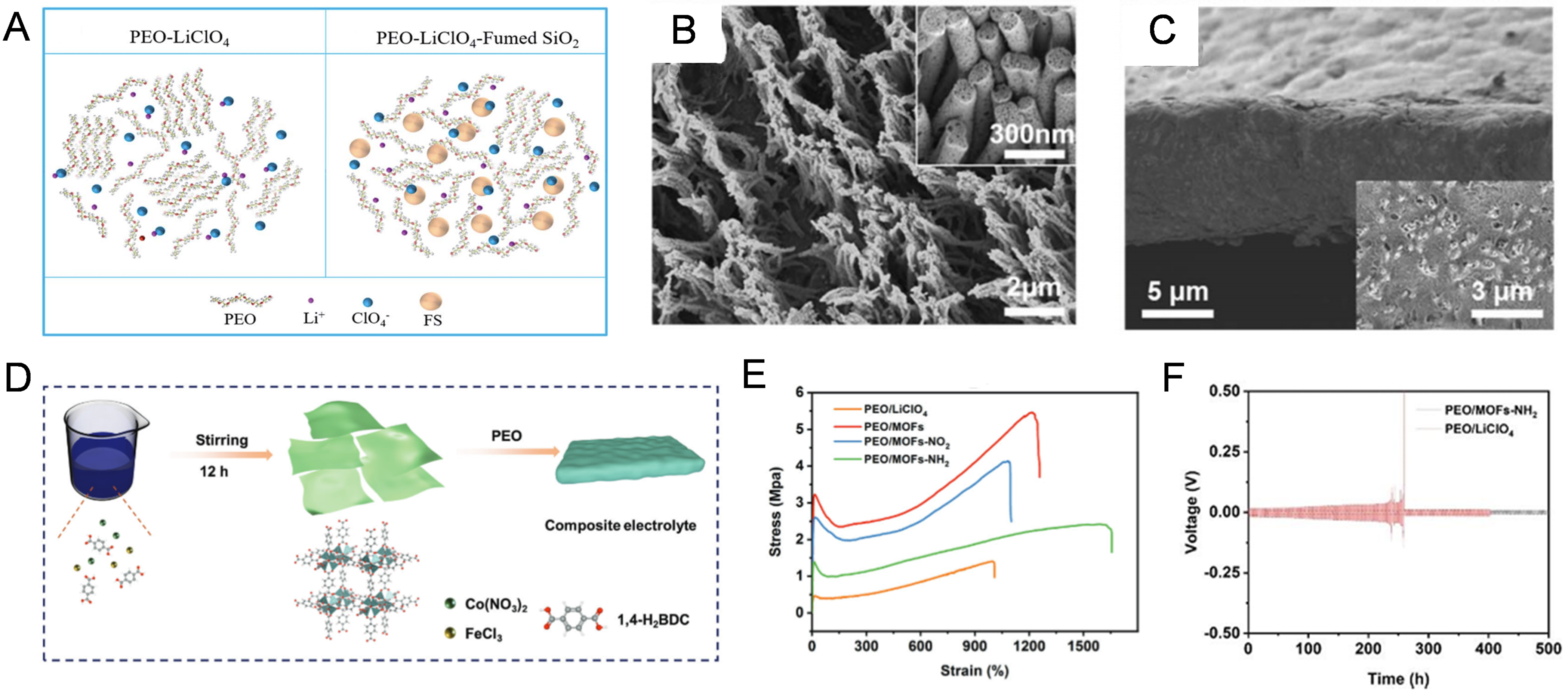
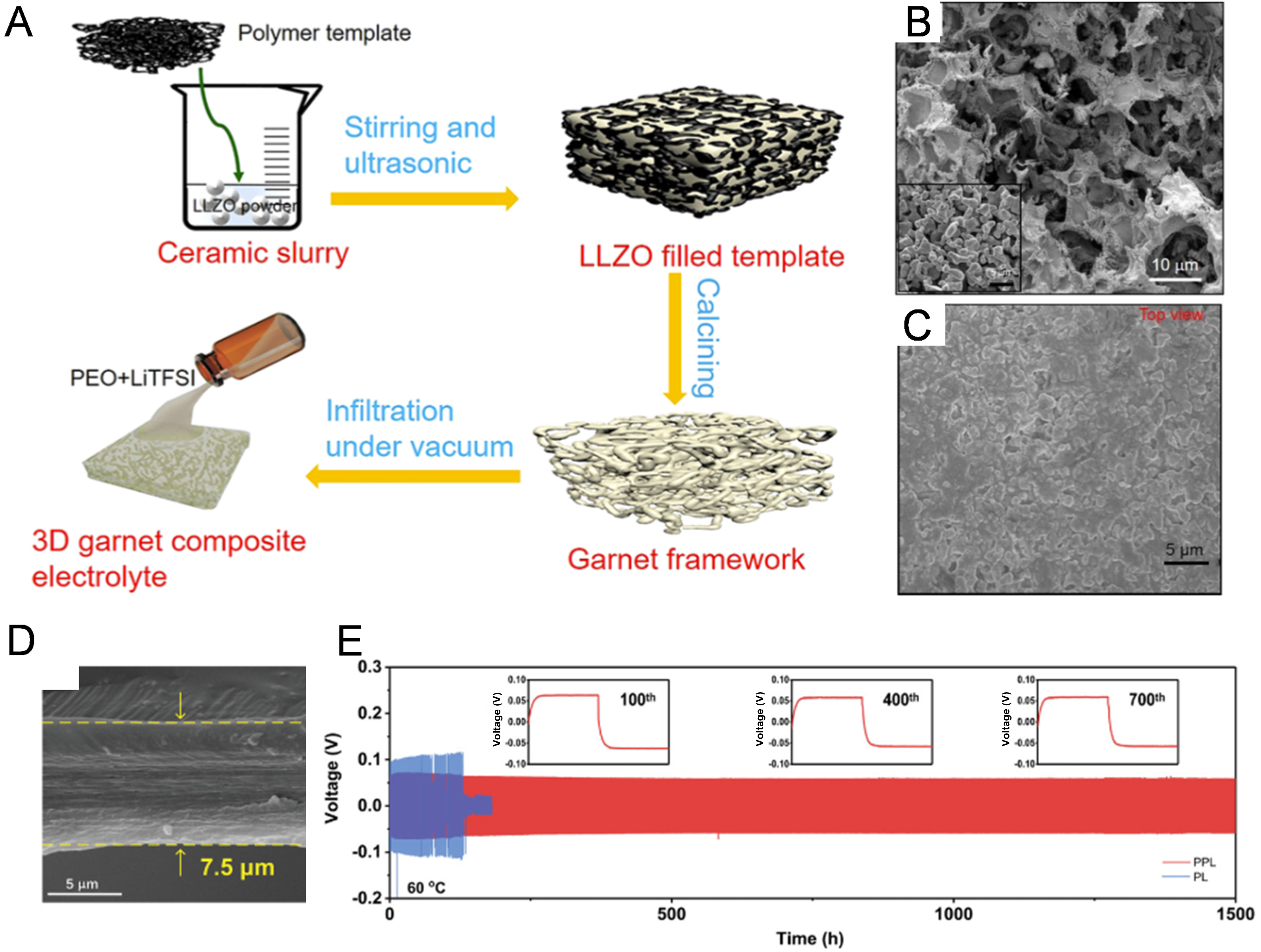
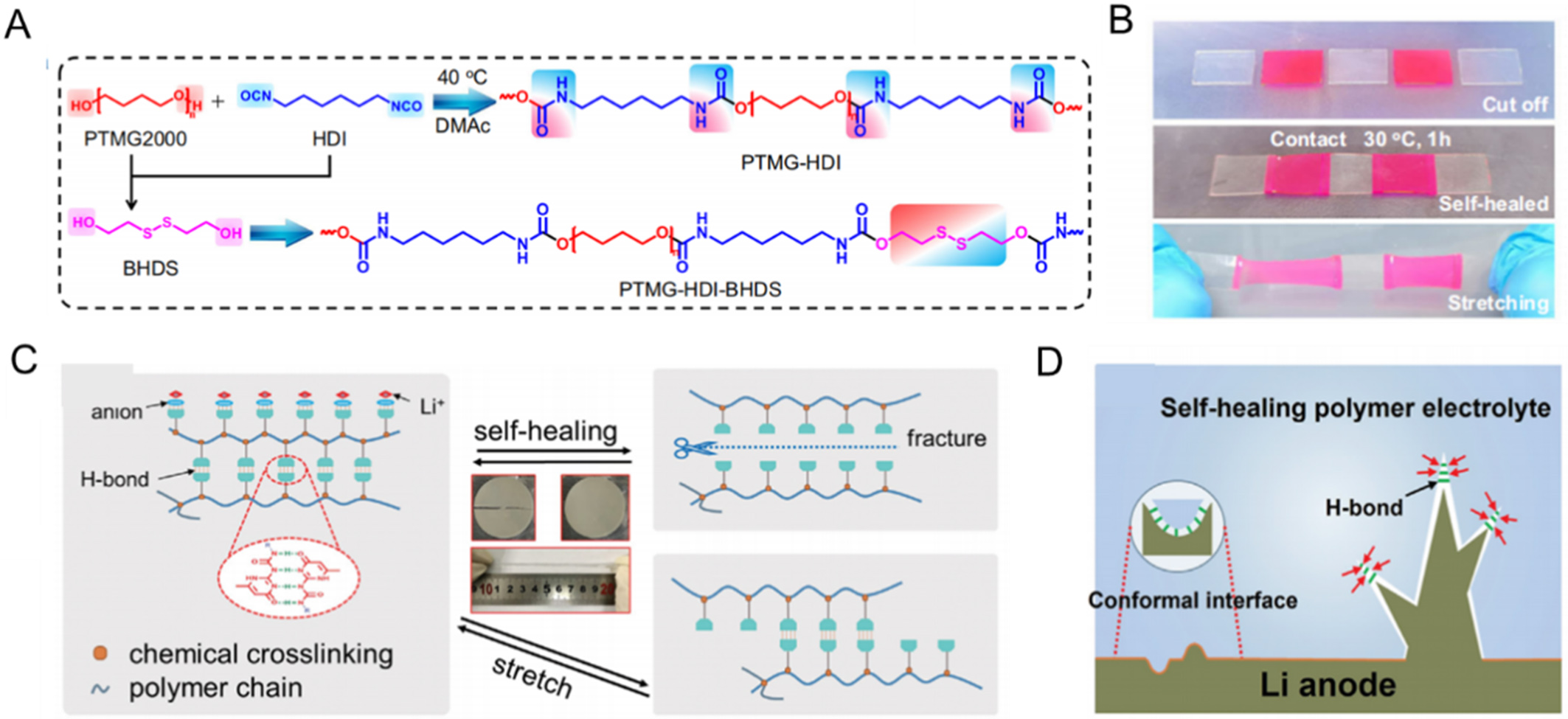
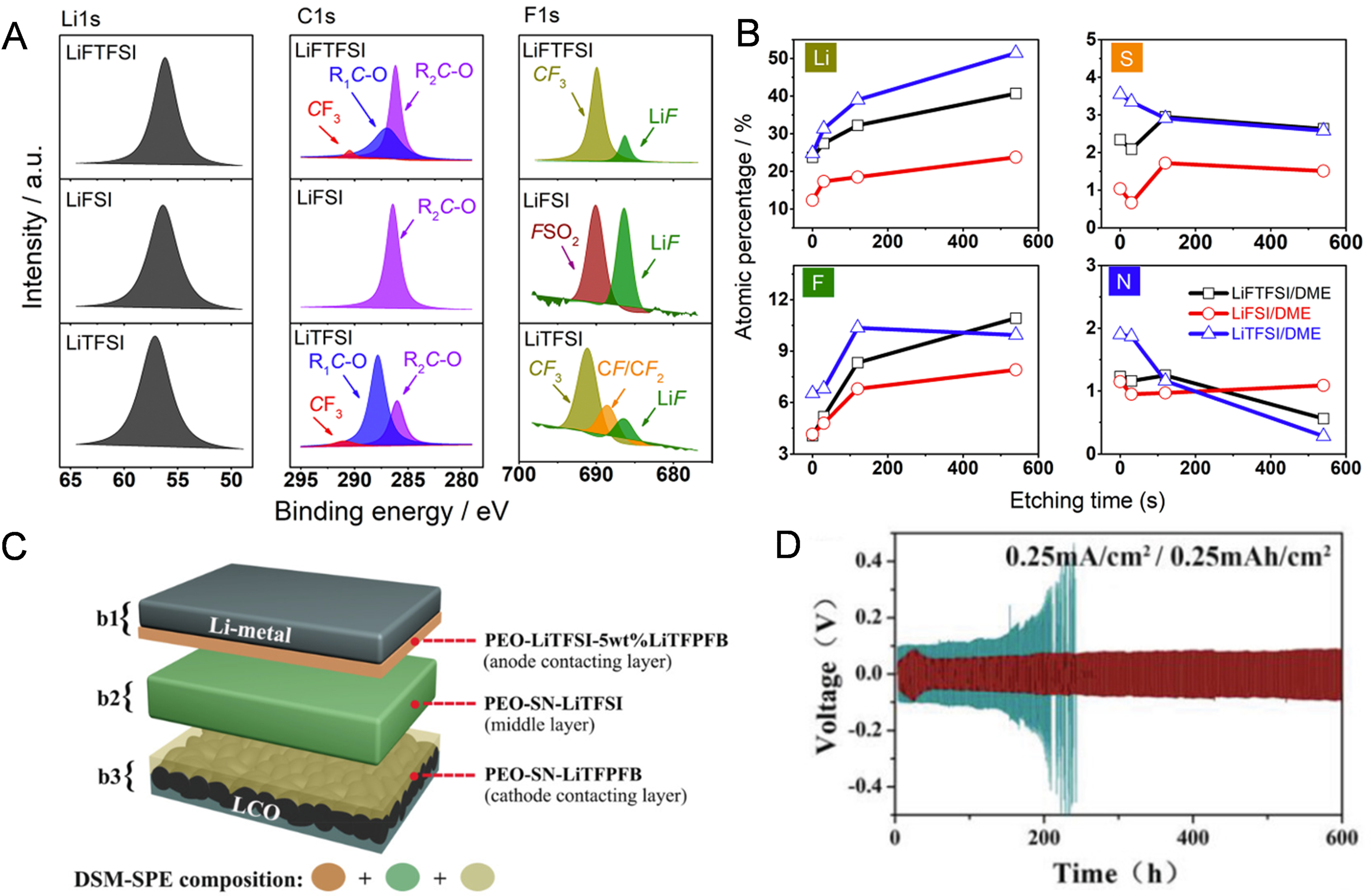
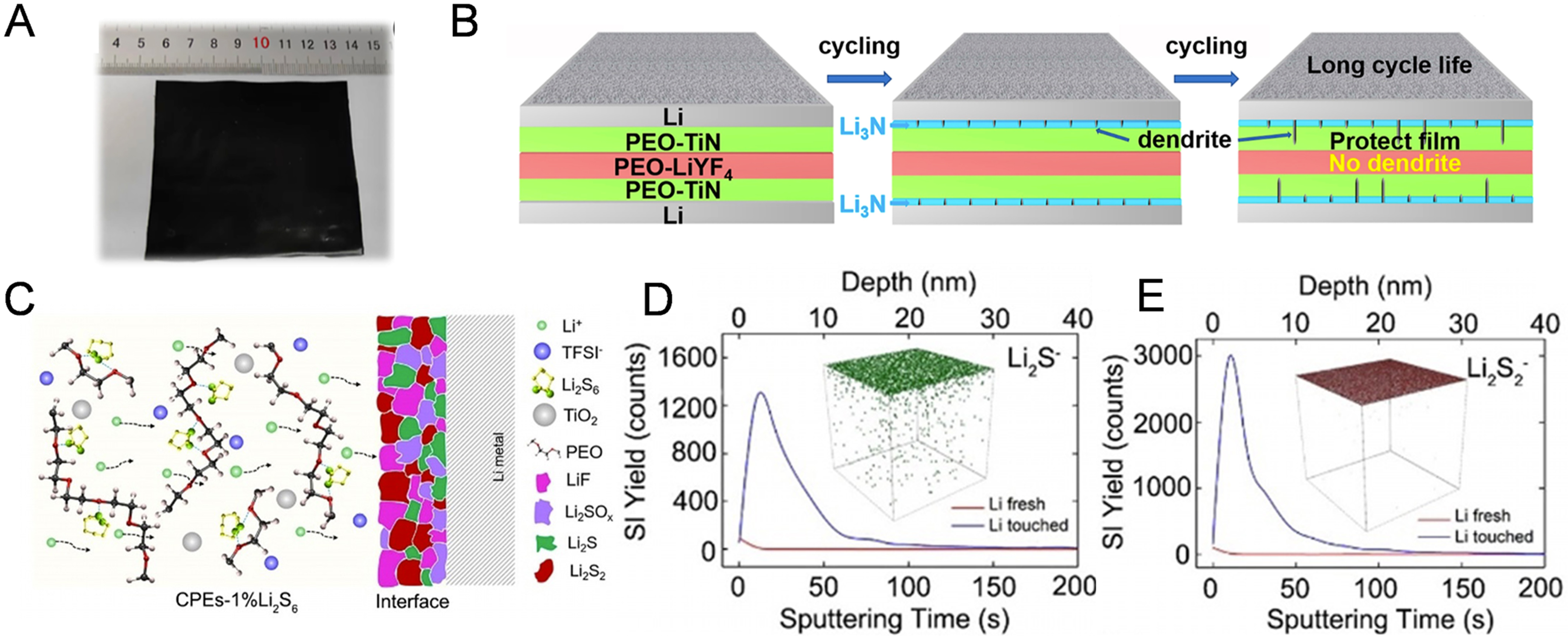
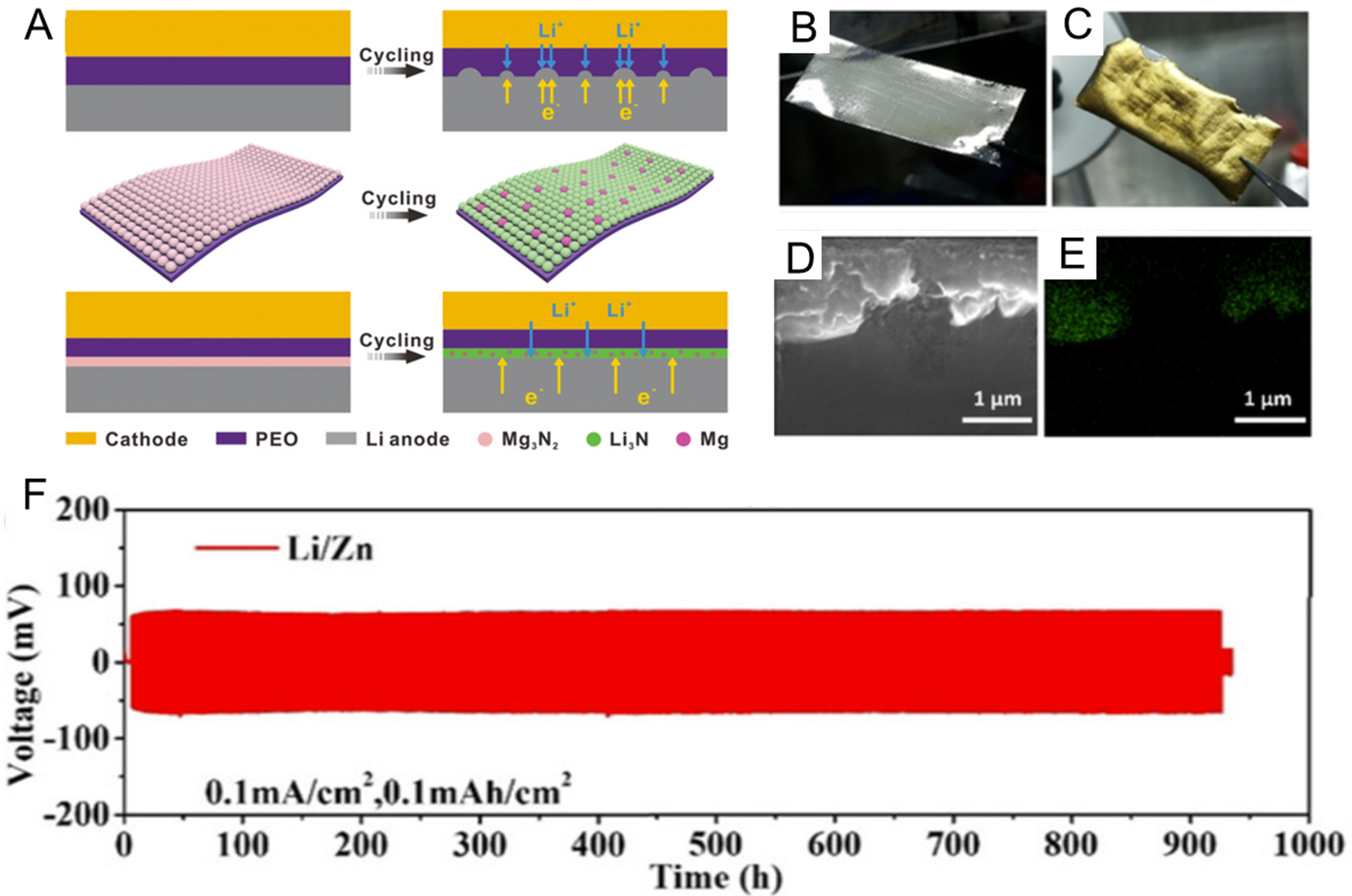
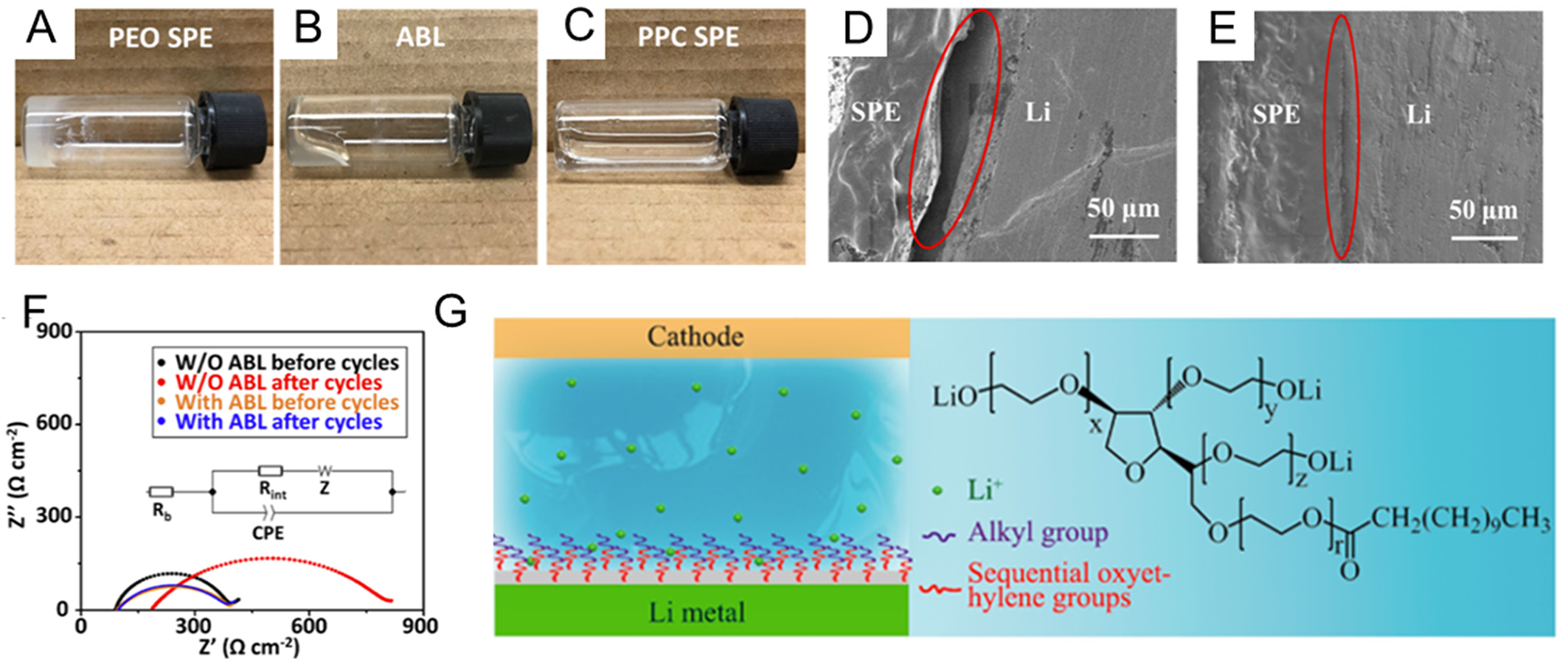
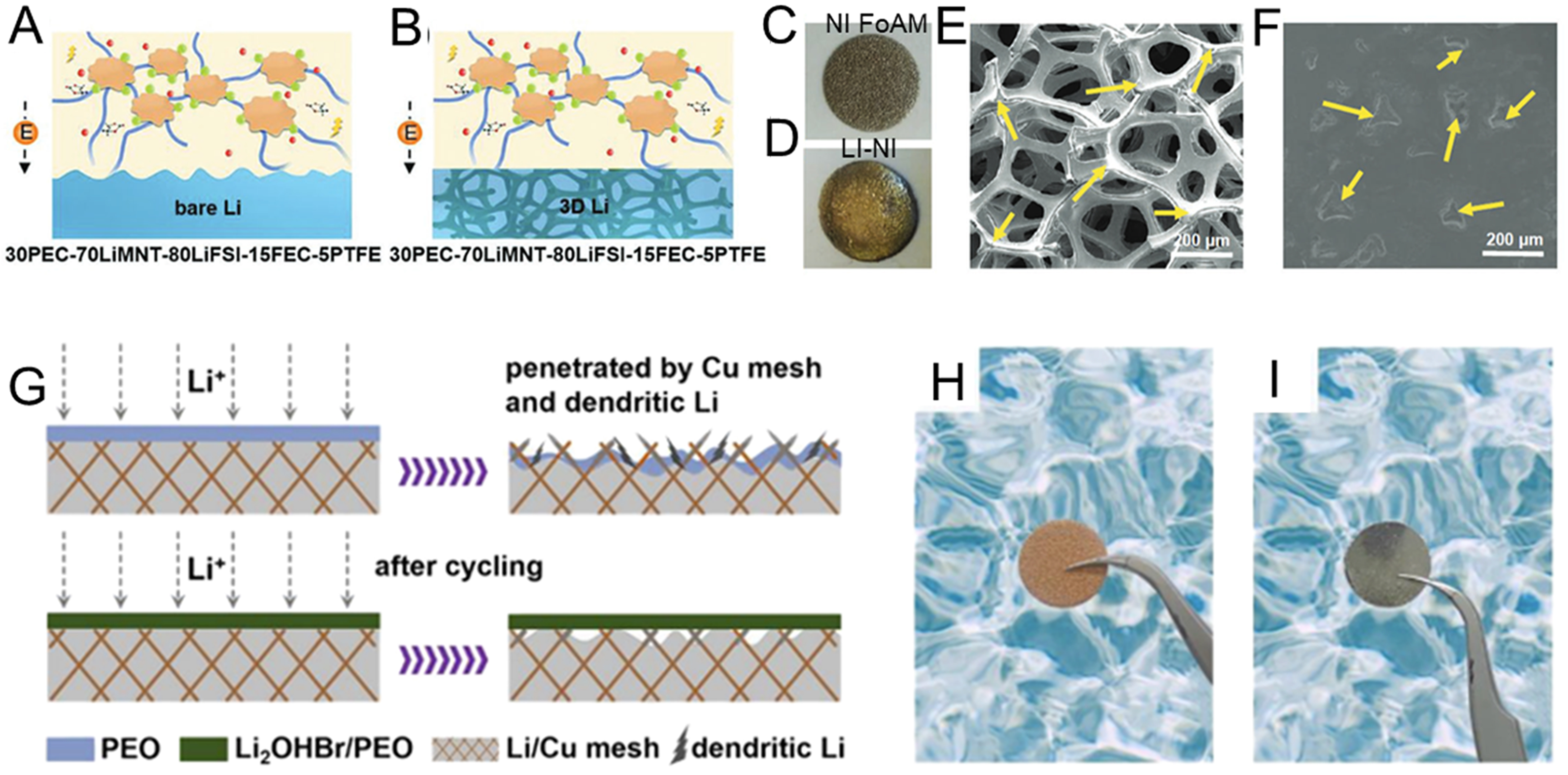
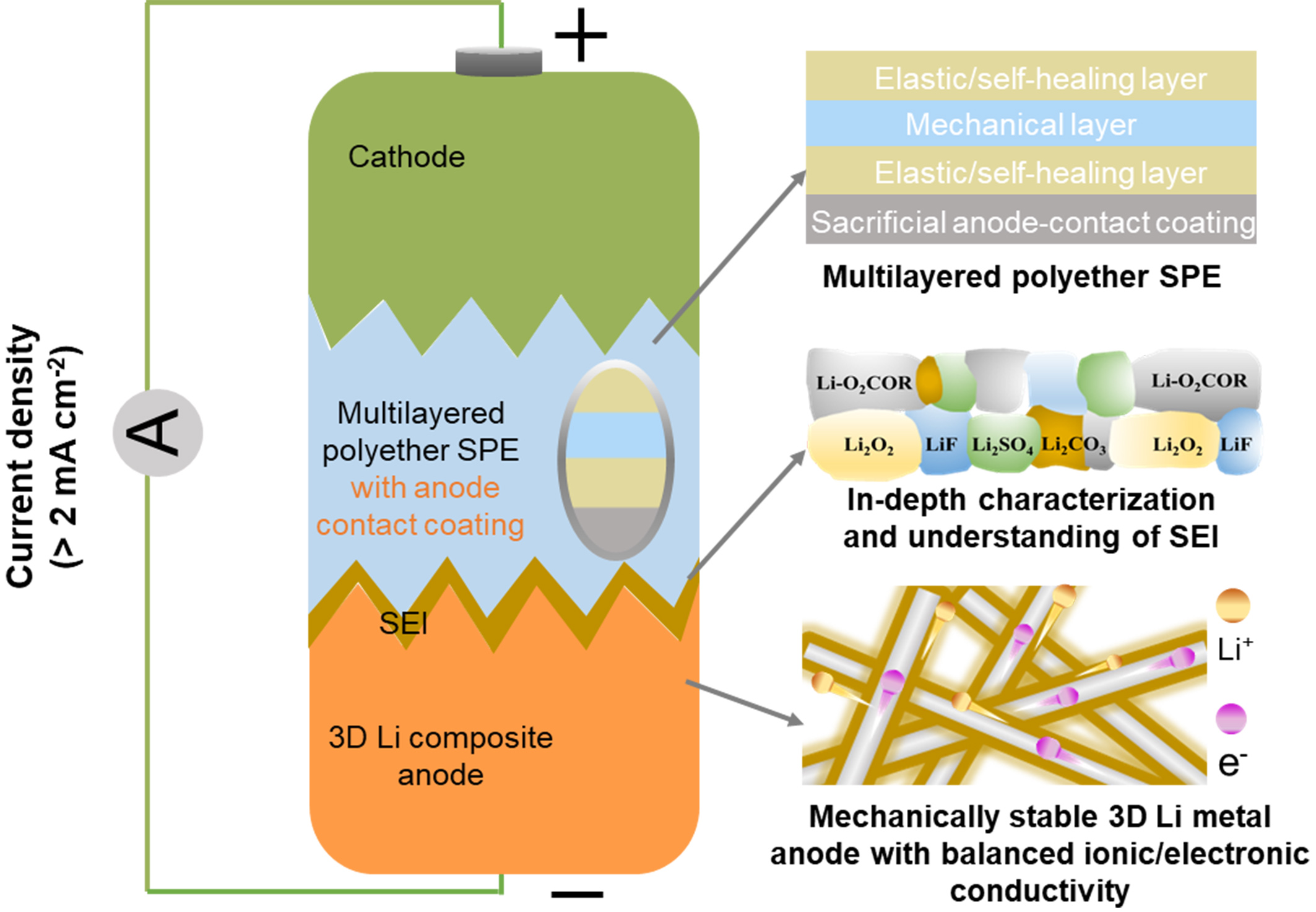








Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.