fig3
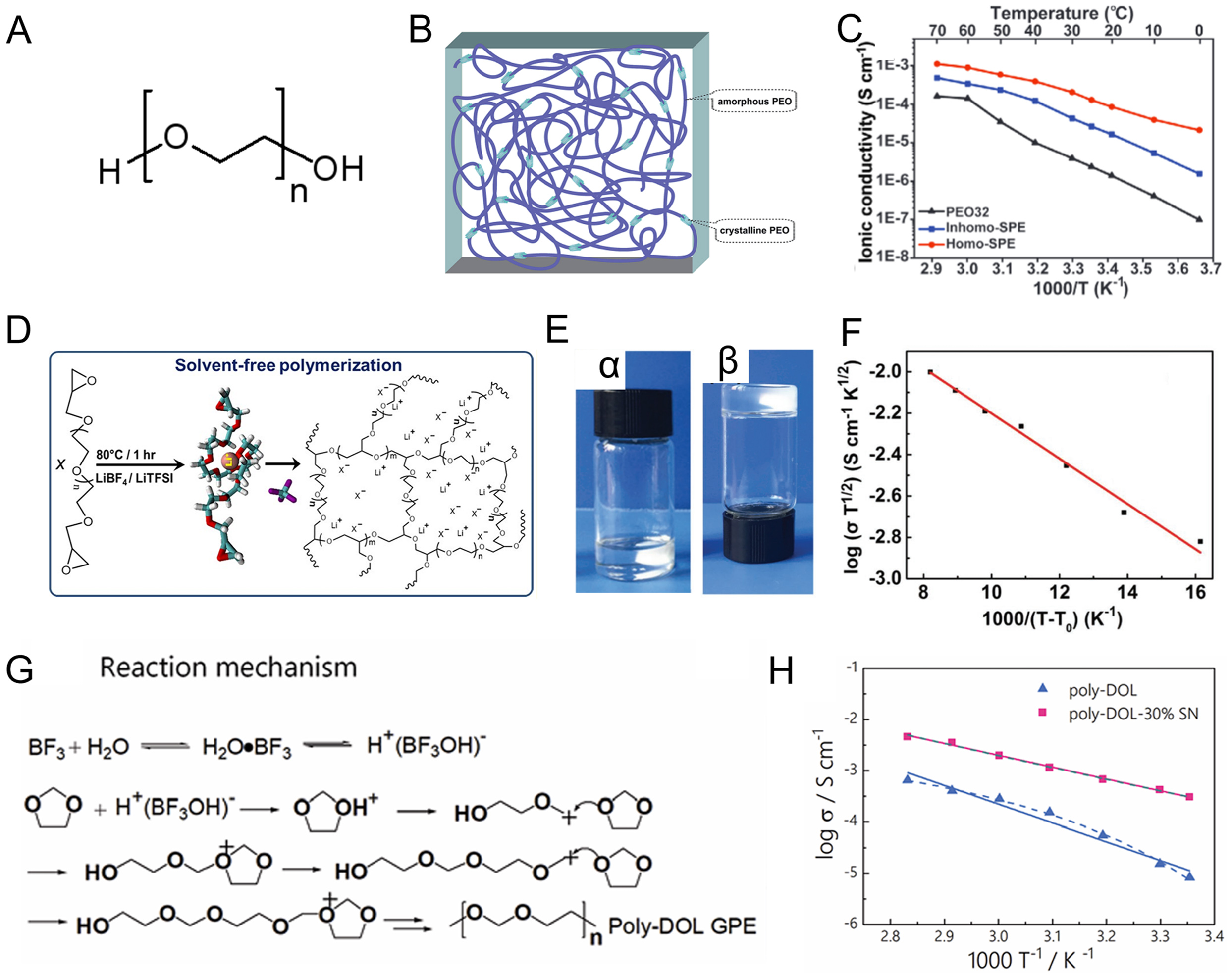
Figure 3. (A) Chemical structure of PEO; (B) Scheme of semi-crystalline PEO; Figure 3B is reprinted with permission from Ref. [34], Copyright © 2015 Royal Society of Chemistry.; (C) Ionic conductivities of PEO electrolyte with various Li salts. Figure 3C is reprinted with permission from Ref. [35], Copyright © 2020 Wiley; (D) Scheme for the synthesis of P(PEGDE). Figure 3D is reprinted with permission from Ref. [36], Copyright © 2019 American Chemical Society; (E) The optical images of (α) PEGDE solution and (β) crosslinked solid electrolyte pure P(PEGDE); (F) The temperature dependence of ionic conductivity for the composite P(PEGDE) polymer electrolytes. Figure 3E and F is reprinted with permission from Ref. [37], Copyright © 2017 Wiley; (G) Reaction mechanism illustrating how LiDFOB initiates polymerization of DOL; (H) Ionic conductivity of the poly-DOL electrolyte versus temperature. Figure 3G and H is reprinted with permission from Ref. [38], Copyright © 2020 Royal Society of Chemistry. PEO: Polyethylene oxide; P(PEGDE): poly[poly(ethylene glycol) diglycidyl ether]; PEGDE: poly(ethylene glycol) diglycidyl ether; LiDFOB: lithium difluoro(oxalato)borate; DOL: 1,3-dioxolane.