Liquid-like ionic conductivity in the solid state
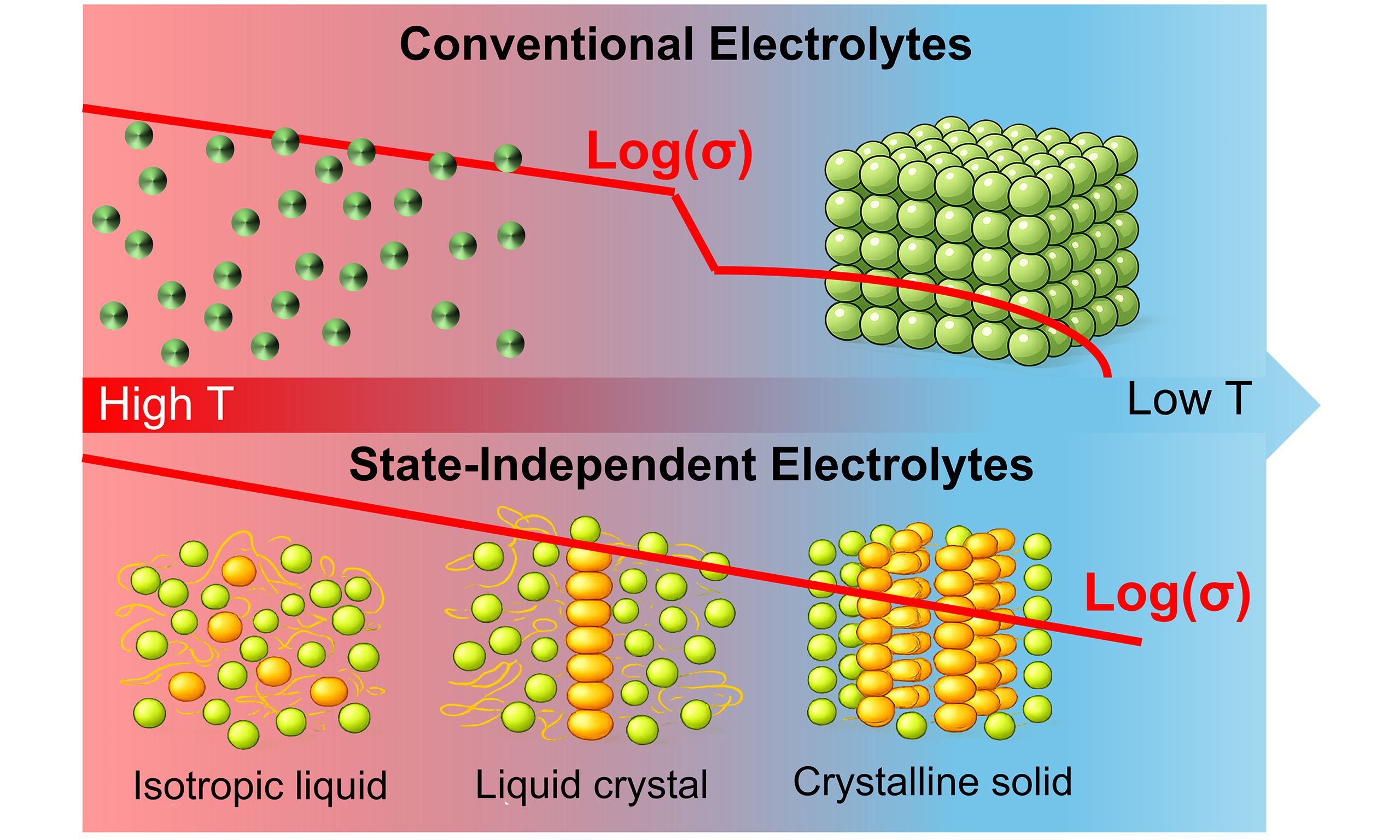
In conventional electrolyte systems, ionic conductivity is strongly state-dependent. When a liquid electrolyte transitions into an ordered solid, ion migration typically proceeds through defect-mediated hopping within relatively static crystalline frameworks, where transport is strongly coupled to lattice energetics and structural rigidity[1]. The enhanced structural rigidity restricts ion motion, sharply increasing diffusion barriers and causing conductivity to collapse [Figure 1A][2]. This fundamental limitation has long forced battery designers to choose between high-conductivity liquids and safer, structurally robust solids[3]. Writing in Science, Barclay et al. challenge this paradigm by demonstrating organic salts that retain the same ionic conduction mechanism across three distinct states of matter. Remarkably, these “state-independent electrolytes” (SIEs) sustain Arrhenius-type ion transport from the isotropic liquid to the liquid-crystal mesophase and finally to the crystalline solid, effectively preserving liquid-like conduction even after solidification upon cooling[4].
Figure 1. (A) The typical temperature dependence of ionic conductivity (σ) in a conventional electrolyte, contrasted with that of a SIE; (B) Schematic illustration of the solid-state superstructure and properties of SIEs; (C) Structural formula of the SIEn·X series of SIEs. This figure is adapted with permission from Ref.[4]. SIE: State-independent electrolyte.
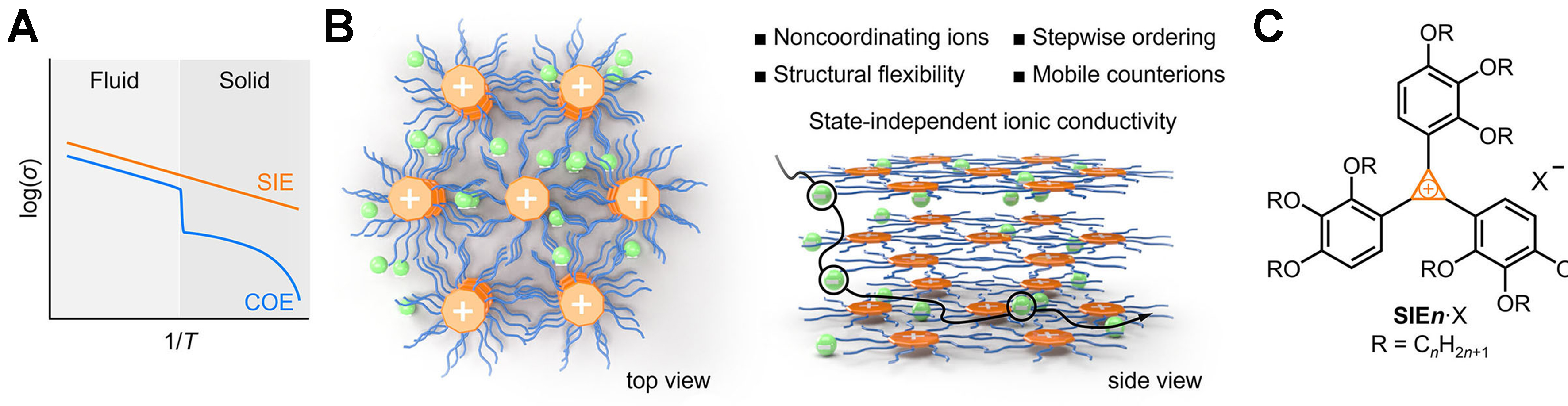
The authors propose three rational design criteria for achieving state-independent ionic conductivity [Figure 1B]. First, cations and anions should interact weakly, thereby minimizing ion pairing. Second, the solid framework should provide numerous energetically equivalent sites to enable continuous ion hopping. Third, structural flexibility should be retained in the solid state to prevent rigid trapping. These principles are elegantly realized using tris(trialkoxyphenyl) cyclopropenium cations (SIEn+, as shown in Figure 1C), which feature a highly delocalized positive charge and threefold rotational symmetry, surrounded by flexible long alkyl chains. Figure 2A shows electrostatic surface potential (ESP) simulations demonstrating that SIEn+ distributes positive charge diffusely, dramatically reducing Coulombic attraction to counterions compared to classical ionic-liquid cations. Molecular modeling further reveals that when SIEn+ disks assemble into columnar stacks, their aromatic cores become sterically shielded [Figure 2B], forcing halide anions to reside in a peripheral “halo” region. This architecture inherently weakens cation-anion binding and preserves anion mobility.
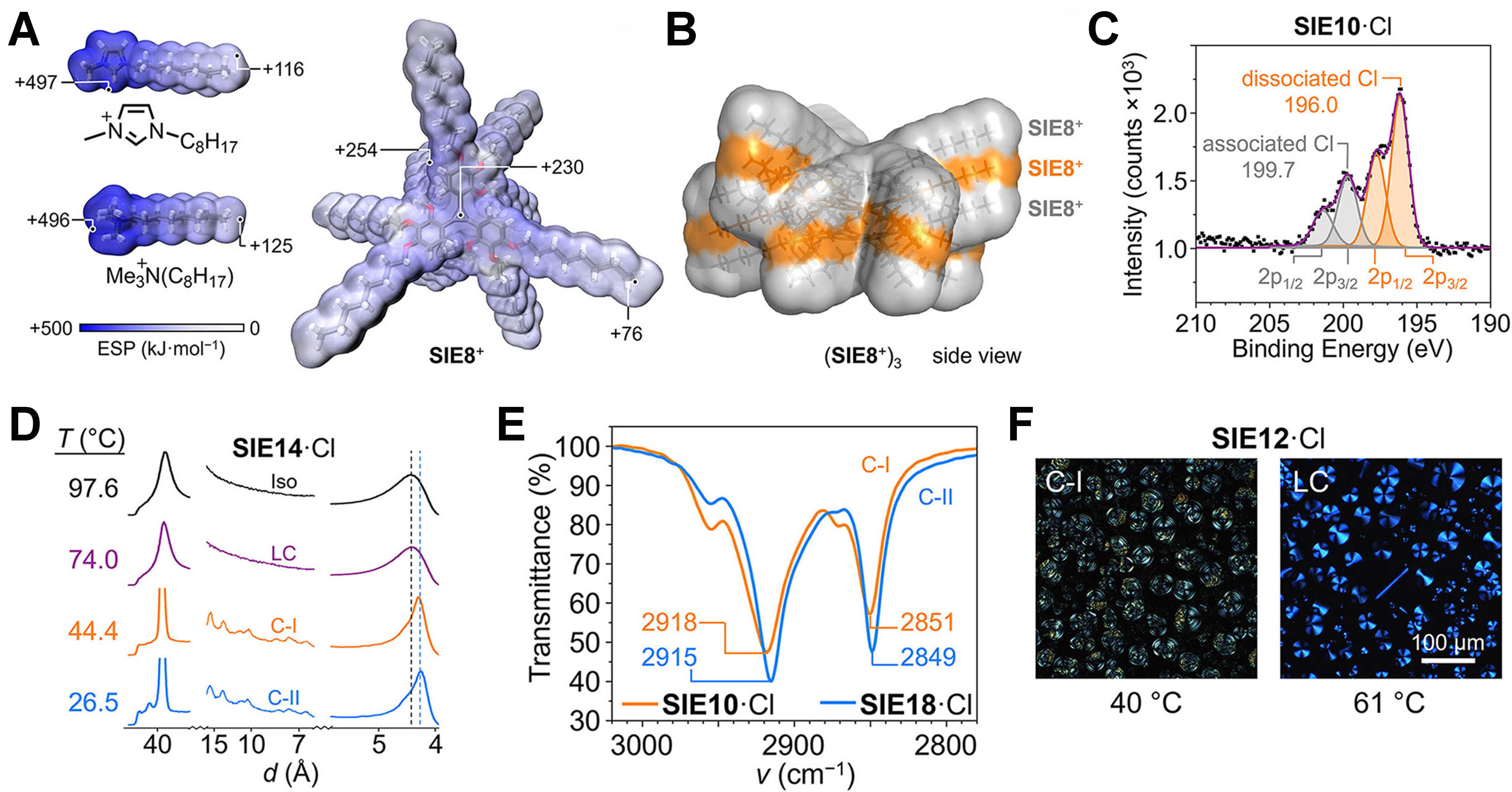
Figure 2. (A) Comparison of calculated ESPs at the [B3LYP/6-31G(d,p)] level for SIE8+ and common cationic imidazolium and ammonium motifs used in ionic liquids and ionic liquid crystals; (B) Surface accessible to a 1.81-Å-radius sphere (chloride radius) around a (SIE8+)3 trimer, calculated at the B3LYP/D3(BJ)/3-21G level; (C) Cl 2p region of the XPS spectrum of SIE10·Cl; (D) Variable-temperature X-ray scattering patterns of SIE14·Cl; (E) Ambient FTIR spectra of SIE10·Cl (C-I) and SIE18·Cl (C-II) in the CH2 stretching region; (F) POM images of crystalline SIE12·Cl (C-I). This figure is adapted with permission from Ref.[4]. ESPs: Electrostatic surface potentials; SIE: state-independent electrolyte; XPS: X-ray photoelectron spectroscopy; FTIR: Fourier-transform infrared; POM: polarized optical microscopy.
A critical question is whether structural transitions disrupt conduction. This study provides compelling evidence that, although these salts undergo multiple ordered phase transitions upon cooling, they retain partial structural flexibility even deep within the crystalline state. X-ray photoelectron spectroscopy (XPS) [Figure 2C] reveals chloride environments dominated by unusually low binding energies, characteristic of uncoordinated or weakly bound halides, thereby confirming minimal ion-pairing strength. Differential scanning calorimetry and X-ray scattering [Figure 2D] further show that ordering progresses stepwise - from isotropic liquid to liquid-crystalline (LC) phase to multiple crystalline polymorphs - while maintaining molecular mobility within the alkyl side-chain regions. Infrared spectroscopy [Figure 2E] confirms this observation, with CH2 stretching vibrations indicating that even the crystalline C-I phase preserves notable chain flexibility compared to the more rigid C-II structures. Polarized optical microscopy (POM) images [Figure 2F] reveal hexagonal columnar liquid-crystalline textures, demonstrating ordered yet compliant mesophases. Together, these results prove that the SIE framework introduces order gradually, thereby avoiding the abrupt “freezing” that typically disrupts ion transport pathways.
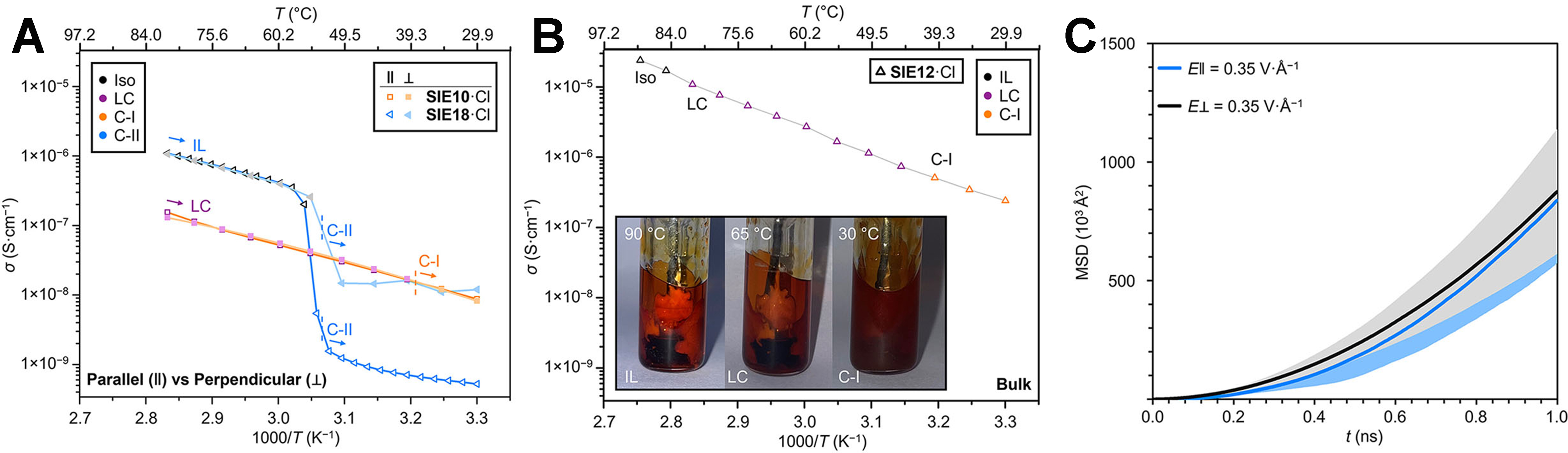
Figure 3A shows that the ionic conductivity of SIEn+ salts with shorter alkyl chains follows an uninterrupted Arrhenius relationship across phase transitions, indicating that the same transport mechanism persists regardless of structural state. In contrast, for longer-chain salts that crystallize directly into more rigid C-II phases, an expected drop in conductivity is observed - clarifying that structural flexibility is indeed critical. For salt forming a C-I solid phase, conductivity does not collapse upon cooling; instead, it decreases smoothly and predictably with temperature, just as in a liquid. This sharply contrasts with traditional organic electrolytes, which exhibit dramatic decreases in conductivity upon solidification. Bulk electrochemical impedance analysis [Figure 3B] confirms that this behavior persists at macroscopic scales, with a conductivity comparable to that of certain glassy inorganic solid electrolytes at room temperature. Molecular dynamics (MD) simulations [Figure 3C] provide mechanistic insight: chloride ions hop efficiently between equivalent peripheral environments without directional bias, exhibiting comparable Mean squared displacements (MSDs) parallel and perpendicular to the columnar axis. This finding confirms that anion transport remains isotropic even within the anisotropically ordered framework.
Figure 3. (A) Comparison of ionic conductivities measured parallel and perpendicular to the alignment of the columnar phases; (B) Direct current conductivity measured using ~0.5 g SIE12·Cl in a bulk electrochemical cell; (C) MSD of Cl- anions obtained from a MD simulation of the crystalline phase of SIE10·Cl. This figure is adapted with permission from Ref.[4]. MSD: Mean squared displacement; SIE: state-independent electrolyte; MD: molecular dynamics; IL: ionic liquid; LC: liquid-crystalline.
Traditionally, liquids provide conductivity through molecular disorder, whereas solids require specific structural motifs (e.g., fast ion conductors)[5]. This work reveals a third pathway in which careful molecular design decouples ionic mobility from the thermodynamic state. The key mechanism is that halide ions are neither tightly bound nor confined within lattice wells; instead, they migrate through conformationally flexible alkyl environments that remain dynamic even in crystalline phases. Importantly, the conduction mechanism is conserved across isotropic, liquid-crystalline, and C-I solid states. Arrhenius slopes remain constant, indicating that the activation energy does not change despite increasing structural order. This finding fundamentally challenges the assumption that freezing necessarily changes conduction physics.
This discovery holds profound technological implications. Materials that conduct ions effectively in both liquid and solid states enable electrolytes that can be processed as fluids but operate as safe as in solid systems. Such properties are attractive for flexible energy storage, solid-state batteries, soft electronics, ionic plastic crystals, and anion-exchange technologies[6]. Moreover, this framework offers a universal design blueprint: employing diffuse ionic charge, weak ion coordination, and stepwise structural ordering to engineer soft-solid electrolytes with fluid-like transport. By proving that ionic conductivity can be state-independent, Barclay et al. establish a new paradigm in materials physics. Their work demonstrates that ion motion is not inherently dictated by whether a material is liquid or solid, opening both conceptual and technological avenues for next-generation solid electrolytes and multifunctional ionic materials[4].
Although the work by Barclay et al. represents a notable breakthrough, the behavior observed thus far also reflects a delicate balance between molecular flexibility and structural order[4]. Continuous Arrhenius-type transport is most evident in systems that adopt the more compliant C-I phase, whereas salts that crystallize directly into the more rigid C-II form exhibit reduced mobility. Within the reported SIEn-X series, a clear structural dependence emerges: materials bearing longer alkyl chains (n ≳ 16) preferentially form the more densely packed C-II phase, while shorter-chain analogs (n = 8-14) more readily stabilize the C-I phase that preserves isotropic ion transport. This trend suggests that structural features promoting stronger van der Waals cohesion and side-chain interdigitation - such as increased chain length or more efficient packing symmetry - tend to bias the system toward the less mobile C-II regime. The sensitivity to polymorph selection suggests that the extent of side-chain motion remains a key factor governing transport in the solid state. At the same time, long alkyl substituents that facilitate weak ion coordination may influence thermal stability, electrochemical stability, and mechanical integrity[7,8], highlighting the multidimensional trade-offs inherent to soft ionic materials.
From a synthetic chemistry perspective, the most important advance is that state-independent ionic conductivity emerges as a molecularly programmable property rather than a fixed material constraint. The SIEn+ framework provides multiple chemically accessible design parameters, including alkyl chain length and branching, the electronic structure of the aromatic core, and counter-ion identity, all of which can be systematically varied to balance conformational flexibility, packing order, and ion–framework interactions. Such synthetic tunability opens a pathway toward a broader class of soft solid electrolytes whose transport properties can be rationally engineered at the molecular level. More broadly, this work suggests that future electrolyte design may benefit from targeting dynamically disordered, weakly coordinating ion environments rather than relying exclusively on rigid crystalline conduction pathways, thereby expanding the design space for next-generation solid and quasi-solid ionic conductors.
DECLARATIONS
Authors’ contributions
Conceived the Highlight: Su, B. L.; Qu, H.
Drafted the manuscript: Qu, H.
Supervised the work and revised the manuscript: Su, B. L.
Both authors read and approved the final manuscript.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
The authors acknowledge the Wallonia Government for financial support under the “Plan de Relance” (2310153-BatFactory).
Conflicts of interest
Su, B. L. is the Editor-in-Chief of the journal Chemical Synthesis. Su, B. L was not involved in any steps of the editorial process, notably including reviewer selection, manuscript handling, or decision making. Qu, H. declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Zhu, X.; Wu, J.; Liu, J. Insight into inorganic solid-state electrolytes: ionic transport and failure mechanisms. Adv. Funct. Mater. 2024, 34, 2409547.
2. Sotoudeh, M.; Baumgart, S.; Dillenz, M.; et al. Ion mobility in crystalline battery materials. Adv. Energy. Mater. 2024, 14, 2302550.
3. Li, H.; Li, M.; Weng, S.; et al. Autonomous ion-highways quasi-solid electrolytes toward high-voltage lithium metal batteries. Natl. Sci. Rev. 2025, 12, nwaf363.
4. Barclay, J.; Williamson, J. M.; Litt, H.; et al. State-independent ionic conductivity. Science 2025, 390, 1254-8.
5. He, X.; Zhu, Y.; Mo, Y. Origin of fast ion diffusion in super-ionic conductors. Nat. Commun. 2017, 8, 15893.
6. Wang, H.; Yan, W.; Zhu, M. Revolutionizing fiber batteries with polymer gel electrolyte: a groundbreaking innovation in wearable energy. Chem. Synth. 2024, 4, 57.
7. Ruan, Q.; Yao, M.; Yuan, D.; et al. Ionic liquid crystal electrolytes: fundamental, applications and prospects. Nano. Energy. 2023, 106, 108087.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.