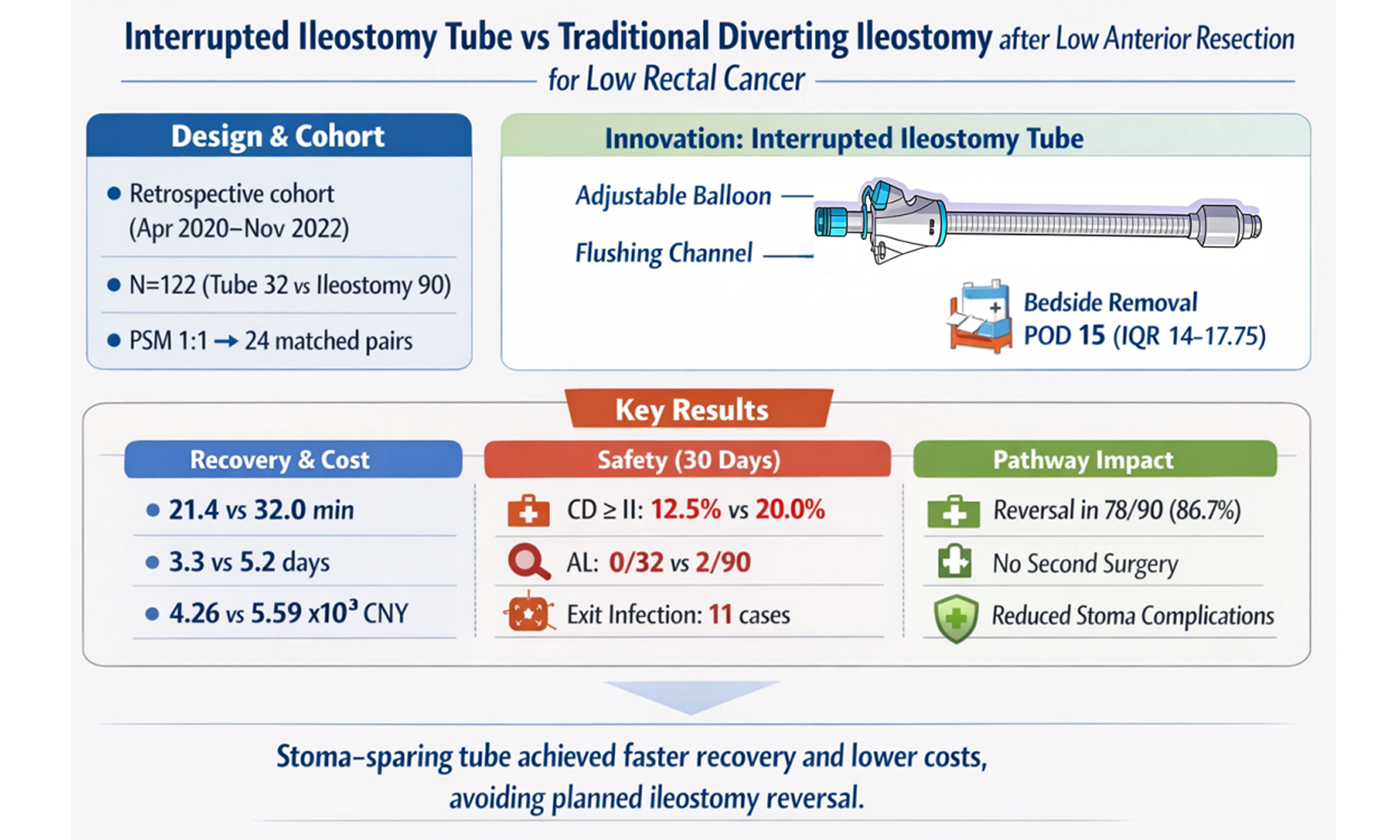
Interrupted Ileostomy Tube vs. traditional diverting ileostomy in low rectal cancer: a propensity score–matched retrospective study
Abstract
Aim: Temporary diverting ileostomy protects low colorectal/rectal anastomoses but adds stoma morbidity and a planned reversal. We evaluated an Interrupted Ileostomy Tube as a stoma-sparing diversion strategy.
Methods: We retrospectively analyzed 122 patients undergoing radical resection for low rectal cancer with prophylactic diversion (April 2020 - November 2022): traditional diverting ileostomy (n = 90) vs. Interrupted Ileostomy Tube (n = 32). Outcomes were compared before and after 1:1 propensity-score matching.
Results: The Tube group had shorter diversion-creation time (21.38 ± 4.53 vs. 32.03 ±
Conclusion: In this retrospective analysis, the Interrupted Ileostomy Tube was associated with faster recovery and lower index costs and avoided planned stoma reversal and late stoma morbidity without an apparent increase in early complications.
Keywords
INTRODUCTION
Colorectal cancer is one of the most common malignant tumors worldwide, and the proportion of low rectal/low colorectal cancers has been increasing in recent years[1,2]. Radical surgery, including sphincter-preserving procedures and abdominoperineal resection, remains the cornerstone of curative treatment[3-5]. With advances in minimally invasive techniques, ultra-low sphincter-preserving surgery has become increasingly feasible, allowing many patients to avoid a permanent abdominal stoma. However, low rectal anastomoses carry a substantial risk of anastomotic leakage (AL), which can lead to sepsis, reoperation, prolonged hospitalization, and impaired quality of life[6]. Prevention and optimal management of AL therefore remain key challenges in low rectal cancer surgery[7,8].
Temporary diverting ileostomy is widely used as a prophylactic measure to reduce the clinical consequences of AL[9,10]. Although effective in protecting the anastomosis, a diverting stoma is associated with stoma-related complications, the need for a planned second operation for stoma closure, additional hospitalizations and healthcare costs, and considerable physical and psychological burden for patients. Moreover, there is ongoing controversy regarding which patients truly benefit from prophylactic diversion, and how to balance its protective effect against its inherent morbidity and resource use[11,12]. In an attempt to avoid a formal stoma, various catheter-based diversion techniques (e.g., Foley catheters, gastrostomy tubes, or modified drainage tubes placed in the ileum) have been explored[13]. However, these devices were not specifically designed for this purpose and often suffer from limitations such as easy dislodgement, poor conformity to the intestinal lumen, and unreliable diversion.
The Interrupted Ileostomy Tube is a dedicated catheter-based diversion device designed to achieve temporary fecal diversion while avoiding creation of a conventional abdominal stoma. By enabling bedside tube removal after recovery of bowel function, this approach has the potential to reduce the need for secondary surgery and to mitigate stoma-related complications and burden on patients. However, its safety and efficacy in the setting of low rectal cancer surgery have not been fully characterized. Therefore, in this retrospective study, we compared perioperative outcomes and postoperative complications between the Interrupted Ileostomy Tube and traditional diverting ileostomy in patients undergoing radical surgery for low rectal cancer, with the aim of providing evidence to guide patient selection and optimize diversion strategies in clinical practice.
METHODS
Study population
This was a single-center retrospective cohort study. A total of 122 consecutive patients who underwent radical resection for low rectal cancer with intestinal diversion at the Department of Gastrointestinal Surgery, The First Affiliated Hospital of Wannan Medical College (Yijishan Hospital), Wuhu, China, between April 2020 and November 2022 were included. According to the type of diversion, patients were divided into two groups: (1) Traditional Fistula Group: 90 patients who received a conventional diverting ileostomy; (2) Interrupted Ileostomy Tube Group: 32 patients who received diversion using the Interrupted Ileostomy Tube. Baseline clinical data were collected from the electronic medical record, including sex, age, height, weight, body mass index (BMI), medication history, hypertension, diabetes, and other comorbidities, as well as tumor- and surgery-related characteristics. This retrospective observational study is reported in accordance with the STROBE (Strengthening the Reporting of Observational Studies in Epidemiology) statement, and the completed checklist is provided in the Supplementary Materials.
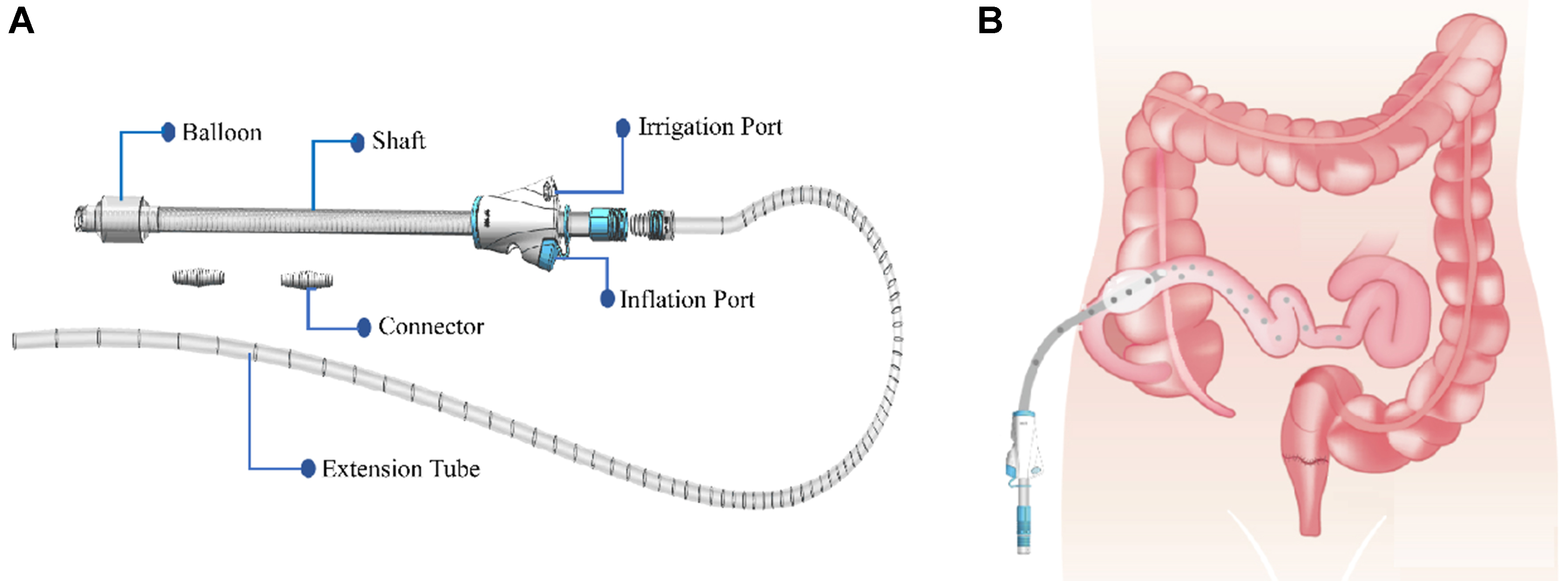
All patients and their families were informed of the overall surgical procedure, potential complications, and the details of the consumables and implanted devices, including the Interrupted Ileostomy Tube (patent No. ZL 201720799741.X; medical device production license No. 20170047; medical device registration certificate No. Anhui Device Registration Approval No. 20212140096; registrant: Anhui Yichuanglian Medical Technology Co., Ltd.; entrusted manufacturer: Anhui Zhongrong Medical Device Co., Ltd.; Figure 1). Written informed consent for surgery and use of anonymized clinical data was obtained from all patients. The study protocol was approved by the Ethics Committee of The First Affiliated Hospital of Wannan Medical College [approval No. (2017) Lunshenxin-02] and was conducted in accordance with the Declaration of Helsinki. For this retrospective analysis, only anonymized data were used. A detailed conflict-of-interest statement is provided in the Declarations.
Figure 1. Schematic diagram of the Interrupted Ileostomy Tube and its functional design. (A) Illustrates the structure of the Interrupted Ileostomy Tube, including the adjustable balloon for modulation of luminal occlusion and patient comfort, and the internal flushing channel to clear intraluminal blockage; (B) Shows the overall concept of temporary ileal diversion using the device. Created in BioRender. Zhao, H. (2026) https://BioRender.com/zaqpnn1.
Inclusion and exclusion criteria
Inclusion criteria
Patients were included if they met all of the following criteria: Low rectal cancer confirmed by colonoscopy and histopathology, with the lower tumor margin < 8 cm from the anal verge; No distant metastasis on preoperative staging [chest and abdominal computed tomography (CT) and pelvic magnetic resonance imaging (MRI), with/without chest radiography per institutional routine]; Elective curative-intent radical resection with primary colorectal/coloanal anastomosis performed according to current guidelines; Prophylactic fecal diversion performed at the index operation (Interrupted Ileostomy Tube or traditional diverting ileostomy) based on institutional criteria and surgeon discretion; Written informed consent and complete clinical data available for analysis.
Exclusion criteria
Patients were excluded if they had: Previous or synchronous malignancy; Distant metastasis or suspected tumors in other organs on preoperative evaluation; Emergency surgery; Severe frailty or major contraindications to surgery; No prophylactic diversion ultimately performed based on intraoperative assessment; Concurrent major procedures beyond the planned rectal resection; Pregnancy or inability to provide informed consent (e.g., cognitive impairment or severe psychiatric illness); Known hypersensitivity to device constituent materials [e.g., polyethylene (PE)/polypropylene (PP)/polyvinyl chloride (PVC)]; Inability to safely complete the planned procedure (e.g., extensive pelvic/abdominal adhesions); or Incomplete key clinical data required for analysis.
Preoperative examination and preparation
All patients underwent standard preoperative evaluation, including complete blood count, serum biochemistry, blood typing, and coagulation profile, as well as electrocardiogram, chest X-ray, and abdominal CT to confirm diagnosis and assess surgical risk.
Mechanical bowel preparation was performed twice using oral magnesium sulfate 100 g dissolved in 1,000 mL of water, administered once in the morning and once in the evening on the day before surgery. Patients fasted from solid food for 24 h and from clear liquids for 8 h before surgery. Prophylactic intravenous antibiotics were administered 2 h before skin incision or at induction of anesthesia, according to institutional protocol[14].
Surgical procedures
Radical surgery for low rectal/low rectal cancer (open or minimally invasive) was performed according to standard oncologic principles. After resection of the diseased bowel segment and completion of the rectal or coloanal anastomosis, the diversion strategy was non-randomized in this retrospective cohort and was selected through shared decision-making. The final decision was made by the attending surgical team after standardized preoperative counseling regarding the expected benefits and burdens of each diversion approach and was confirmed intraoperatively. In general, the choice of diversion took into account the preoperative risk profile and intraoperative assessment of anastomotic risk (e.g., anastomotic height and operative difficulty), as well as patient preference, as documented in the informed consent and operative records.
Traditional diverting ileostomy
For the Traditional Fistula Group, a conventional loop ileostomy was created through a curved lower abdominal incision or a right lower rectus abdominis incision. A vertically oriented, spindle-shaped skin incision was made, with a diameter sufficient to accommodate approximately three transverse fingers. Approximately 4 cm of the proximal ileum was delivered through the abdominal wall. The seromuscular layer of the ileum was then sutured in an interrupted fashion to the peritoneum, fascial layer, and skin to prevent retraction of the stoma and prolapse of adjacent bowel. The stoma was temporarily covered and fully opened at an appropriate time within 48 h postoperatively to reduce contamination of the surrounding skin and incision[15,16].
Interrupted Ileostomy Tube placement
In the Interrupted Ileostomy Tube Group, after completion of the anastomosis, the small intestine was inspected to exclude adhesions or deformities that might interfere with tube placement. All “laparoscopic” cases in this study were laparoscopic-assisted radical resections, and the tube was placed through the original right lower-quadrant auxiliary incision using the same steps as in open surgery; thus, the main difference between approaches was incision size (larger in open surgery vs. smaller auxiliary incision in laparoscopic-assisted surgery). A small abdominal wall opening was then created at McBurney’s point in the right lower quadrant, or at an equivalent site chosen according to the cecal position to avoid acute angulation of the ileum. The incision technique was similar to that used for laparoscopic trocar placement. The diameter of the abdominal wall tract was kept slightly smaller than that of the tube (approximately 1 cm), to ensure a snug fit and reduce leakage of intestinal contents.
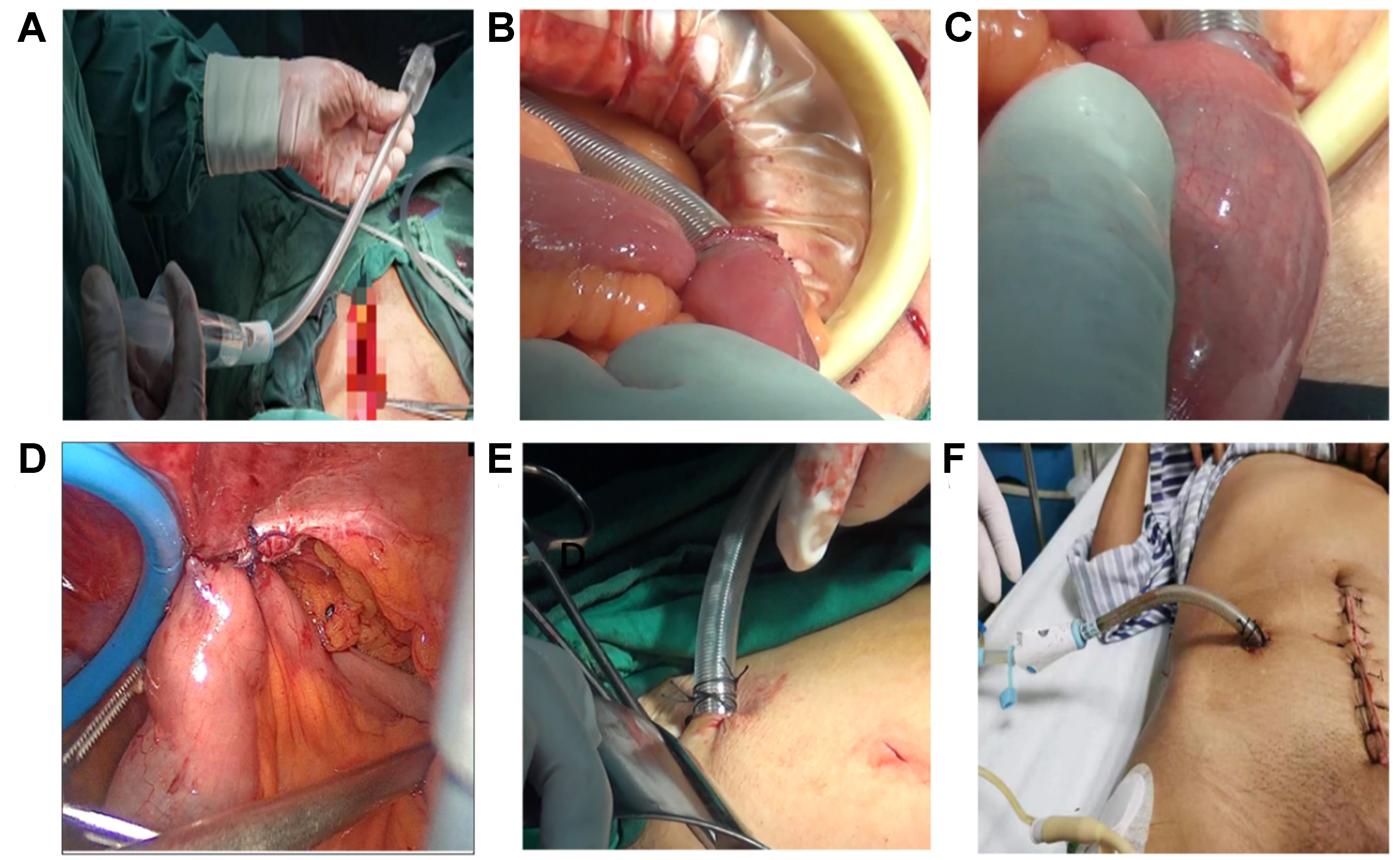
Before insertion, the tube was inspected to confirm its integrity, patency of the flushing channel, and proper function of the balloon. The balloon was inflated with saline and deflated to ensure it could be filled and emptied smoothly [Figure 2A]. A 10-15 cm segment of terminal ileum was then selected, and a 1-cm longitudinal enterotomy was made on the antimesenteric border. A purse-string suture was placed around the enterotomy [Figure 2B]. The Interrupted Ileostomy Tube was advanced retrogradely through the enterotomy into the ileal lumen until the balloon passed beyond the purse-string. The purse-string was tightened and tied to secure the tube and minimize leakage of intestinal contents around the catheter. A second reinforcing purse-string suture was placed between the first and second rings to further reduce the risk of leakage.
Figure 2. Intraoperative steps for Interrupted Ileostomy Tube placement. (A) Balloon inflation test; (B) Creation of purse-string and enterotomy with tube insertion; (C) Balloon inflation until the bowel wall turns pale pink; (D) Fixation of ileum to the abdominal wall; (E) External fixation of the tube and closure of the skin; (F) Final appearance of the tube connected to a drainage system.
The balloon was then inflated gradually with approximately 15 mL of saline until the intestinal wall around the balloon appeared gently blanched, indicating adequate apposition without excessive tension [Figure 2C]. Patency of the lumen and functional occlusion were confirmed intraoperatively by observing effluent through the tube and performing gentle flushing when needed. The ileum was anchored to the peritoneum and inner abdominal wall with 3-4 interrupted sutures to prevent torsion and kinking and to reduce the risk of catheter migration/displacement [Figure 2D]. The external portion of the tube was secured to the skin, and the abdominal wall incision was reinforced with additional sutures as needed [Figure 2E]. The distal end of the tube was connected to a drainage bag or disposable negative-pressure drainage device to collect effluent [Figure 2F].
Postoperative management
Postoperative management followed a standardized institutional postoperative pathway incorporating selected enhanced-recovery elements. Oral intake was advanced in a stepwise manner in this cohort of low rectal cancer patients undergoing prophylactic diversion, reflecting local practice and tolerance considerations. All patients were initially kept fasting, and intestinal function was monitored by assessing bowel sounds, flatus, and clinical tolerance (nausea/abdominal distension). Once bowel function had resumed, patients were gradually advanced from water intake to a liquid and then semi-solid diet, according to tolerance. Wounds and diversion sites (stoma or tube exit site) were inspected regularly. Dressings were changed as needed, and drainage systems were maintained or replaced according to the volume and character of effluent.
Patients in the Traditional Fistula Group were discharged once they were afebrile, tolerated oral intake, and had stable stoma function. They were routinely re-evaluated 3-6 months after the index surgery to determine timing of elective ileostomy reversal based on oncologic status and overall recovery[17].
In the Interrupted Ileostomy Tube Group, the tube was typically removed at approximately two weeks postoperatively, either during the index hospitalization or after discharge at scheduled outpatient follow-up, depending on clinical recovery and local care pathways. Before removal, the following criteria had to be met: recovery of bowel function, tolerance of a regular oral diet, absence of clinical signs suggestive of AL or intra-abdominal infection (e.g., persistent fever, pelvic pain, rising inflammatory markers, suspicious pelvic drainage), and stable/decreasing tube output with unobstructed drainage into the collection device (including the presence of loose effluent via the tube/drainage bag when applicable). The previously stated criterion “passage of loose stool per anus” was a wording error and has been corrected to reflect tube/drainage-bag effluent rather than anal stool passage.
Balloon-occlusion management was primarily guided by bedside clinical assessment and tube-output monitoring. The balloon volume was documented after placement. If clinically indicated (e.g., abdominal distension or intolerance), occlusion could be titrated by stepwise deflation in small increments with interval reassessment, while continuing to monitor symptoms and tube output. Imaging and/or endoscopic evaluation was performed when clinically indicated (e.g., suspicion of leakage/infection), rather than mandated routinely prior to tube removal.
For removal, the balloon was completely deflated, and the tube was gently rotated and withdrawn at the bedside. The puncture site was covered with Vaseline gauze and a sterile dressing. If local healing was delayed or mild exudation persisted, dressings were changed every 2-3 days; in most cases, the tract closed within about 1 week. For tube-/puncture-site events, “puncture-site infection” was defined as localized erythema and/or minor exudation with mild tenderness at the exit/tract site without systemic signs. These diversion-site events were graded using the Clavien-Dindo classification and reported separately from the composite 30-day morbidity endpoint (Clavien-Dindo ≥ II). All patients were followed up by monthly telephone contact to document readmissions, secondary surgeries, delayed complications (including stoma- or tube-related events), and overall recovery.
Study endpoints
The primary endpoint was clinically relevant AL within 30 days after surgery, defined as International Study Group of Rectal Cancer (ISREC) grade B or C leakage[18]. Key secondary endpoints included (1) 30-day postoperative morbidity (Clavien-Dindo grade ≥ II)[19]; (2) perioperative recovery metrics [time to first oral intake and length of stay (LOS)]; (3) direct in-hospital costs; and (4) diversion-pathway outcomes, including stoma reversal (rate and time to reversal) in the Traditional group and time to tube removal in the Tube group. Diversion-site complications were reported separately according to the procedure-specific profile (stoma-related complications vs. tube/puncture-site–related events) and were not combined into a single composite endpoint. For descriptive purposes, “late” diversion-related events were defined as events occurring > 30 days after surgery. Cost analysis was restricted to direct in-hospital costs during the index admission; costs related to stoma reversal hospitalization and management of stoma-associated complications were not captured in this study.
Assessment of AL. Postoperative surveillance included routine clinical assessment (vital signs, abdominal/pelvic symptoms, and drain output) and laboratory markers of inflammation as clinically indicated. Suspected AL (e.g., fever, pelvic/abdominal pain, peritonitis, abnormal/purulent or feculent drainage, ileus, or unexplained inflammatory response) was evaluated with abdominopelvic CT with intravenous contrast (with rectal water-soluble contrast when appropriate) and/or endoscopic assessment (flexible sigmoidoscopy/rectoscopy) at the surgeon’s discretion. Routine imaging prior to ileostomy tube removal was not mandatory; imaging/endoscopy was performed when clinically indicated to exclude AL. Tube removal was undertaken only when patients were clinically stable without signs suggestive of AL.
Follow-up and outcome ascertainment
Patients were followed through monthly telephone contact and review of outpatient and inpatient medical records. Follow-up time was calculated from the date of the index surgery to the date of the last successful contact (telephone follow-up or clinic visit) or the last verifiable medical record entry, whichever occurred later. Follow-up completeness was assessed as the proportion of patients with at least one documented post-discharge follow-up, and loss to follow-up was defined as failure to contact a patient for ≥ 3 consecutive scheduled calls with no subsequent medical record information available. Diversion-related events and late complications were first captured through structured telephone interviews and were cross-validated against the electronic medical record whenever applicable. Outcomes were ascertained up to the date of the last successful contact or the last verifiable medical record entry.
Statistical methods
All statistical analyses were performed using R software (version 4.5.1; R Foundation for Statistical Computing, Vienna, Austria). Continuous variables were summarized as mean ± standard deviation (SD) when approximately normally distributed or as median (interquartile range, IQR) otherwise; categorical variables were summarized as counts and percentages. Distributional assumptions were assessed using histograms, Q–Q plots, and the Shapiro–Wilk test.
Comparisons between the Interrupted Ileostomy Tube Group and the Traditional Fistula Group were conducted using Student’s t test for normally distributed continuous variables and the Wilcoxon rank-sum test for non-normally distributed variables. Categorical variables were compared using the chi-square test or Fisher’s exact test, as appropriate. All tests were two-sided, and P values < 0.05 were considered statistically significant. Descriptive statistics, hypothesis testing, and formatted summary tables and plots were generated using base R together with the tableone, gtsummary, and ggplot2 packages.
To reduce potential selection bias, propensity score matching (PSM) was performed using the MatchIt and cobalt packages. The propensity score for each patient - defined as the probability of receiving the Interrupted Ileostomy Tube - was estimated using a multivariable logistic regression model that included the following baseline covariates: sex, age, BMI, Eastern Cooperative Oncology Group (ECOG) performance status, distance from the tumor to the anal verge (> 5 cm vs. ≤ 5 cm), preoperative carcinoembryonic antigen (CEA), cancer antigen (CA) 19-9 and CA125, hemoglobin, albumin, American Society of Anesthesiologists (ASA) grade, operative approach (open vs. minimally invasive), histological grade, tumor–node–metastasis (TNM) stage, and comorbidities (cardiovascular disease, hypertension, diabetes, and other comorbidities). A 1:1 nearest-neighbor matching algorithm without replacement was applied, with a caliper width of 0.2 of the SD of the logit of the propensity score. Covariate balance before and after matching was evaluated using absolute standardized mean differences (SMDs) and Love plots, with an |SMD| < 0.10 interpreted as good balance and |SMD| ≤ 0.20 as acceptable balance.
The primary endpoint (30-day clinically relevant AL, ISREC grade B/C) was compared between groups using Fisher’s exact test (or χ2 test, as appropriate), and effect sizes were reported as odds ratios (ORs) with 95% confidence intervals (CIs) from logistic regression when estimable. Secondary endpoints were analyzed using appropriate parametric or nonparametric tests as described above. Matched-cohort analyses were conducted as sensitivity analyses to assess robustness, and secondary outcomes were interpreted cautiously without formal adjustment for multiple comparisons.
RESULTS
Baseline clinicopathologic characteristics before and after PSM
A total of 122 patients who underwent radical resection of low rectal cancer were included, of whom 90 received traditional prophylactic ileostomy (Traditional Fistula Group) and 32 received an Interrupted Ileostomy Tube (Interrupted Ileostomy Tube Group). In the overall cohort, most baseline clinicopathologic characteristics were broadly comparable between the two groups, including sex, age, BMI, ECOG performance status, distance from the tumor to the anal verge, hemoglobin and albumin levels, ASA grade, operative approach, histological grade, TNM stage, and major comorbidities (cardiovascular disease, hypertension, diabetes, and other conditions) [Table 1]. In contrast, tumor markers showed a modest imbalance: patients in the Interrupted Ileostomy Tube Group had slightly lower CEA, CA19-9, and CA125 levels than those in the Traditional Fistula Group (all P < 0.05; Table 1), indicating modest baseline imbalance in tumor marker levels. However, given the non-randomized allocation of diversion strategy, PSM was performed to further reduce measured confounding and improve comparability between groups.
Baseline clinicopathologic characteristics before and after PSM
| Before matching | After nearest-neighbor matching | |||||||
| Baseline and perioperative characteristics1 | Overall N = 122 | Traditional Fistula Group N = 90 | Interrupted Ileostomy Tube Group N = 32 | P-value2 | Overall N = 48 | Traditional Fistula Group N = 24 | Interrupted Ileostomy Tube Group N = 24 | P-value2 |
| Sex | 0.442 | 0.525 | ||||||
| Female | 45 (37%) | 35 (39%) | 10 (31%) | 14 (29%) | 8 (33%) | 6 (25%) | ||
| Male | 77 (63%) | 55 (61%) | 22 (69%) | 34 (71%) | 16 (67%) | 18 (75%) | ||
| Age (years) | 67.19 ± 5.99 | 67.79 ± 4.87 | 65.50 ± 8.26 | 0.112 | 65.02 ± 6.84 | 65.71 ± 4.65 | 64.33 ± 8.54 | 0.438 |
| BMI (kg/m2) | 23.51 ± 2.56 | 23.42 ± 2.82 | 23.76 ± 1.64 | 0.481 | 23.85 ± 2.46 | 23.87 ± 3.09 | 23.84 ± 1.68 | 0.588 |
| ECOG performance status | 0.790 | 0.494 | ||||||
| ECOG 0 | 89 (73%) | 67 (74%) | 22 (69%) | 36 (75%) | 17 (71%) | 19 (79%) | ||
| ECOG 1 | 28 (23%) | 19 (21%) | 9 (28%) | 11 (23%) | 7 (29%) | 4 (17%) | ||
| ECOG 2 | 5 (4.1%) | 4 (4.4%) | 1 (3.1%) | 1 (2.1%) | 0 (0%) | 1 (4.2%) | ||
| Distance from tumor to anal verge | 0.742 | 0.763 | ||||||
| < 5 cm | 41 (34%) | 31 (34%) | 10 (31%) | 17 (35%) | 9 (38%) | 8 (33%) | ||
| ≥ 5 cm | 81 (66%) | 59 (66%) | 22 (69%) | 31 (65%) | 15 (63%) | 16 (67%) | ||
| CEA (ng/mL) | 27.70 ± 4.51 | 28.26 ± 3.70 | 26.11 ± 6.05 | 0.032 | 26.98 ± 4.77 | 26.89 ± 4.25 | 27.07 ± 5.33 | 0.943 |
| CA19-9 (U/mL) | 57.17 ± 6.48 | 57.96 ± 5.54 | 54.96 ± 8.30 | 0.034 | 56.10 ± 6.71 | 56.05 ± 6.24 | 56.14 ± 7.28 | 0.926 |
| CA125 (U/mL) | 90.08 ± 4.95 | 90.59 ± 4.18 | 88.62 ± 6.52 | 0.032 | 89.42 ± 5.46 | 89.39 ± 5.03 | 89.46 ± 5.96 | 0.951 |
| Hemoglobin (g/L) | 119.05 ± 8.06 | 118.46 ± 7.97 | 120.72 ± 8.20 | 0.162 | 120.49 ± 8.03 | 119.84 ± 7.12 | 121.13 ± 8.95 | 0.404 |
| Albumin (g/L) | 39.77 ± 2.26 | 39.94 ± 2.09 | 39.30 ± 2.67 | 0.111 | 39.43 ± 2.16 | 39.34 ± 1.53 | 39.53 ± 2.68 | 0.797 |
| ASA grade | 0.782 | > 0.999 | ||||||
| ASA I | 70 (57%) | 51 (57%) | 19 (59%) | 32 (67%) | 16 (67%) | 16 (67%) | ||
| ASA II | 32 (26%) | 23 (26%) | 9 (28%) | 11 (23%) | 6 (25%) | 5 (21%) | ||
| ASA III | 20 (16%) | 16 (18%) | 4 (13%) | 5 (10%) | 2 (8.3%) | 3 (13%) | ||
| Cardiovascular disease | 8 (6.6%) | 6 (6.7%) | 2 (6.3%) | > 0.999 | 3 (6.3%) | 1 (4.2%) | 2 (8.3%) | > 0.999 |
| Hypertension | 30 (25%) | 22 (24%) | 8 (25%) | 0.950 | 14 (29%) | 8 (33%) | 6 (25%) | 0.525 |
| Diabetes | 17 (14%) | 13 (14%) | 4 (13%) | > 0.999 | 7 (15%) | 4 (17%) | 3 (13%) | > 0.999 |
| Other comorbidities | 5 (4.1%) | 3 (3.3%) | 2 (6.3%) | 0.606 | 2 (4.2%) | 1 (4.2%) | 1 (4.2%) | > 0.999 |
| Operative approach | 0.084 | > 0.999 | ||||||
| Laparotomy | 46 (38%) | 38 (42%) | 8 (25%) | 16 (33%) | 8 (33%) | 8 (33%) | ||
| Laparoscopic surgery | 76 (62%) | 52 (58%) | 24 (75%) | 32 (67%) | 16 (67%) | 16 (67%) | ||
| Histological grade | 0.298 | 0.840 | ||||||
| Well differentiated | 46 (38%) | 36 (40%) | 10 (31%) | 17 (35%) | 8 (33%) | 9 (38%) | ||
| Moderately differentiated | 29 (24%) | 23 (26%) | 6 (19%) | 11 (23%) | 5 (21%) | 6 (25%) | ||
| Poorly differentiated | 47 (39%) | 31 (34%) | 16 (50%) | 20 (42%) | 11 (46%) | 9 (38%) | ||
| TNM stage | 0.220 | 0.634 | ||||||
| Stage I | 22 (18%) | 15 (17%) | 7 (22%) | 9 (19%) | 3 (13%) | 6 (25%) | ||
| Stage II | 58 (48%) | 40 (44%) | 18 (56%) | 25 (52%) | 13 (54%) | 12 (50%) | ||
| Stage III | 42 (34%) | 35 (39%) | 7 (22%) | 14 (29%) | 8 (33%) | 6 (25%) | ||
To reduce measured confounding, we constructed a propensity score model including sex, age, BMI, ECOG performance status, distance from the tumor to the anal verge, CEA, CA19-9, CA125, hemoglobin, albumin, ASA grade, operative approach, histological grade, TNM stage, and comorbidities (cardiovascular disease, hypertension, diabetes, and other conditions). A 1:1 nearest-neighbor matching without replacement was then performed using a caliper width of 0.2 on the logit of the propensity score. After matching, 24 patients in the Interrupted Ileostomy Tube Group were successfully matched to 24 patients in the Traditional Fistula Group, yielding a matched analytic cohort of 48 patients [Table 1].
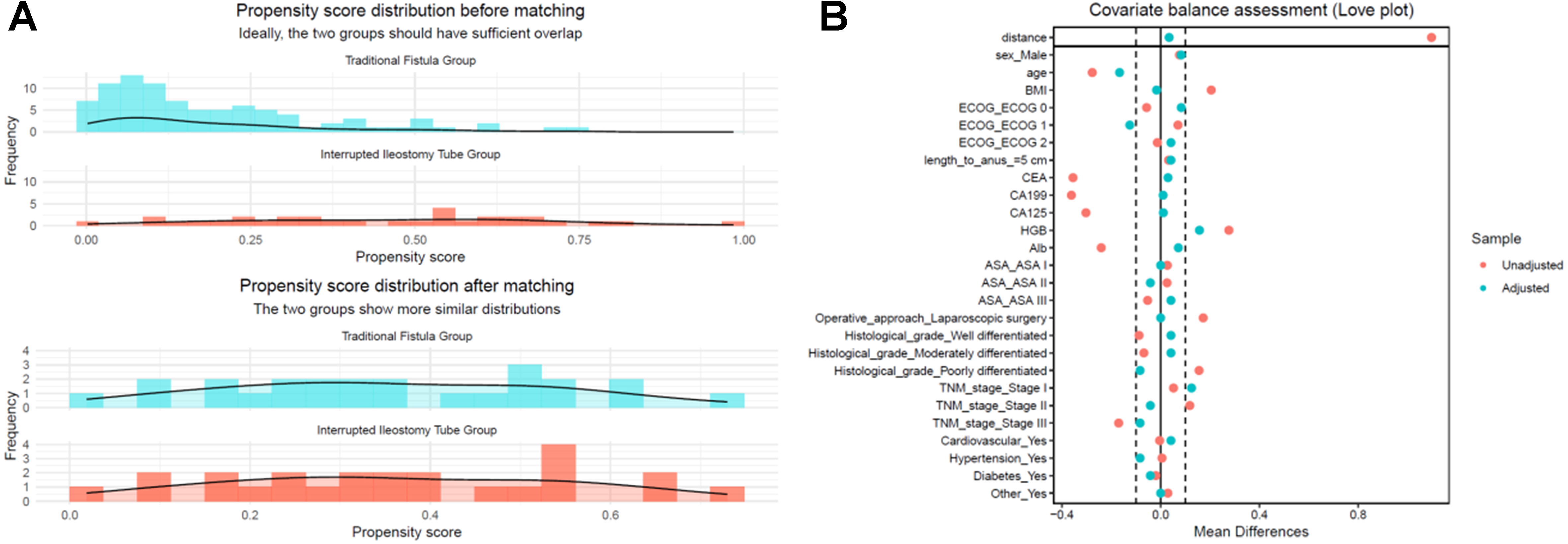
In the matched cohort, baseline covariates achieved improved balance as assessed by absolute SMDs, rather than hypothesis testing. After matching, most key continuous covariates reached good/excellent balance (|SMD| < 0.10), including BMI (0.011), CEA (0.037), CA19-9 (0.012), CA125 (0.013), and albumin (0.088). Two variables remained within the acceptable range but above the stringent threshold, namely age (|SMD| = 0.200) and hemoglobin (|SMD| = 0.159); importantly, all evaluated covariates met the prespecified acceptable balance criterion (|SMD| ≤ 0.20). The Love plot demonstrated a clear improvement in covariate balance before vs. after matching, and the distribution of propensity scores showed markedly greater overlap between the two treatment groups following matching [Figure 3A and B].
Figure 3. PSM diagnostics. (A) Love plot of absolute SMDs for baseline covariates before and after PSM; most covariates move below the prespecified balance threshold (|SMD| < 0.10) after matching; (B) Propensity score distributions for the Traditional Fistula Group and the Interrupted Ileostomy Tube Group before and after 1:1 nearest-neighbor matching, showing improved overlap in the matched cohort. PSM: Propensity score matching; SMDs: standardized mean differences; BMI: body mass index; ECOG: Eastern Cooperative Oncology Group; CEA: carcinoembryonic antigen; ASA: American Society of Anesthesiologists; TNM: tumor–node–metastasis.
Follow-up and completeness
The median follow-up duration in the overall cohort was 9.5 months (IQR 7.0-13.0; range, 3.0-18.0). Three patients were lost to follow-up, all in the Traditional group (Traditional: n = 3; Tube: n = 0). In the propensity-score–matched cohort (N = 48), the median follow-up duration was 9.0 months (IQR 7.0-12.0). Follow-up ≥ 6 months was available for 95/122 (77.9%) patients in the overall cohort and 41/48 (85.4%) patients in the matched cohort. For long-term outcomes, events were assessed up to the date of last successful contact.
Perioperative outcomes before and after matching
Before matching, patients in the Interrupted Ileostomy Tube Group had more favorable perioperative profiles than those in the Traditional Fistula Group. Specifically, they required a shorter time for stoma formation (21.38 ± 4.53 vs. 32.03 ± 8.51 min), achieved earlier first oral intake (3.26 ± 0.99 vs. 5.19 ± 0.95 days), had a shorter length of hospital stay (8.73 ± 1.68 vs. 11.50 ± 2.99 days), and incurred lower hospitalization costs [4.26 ± 0.62 vs. 5.59 ± 0.89 × 104 CNY (Chinese Yuan)] (all P < 0.001; Table 2). Time to first flatus was comparable between groups (3.44 ± 1.08 vs. 3.62 ± 1.06 days, P = 0.650).
Perioperative outcomes before and after PSM
| Before matching | After nearest-neighbor matching | |||||||
| Perioperative outcomes1 | Overall N = 122 | Traditional Fistula Group N = 90 | Interrupted Ileostomy Tube Group N = 32 | P-value2 | Overall N = 48 | Traditional Fistula Group N = 24 | Interrupted Ileostomy Tube Group N = 24 | P-value2 |
| Time of stoma formation (min) | 29.24 ± 8.98 | 32.03 ± 8.51 | 21.38 ± 4.53 | < 0.001 | 27.52 ± 8.87 | 33.54 ± 8.11 | 21.50 ± 4.38 | < 0.001 |
| Time to first flatus (days) | 3.57 ± 1.06 | 3.62 ± 1.06 | 3.44 ± 1.08 | 0.650 | 3.46 ± 1.00 | 3.52 ± 0.98 | 3.39 ± 1.03 | 0.796 |
| Time to first oral intake (days) | 4.68 ± 1.28 | 5.19 ± 0.95 | 3.26 ± 0.99 | < 0.001 | 4.21 ± 1.55 | 5.34 ± 1.07 | 3.07 ± 1.03 | < 0.001 |
| Length of hospital stay (days) | 10.78 ± 2.97 | 11.50 ± 2.99 | 8.73 ± 1.68 | < 0.001 | 9.82 ± 2.77 | 10.94 ± 3.20 | 8.70 ± 1.67 | 0.013 |
| Hospitalization cost (10,000 CNY) | 5.24 ± 1.02 | 5.59 ± 0.89 | 4.26 ± 0.62 | < 0.001 | 4.95 ± 1.00 | 5.64 ± 0.88 | 4.26 ± 0.53 | < 0.001 |
These perioperative advantages remained robust after PSM. In the matched cohort (24 vs. 24 patients), the Interrupted Ileostomy Tube Group continued to show a markedly shorter stoma formation time (21.50 ± 4.38 vs. 33.54 ± 8.11 min), earlier first oral intake (3.07 ± 1.03 vs. 5.34 ± 1.07 days), shorter hospital stay (8.70 ± 1.67 vs. 10.94 ± 3.20 days), and lower hospitalization cost (4.26 ± 0.53 vs. 5.64 ± 0.88 × 104 CNY) compared with the Traditional Fistula Group (all P ≤ 0.013; Table 2). Time to first flatus remained similar between groups after matching (3.39 ± 1.03 vs. 3.52 ± 0.98 days, P = 0.796).
Early postoperative complications
Early postoperative complications were uncommon in both groups [Table 3]. Overall, early postoperative complications occurred in four of 32 patients (12.5%) in the Interrupted Ileostomy Tube Group and in 18 of 90 patients (20.0%) in the Traditional Fistula Group (P = 0.343).
Early postoperative complications
| Complication1 | Interrupted Ileostomy Tube Group N = 32 | Traditional Fistula Group N = 90 | P value2 |
| 30-day postoperative morbidity (Clavien-Dindo ≥ II), n (%) | 4 (12.5%) | 18 (20%) | 0.343 |
| Anastomotic fistula, n (%) | 0 | 2 (2.2%) | 0.968 |
| Stoma complications, n (%) | 0 | 5 (5.6%) | - |
| Surgical site infection, n (%) | 1 (3.1%) | 4 (4.4%) | 1 |
| Intestinal obstruction, n (%) | 2 (6.2%) | 5 (5.6%) | 1 |
| Postoperative bleeding, n (%) | 0 | 0 | - |
| Abdominal effusion, n (%) | 0 | 1 (1.1%) | 1 |
| 30-day readmission, n (%) | 1 (3.1%) | 1 (1.1%) | 0.457 |
In the Traditional Fistula Group, complications included AL (2/90, 2.2%), stoma-related events (5/90, 5.6%), surgical site infection (4/90, 4.4%), intestinal obstruction (5/90, 5.6%), and abdominal effusion (1/90, 1.1%). In the Interrupted Ileostomy Tube Group, one patient (3.1%) developed a surgical site infection and two patients (6.2%) developed intestinal obstruction; no AL, stoma-related complications, postoperative bleeding, or abdominal effusion were observed in this group. Thirty-day readmission rates were low and similar between the two groups (3.1% vs. 1.1%, P = 0.457).
Long-term outcomes: secondary surgery and diversion-related complications
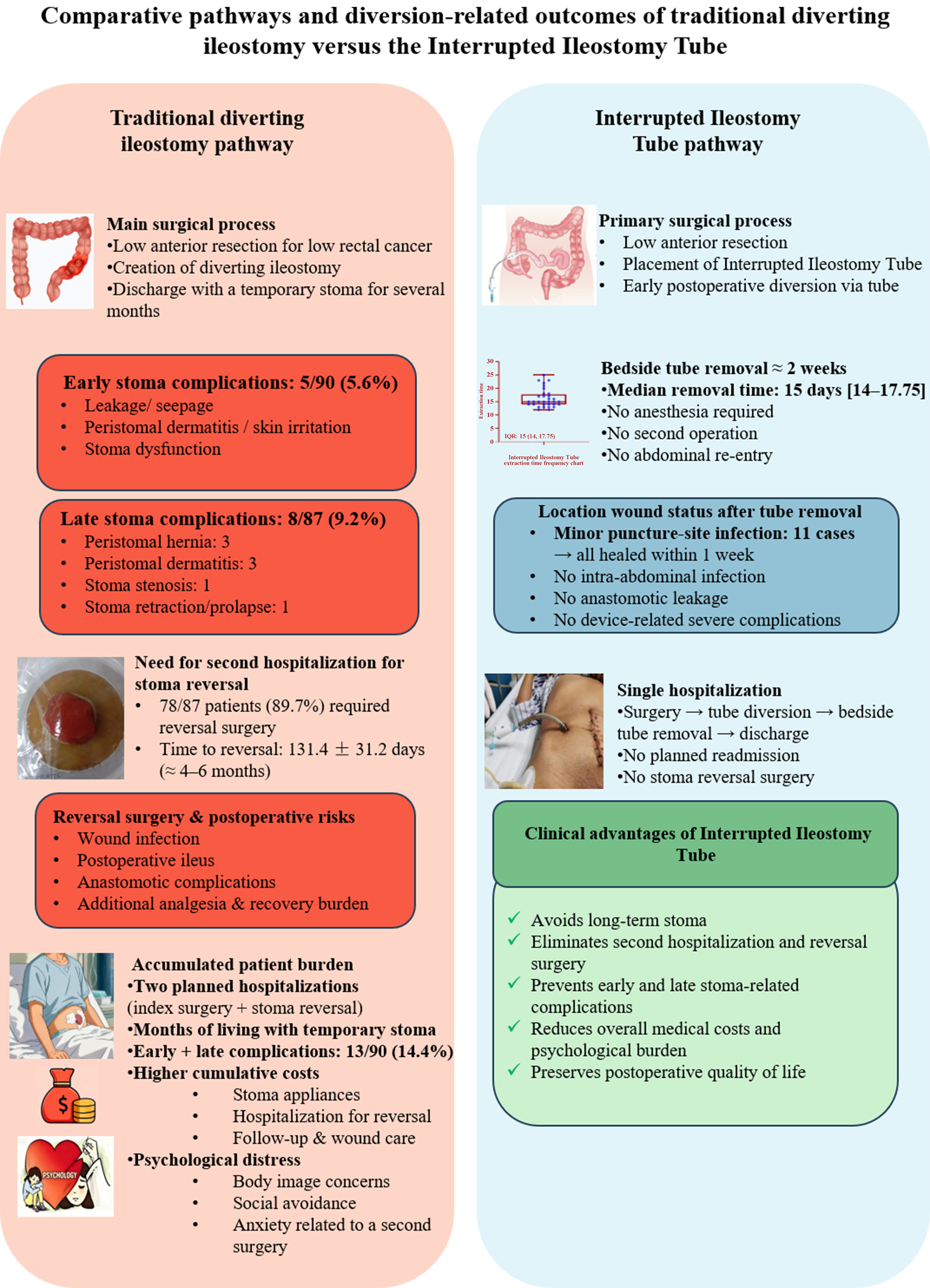
During follow-up, the downstream course of intestinal diversion differed markedly between the two groups. In the Traditional Fistula Group, most patients required a second hospitalization for elective ileostomy reversal 3-6 months after the index surgery. Among patients with available follow-up in the Traditional Fistula Group (n = 87), 78 underwent elective stoma closure during follow-up, with a mean interval of 131.4 ± 31.2 days from the primary operation. In contrast, no patient in the Interrupted Ileostomy Tube Group underwent a second abdominal procedure. Once bowel function and anal defecation had recovered, the tube was removed at the bedside, either during the index hospitalization or after discharge at scheduled outpatient follow-up, with a median time to removal of 15 days (IQR 14-17.75) measured from the date of surgery (postoperative days), as summarized in Figure 4. Notably, the index length of hospital stay in the Interrupted Ileostomy Tube group was 8.73 ± 1.68 days in the overall cohort (and 8.70 ± 1.67 days after PSM; Table 2). It is shorter than the median time to tube removal, indicating that tube removal was frequently completed after discharge during outpatient reassessment once predefined criteria were met.
Figure 4. Schematic comparison of long-term diversion pathways between traditional diverting ileostomy and the Interrupted Ileostomy Tube. The traditional pathway involves discharge with a temporary stoma, early stoma-related events (5/90) and late stoma-related complications (8/87), and a second hospitalization for stoma reversal after approximately 4-6 months (78/87; mean 131.4 ± 31.2 days), with additional operative, economic, and psychological burden. In contrast, the Interrupted Ileostomy Tube allows bedside tube removal either during the index hospitalization or after discharge at outpatient follow-up, with a median time to removal of 15 postoperative days (IQR 14-17.75); because the index LOS is shorter than the median removal time [Table 2], tube removal was frequently completed after discharge once predefined criteria were met, with no formal stoma, no planned second surgery, and only minor puncture-site infections that resolved with simple dressings. Created in BioRender. Zhao, H. (2026) https://BioRender.com/mmwdh62. IQR: Interquartile range; LOS: length of stay.
Late stoma-related morbidity occurred exclusively in the Traditional Fistula Group. Among patients with available follow-up in the Traditional Fistula Group (n = 87), 8 (9.2%) developed late complications at the stoma site, including parastomal hernia (3 cases), peristomal dermatitis (3 cases), stoma stenosis (1 case), and stoma retraction or prolapse (1 case). No corresponding stoma-related complications were observed in the Interrupted Ileostomy Tube Group, in which diversion was achieved without creation of a permanent abdominal stoma. In this group, 11 patients developed localized puncture-/exit-site infection after tube removal, defined as localized erythema with mild tenderness and/or minor exudation at the puncture/exit site without systemic signs of infection. All lesions were managed conservatively with local wound care (dressing changes) and resolved within approximately one week, with no invasive intervention, reoperation, readmission, or long-term sequelae; these events were classified as Clavien-Dindo grade I. Collectively, these findings indicate that the Interrupted Ileostomy Tube strategy completes diversion without a planned second abdominal surgery, avoids stoma reversal-related readmission, and prevents late stoma-related complications that are inherent to conventional diverting ileostomy, while maintaining an acceptable local wound profile after tube removal [Figure 4].
DISCUSSION
Radical resection remains the cornerstone of curative treatment for low rectal cancer, but the increasing emphasis on sphincter preservation and minimally invasive techniques has brought anastomotic integrity and postoperative quality of life to the forefront of clinical decision-making[20,21]. AL after low anterior resection occurs in approximately 3%-10% of cases and is associated with reoperation, permanent stoma formation, prolonged hospitalization, and impaired long-term function[22-24]. To mitigate this risk, temporary diverting ileostomy is widely used as a prophylactic measure[25,26], yet it introduces a distinct set of stoma-related complications and mandates a second operation for stoma reversal in most patients, raising concerns about overtreatment, costs, and psychosocial burden[27,28]. Against this background, the Interrupted Ileostomy Tube was developed as a device-based alternative that provides temporary diversion without the creation of a formal abdominal stoma.
In this retrospective propensity score–matched analysis, we compared the Interrupted Ileostomy Tube with traditional diverting ileostomy in patients undergoing low anterior resection for low rectal cancer. Baseline characteristics were generally balanced between groups, and any modest differences in tumor marker profiles were effectively attenuated by PSM. After matching, no significant imbalances in age, comorbidities, disease stage, or operative approach were observed, and the direction and magnitude of treatment effects on perioperative outcomes remained largely unchanged. These findings suggest that the observed benefits of the Interrupted Ileostomy Tube are unlikely to be explained solely by baseline differences and support the robustness of our primary results.
From a perioperative standpoint, the Interrupted Ileostomy Tube was associated with a consistently more favorable short-term profile. Compared with the traditional diverting ileostomy, the tube group had significantly shorter stoma formation time, earlier oral intake, reduced length of hospital stays, and lower hospitalization costs, both in the overall cohort and in the propensity-matched cohort. LOS refers to the index hospitalization and in our data was 8.7 days in the Tube group and 11.5 days in the Traditional group (10.9 days after matching). Because both groups were managed under the same institutional pathway, these between-group comparisons are internally valid within this real-world context. Although major postoperative morbidity was infrequent, a longer index LOS in the conventional diverting ileostomy cohort can be driven by stoma-related care requirements rather than surgical complications per se. Newly created ileostomies often have variable early output and carry a well-recognized risk of dehydration and electrolyte/renal derangements - one of the leading causes of stoma-related readmissions - prompting many institutions to monitor intake/output and biochemical parameters and to optimize hydration strategies before discharge. In addition, discharge readiness in new ostomates depends on achieving basic self-care competence (pouch emptying and changes, peristomal skin protection) and securing supplies and follow-up support, which has been identified as a common contributor to delayed discharge even after otherwise uneventful colorectal resection. These practical considerations may partly explain the longer hospitalization observed in patients managed with a traditional diverting ileostomy. Importantly, hospital discharge was not contingent on tube removal; patients could be discharged once clinically stable, with tube removal performed later at bedside during outpatient reassessment when appropriate. Mechanistically, these advantages are plausible. The tube-based technique avoids a large abdominal wall incision and circumferential fixation of an exteriorized ileal loop, instead relying on a balloon-occlusion catheter anchored intraluminally and at the abdominal wall. This simplifies the operative steps, shortens anesthesia time, and minimizes additional tissue trauma around the stoma site. In line with expert consensus on postoperative gastrointestinal function, shorter operative and anesthesia times, together with effective decompression via the tube, likely contributed to the more rapid recovery of bowel function and earlier advancement of diet observed in the Interrupted Ileostomy Tube group.
A key innovation of the Interrupted Ileostomy Tube lies in its device design. The adjustable balloon enables patient-tailored, near-complete intraluminal occlusion during the early postoperative phase, while allowing fine titration when clinically indicated (e.g., to optimize apposition or improve tolerance) and stepwise deflation during the weaning phase once bowel function recovers. This design helps maintain stable contact with the intestinal wall and may reduce leakage around the catheter, thereby supporting functional diversion more reliably than improvised catheters such as Foley tubes or nasogastric drains[29,30]. The dedicated flushing channel facilitates clearance of intraluminal debris and may decrease the likelihood of catheter blockage, allowing for more stable diversion during the early postoperative period. These technical features translate into practical advantages at the bedside, including easier nursing care, reduced need for appliance changes, and better cosmetic acceptability.
The most clinically meaningful difference between the two strategies emerged during follow-up. In the Traditional Fistula Group, 78 of 90 patients (86.7%) ultimately underwent stoma reversal after a mean interval of approximately 4-5 months, necessitating a second hospitalization and another abdominal procedure. Late stoma-related complications were observed exclusively in this group, with eight of 90 patients (8.9%) developing parastomal hernia, peristomal dermatitis, stoma stenosis, or stoma retraction/prolapse. These events are well-recognized sequelae of conventional diverting ileostomy and have been linked to impaired body image, social embarrassment, and long-term healthcare utilization in prior studies[31]. By contrast, no patient in the Interrupted Ileostomy Tube group required a planned second surgery. Diversion was terminated by bedside tube removal once bowel function and anal defecation had recovered, with a median removal time of 15 days (IQR 14-17.75). Localized puncture-site infections occurred in 11 patients but resolved with simple dressing changes within approximately one week, without reoperation or lasting disability. Thus, the tube strategy effectively decouples the benefits of temporary diversion from the long-term liabilities of a formal stoma and its reversal. Notably, the median time to tube removal (15 days) exceeded the mean index length of hospital stay (10.78 ± 2.97 days), indicating that tube removal was often completed after discharge during scheduled outpatient follow-up once predefined clinical criteria were met. This practice may help optimize inpatient resource utilization without requiring a second abdominal procedure.
Importantly, early postoperative safety was preserved. Our primary endpoint was 30-day clinically relevant AL (ISREC grade B/C). In the present cohort, clinically relevant AL was uncommon and did not appear to differ between groups (0/32 in the Interrupted Ileostomy Tube group vs. 2/90 in the traditional diverting ileostomy group). Within the limits of this retrospective analysis, we did not observe a signal of increased risk of clinically relevant AL with the tube strategy. However, clinically relevant leakage was rare, and the study was not designed or powered for non-inferiority or equivalence; thus, these results should be interpreted as evidence of feasibility and safety signals rather than definitive proof of comparable leak risk. Mechanistically, the tube may confer protection by providing functional diversion and continuous decompression, thereby reducing distal fecal stream exposure and intraluminal pressure at the anastomosis during the early healing phase. Future prospective, adequately powered multicenter studies with standardized postoperative imaging and adjudication are warranted to more precisely define the effect of this strategy on AL-related outcomes. Overall complication rates were low in both groups, and we did not observe an increased incidence of AL, ileus, or intra-abdominal sepsis in the Interrupted Ileostomy Tube group compared with the traditional stoma group. Moreover, no device-specific adverse events such as balloon rupture–related perforation or severe catheter-induced erosion were recorded. Although our study was not powered to detect small differences in rare events, the consistency of early outcomes between groups, together with the clear reduction in late stoma-related morbidity, supports the feasibility and safety of adopting the Interrupted Ileostomy Tube as an alternative diversion strategy in appropriately selected patients.
Beyond clinical endpoints, the Interrupted Ileostomy Tube also has implications for resource allocation and patient-centered care. Because reversal admissions and stoma-related complication costs were not included, our cost comparison likely underestimates the total pathway cost of conventional diverting ileostomy; notably, we report the reversal rate and time-to-reversal in Section “Long-term outcomes: secondary surgery and diversion-related complications” as downstream resource-use indicators. Therefore, the observed reduction in index-hospitalization cost should be interpreted alongside the downstream care pathway. By obviating the need for routine stoma reversal, this approach reduces cumulative hospital days, operative workload, and direct costs, allowing surgical and inpatient resources to be redirected to patients with greater need. For patients, avoiding a visible stoma and a second abdominal surgery may alleviate the psychological distress associated with altered body image, dependence on ostomy appliances, and uncertainty about timing of closure, thereby improving overall quality of life during the recovery period[32,33]. Although we did not formally measure patient-reported outcomes in this study, the observed patterns of reduced late morbidity and avoidance of reoperation are consistent with a more favorable psychosocial trajectory.
Limitations
Several limitations should be acknowledged. First, this was a single-center, retrospective study with a relatively modest sample size, which introduces potential selection bias and limits the precision of effect estimates. We attempted to mitigate confounding through PSM, but residual confounding by unmeasured variables cannot be fully excluded. Importantly, the diversion strategy was not randomly allocated and was influenced by shared decision-making, intraoperative judgment, and patient preference; therefore, confounding by indication remains a key concern despite adjustment. Although we demonstrated improved covariate balance after PSM, unmeasured factors (e.g., nuanced intraoperative assessment, surgeon preference, and perioperative details not captured in structured data) may still have affected both treatment selection and outcomes. Accordingly, the observed associations should be interpreted as comparative effectiveness signals rather than definitive causal effects. In addition, clinically relevant AL was rare, which limited statistical precision; accordingly, this study was not designed or powered to establish non-inferiority or equivalence with respect to leak risk. Second, the study population consisted of patients treated at a specialized rectal surgery unit, and the findings may not be directly generalizable to centers with different case mixes, surgical expertise, or perioperative protocols. Third, postoperative imaging and/or endoscopic evaluation for AL was performed when clinically indicated rather than mandated uniformly for all patients (including prior to tube removal), which may introduce variability in detection and classification of leakage events. Fourth, we did not include standardized health-related quality of life or cost-effectiveness measures, which would be valuable for quantifying the broader impact of the Interrupted Ileostomy Tube on patient experience and healthcare systems. Prospective, multicenter studies incorporating patient-reported outcomes, longer follow-up, and economic analyses are warranted to confirm and extend our findings. Future studies should also incorporate standardized postoperative surveillance and independent endpoint adjudication to more precisely define anastomotic leak–related outcomes.
Despite these limitations, our data indicate that the Interrupted Ileostomy Tube provides effective temporary diversion without compromising early postoperative safety, while simultaneously avoiding planned second surgery and eliminating late stoma-related complications inherent to conventional diverting ileostomy. In the context of modern low rectal cancer surgery - where sphincter preservation, functional outcomes, and quality of life are increasingly prioritized - this tube-based strategy may represent a valuable alternative for selected patients at risk of AL.
In conclusion, Interrupted Ileostomy Tube provides effective temporary fecal diversion for low rectal cancer surgery while maintaining early postoperative safety comparable to traditional diverting ileostomy. In our cohort, this technique was associated with shorter hospital stay, faster return to oral intake, lower hospitalization costs, and complete avoidance of planned stoma reversal and late stoma-related complications. These findings support Interrupted Ileostomy Tube as a practical alternative to conventional diverting ileostomy in carefully selected patients requiring prophylactic diversion.
DECLARATIONS
Acknowledgments
The graphical abstract was created with BioRender.com [Created in BioRender. Zhao, H. (2026) https://BioRender.com/8yfmaup].
Authors’ contributions
Contributed to the conception and design of the study, acquisition of data, data analysis and interpretation, drafting of the manuscript, and approval of the final version: Zhao H
Contributed to the acquisition of data, data analysis and interpretation, drafting of the manuscript, and approval of the final version: Liu Y
Contributed to the conception and design of the study, acquisition of data, data analysis and interpretation, and approval of the final version: Jin Y, Jiang D
Contributed to the conception and design of the study, acquisition of data, and approval of the final version: Zhao G, Li Y
Contributed to the conception and design of the study, acquisition of data, data analysis and interpretation, drafting and critical revision of the manuscript, and approval of the final version: Zhao J
Availability of data and materials
The datasets used and analyzed during the current study are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This study was supported by the following grants: 2023 Anhui Provincial Health and Wellness Research Project (No. AHWJ2023Bba20065); Anhui Province Clinical Medicine Research and Transformation Program (No. 202427b10020034); 2024 University-Enterprise Integrated Development “Qilu Research Fund” Project (No. XQQL202408); 2024 Key Research Project Fund (Natural Sciences Category) (No. WK2024ZZD28).
Conflicts of interest
Jin Y is the Guest Editor of the Special Issue entitled “Recent Advances in Gastrointestinal Tumour and Bariatric-Metabolic Surgery” in the journal Mini-invasive Surgery. Jin Y was not involved in any steps of the editorial process, notably including reviewers’ selection, manuscript handling, or decision-making. The other authors declare that there are no conflicts of interest.
Ethical approval and consent to participate
Ethical clearance for this study was obtained from the Ethics Committee of The First Affiliated Hospital of Wannan Medical College (Yijishan Hospital), approval number 2017 Lun Shen Xin (02). All patients provided written informed consent to participate in the study.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Sung H, Ferlay J, Siegel RL, Laversanne M, Soerjomataram I, Jemal A, Bray F. Global cancer statistics 2020: GLOBOCAN estimates of incidence and mortality worldwide for 36 cancers in 185 countries. CA Cancer J Clin 2021;71:209-49.
2. Szczepkowski M, Banasiewicz T, Krokowicz P, et al. Polish consensus statement on the protective stoma. Pol Przegl Chir 2014;86:391-404.
3. Wang S, Li AJ, Jiang HH, Lin Y, Ding HB. Sphincter-preserving surgical techniques in low rectal cancer management: a systematic review of contemporary evidence. World J Gastrointest Surg 2025;17:107525.
4. Rouanet P, Rivoire M, Gourgou S, et al. Sphincter-saving surgery after neoadjuvant therapy for ultra-low rectal cancer where abdominoperineal resection was indicated: 10-year results of the GRECCAR 1 trial. Br J Surg 2020;108:10-3.
5. Chitul A, Ciofic E, Burcoş T, Cristian D, Grama F. Functional outcomes and patient satisfaction after abdominoperineal resection versus sphincter-preserving techniques for low rectal cancer: a retrospective single-centre study. Chirurgia 2025;120:409-15.
6. Messias BA, Botelho RV, Saad SS, Mocchetti ER, Turke KC, Waisberg J. Serum C-reactive protein is a useful marker to exclude anastomotic leakage after colorectal surgery. Sci Rep 2020;10:1687.
7. Park JS, Choi GS, Kim SH, et al. Multicenter analysis of risk factors for anastomotic leakage after laparoscopic rectal cancer excision: the Korean laparoscopic colorectal surgery study group. Ann Surg 2013;257:665-71.
8. Lindgren R, Hallböök O, Rutegård J, Sjödahl R, Matthiessen P. What is the risk for a permanent stoma after low anterior resection of the rectum for cancer? A six-year follow-up of a multicenter trial. Dis Colon Rectum 2011;54:41-7.
9. Monzón-Abad A, Gracia-Roche C, Martínez-Germán A, Barranco-Domínguez I, Sánchez-Fuentes N. A preliminary study of transcaecal ileostomy as an alternative to defunctioning ostomies. Colorectal Dis 2014;16:130-3.
10. Clark DA, Stephensen B, Edmundson A, Steffens D, Solomon M. Geographical variation in the use of diverting loop ileostomy in Australia and New Zealand colorectal surgeons. Ann Coloproctol 2021;37:337-45.
11. Hanna M, Vinci A, Pigazzi A. Diverting ileostomy in colorectal surgery: when is it necessary? Langenbecks Arch Surg 2015;400:145-52.
12. Kawai M, Sakamoto K, Honjo K, et al. Benefits and risks of diverting stoma creation during rectal cancer surgery. Ann Coloproctol 2022;40:467-73.
13. Zhou X, Lin C, Chen W, Lin J, Xu J. Completely diverted tube ileostomy compared with loop ileostomy for protection of low colorectal anastomosis: a pilot study. Colorectal Dis 2014;16:O327-31.
14. Tan J, Ryan ÉJ, Davey MG, et al. Mechanical bowel preparation and antibiotics in elective colorectal surgery: network meta-analysis. BJS Open 2023;7:zrad040.
15. Shiomi A, Ito M, Maeda K, et al. Effects of a diverting stoma on symptomatic anastomotic leakage after low anterior resection for rectal cancer: a propensity score matching analysis of 1,014 consecutive patients. J Am Coll Surg 2015;220:186-94.
16. Cai L, Qiu G, Jiang Z, et al. Linea alba support method of prophylactic loop ileostomy via lower abdominal midline incision for rectal cancer: a retrospective cohort study. Surg Endosc 2025;39:1351-61.
17. Kisielewski M, Pisarska-Adamczyk M, Dowgiałło-Gornowicz N, et al. Timing of loop ileostomy closure does not play a pivotal role in terms of complications - results of the liquidation of iLEOstomy (LILEO) study. J Pers Med 2024;14:934.
18. van Helsdingen CP, Jongen AC, de Jonge WJ, Bouvy ND, Derikx JP. Consensus on the definition of colorectal anastomotic leakage: a modified Delphi study. World J Gastroenterol 2020;26:3293-303.
19. Clavien PA, Barkun J, de Oliveira ML, et al. The Clavien-Dindo classification of surgical complications: five-year experience. Ann Surg 2009;250:187-96.
20. Negro S, Bergamo F, Dell’Atti L, et al. Quality of life in rectal cancer treatments: an updated systematic review of randomized controlled trials (2013-2023). Cancers 2025;17:2310.
21. Sun G, Zang Y, Ding H, et al. Comparison of anal function and quality of life after conformal sphincter preservation operation and intersphincteric resection of very low rectal cancer: a multicenter, retrospective, case-control analysis. Tech Coloproctol 2023;27:1275-87.
22. Lu ZR, Rajendran N, Lynch AC, Heriot AG, Warrier SK. Anastomotic leaks after restorative resections for rectal cancer compromise cancer outcomes and survival. Dis Colon Rectum 2016;59:236-44.
23. Hammond J, Lim S, Wan Y, Gao X, Patkar A. The burden of gastrointestinal anastomotic leaks: an evaluation of clinical and economic outcomes. J Gastrointest Surg 2014;18:1176-85.
24. Turrentine FE, Denlinger CE, Simpson VB, et al. Morbidity, mortality, cost, and survival estimates of gastrointestinal anastomotic leaks. J Am Coll Surg 2015;220:195-206.
25. Li SY, Chen G, Bai X, et al. Anus-preserving rectectomy via telescopic colorectal mucosal anastomosis for low rectal cancer: experience from a Chinese cohort. World J Gastroenterol 2013;19:3841-6.
26. Sheng QS, Hua HJ, Cheng XB, et al. A modified spontaneously closed defunctioning tube ileostomy after anterior resection of the rectum for rectal cancer with a low colorectal anastomosis. Indian J Surg 2016;78:125-9.
27. Lin L, Wang Z, Zhang Q, Wang C, Zhang Z. Application of transumbilical laparoscopic surgery on low/ultralow rectal cancer for anal sphincter preservation. J Laparoendosc Adv Surg Tech A 2022;32:740-6.
29. Wang H, Chen W, Zhou X, Ye T, Gong L, Cai Y. Application of the intestine diversion tube with a double-balloon without ileostomy in low rectal cancer. J Gastrointest Oncol 2023;14:213-9.
30. Patil V, Vijayakumar A, Ajitha MB, Kumar LS. Comparison between tube ileostomy and loop ileostomy as a diversion procedure. ISRN Surg 2012;2012:547523.
31. Morks AN, Havenga K, Ploeg RJ. Can intraluminal devices prevent or reduce colorectal anastomotic leakage: a review. World J Gastroenterol 2011;17:4461-9.
32. Song L, Han X, Zhang J, Tang L. Body image mediates the effect of stoma status on psychological distress and quality of life in patients with colorectal cancer. Psychooncology 2020;29:796-802.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.