fig3
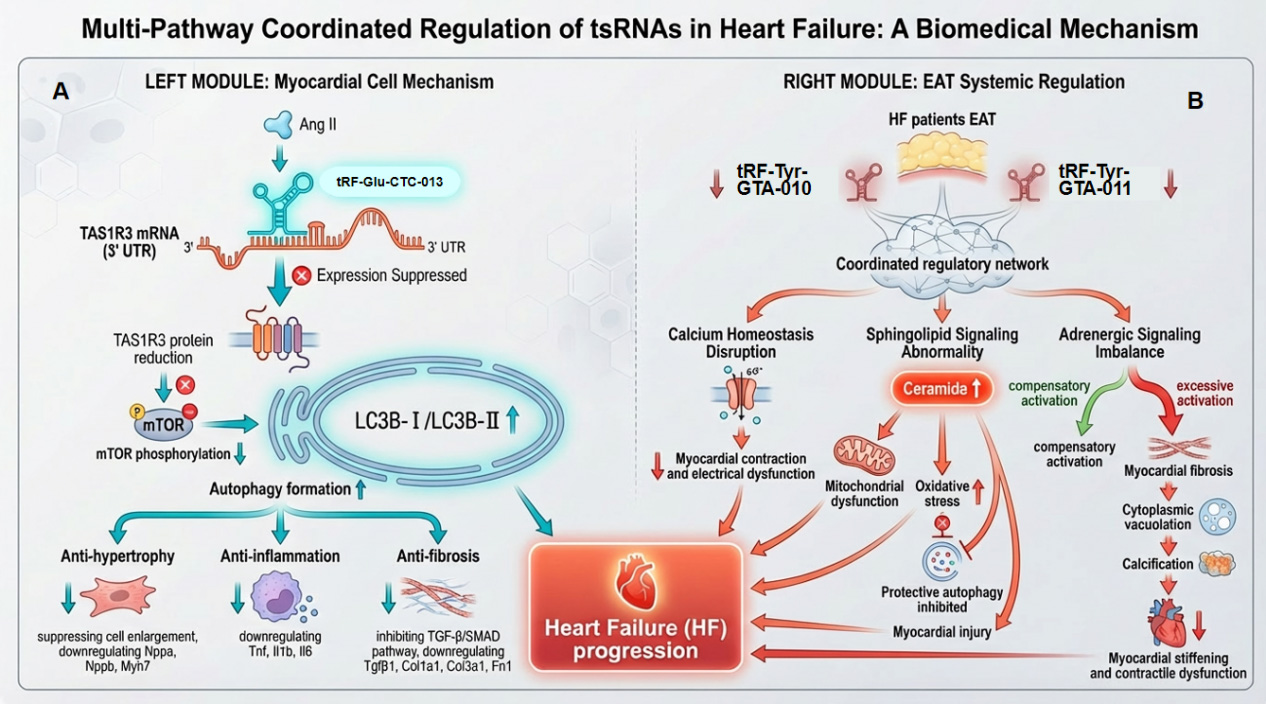
Figure 3. Multi-pathway coordinated regulation of tsRNAs in HF. (A) Myocardial cell-intrinsic protective mechanism. The tsRNA tRF-Glu-CTC-013 binds to the 3’UTR of TAS1R3 mRNA, suppressing TAS1R3 protein expression. Reduced TAS1R3 leads to mTOR phosphorylation inhibition and promotes the conversion of LC3B-I to LC3B-II, thereby inducing autophagy. This autophagic response exerts anti-hypertrophic effects (suppressing cell enlargement and downregulating Nppa, Nppb, Myh7), anti-inflammatory effects (downregulating Tnf, Il1b, Il6), and inhibits the TGF-β/SMAD pathway (downregulating Tgfb1, Col1a1, Col3a1, Fn1). Collectively, this pathway counteracts myocardial hypertrophy, fibrosis, metabolic dysfunction, and subsequent contractile and electrical dysfunction; (B) Systemic regulatory mechanism via epicardial adipose tissue (EAT). In the EAT of HF patients, tsRNAs (IRF-Tyr-GTA-010, IRF-Tyr-GTA-011) form a coordinated regulatory network. This network induces sphingolipid signaling abnormality (increased ceramide) and adrenergic signaling imbalance. These disturbances lead to MF, cytoplasmic vacuolation (fatty degeneration), and calcification, ultimately contributing to myocardial stiffening and contractile dysfunction. tsRNAs: tRNA-derived small RNAs; HF: heart failure; mTOR: mammalian target of rapamycin; TGF-β: transforming growth factor-β; MF: myocardial fibrosis; SMAD: mothers against decapentaplegic homolog.