Smart nanomaterial-driven innovations in combined cancer immunotherapy
Abstract
Conventional cancer immunotherapy frequently encounters limitations such as suboptimal clinical responses, systemic adverse effects, and acquired immunologic tolerance. Rationally designed smart nanomaterials, engineered to recognize tumor-specific stimuli, target immune cells, and remodel the tumor microenvironment, offer significant potential in overcoming these formidable challenges in metastatic tumors. In this review, we summarize the latest advancements in efficacious therapeutic interventions for the purpose of improving pharmaceutical properties, remodeling tumor immune microenvironment, and achieving combined immunotherapy. Then, we provide a brief overview of the most recent combinational immunotherapy corresponding clinical management, highlighting the clinically validated combinations that elicit systemic antitumor immunity and long-term immunomodulatory memory. Furthermore, we outline key molecular mechanisms and signaling pathways for organ-specific metastasis, and discuss certain preclinical advancements in the realm of smart nanomaterials integrated with existing treatment modalities and immunomodulatory strategies. Additionally, the discussion includes present challenges and future opportunities in designing functional nanomaterials, emphasizing the critical factors related to material design, safety concerns, and regulatory mechanisms. Overall, this review synthesizes preclinical and clinical findings to demonstrate how smart nanomaterials can enhance the therapeutic index through multi-target immunomodulation, and it examines the major challenges and future possibilities of immunotherapy combinations.
Keywords
INTRODUCTION
Metastasis is a crucial hallmark of malignancy, posing substantial challenges to effective clinical management[1]. Immunotherapy has demonstrated potential in treating certain metastatic tumors characterized by high mutational burden[2]. Cancer immunotherapy, particularly immune checkpoint inhibitors, has been approved for clinical use for durable remissions in many metastatic tumors[3-5]. Distinct from conventional therapeutic modalities, its core advantage is activating the body’s intrinsic immune system for precise tumor elimination, with lower systemic toxicities, durable efficacy, and potential synergistic effects[6,7]. Despite notable advancements, cancer immunotherapy still confronts unresolved dilemmas, encompassing strategies to augment therapeutic effectiveness across various malignancies and patients, as well as methodologies to forecast patient responsiveness or resistance[8,9]. Therefore, elucidating the molecular mechanisms underlying clinical outcomes and therapeutic resistance is pivotal to broadening the scope of beneficial impacts derived from immunotherapeutic interventions[10-12]. Mechanistic insights from the cancer-immunity cycle underscore the potential to enhance immunotherapy efficacy while mitigating acquired immunological tolerance[13,14]. Effective therapeutic interventions should target resistance mechanisms in malignancies to boost long-term T cell responses and facilitate their iterative expansion[15,16].
The iterative nature of the antitumor immune response can partially be explained by the cancer-immunity cycle, originally conceptualized in 2013, which refers to a series of stepwise events driving T cell responses to kill tumors in a self-sustaining manner[17,18]. In brief, the process begins with the release of tumor-associated antigens from dying tumor cells, followed by the processing and presentation of these antigens by antigen-presenting cells, particularly dendritic cells (DCs)[19-21]. Therapeutic interventions have been employed to promote the release of tumor-associated antigens by inducing tumor death in an immunogenic pathway, involving current clinical scenarios such as chemotherapy[22], radiotherapy (RT)[23], and targeted therapy[24]. In the subsequent steps, vaccines have been recognized as an effective modality to enhance the presentation of antigenic peptides. Meanwhile, blockade of the immune checkpoint cytotoxic Tlymphocyte-associated protein 4 (CTLA4) proves efficient in facilitating T cell priming and activation[25]. Next, activated effector T cells traffic and infiltrate into tumors and stroma, interact with intratumoral immune cells, and maintain their effector function[26,27]. Targeting these steps, removal of pathophysiological barriers within the tumor microenvironment (TME) may enhance T cell infiltration, comprising dense extracellular matrix, abnormal angiogenesis, and metabolic derangements[28,29]. In addition, increasing chemokine levels or inhibiting transforming growth factor also contributes to T cells’ trafficking and infiltration[30]. Finally, intratumoral effector T cells potentially recognize and eliminate cancer cells, and release additional tumor-associated antigens to favor a self-sustained cycle[31,32]. Immunotherapeutic approaches such as chimeric antigen receptor T (CAR-T) cell therapy [33] and T-cell receptor-engineered T-cell (TCR-T) therapy[34] are able to yield T cell-mediated cytotoxic responses. Conceivably, programmed cell death ligand-1 (PD-L1) has an inhibitory function that primarily acts to modulate tumor killing[35,36].
Considering the intricate interplay of numerous factors within the TME, combination therapies are strategically positioned to synergistically complement and sustain the cancer-immunity cycle[37,38]. Rational integration of multiple treatment modalities, including chemotherapy, phototherapy, gene therapy, and immunotherapy, has been proposed to restore immunosurveillance and reinvigorate pre-existing antitumor immune responses[39-41]. Engineered nanoplatforms have been designed to act across multiple stages of the cancer-immunity cycle, capitalizing on the pharmaceutical advantages of targeting specificity and stimuli responsiveness[42-44]. The rapid advancement of nanomaterials offers unprecedented opportunities for precise immune modulation with tolerable immune-related adverse events[45,46].
To date, a variety of nanoplatforms have entered clinical trials, involving lipid nanoparticles (LNPs)[47,48], polymeric nanoparticles[49], inorganic nanomaterials[50], and exosome-mimetic nanovesicles[51]. For example, LNPs, notable for their adjustable size, easy surface modification, and biocompatibility, have emerged as pivotal tools in facilitating the controlled release of immunostimulatory agents (BNT122, NCT03289963) and potent vaccination (mRNA-4157, NCT03897886)[52-54]. The polymeric nanoformulation of AZD4635 is currently undergoing Phase II trials (NCT04089553), demonstrating outstanding biosafety and precise drug release capabilities in cancer immunotherapy[55]. Research into metalloimmunotherapy involves inorganic nanomaterials such as MnO2, and gold nanoparticles have been approved for image-guided radioimmunotherapy[56,57]. Mesenchymal stromal cell-derived iExosomes (NCT03608631) or DC-derived exosome (NCT01159288) exhibit unique advantages in tumor targeting and immune modulation in clinical settings[58-60]. Although certain nanomedicines have gained approval for clinical trials, the issue of metastases caused by hematogenous dissemination of circulating tumor cells remains a tough clinical issue[61]. Smart nanomaterials possess natural immune-stimulating properties or incorporate immunomodulatory adjuvants, and have precipitated a transformative breakthrough in overcoming delivery barriers and optimizing therapeutic efficacy[62,63].
By integrating diverse therapeutic modalities, nanomaterials effectively address critical challenges in cancer immunotherapy, particularly in microenvironment remodeling, therapeutic optimization, and the suppression of tumor metastasis. This review elucidates the synergistic potential of nanomaterials in reinitiating the cancer-immunity cycle when combined with chemotherapy, RT, phototherapy, sonodynamic therapy, metabolic regulation, and neuro-immune interactions, with a specific focus on strategies to target and eliminate disseminated tumor cells at metastatic sites [Figure 1]. Furthermore, the rational design of functionalized nanoplatforms, encompassing material selection, structural configuration, and the application of artificial intelligence (AI), is discussed in terms of overcoming physiological barriers, counteracting the immunosuppressive microenvironment, and preventing metastatic colonization. Current challenges and future opportunities in engineering nanomaterials to synergistically modulate the immunosuppressive microenvironment and restore antitumor immunity are also discussed. This review highlights nanomaterial-supported combined immunotherapy methods, offering crucial insights for optimizing treatment outcomes, inhibiting distant metastasis, and moving towards clinical translation.
Figure 1. Schematic representation of smart nanomaterials-enabled multimodal combinatorial cancer immunotherapy. Multifunctional nanoplatforms integrating chemotherapy, radiotherapy, phototherapy, sonodynamic therapy, metabolic regulation, and immune regulation are designed with artificial intelligence (AI)-assisted optimization to synergistically activate systemic antitumor immunity, remodel the TME, target distant metastatic sites, and optimize therapeutic intervention. Drawn by Figdraw (ID: TRWRA36009). TME: Tumor microenvironment.
CLINICALLY VALIDATED COMBINATIONS IN CANCER IMMUNOTHERAPY
Clinical evidence has shown the shortcomings of monotherapies in handling metastatic tumors, prompting the swift advancement of combined treatment approaches recently. Clinically validated combinations have demonstrated notable potential in different solid tumors, improving progression-free survival (PFS) and objective response rate (ORR) [Table 1]. Typically, combining immune checkpoint inhibitors with chemotherapy has become a standard of care for non-small cell lung cancer (NSCLC), triple-negative breast cancer (TNBC), and head and neck squamous cell carcinoma (HNSCC).
Most recent clinical trials of immunotherapy combinations
| Treatment | Tumor type | Outcomes | Identifier |
| Zolbetuximab + mFOLFOX6 | Metastatic gastric carcinoma | PFS: 10.6 months | NCT03504397 Phase III, 2023 |
| αPD-1 + capecitabine + cyclophosphamide + vinorelbine | Metastatic HER2-negative breast cancer | ORR: 18.5%; PFS: 6.6 months; OS: 42.6 months | NCT04389073 Phase II, 2024 |
| αPD-1 + lenvatinib + TACE | Unresectable HCC | SLR: 59.2%; ORR: 78.9%; PFS: 20.03 months | NCT04997850 Phase II, 2026 |
| αPD-L1 + SBRT | Metastatic NSCLC | PFS: 15.56 months; OS: 40.5 months | NCT02492568 Phase II, 2025 |
| Sintilimab + lenvatinib + cryoablation | Advanced/metastatic intrahepatic cholangiocarcinoma | ORR: 75%; PFS: 16.8 months; OS: 25.4 months | NCT05010668 Phase II, 2025 |
| αPD-1 + dasatinib + quercetin | HNSCC | OS: up to 5 years | NCT05724329 Phase II, 2025 |
| Sintilimab + carboplatin + nab-paclitaxel | Oral/oropharyngeal squamous cell carcinoma | ORR: 64.6%; OS: 24 months | NCT04718415 Phase II, 2025 |
| Pembrolizumab + axitinib + FMT | Metastatic renal cell carcinoma | ORR: 52%; PFS: 24 months | NCT04758507 Phase II, 2025 |
| αPD-1 + FMT | NSCLC | ORR: 80%; | NCT04951583 Phase II, 2025 |
| Melanoma | ORR: 75%; |
For patients with unresectable hepatocellular carcinoma (HCC), transarterial chemoembolization (TACE) appears to be superior first-line therapy, while the conversion rate from this locoregional therapy is only about 10%. Promising results from the complex triple combination of lenvatinib, TACE, and PD-1 inhibitors (LEN-TAP) regimen, have led to a higher rate of salvage liver resection (SLR) than TACE alone, at 59.2% compared to 18.3%[64]. In another phase II trial in metastatic breast cancer, combinational treatment of metronomic chemotherapy plus anti-PD-1 (αPD-1) significantly prolonged the patient’s PFS[65]. This regimen demonstrates favorable safety and systemic immune reprogramming in refractory malignancies, representing a promising option for patients with aggressive, typically immunologically cold breast cancer.
Radio-immunotherapy has demonstrated its efficacy in patients with a low tumor mutation burden, and may expand the therapeutic window of checkpoint blockade for low response rates[66,67]. In cases of metastatic NSCLC, the combination of stereotactic body radiation therapy (SBRT) followed by pembrolizumab (NCT02492568) resulted in a longer PFS compared to SBRT alone (15.56 vs. 4.39 months)[68]. This approach exhibits superior efficacy in immunologically cold tumors characterized by low tumor mutation burden, PD-L1 negativity, and Wnt mutations. In a landmark study, minimally invasive cryoablation may also affect host immune responses and sensitivity to therapeutic interventions[69]. When combined with checkpoint inhibitor sintilimab and lenvatinib, partial cryoablation in advanced or metastatic intrahepatic cholangiocarcinoma significantly increased the ORR[70]. However, it also faces several major limitations, including a small single-arm population, lack of independent central review, and unvalidated efficacy in patients with the combined treatment regimen of gemcitabine, cisplatin, and immune checkpoint inhibitors (GemCis-ICI) refractory disease.
The gut microbiota is increasingly recognized as a pivotal factor in cancer pathogenesis and treatment response. Fecal microbiota transplantation (FMT) has emerged as a promising strategy to modulate the microbiome and reshape the immune landscape[71]. Results from the Phase 2 TACITO trial demonstrated that FMT combined with immune checkpoint inhibitors for the treatment of metastatic renal cell carcinoma was associated with a significant increase in PFS (24.0 months vs. 9.0 months) and ORR (52% vs. 32%) compared with patients receiving placebo therapy[72]. Additionally, microbiome profiling confirmed the engraftment of donor strains, along with increased α-diversity and more pronounced shifts in microbiome composition of β-diversity relative to baseline in the FMT-treated group. This combination therapy offers durable clinical benefits and effective microbiome reprogramming via successful donor engraftment, yet is constrained by small sample size, insufficient baseline balance, reliance on a single donor, and challenges in stable stool supply.
Given the elevated expression of mesothelin, nanobody-functionalized CAR-T cells targeting mesothelin and equipped with αPD-1 nanobodies (referred to as NAC-T) have been strategically constructed[73]. Recently, a first-in-human clinical trial was performed in 11 patients with refractory malignant mesothelioma who had failed standard therapies. The NAC-T treatment demonstrated favorable safety, with an overall response rate of 63.6%, and the disease control rate reached 100%. The median PFS was 5.0 months, and the median overall survival was prolonged to 25.6 months. These clinical results underscore the promising translational potential of combinational nanobody-mediated immune checkpoint inhibition with CAR-T therapy.
Despite advances in clinical management, effectively inhibiting distant tumor metastasis remains one of the most formidable challenges in contemporary oncology[74,75]. The primary obstacles for combinatorial antimetastatic regimens are major off-target effects[76]. Non-specific biodistribution and off-target drug release may initiate excessive inflammation and lead to immune-related adverse events[77]. By using specific ligands for surface modification, engineered nanoparticulate formulations actively target metastatic tumors, leading to increased accumulation at the tumor site and reduced unintended exposure to healthy tissues[78]. Nanocarriers that respond to the TME allow for controlled drug release triggered by pathological characteristics, thereby minimizing early leakage and off-target effects[79,80]. Emerging nanomaterials for cargo delivery have demonstrated compelling potential in improving therapeutic selectivity and reducing immune-related adverse events[81,82].
PRECLINICAL PROGRESS OF NANOMATERIAL-ENABLED COMBINATIONAL IMMUNOTHERAPY
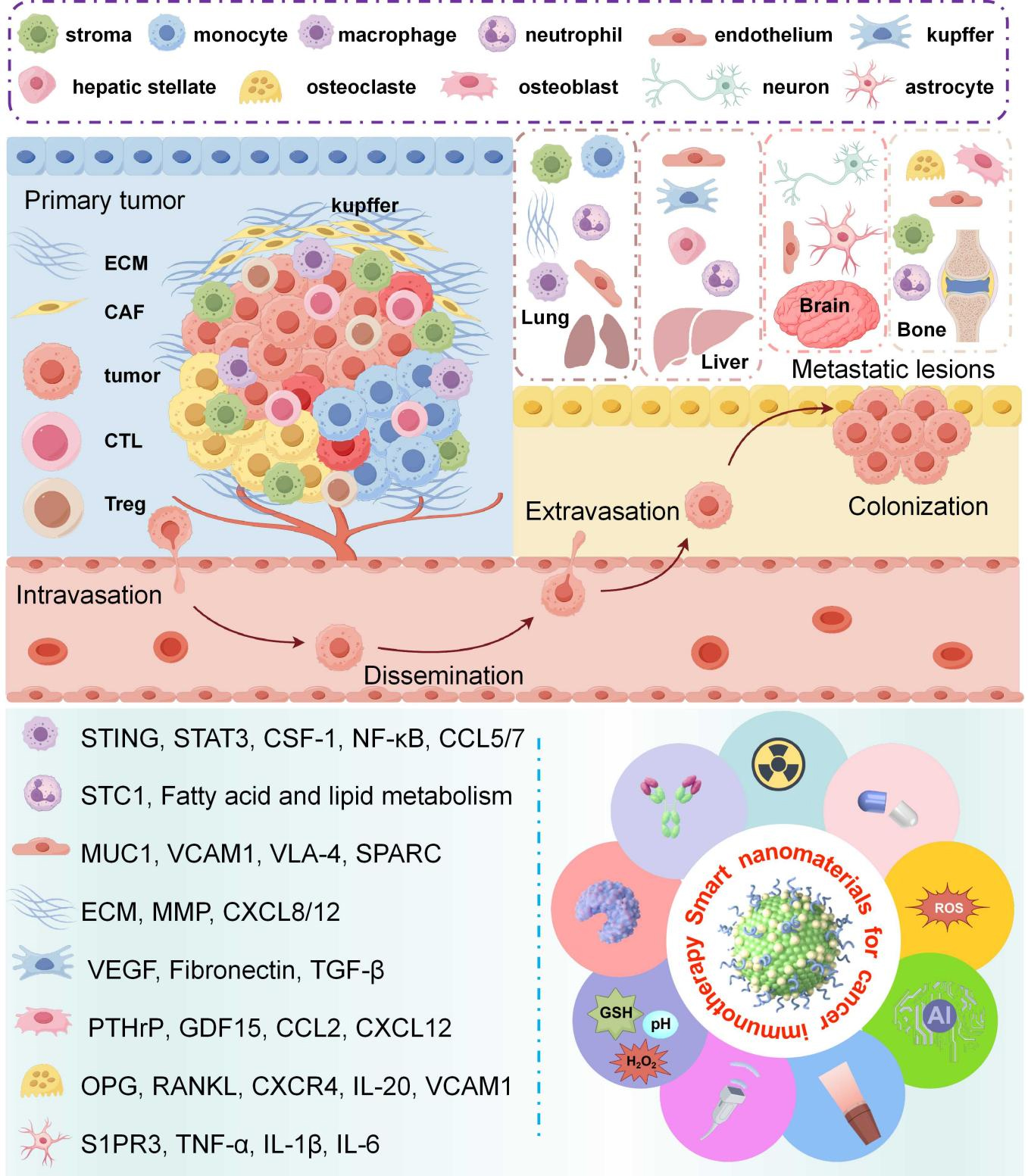
The metastatic process typically encompasses several stages, beginning with the intravasation of primary tumor cells into the vasculature, followed by hematogenous dissemination, extravasation into distant organ sites, and culminating in the colonization and proliferation of secondary lesions [Figure 2]. Malignant progression often results in the development of secondary lesions in distant organs such as the lungs, liver, bones, and brain, significantly compromising patient outcomes and diminishing long-term survival.
Figure 2. Schematic illustration of the metastatic cascade and nanomaterial-driven combinatorial immunotherapeutic strategies targeting organ-specific metastasis. This diagram depicts the multistep metastatic process including intravasation, dissemination, extravasation, and colonization at distant organs (lung, liver, bone, brain). Smart nanomaterials interrupt the metastatic cascade by remodeling the immunosuppressive TME, inhibiting ECM remodeling and CAF activity, blocking key oncogenic signaling pathways, and suppressing the formation of metastatic niches. Meanwhile, nanoplatforms trigger ICD, promote CTL infiltration, reduce Treg abundance, and reactivate systemic antitumor immune responses to inhibit tumor invasion and distant metastasis. CAF: Cancer-associated fibroblast; CTL: cytotoxic T-lymphocyte; ECM: extracellular matrix; ICD: immunogenic cell death; MMP: matrix metalloproteinase; MUC1: Mucin-1; OPG: osteoprotegerin; PTHrP: parathyroid hormone related protein; RANKL: receptor activator of nuclear factor-κB ligand; S1PR3: sphingosine-1-phosphate receptor 3; STC1: stanniocalcin 1; STING: stimulator of interferon gene; Treg: T regulatory cell; VEGF: vascular endothelial growth factor; TME: tumor microenvironment; ROS: reactive oxygen species; AI: artificial intelligence; STAT3: signal transducer and activator of transcription 3; CSF-1: colony-stimulating factor 1; NF-κB: nuclear factor kappa B; CCL5/7: chemokine ligand 5/7; VCAM1: vascular cell adhesion molecule-1; VLA-4: very late appearing antigen-4; SPARC: secreted protein acidic and rich in cysteine; CXCL8/12: C-X-C motif chemokine ligand 8/12; TGF-β: transforming growth factor beta; GDF15: growth differentiation factor 15; CCL2: chemokine ligand 2; PANKL: primary adrenal NK/T cell lymphoma; CXCR4: Chemokine receptor 4; IL-1β: interleukin-1 beta; IL-6: interleukin-6; TNF-α: tumor necrosis factor-α; GSH: glutathione. Drawn by Figdraw (ID: UPRTY104f0).
Nanomaterials synergizing with immunogenic cell death induction
Nanoplatform-mediated therapeutic interventions, including reactive oxygen species (ROS) generation, DNA damage induction, and mild photothermal stimulation, effectively trigger immunogenic cell death (ICD), accompanied by the release of tumor-associated antigens and damage-associated molecular patterns[83,84]. Targeting activation of the STING (stimulator of interferon gene) pathway has shown efficacy in amplifying ICD and RT sensitization. In this regard, He et al. constructed a metformin-based multifunctional nanocomplex to promote cytosolic DNA accumulation for maximizing radio-immunotherapy[85]. In this nanocomplex, metformin modification not only significantly enhances the transfection efficiency of siTREX1 but also exhibits pharmacological inhibition of DNA repair by targeting the DNA homologous recombination repair protein recombinase. Attributed to the obvious ICD effect, the CS-MET/siTREX1 nanocomplex achieved 99.1% primary tumor growth inhibition and 88.2% distant metastasis suppression under 20 Gy irradiation. Moreover, siTREX1 contributes to the cytosolic DNA accumulation, leading to a nearly 2.7-fold increase in cytosolic DNA levels in comparison to those without gene silencing. According to western blotting assays, the expression of TREX1 and RAD51 was downregulated, while the DNA breakage biomarker and STING-related proteins were dramatically upregulated when cells were treated with the nanocomplex and irradiation.
Mild-temperature photothermal therapy, generally 42-45 °C , has shown significant advantages in initiating biological effects, such as loosening the extracellular matrix, enhancing blood perfusion, and stimulating in situ vaccination[86]. However, this mild hyperthermia is not enough for localized tumor ablation because of the self-defense mechanisms derived from heat shock response or autophagy[87]. In this regard, engineered photothermal-targeting chimeras (PTTACs) were proposed to achieve tumor-specific degradation of PD-L1 and bromodomain-containing protein 4 (BRD4)[88]. To achieve spatiotemporally confined degradation of the protein of interest, PTTACs were loaded into micellar nanoparticles responsive to extracellular acidity and intracellular acidity, respectively. Moreover, PTTACs exhibit high photothermal conversion efficiency and protein binding affinity, and self-assemble into lamellar nanosheets with improved photothermal conversion and selective protein binding. Exposure to an 808 nm laser led the PTTAC nanoplatform to reduce PD-L1+ tumor cells by 3.5-fold. As a result, this combined strategy significantly suppressed tumor growth and metastasis by degrading PD-L1/BRD4, inducing robust ICD effects, and eliciting potent antitumor immunity as well.
Nanomaterials synergizing with TME remodeling
Through reversal of metabolic disorders, normalization of the extracellular matrix, and disruption of neuro-immune inhibitory signals, rationally designed nanomaterials are capable of reestablishing and sustaining anti-tumor immunity[89,90]. Abnormal metabolism in tumor cells supports their proliferation, enables them to adapt to stresses, and helps them evade immune surveillance[91]. Interfering with these metabolic processes, specifically aerobic glycolysis, which increases energy supply and lactate accumulation, is beneficial for reversing immune suppression[92]. To counteract these metabolic abnormalities, a programmed nanosystem is designed to rectify the glucose uptake and aerobic glycolysis[93]. T-AsiG-CPL disassembles to release the nuclear factor κB (NF-κB) inhibitor, decreasing extracellular matrix deposition and facilitating deep penetration of CD71 aptamer-linked glucose transporter 1 small interfering RNA (AsiG) in response to tumor acidity. Mechanistically, this silences glucose transporter 1, suppressing glycolysis and mitochondrial respiration by 78.19% and 88.60%, respectively. Moreover, T-AsiG-CPL treatment resulted in a 79.13% reduction in tumor weight and an 82.79% decrease in tumor volume, and completely eradicated metastasis foci in the mesentery, liver, and lung. The advantage in preventing metastasis stems from normalized stromal homeostasis and interrupted metabolic crosstalk, thereby restraining the invasive and disseminating potential of tumor cells.
Excessive activation of the sympathetic nervous system is responsible for T cell exhaustion, and this mechanistic insight into neuro-immune interactions offers opportunities for therapeutic intervention[94,95]. Recently, a ROS-responsive liposomal nanoplatform (LipoNCs@pGSDMB) was developed for bone metastasis treatment through neuroimmune modulation[96]. The formed liposome co-delivers a polymeric STING agonist and gasdermin B plasmid, which is selectively activated in metastatic bone niches to block neurosignaling via the voltage-gated calcium channel. The LipoNCs@pGSDMB nanoplatform elevated the proportion of mature DCs in draining lymph nodes to 41.3%, nearly doubled CD8+ cytotoxic T cell infiltration, and reduced T regulatory cells by ~72%. In a murine model of 4T1 breast cancer bone metastasis, LipoNCs@pGSDMB treatment achieved 94% tumor suppression and complete neuropathic pain relief through the synergy of immunomodulation and neuroregulation.
Nanomaterials synergizing with checkpoint blockage
Combining nanomaterials for immunomodulator delivery with immune checkpoint blockade can synergistically improve treatment effectiveness[97-99]. Assisted by ultrasound (US)-induced oxidative forces, a polypeptide-based sono-adjuvant library with customizable properties was developed for targeted delivery to lymph nodes[100]. The sono-adjuvant-based vaccine with US stimulation (SONV+US) markedly prolonged the median survival, and frequencies of matured DCs and CD8+ T cells were much higher than those of conventional adjuvant-based vaccines. When combined with αPD-L1, neoantigen-based sonosensitive vaccine (Ne-SONV) boosted IFN-γ+CD8+ and TNF-α+CD8+ T cell populations by 5.8- and 4.0-fold relative to the admixture of neoantigen peptide with CpG adjuvant. This sono-adjuvant platform achieved 100% survival in orthotopic HCC models, exhibiting great potential for personalized cancer vaccination.
In clinical settings, chemotherapy remains a mainstream approach in the management of malignant tumors. Recently, the in-depth integration of nanotechnology and AI has emerged as a core driving force that accelerates the development of integrated and personalized therapeutic strategies for oral cancer[101]. By using machine learning, a clinically approved alkylating agent at a tolerable dose was employed to promote tumor-derived double-stranded DNA production, intratumoral accumulation, and optimize its availability[102]. The AI-assisted immune nanoenhancer platform, utilizing low-dose alkylating agents, achieved 97.64% tumor growth inhibition and 89.66% reduction in pulmonary metastases. This STING activation mechanism markedly increased the proportions of splenic effector memory T cells and central memory T cells to 17.20% and 39.16%, respectively. For AI-assisted nanomedicine design, this work establishes a versatile paradigm by employing machine learning algorithms to predict chemotherapeutic encapsulation and suitable dosage for advanced immunotherapy.
Nanomaterials synergizing with CAR-T
Emerging nanomaterial-assisted immunomodulation and therapeutic optimization have been ingeniously integrated with engineered cellular therapies to enhance antitumor effectiveness and translational potential[103]. The effectiveness of CAR-T in hematologic malignancy is impressive, yet the application in solid tumors is severely constrained[104]. For glioblastoma treatment, tumor-responsive cellular formulations were prepared by conjugating pexidartinib-containing liposomes (PLX-Lip) to the engineered CAR-T cells[105]. The formed hypoxia-responsive moiety azobenzene (AZO) modified PLX-Lip/AZO-T cells could actively transmigrate across the blood-brain barrier, which was 5.8-fold higher than that of non-integrated PLX-Lip/T cells. In the hypoxic microenvironment, over 70% of PLX-Lip were detached from CAR-T cells at 12 h post-infiltration, and the uptake of PLX-Lip by tumor-associated macrophages (TAMs) and microglia was 4.7-fold higher than the control group. In orthotopic mouse models, PLX-Lip/AZO-T cell treatment achieved complete tumor eradication in 60% of mice, with 80% of treated mice surviving over 50 days. Importantly, this combined therapy demonstrated good safety profiles without significant organ toxicity or cytokine release syndrome, underscoring its strong potential for clinical application.
CONCLUSION
Nanomaterial-enabled combinational immunotherapies can effectively enhance the delivery of immunomodulatory agents, target immune cells, modulate the TME, and overcome tumor immune evasion. Specifically, nanoplatforms have shown remarkable efficacy in amplifying DNA damage, inducing ICD, activating the STING pathway, and synergizing with various immunotherapeutic modalities to suppress tumor growth and metastasis. Additionally, the integration of nanotechnology with AI has provided a novel paradigm for personalized therapeutic design, optimizing drug delivery and dosage to further improve treatment outcomes. However, current challenges still hinder the practical translation of these nanomaterial-driven immunotherapies into clinical applications.
Despite significant advancements in the field of smart nanomaterial-driven combinatorial immunotherapy, several critical challenges continue to impede its clinical translation. Firstly, the scalable synthesis of these materials with consistent batch-to-batch reproducibility remains a significant bottleneck, as standardized manufacturing protocols and quality control systems for combinatorial regimens are still underdeveloped. Moreover, the heterogeneous nature of the TME results in variable therapeutic responses, with inadequate tumor selectivity leading to off-target effects. Issues such as nonspecific accumulation of nanocarriers in healthy tissues, uncontrolled co-release of therapeutic payloads, and aberrant immune activation collectively undermine the safety and efficacy of treatments. Besides, therapeutic resistance induced by the TME and the complex interplay between distinct immunotherapeutic modalities present additional obstacles to achieving effective treatment outcomes. Furthermore, the complexities of combination therapies, such as optimizing the ratio of nanomaterials to immunomodulatory agents, coordinating the timing of different therapeutic modalities, and addressing potential drug-drug interactions, add layers of difficulty to clinical implementation. Absence of unified regulatory standards and clinical evaluation protocols specifically designed for nanomaterial-based combined immunotherapies further delays their clinical approval and commercialization. A comprehensive understanding of metastatic biology, key signaling pathways, and emerging therapeutic strategies is thus critical to improving the diagnosis and treatment of metastatic malignancies.
Future directions should focus on addressing these existing challenges to bridge the gap between laboratory successes and clinical applications. Refining nanoplatform engineering with AI will be crucial for improving targeting precision, minimizing adverse effects, and optimizing therapeutic efficacy, as AI-driven design can predict nanomaterial behavior and tailor formulations to individual patients. Establishing standardized manufacturing protocols and quality control systems to ensure reproducibility of combined immunotherapies is necessary to optimize scalable synthesis and ensure consistency. Conducting large-scale animal studies is essential to establish the long-term safety and efficacy of nanomaterial-enabled immunotherapies, strengthening interdisciplinary collaboration between nanotechnology and immunology to optimize synergistic interactions, thereby advancing the clinical translation of combinatorial immunotherapies and improving patient outcomes.
DECLARATIONS
Authors’ contributions
Conceptualization and writing: Wang W
Figure preparation: Yan Z
Data collection: Yan K
Coordination and revision: Lai Y
Proofreading and supervision: Xu W
Coordination, revision and editing: Zhou M
Conceptualization, revision, supervision, and funding acquisition: Yu H
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool Home for researchers (released 2026-03-29) was used solely for language editing. The tool did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (U22A20328 to Yu H); Science and Technology Commission of Shanghai Municipality (23ZR1475000 and 20430711800 to Yu H.).
Conflicts of interest
Wang W, Lai Y, Xu W and Yu H are the Guest Editors of the Special Issue “Smart Nanomaterials for Cancer Immunotherapy” of Journal of Cancer Metastasis and Treatment. Yu H is an Associate Editor of Journal of Cancer Metastasis and Treatment. They were not involved in any steps of the editorial process, notably including reviewers’ selection, manuscript handling, or decision-making. The other authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication.
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Shi X, Wang X, Yao W, et al. Mechanism insights and therapeutic intervention of tumor metastasis: latest developments and perspectives. Sig Transduct Target Ther. 2024;9:192.
2. Pallares RM, Consolino L, Wang A, et al. Targeting metastasis with nanomedicine. Nat Rev Bioeng. 2025;4:47-66.
3. Yi M, Li T, Niu M, et al. Exploiting innate immunity for cancer immunotherapy. Mol Cancer. 2023;22:187.
4. Hayday A, Dechanet-merville J, Rossjohn J, Silva-santos B. Cancer immunotherapy by γδ T cells. Science. 2024;386:eabq7248.
5. Geng Q, Xu J, Du C, et al. Small molecules targeting immune checkpoint proteins for cancer immunotherapy: a patent and literature review (2020-2024). Expert Opin Ther Pat. 2025;35:409-40.
6. Yang K, Halima A, Chan TA. Antigen presentation in cancer - mechanisms and clinical implications for immunotherapy. Nat Rev Clin Oncol. 2023;20:604-23.
7. Borgers JSW, Heimovaara JH, Cardonick E, et al. Immunotherapy for cancer treatment during pregnancy. Lancet Oncol. 2021;22:e550-61.
8. Heras-murillo I, Adán-barrientos I, Galán M, Wculek SK, Sancho D. Dendritic cells as orchestrators of anticancer immunity and immunotherapy. Nat Rev Clin Oncol. 2024;21:257-77.
9. Yap TA, Parkes EE, Peng W, Moyers JT, Curran MA, Tawbi HA. Development of immunotherapy combination strategies in cancer. Cancer Discovery. 2021;11:1368-97.
10. Oliveira G, Wu CJ. Dynamics and specificities of T cells in cancer immunotherapy. Nat Rev Cancer. 2023;23:295-316.
11. Ye F, Dewanjee S, Li Y, et al. Advancements in clinical aspects of targeted therapy and immunotherapy in breast cancer. Mol Cancer. 2023;22:105.
12. Yang Z, Liu X, Zhu J, et al. Inhibiting intracellular CD28 in cancer cells enhances antitumor immunity and overcomes anti-PD-1 resistance via targeting PD-L1. Cancer Cell. 2025;43:86-102.e10.
13. Chen Z, Han F, Du Y, Shi H, Zhou W. Hypoxic microenvironment in cancer: molecular mechanisms and therapeutic interventions. Sig Transduct Target Ther. 2023;8:70.
14. Ghorani E, Swanton C, Quezada SA. Cancer cell-intrinsic mechanisms driving acquired immune tolerance. Immunity. 2023;56:2270-95.
15. Huppert LA, Green MD, Kim L, et al. Tissue-specific Tregs in cancer metastasis: opportunities for precision immunotherapy. Cell Mol Immunol. 2021;19:33-45.
16. Wood H, Acharjee A, Pearce H, et al. Breastfeeding promotes early neonatal regulatory T‐cell expansion and immune tolerance of non‐inherited maternal antigens. Allergy. 2021;76:2447-60.
17. Chen DS, Mellman I. Oncology meets immunology: the cancer-immunity cycle. Immunity. 2013;39:1-10.
18. Ma J, Hua L, Zhu Y, Mao G, Fu C, Qin S. Photo-thermally controllable tumor metabolic modulation to assist T cell activation for boosting immunotherapy. Int J Nanomedicine. 2024;19:11181-94.
19. Sharma P, Zhang X, Ly K, et al. Hyperglycosylation of prosaposin in tumor dendritic cells drives immune escape. Science. 2024;383:190-200.
20. Galassi C, Chan TA, Vitale I, Galluzzi L. The hallmarks of cancer immune evasion. Cancer Cell. 2024;42:1825-63.
21. Gao J, Wang W, Pei Q, Lord MS, Yu H. Engineering nanomedicines through boosting immunogenic cell death for improved cancer immunotherapy. Acta Pharmacol Sin. 2020;41:986-94.
22. Li D, Liu S, Ma Y, Liu S, Liu Y, Ding J. Biomaterials that induce immunogenic cell death. Small Methods. 2023;7:2300204.
23. Lynch C, Pitroda SP, Weichselbaum RR. Radiotherapy, immunity, and immune checkpoint inhibitors. Lancet Oncol. 2024;25:e352-62.
24. Galluzzi L, Guilbaud E, Schmidt D, Kroemer G, Marincola FM. Targeting immunogenic cell stress and death for cancer therapy. Nat Rev Drug Discovery. 2024;23:445-60.
25. Lauss M, Phung B, Borch TH, et al. Molecular patterns of resistance to immune checkpoint blockade in melanoma. Nat Commun. 2024;15:3075.
26. Giles JR, Globig A, Kaech SM, Wherry EJ. CD8+ T cells in the cancer-immunity cycle. Immunity. 2023;56:2231-53.
27. Van Elsas MJ, Middelburg J, Labrie C, et al. Immunotherapy-activated T cells recruit and skew late-stage activated M1-like macrophages that are critical for therapeutic efficacy. Cancer Cell. 2024;42:1032-1050.e10.
28. Mellman I, Chen DS, Powles T, Turley SJ. The cancer-immunity cycle: indication, genotype, and immunotype. Immunity. 2023;56:2188-205.
29. Xia D, Zhang X, Hao H, et al. Strategies to prolong drug retention in solid tumors by aggregating Endo-CMC nanoparticles. J Control Release. 2023;360:705-17.
30. Luo X, Huang W, Li S, et al. SOX12 facilitates hepatocellular carcinoma progression and metastasis through promoting regulatory T‐cells infiltration and immunosuppression. Adv Sci. 2024;11:2310304.
31. Chen J, Feng W, Sun M, et al. TGF-β1-induced SOX18 elevation promotes hepatocellular carcinoma progression and metastasis through transcriptionally upregulating PD-L1 and CXCL12. Gastroenterology. 2024;167:264-80.
32. Zhao Y, Pei L, Liu B, et al. Macrophage membrane-coated nanomedicine enhances cancer immunotherapy by activating macrophages and T cells. Mol Pharm. 2025;22:2402-12.
33. Chung JB, Brudno JN, Borie D, Kochenderfer JN. Chimeric antigen receptor T cell therapy for autoimmune disease. Nat Rev Immunol. 2024;24:830-45.
34. Pang Z, Lu M, Zhang Y, et al. Neoantigen-targeted TCR-engineered T cell immunotherapy: current advances and challenges. Biomark Res. 2023;11:104.
35. Zhang L, Lin H, Liang J, et al. Programmed death-ligand 1 regulates ameloblastoma growth and recurrence. Int J Oral Sci. 2025;17:29.
36. Fan Z, Wu C, Chen M, et al. The generation of PD-L1 and PD-L2 in cancer cells: from nuclear chromatin reorganization to extracellular presentation. Acta Pharm Sin B. 2022;12:1041-53.
37. Ma K, Wang L, Li W, et al. Turning cold into hot: emerging strategies to fire up the tumor microenvironment. Trends Cancer. 2025;11:117-34.
38. Peng S, Xiao F, Chen M, Gao H. Tumor‐microenvironment‐responsive nanomedicine for enhanced cancer immunotherapy. Adv Sci. 2021;9:2103836.
39. Yi M, Zheng X, Niu M, Zhu S, Ge H, Wu K. Combination strategies with PD-1/PD-L1 blockade: current advances and future directions. Mol Cancer. 2022;21:28.
40. Goswami S, Pauken KE, Wang L, Sharma P. Next-generation combination approaches for immune checkpoint therapy. Nat Immunol. 2024;25:2186-99.
41. Song L, Hao Y, Wang C, et al. Liposomal oxaliplatin prodrugs loaded with metformin potentiate immunotherapy for colorectal cancer. J Control Release. 2022;350:922-32.
42. Fang M, Zheng J, Song Q, et al. Breaking apoptosis‐induced immune silence: ultrasound‐activated nano‐oncolytic therapy reinvigorates antitumor immunity. Adv Mater. 2025;37:2508681.
43. Yuan Y, Zhuoga C, Zeng C, et al. Advances in nanobody-based platforms for precision cancer diagnosis and therapy. Polym Sci Technol. 2025;1:692-715.
44. Karaosmanoglu S, Zhou M, Shi B, Zhang X, Williams GR, Chen X. Carrier-free nanodrugs for safe and effective cancer treatment. J Control Release. 2021;329:805-32.
45. Ma X, Zhang MJ, Wang J, et al. Emerging biomaterials imaging antitumor immune response. Adv Mater. 2022;34:2204034.
46. Yin Y, Chen Z, Yang C, et al. Spatiotemporally controlled Co-treatment of lung cancer and pulmonary immune-related adverse events by inhalable albumin-chaperoned ultrathin MnO2 nanosheets. ACS Nano. 2025;19:19730-43.
47. Fang H, You P, Lin S, et al. Annexin A1 mRNA-loaded liposomes alleviate acute pancreatitis by suppressing STING pathway and promoting efferocytosis in macrophages. Nat Nanotechnol. 2025;20:1514-25.
48. Kim M, Lee JS, Kim W, et al. Aptamer-conjugated nano-liposome for immunogenic chemotherapy with reversal of immunosuppression. J Control Release. 2022;348:893-910.
49. Jia W, Wu Y, Xie Y, Yu M, Chen Y. Advanced polymeric nanoparticles for cancer immunotherapy: materials engineering, immunotherapeutic mechanism and clinical translation. Adv Mater. 2025;37:2413603.
50. Meng Q, Ding B, Ma P, Lin J. Inorganic nanobiomaterials boost tumor immunotherapy: strategies and applications. Acc Chem Res. 2025;58:1210-23.
51. Liu J, Ren L, Li S, et al. The biology, function, and applications of exosomes in cancer. Acta Pharm Sin B. 2021;11:2783-97.
52. Albertsen C, Kulkarni JA, Witzigmann D, Lind M, Petersson K, Simonsen JB. The role of lipid components in lipid nanoparticles for vaccines and gene therapy. Adv Drug Delivery Rev. 2022;188:114416.
53. Bauman J, Burris H, Clarke J, et al. 798 Safety, tolerability, and immunogenicity of mRNA-4157 in combination with pembrolizumab in subjects with unresectable solid tumors (KEYNOTE-603): an update. J Immunother Cancer. 2020;8:A1-A559.
54. Lopez J, Powles T, Braiteh F, et al. Autogene cevumeran with or without atezolizumab in advanced solid tumors: a phase 1 trial. Nat Med. 2025;31:152-64.
55. Falchook GS, Reeves J, Gandhi S, et al. A phase 2 study of AZD4635 in combination with durvalumab or oleclumab in patients with metastatic castration-resistant prostate cancer. Cancer Immunol Immunother. 2024;73:72.
56. Su Z, Xu D, Hu X, et al. Biodegradable oxygen-evolving metalloantibiotics for spatiotemporal sono-metalloimmunotherapy against orthopaedic biofilm infections. Nat Commun. 2024;15:8058.
57. Zhu Y, Chen P, Hu B, et al. MDSC-targeting gold nanoparticles enhance PD-1 tumor immunotherapy by inhibiting NLRP3 inflammasomes. Biomaterials. 2024;307:122533.
58. Hollingsworth D, Srivastava S, Akter S, et al. Colostrum extracellular vesicles are neuroprotective in models of Parkinson’s disease. Theranostics. 2026;16:4190-223.
59. Wang F, Yin L, Hu Y. Progress of extracellular vesicles-based system for tumor therapy. J Control Release. 2025;381:113570.
60. Zhao L, Gu C, Gan Y, Shao L, Chen H, Zhu H. Exosome-mediated siRNA delivery to suppress postoperative breast cancer metastasis. J Control Release. 2020;318:1-15.
61. Li Y, Liu F, Cai Q, et al. Invasion and metastasis in cancer: molecular insights and therapeutic targets. Sig Transduct Target Ther. 2025;10:57.
62. Zhang J, Liu J, Wang Y, et al. Macrophage hitchhiking for smart nanomedicine: bridging immune navigation and targeted therapy. J Control Release. 2025;386:114155.
63. Luo J, Chen J, Zhou J, et al. Harnessing smart nanomaterials to reprogram neutrophil plasticity in immune modulation. J Nanobiotechnol. 2025;23:666.
64. Zhang X, Cai H, Peng W, et al. Lenvatinib plus transarterial chemoembolization and PD-1 inhibitors as conversion therapies for unresectable intermediate-advanced hepatocellular carcinoma: a phase 2 trial and exploratory biomolecular study. Sig Transduct Target Ther. 2026;11:37.
65. Mo H, Yu Y, Sun X, et al. Metronomic chemotherapy plus anti-PD-1 in metastatic breast cancer: a Bayesian adaptive randomized phase 2 trial. Nat Med. 2024;30:2528-39.
66. Wang Y, Dong A, Man J, et al. TREM2 scFv‐engineering escherichia coli displaying modulation of macrophages to boost cancer radio‐immunotherapy. Adv Mater. 2025;37:2417920.
67. Zhang Z, Zhang X, Chen D. Using triple radio-immunotherapy to overcome cancer immunotherapy resistance. Cancer Biol Med. 2023;20:788.
68. Huang J, Theelen WSME, Belcaid Z, et al. Combination of pembrolizumab and radiotherapy induces systemic antitumor immune responses in immunologically cold non-small cell lung cancer. Nat Cancer. 2025;6:1676-92.
69. Gu C, Wang X, Wang K, et al. Cryoablation triggers type I interferon-dependent antitumor immunity and potentiates immunotherapy efficacy in lung cancer. J Immunother Cancer. 2024;12:e008386.
70. Gu S, Luo Q, Zhang Y, et al. Cryoablation plus sintilimab and lenvatinib in advanced or metastatic intrahepatic cholangiocarcinoma: a phase 2 trial. Nat Cancer. 2025;7:60-79.
71. Hajjar R, Mars RAT, Kashyap PC. Harnessing the microbiome for cancer therapy. Nat Rev Microbiol. ;2026:1268.
72. Porcari S, Ciccarese C, Heidrich V, et al. Fecal microbiota transplantation plus pembrolizumab and axitinib in metastatic renal cell carcinoma: the randomized phase 2 TACITO trial. Nat Med. 2026;32:1316-24.
73. Sun Y, Yang H, Xu Q, et al. Anti‐PD‐1 nanobody‐armored MSLN CAR‐T Therapy for malignant mesothelioma: preclinical and clinical studies. Adv Sci. ;2025:e08754.
74. Wang Y, Jia J, Wang F, et al. Pre-metastatic niche: formation, characteristics and therapeutic implication. Sig Transduct Target Ther. 2024;9:236.
75. Cambria E, Coughlin MF, Floryan MA, Offeddu GS, Shelton SE, Kamm RD. Linking cell mechanical memory and cancer metastasis. Nat Rev Cancer. 2024;24:216-28.
76. Cooper AJ, Sequist LV, Lin JJ. Third-generation EGFR and ALK inhibitors: mechanisms of resistance and management. Nat Rev Clin Oncol. 2022;19:499-514.
77. Liu H, Zhao H, Sun Y. Tumor microenvironment and cellular senescence: understanding therapeutic resistance and harnessing strategies. Semin Cancer Biol. 2022;86:769-81.
78. Jin Y, Huang Y, Ren H, et al. Nano-enhanced immunotherapy: Targeting the immunosuppressive tumor microenvironment. Biomaterials. 2024;305:122463.
79. Fan D, Cao Y, Cao M, Wang Y, Cao Y, Gong T. Nanomedicine in cancer therapy. Sig Transduct Target Ther. 2023;8:293.
80. Maksymova L, Pilger YA, Nuhn L, Van Ginderachter JA. Nanobodies targeting the tumor microenvironment and their formulation as nanomedicines. Mol Cancer. 2025;24:65.
81. Liu N, Wang X, Wang Z, et al. Nanomaterials-driven in situ vaccination: a novel frontier in tumor immunotherapy. J Hematol Oncol. 2025;18:45.
82. Yu M, Yang W, Yue W, Chen Y. Targeted cancer immunotherapy: nanoformulation engineering and clinical translation. Adv Sci. 2022;9:2204335.
83. Sun S, Yu M, Yu L, et al. Nrf2 silencing amplifies DNA photooxidative damage to activate the STING pathway for synergistic tumor immunotherapy. Biomaterials. 2023;296:122068.
84. Lei L, Dong Z, Xu L, et al. Metal-fluorouracil networks with disruption of mitochondrion enhanced ferroptosis for synergistic immune activation. Theranostics. 2022;12:6207-22.
85. He S, Huang Y, Liu J, et al. A Metformin-based multifunctional nanoplatform as a DNA damage amplifier for maximized radio-immunotherapy to overcome radiotherapy resistance. ACS Nano. 2025;19:14848-64.
86. Meng M, Wu J, Guo X, et al. An injectable photothermal-fusing hydrogel: achieving temperature-controllable mild photothermal therapy to reverse chemotherapy-induced immune tolerance. Nano Lett. 2024;24:13044-53.
87. Zhou F, Gao J, Tang Y, et al. Engineering chameleon prodrug nanovesicles to increase antigen presentation and inhibit PD‐L1 expression for circumventing immune resistance of cancer. Adv Mater. 2021;33:2102668.
88. Dang Y, Hou B, Pan J, et al. In Situ Transformable photothermal targeting chimeras for spatiotemporally controllable protein degradation and combination immunotherapy for cancer. J Am Chem Soc. 2026;148:2388-404.
89. Hu B, Stewart W, Chen Q, et al. Modulating tumor collagen fiber alignment for enhanced lung cancer immunotherapy via inhaled RNA. Nat Commun. 2025;16:8120.
90. Kong X, Xie X, Wu J, et al. Combating cancer immunotherapy resistance: a nano‐medicine perspective. Cancer Commun. 2025;45:813-40.
91. Hao X, Shen Y, Liu J, et al. Solid tumour-induced systemic immunosuppression involves dichotomous myeloid-B cell interactions. Nat Cell Biol. 2024;26:1971-83.
92. Lan J, Li H, Xue J, Duan Y, Sun J, Niu M. The immune microenvironment in liver cancer: from analysis to targeting. Adv Sci. 2025;12:e18487.
93. Wu B, Wang Z, Liu J, et al. Dual rectification of metabolism abnormality in pancreatic cancer by a programmed nanomedicine. Nat Commun. 2024;15:10526.
94. Pu T, Sun J, Ren G, Li H. Neuro-immune crosstalk in cancer: mechanisms and therapeutic implications. Sig Transduct Target Ther. 2025;10:176.
95. Amit M, Eichwald T, Roger A, et al. Neuro-immune cross-talk in cancer. Nat Rev Cancer. 2025;25:573-89.
96. Zhang Z, Chen P, Zheng Y, et al. Oxidation-activated nanotherapy boosts tumor immunity and disrupts tumor-nerve crosstalk to combat bone metastases and cancer pain. Sci. Adv. 2026;12:eady1292.
97. Masoumi E, Tahaghoghi-hajghorbani S, Jafarzadeh L, Sanaei M, Pourbagheri-sigaroodi A, Bashash D. The application of immune checkpoint blockade in breast cancer and the emerging role of nanoparticle. J Control Release. 2021;340:168-87.
98. Hunter TL, Bao Y, Zhang Y, et al. In vivo CAR T cell generation to treat cancer and autoimmune disease. Science. 2025;388:1311-7.
99. Tao Z, Kuai X, Wang G, et al. Combination of chemotherapy and immune checkpoint therapy by the immunoconjugates-based nanocomplexes synergistically improves therapeutic efficacy in SCLC. Drug Delivery. 2022;29:1571-81.
100. Chen F, Zhang H, Li S, et al. Polypeptide sono-adjuvant for ultrasound-activatable regulation of innate immunity and cancer vaccination therapy. Nat Commun. 2025;17:272.
101. Tian W, Xie Y, Su H, Zheng C, Li M. Explainable artificial intelligence-driven salivary exosome spectroscopic profiling for clinical diagnosis and metastasis detection of oral cancer. Nano Lett. 2026;26:2279-88.
102. Liu Y, Li Y, Gong W, et al. An immune nanoenhancer revitalizes chemotherapeutics to tailor tumor‐derived dsDNA for anticancer immunoengineering. Adv Mater. 2025;38:e16524.
103. Pan Y, Cheng J, Zhu Y, Zhang J, Fan W, Chen X. Immunological nanomaterials to combat cancer metastasis. Chem Soc Rev. 2024;53:6399-444.
104. Du B, Qin J, Lin B, Zhang J, Li D, Liu M. CAR-T therapy in solid tumors. Cancer Cell. 2025;43:665-79.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.