Bioaccumulation- and exposure-informed environmental thresholds for antibiotics within a One Health framework
Abstract
Antibiotics are widely detected in aquatic environments; however, ecological water quality criterias (WQCs) and human health-based limits, such as maximum residue limits, are derived under separate regulatory frameworks, potentially leaving gaps in achieving integrated One Health protection. Here, we propose a complementary approach that integrates ecological processes, including bioaccumulation and trophic transfer, with food safety-based human exposure limits to evaluate health-protective environmental thresholds. Using trimethoprim as a proof of concept, we illustrate how food safety limits for aquatic products can be translated into organism-level thresholds and further linked to water-phase concentrations using reported bioaccumulation factors. The resulting threshold range is substantially lower than the conventional species sensitivity distribution-derived WQC, suggesting that ecotoxicity-based criteria alone may underestimate risks associated with human dietary exposure. Rather than replacing existing WQCs, this framework provides an additional line of evidence for comparing thresholds and identifying the more protective values. By explicitly linking environmental contamination, ecological processes, and human dietary exposure, this approach offers a practical pathway for operationalizing One Health principles in environmental risk management.
Keywords
Antibiotics are among the most widely prescribed and consumed pharmaceuticals worldwide, and their continuous discharge into aquatic environments via wastewater effluent, agricultural runoff, and aquaculture has raised growing ecological and public health concerns[1,2]. From a One Health perspective, regulating antibiotics in the environment is primarily motivated by the need to minimize the emergence and dissemination of antibiotic resistance genes (ARGs)[3], which the World Health Organization (WHO) recognizes as a major global health threat[4]. The One Health framework emphasizes the interconnectedness of environmental, animal, and human health systems and advocates coordinated, cross-sectoral risk management rather than isolated regulatory decision-making within a single domain.
However, quantitative dose-response relationships between environmental antibiotic concentrations and ARG selection remain poorly characterized, limiting the scientific basis for establishing precise regulatory thresholds. In the absence of robust evidence, current control strategies generally follow the conventional chemical pollutant paradigm, relying primarily on ecotoxicological endpoints. Although effective for protecting aquatic ecosystems, this approach often insufficiently accounts for human exposure and health implications, thereby falling short of the integrated protection envisioned under the One Health framework.
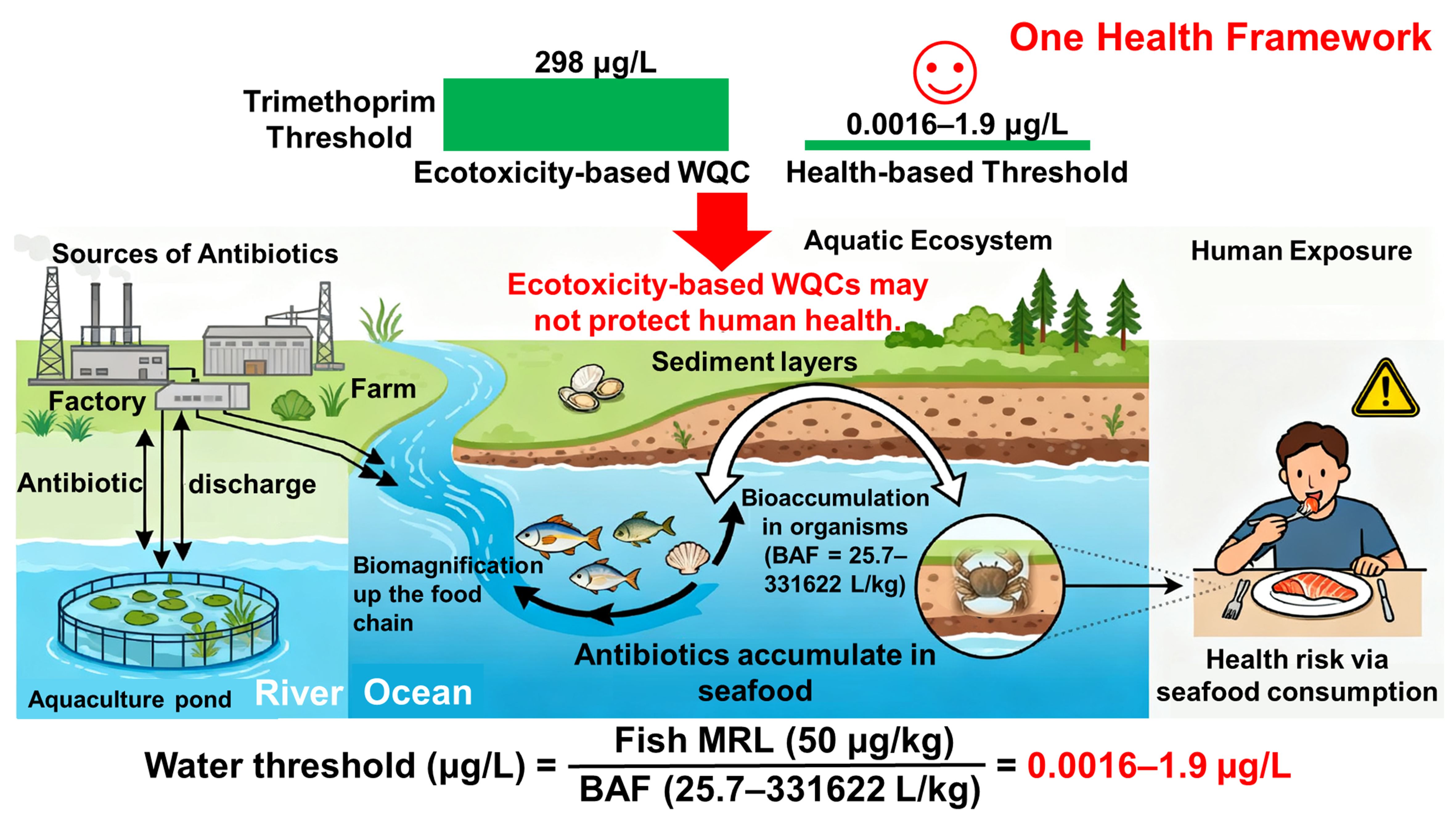
Trimethoprim provides a representative example. This extensively used synthetic antibiotic[5] has water quality criteria (WQC) set at 4,672 µg/L for acute toxicity and 298 µg/L for chronic toxicity, based on species sensitivity distribution (SSD) modeling[6]. These WQCs are designed to protect aquatic organisms from direct toxic effects[7]. Implicitly, this ecotoxicity-centric approach assumes that protecting aquatic life will indirectly protect human health. However, this assumption may not hold for bioaccumulative substances, for which dietary intake constitutes the dominant exposure pathway.
In parallel, food safety regulators have long recognized the human health implications of dietary antibiotic exposure[8]. Authorities such as China’s Ministry of Agriculture and Rural Affairs have established acceptable daily intakes (ADIs) and maximum residue limits (MRLs) for antibiotics in animal-derived foods, including fish and shellfish. Most ADIs and MRLs are derived from toxicological studies conducted primarily in mammals. For trimethoprim, the MRL for fish is set at 50 µg/kg[9]. This value was derived by integrating Chinese dietary consumption data and body weight assumptions with an ADI based on microbiological studies that explicitly account for the potential selection of resistant bacteria in the human gut microbiota under in vitro conditions[10]. These food safety standards are intended to ensure that lifelong dietary exposure remains within acceptable bounds. However, they are rarely connected to water or sediment quality standards, resulting in parallel yet disconnected regulatory frameworks. This lack of integration could leave protection gaps, particularly in regions characterized by high seafood consumption and persistent environmental contamination.
Here, we argue that these two frameworks should be jointly considered to provide a more comprehensive and One Health-protective basis for antibiotic regulation. Importantly, we do not propose that a single numerical threshold should universally apply to all biological receptors. Rather, consistent with the One Health definition, we advocate coordinated evaluation across regulatory domains to ensure that protection afforded to one receptor group does not inadvertently leave another insufficiently protected.
Field studies in coastal waters of China have detected measurable levels of trimethoprim in fish muscle tissue, demonstrating that aquatic organisms can serve as vectors for human exposure[11]. Meanwhile, MRLs are commonly established for fishery products, bioaccumulation data are relatively abundant, and fish consumption constitutes a major dietary exposure route for many chemicals. By combining the fish MRL
Figure 1. Bioaccumulation- and exposure-based approaches for deriving environmental thresholds within a One Health framework. Figure created with Microsoft PowerPoint. WQC: Water quality criteria; BAF: bioaccumulation factor; MRL: maximum residue limit.
This discrepancy highlights that ecotoxicity-based WQCs alone may not be sufficiently protective of human health, particularly where seafood consumption represents a major exposure pathway[15]. Reverse-derivation approaches enable incorporation of real-world bioaccumulation and biomagnification processes, which are rarely captured in laboratory toxicity tests but can substantially amplify human exposure risks. Similar evidence has been reported for other antibiotic classes, such as tetracyclines and sulfonamides, which accumulate in benthic organisms and biomagnify through coastal and estuarine food webs[16].
Future applications of this framework could systematically extend this back-calculation approach to additional antibiotic classes by integrating compound-specific ADIs or MRLs, experimentally or field-derived BAFs, and population-specific exposure parameters. Particular attention should be given to antibiotics exhibiting persistence, trophic transfer potential, or high usage volumes, as these characteristics may increase the likelihood of dietary exposure relevance. Such an extension would facilitate cross-class comparisons and help identify compounds for which ecotoxicity-based thresholds diverge substantially from human health-informed benchmarks.
It should also be acknowledged that, for some chemicals, ecotoxicity-based criteria may be more stringent than thresholds derived from human exposure considerations. In such cases, ecotoxicity-based limits may already provide sufficient protection for human health, even though they are not explicitly derived from exposure assessment. This outcome is fully consistent with the One Health objective of achieving simultaneous protection of ecosystems and human populations. Accordingly, this commentary does not advocate replacing ecotoxicological approaches but rather emphasizes the importance of evaluating environmental thresholds using both ecological and human health evidence. In this context, One Health functions as an integrative decision-support framework rather than a mandate for numerical uniformity across species with inherently different toxicological sensitivities. Where multiple thresholds are available, adopting the more protective value represents a pragmatic strategy for integrated protection.
The trimethoprim example is intended solely to demonstrate the feasibility and relevance of the proposed framework, not to serve as a definitive regulatory recommendation. Formal derivation would require broader datasets to characterize uncertainty and sensitivity, including regional variability in dietary consumption patterns, population characteristics, inter-species differences in accumulation capacity, variability in BAFs across environmental conditions, and inconsistencies among bioaccumulation indicators across regulatory contexts[17,18]. Incorporating these uncertainty and sensitivity analyses would enable the derived thresholds to encompass the majority of realistic exposure scenarios and population groups, thereby enhancing their protective robustness.
This integrated approach offers several advantages. First, it grounds environmental risk assessment in real-world exposure scenarios by explicitly considering the food-web transfer. Second, it enables regional refinement through incorporation of local bioaccumulation data, site-specific contamination profiles, and population-specific dietary habits, particularly where national-level criteria may inadequately capture local exposure risks. Third, it provides a pragmatic solution for linking ecological and human health protection while scientific understanding of exposure-ARG dose-response relationships continues to evolve. Because the ultimate purpose of environmental antibiotics regulation is to minimize the emergence and spread of ARGs, translating microbiologically derived ADIs or MRLs into water and sediment thresholds provides a direct and health-protective strategy aligned with the core driver of antibiotic risk management.
Beyond antibiotics, this conceptual framework has broader applicability. Many emerging contaminants share characteristics of persistence, bioaccumulation, and dietary exposure relevance[19,20]. For example, perfluorooctane sulfonic acid (PFOS) and perfluorooctanoic acid (PFOA), two representative per- and polyfluoroalkyl substances (PFAS), exhibit reported log BAFs of 3.55 ± 0.83 and 2.16 ± 0.85, respectively, in aquatic organisms[21]. Given that human exposure to PFAS is dominated by fish and seafood consumption[22], ADI/MRL-based calculation combined with bioaccumulation and trophic magnification data is also applicable to derive thresholds. A similar exposure-dominant pattern exists for methylmercury, for which fish consumption represents the primary pathway for human exposure[23]. These examples illustrate how alignment between environmental regulation and food safety policies can reduce regulatory blind spots and facilitate holistic risk management, consistent with the core One Health principle of harmonizing sector-specific standards while accounting for receptor-specific toxicological differences.
We therefore encourage regulators, researchers, and policymakers to consider bioaccumulation- and exposure-informed approaches as a complementary line of evidence when developing or revising water and sediment quality thresholds for antibiotics and other bioaccumulative contaminants within a One Health framework. By explicitly linking environmental contamination, ecological processes, and human dietary exposure, this evaluative strategy provides a practical pathway to operationalize the One Health concept in environmental standard setting. In practice, ecotoxicity-based and exposure-informed thresholds can be compared, and the more protective value adopted to ensure integrated protection of both ecosystems and human populations.
DECLARATIONS
Authors’ contributions
Formal analysis, writing - original draft, writing - review and editing, and funding acquisition: Xu, Y. Q.
Conceptualization, supervision, writing - original draft, writing - review and editing, and funding acquisition: Lin, N.
Investigation, formal analysis, and writing - review and editing: Zhang, X.
Writing - review and editing, and funding acquisition: Huang, Y.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Youth Key R&D Program of China (2022YFC3105800) and the Strategic Priority Research Program of the Chinese Academy of Sciences (XDB0750200).
Conflicts of interest
Lin, N. is an Editorial Board Member of the Journal of Environmental Exposure Assessment. Xu, Y. Q. is a Youth Editorial Board Member of the Journal of Environmental Exposure Assessment. They had no involvement in the review or editorial process of this manuscript, including but not limited to reviewer selection, evaluation, or the final decision, while the other authors have declared that they have no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Li, S.; Zhang, Q.; Chen, M.; et al. Antibiotics sources, concentrations, and risk of antimicrobial resistance selection in the Three Gorges Reservoir Basin, China. Environ. Sci. Technol. 2025, 59, 19398-413.
2. OECD. Pharmaceutical residues in freshwater: hazards and policy responses. 2019. https://www.oecd.org/content/dam/oecd/en/publications/reports/2019/11/pharmaceutical-residues-in-freshwater_33f873ac/c936f42d-en.pdf. (accessed 2026-03-17).
3. Lund, D.; Parras-Moltó, M.; Inda-Díaz, J. S.; et al. Genetic compatibility and ecological connectivity drive the dissemination of antibiotic resistance genes. Nat. Commun. 2025, 16, 2595.
4. WHO. Antimicrobial resistance. 2024. https://www.who.int/europe/news-room/fact-sheets/item/antimicrobial-resistance. (accessed 2026-03-17).
5. Afroze, S.; Faisal, M.; Khan, M. N. A.; Barua, H. A comprehensive review of antibiotics and antimicrobial resistance in the aquaculture sector of the world and Bangladesh. Int. J. Microbiol. 2025, 2025, 8818516.
6. Zhao, W.; Wang, B.; Wang, Y.; Deng, S.; Huang, J.; Yu, G. Deriving acute and chronic predicted no effect concentrations of pharmaceuticals and personal care products based on species sensitivity distributions. Ecotoxicol. Environ. Saf. 2017, 144, 537-42.
7. Garner, K. L.; Suh, S.; Lenihan, H. S.; Keller, A. A. Species sensitivity distributions for engineered nanomaterials. Environ. Sci. Technol. 2015, 49, 5753-9.
8. Bacanlı, M. G. The two faces of antibiotics: an overview of the effects of antibiotic residues in foodstuffs. Arch. Toxicol. 2024, 98, 1717-25.
9. Ministry of Agriculture of the People’s Republic of China. GB 31650-2019 National Food Safety Standard - Maximum residue limits for veterinary drugs in foods. 2019. https://food.chemlinked.com/database/view/1661. (accessed 2026-03-17).
10. Webb, S.; Ternes, T.; Gibert, M.; Olejniczak, K. Indirect human exposure to pharmaceuticals via drinking water. Toxicol. Lett. 2003, 142, 157-67.
11. Liu, S.; Zhao, H.; Lehmler, H. J.; Cai, X.; Chen, J. Antibiotic pollution in marine food webs in Laizhou Bay, North China: trophodynamics and human exposure implication. Environ. Sci. Technol. 2017, 51, 2392-400.
12. Hu, T.; Zhang, J.; Xu, X.; et al. Bioaccumulation and trophic transfer of antibiotics in the aquatic and terrestrial food webs of the Yellow River Delta. Chemosphere 2023, 323, 138211.
13. Wu, N. N.; Liu, S.; Xu, R.; et al. New insight into the bioaccumulation and trophic transfer of free and conjugated antibiotics in an estuarine food web based on multimedia fate and model simulation. J. Hazard. Mater. 2024, 465, 133088.
14. Wu, Q.; Pan, C. G.; Wang, Y. H.; Xiao, S. K.; Yu, K. F. Antibiotics in a subtropical food web from the Beibu Gulf, South China: occurrence, bioaccumulation and trophic transfer. Sci. Total. Environ. 2021, 751, 141718.
15. Zhang, H.; Wang, W.; Lin, C.; et al. Decreasing mercury levels in consumer fish over the three decades of increasing mercury emissions in China. Eco. Environ. Health. 2022, 1, 46-52.
16. Li, F.; Bao, Y.; Chen, L.; Su, Z.; Tang, Y.; Wen, D. Screening of priority antibiotics in Chinese seawater based on the persistence, bioaccumulation, toxicity and resistance. Environ. Int. 2023, 179, 108140.
17. Gimeno, S.; Allan, D.; Paul, K.; Remuzat, P.; Collard, M. Are current regulatory log Kow cut-off values fit-for-purpose as a screening tool for bioaccumulation potential in aquatic organisms? Regul. Toxicol. Pharmacol. 2024, 147, 105556.
18. Kuo, D. T. F. Universal bioaccumulation, biomagnification, and toxicokinetic metrics based on equivalent aqueous concentration. Sci. Total. Environ. 2025, 997, 180199.
19. Ackerman Grunfeld, D.; Gilbert, D.; Hou, J.; et al. Underestimated burden of per- and polyfluoroalkyl substances in global surface waters and groundwaters. Nat. Geosci. 2024, 17, 340-6.
20. Tepper, K.; King, J.; Manuneedhi Cholan, P.; et al. Methylmercury demethylation and volatilization by animals expressing microbial enzymes. Nat. Commun. 2025, 16, 1117.
21. Burkhard, L. P. Evaluation of published bioconcentration factor (BCF) and bioaccumulation factor (BAF) data for per- and polyfluoroalkyl substances across aquatic species. Environ. Toxicol. Chem. 2021, 40, 1530-43.
22. Christensen, K. Y.; Raymond, M.; Blackowicz, M.; et al. Perfluoroalkyl substances and fish consumption. Environ. Res. 2017, 154, 145-51.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.