Prenatal exposure to persistent organic pollutants in Wuhan, China: associations with birth outcomes and breastfeeding safety
Abstract
Infants are particularly vulnerable to the adverse effects of persistent organic pollutants (POPs) due to their developing physiological systems and relatively high exposure per unit body weight. This study quantified legacy POPs in breast milk samples collected from 99 lactating mothers in Wuhan, China, and evaluated their associations with neonatal birth outcomes. The predominant organochlorine pesticides (OCPs) detected were 1,2,3,4,5,6-hexachlorocyclohexanes (HCHs) and dichlorodiphenyltrichloroethane (DDT) metabolites. Among polychlorinated biphenyls (PCBs) and polybrominated diphenyl ethers (PBDEs), PCB-52 and BDE-154 were the most abundant congeners. Maternal factors, including parity, body mass index, and dietary patterns, significantly influenced POP concentrations in breast milk. Our results revealed significant associations between specific POPs and birth outcomes. For instance, β-HCH was positively correlated with birth weight, whereas heptachlor exhibited a negative correlation. Additionally, PCB-170 was inversely correlated with infant head circumference, while BDE-28 and BDE-99 showed positive correlations. Our health risk assessment demonstrated that, despite elevated POP levels in a subset of samples, the estimated daily intake for most breastfed infants remained below established safety thresholds. These findings reaffirm that the established nutritional and immunological benefits of breastfeeding far outweigh the potential risks from background POP exposure. Nevertheless, the persistent and widespread detection of these contaminants underscores the need for ongoing biomonitoring and public health interventions to reduce maternal body burdens by mitigating environmental and dietary sources.
Keywords
INTRODUCTION
Persistent organic pollutants (POPs) are anthropogenic chemicals characterized by high toxicity, environmental persistence, lipophilicity, bioaccumulative potential, and the capacity for long-range atmospheric transport. This class includes legacy compounds such as organochlorine pesticides (OCPs), polychlorinated biphenyls (PCBs), and polybrominated diphenyl ethers (PBDEs)[1,2]. Despite global restrictions or bans on many legacy POPs under the Stockholm Convention, their environmental ubiquity and human health relevance persist due to their extraordinary resistance to degradation[3]. These substances continue to cycle through ecosystems and accumulate in food webs, resulting in measurable human exposure decades after their phase-out.
This challenge is further compounded by the ongoing use of chemicals that degrade into regulated POPs or act as inadvertent sources of structurally similar contaminants. While laboratory and bench-scale studies have elucidated the mechanistic pathways involved, such as the formation of unintentionally produced PCBs (UP-PCBs) or polybrominated dibenzo-p-dioxins/furans (PBDD/Fs), field monitoring has confirmed their environmental relevance[4]. However, quantifying the contribution of such precursor-driven pathways to the global burden of legacy POPs remains a critical knowledge gap, hindering accurate risk characterization and regulatory efficacy. Given their endocrine-disrupting, neurotoxic, and immunotoxic properties, POPs pose disproportionate risks during vulnerable life stages, particularly prenatal and early postnatal development[5-7]. Epidemiological evidence links prenatal exposure to adverse birth outcomes and long-term developmental deficits[8-10], underscoring the need for continued surveillance in high-risk populations.
Human breast milk serves a dual role in environmental health research: it is the gold-standard nutritional source for infants and a non-invasive biomonitoring matrix that reflects the integrated maternal body burden of lipophilic contaminants[11,12]. Due to their high lipophilicity and persistence, POPs accumulate in maternal adipose tissue over a lifetime. The concentration of POPs in early postpartum breast milk, particularly in lipid-rich colostrum, therefore represents a practical biomarker that correlates strongly with the maternal body burden during the preceding prenatal period. This is supported by pharmacokinetic rationale and studies demonstrating correlations among levels in maternal serum, breast milk, and cord blood[13-15]. This relationship allows breast milk to serve as both a direct measure of infant dietary exposure and a proxy for historical fetal exposure. Consequently, location-specific assessments of POP profiles in breast milk, which vary regionally due to historical pesticide use, industrial activity, and diet[16], are crucial for informing public health strategies.
The Yangtze River Basin, a hub of industrial and agricultural activity in China, exemplifies the complex interplay between rapid urbanization and environmental contamination. Wuhan, situated in the mid-reaches of the Yangtze River within this basin, has evolved from a traditional heavy-industry center into a dynamic megacity where legacy pollutants coexist with emerging chemical stressors due to intensive manufacturing, urban expansion, and agrochemical use[17,18]. This setting offers a compelling case study for evaluating contemporary human exposure to POPs in a rapidly developing urban context.
Despite extensive toxicological evidence[19], critical gaps remain. Declining global trends may have reduced monitoring efforts, potentially masking residual or synergistic risks[20]. Furthermore, many studies focus on a narrow subset of POPs [e.g., dichlorodiphenyltrichloroethane (DDT), hexachlorocyclohexane (HCH)] and overlook the complex mixtures of PCB and PBDE congeners in human tissues. The influence of maternal sociodemographic and lifestyle factors, modifiable determinants of exposure, is often underexamined. Similarly, congener-specific associations between prenatal POP exposure and precise neonatal outcomes (e.g., birth weight and head circumference) remain poorly characterized, limiting insights into mechanisms of developmental toxicity. Moreover, analytical data are frequently reported without accompanying health risk assessments, which impedes their translation into actionable public health guidance.
To address these gaps, this study presents a comprehensive analysis of legacy OCPs, PCBs, and PBDEs in breast milk from 99 mothers in Wuhan, China. The objectives were to: (1) characterize concentration profiles and potential sources of target POPs; (2) identify maternal factors significantly associated with pollutant levels; (3) evaluate congener-specific relationships between prenatal exposure and key birth outcomes; and (4) conduct a health risk assessment to contextualize infant exposure via breastfeeding. By integrating exposure science, epidemiology, and risk characterization, this work provides timely evidence to support environmental health policy and maternal-child protection initiatives in rapidly urbanizing regions of China and beyond.
MATERIALS AND METHODS
Chemicals and reagents
A standard solution containing a mixture of the target analytes was prepared in n-nonane at a concentration of 1 μg mL-1. The analytes included OCPs, specifically hexachlorobenzene (HCB); DDT and its metabolites dichlorodiphenyldichloroethylene (DDE) and dichlorodiphenyldichloroethane (DDD) (collectively referred to as DDX); HCH isomers; heptachlor and its epoxide isomers (trans- and cis-); aldrin, dieldrin, and endrin (collectively referred to as Drins); and 1,4,5,6,7,7-hexachloro-8,9,10-trinitroborneol-5-ene-2,3-ylidene dimethyl, along with PCB congeners (IUPAC Nos. 18, 28, 44, 52, 66, 77, 81, 101, 105, 114, 118, 123, 126, 128, 138, 153, 156, 157, 169, 170, 180, 187, 189, 195, 206, and 209); and PBDE congeners (IUPAC Nos. 28, 47, 99, 100, 153, 154, and 183). A single stock solution containing all analytes was prepared in n-nonane at a volume ratio of 1:1 (v/v). The exact compositions of the stock solutions are detailed in Supplementary Table 1. All standard compounds were purchased from AccuStandard Inc. (New Haven, CT, USA). All solvents, including dichloromethane (DCM), n-hexane, isooctane, and ethanol, were of high-performance liquid chromatography (HPLC) grade. These materials, along with sodium sulfate, silica gel (70-200 mesh), and formic acid, were obtained from J&K Chemicals (Beijing, China). Concentrated sulfuric acid (95% H2SO4) was purchased from Sigma-Aldrich (Beijing, China). Ultrapure water (ρ = 18.2 MΩ·cm) was produced via a water purification system (Pall-Cascada Ⅲ.Ⅰ, CA, USA).
Study population and sample collection
Since 2015, pregnant women have been recruited as part of a comprehensive project at Wuhan No. 1 Hospital in Wuhan, China. The eligibility requirements are described in a previously published dataset[21]. Briefly, subjects were required to meet the following criteria: (1) residence in Wuhan for at least five years, (2) no clinical records or history of major maternal diseases, and (3) primiparous status. As a subset of this larger project, a total of 99 samples collected between January and December 2019 were selected from the cohort established between 2015 and 2019. A flow chart is provided in Supplementary Figure 1. The sampling procedures and protocols were approved and supervised by the Wuhan No. 1 Hospital Medical Ethics Committee {WH1H [2015] 6} on November 27, 2015. Informed consent was obtained from all participating mothers. The experimental protocols were also approved by the Zhejiang Shuren University Research Ethics Committee, and the results of this project were approved for submission as a scientific paper (202501087).
Human milk was collected within one week after delivery using 30mL clean glass containers. The samples were delivered to an on-site lab for temporary storage, and the sampling protocol is described in[22]. After collection, all human milk samples were transported to a laboratory in Hangzhou using cold-chain logistics and stored at -20 °C until analysis.
Sample pretreatment
The extraction and cleanup procedures were performed as previously described[18].
The process involves liquid-liquid extraction using a mixed solvent of n-hexane and DCM, followed by dehydration, treatment with concentrated sulfuric acid to remove lipids, and cleanup with silica gel. Briefly, each milk sample was spiked with 25 μL of 2,4,5,6-tetrachloro-m-xylene (TCMX) as an internal standard prior to extraction. After extraction and concentration, the final extract was reconstituted in 100 μL of n-nonane for instrumental analysis.
Instrumental analysis
The target pesticides were quantified using an Agilent 7890B gas chromatograph coupled with an Agilent 7010 triple quadrupole mass spectrometer (GC-QqQ-MS; Agilent Technologies, Santa Clara, USA). The mass spectrometer was operated in electron ionization (EI) mode with multiple reaction monitoring (MRM). The injector was set to the normal non-split injection mode, with an injection volume of 2 μL and a temperature of 230 ℃. Detailed information about the GC program, triple quadrupole (QqQ) settings, and quantification settings is provided in Supplementary Table 2.
Quality assurance and quality control
All glassware was meticulously cleaned, dried, and rinsed with n-hexane prior to use. The analytical method was validated before sample analysis. Quantification was performed using the internal standard method (TCMX) based on an 8-point calibration curve, which demonstrated excellent linearity (R2 > 0.999).
The method quantification limit (MQL) and limit of detection (LOD) were established as 10 and 3 times the signal-to-noise ratio (S/N), respectively, in a spiked blank matrix. Concentrations below the MQL were treated as non-detectable. To monitor procedural contamination and analytical performance, one procedural blank and one matrix-spiked blank were processed with every batch of 10 samples. The reported average recoveries for target analytes were obtained from matrix-spiked samples processed with each batch, along with the relative standard deviations (RSDs) for replicate analyses. The results of the quality control measures are presented in Supplementary Tables 3 and 4.
Health risk assessment
The estimated daily intake (EDI, ng kg-1 day-1) of pesticides in the samples was calculated as follows[23]:
where Cmilk represents the wet-weight concentration of pesticides in the samples (μg L-1), IR indicates the daily milk intake for neonates in China (114.6 g day-1)[24], SP represents the density of breast milk
The health risks associated with infant exposure via breastfeeding were evaluated using the hazard quotient (HQ). In this study, the HQ is defined as the ratio of the EDI to the maximum allowable intake dose for humans. An HQ > 1 indicates a potential health risk to infants.
Statistical analysis
Statistical analyses for OCPs, PCBs, and PBDEs were conducted separately, treating each class as an independent group of compounds. A blanket correction for multiple comparisons across the three classes was not applied; however, significance levels were interpreted conservatively within each analysis. Given the lipophilicity of the target analytes, measured wet-weight concentrations were converted to lipid-weight concentrations. However, wet-weight concentrations were used for the calculation of EDI. The normality of data distributions was assessed using the Kolmogorov-Smirnov test. As the data were skewed, a logarithmic transformation was applied to achieve approximate normality for subsequent parametric analyses. Spearman correlation analysis was employed to examine relationships between analyte concentrations and potential influencing factors. All statistical analyses were conducted using IBM Statistical Package for the Social Sciences (SPSS) software version 20.0 (Chicago, IL, USA), with a significance level defined as P < 0.05 for two-tailed tests.
RESULTS AND DISCUSSION
Study population
A total of 99 mother-infant pairs were enrolled in this study, and their comprehensive sociodemographic, maternal, and neonatal characteristics are summarized in Supplementary Table 5. The participating mothers were young women of reproductive age (mean age: 26.3 ± 3.1 years; range: 22-33 years) with a normal pre-pregnancy body mass index (BMI; mean: 21.1 ± 3.1 kg/m2), falling within the World Health Organization’s healthy weight range of 18.5-24.9 kg/m2[25]. The average gestational weight gain was
Among the newborns, males slightly outnumbered females (59.6%, n = 59/99). All key birth outcomes, including birth length (50.1 ± 0.9 cm), head circumference (34.0 ± 1.3 cm), and birth weight (3.3 ± 0.4 kg), were within the established norms for Chinese neonates[26]. Maternal educational attainment was heterogeneous: 35.4% had completed junior high school, 27.3% had some college education, and 24.2% held a bachelor’s degree. Occupational profiles were diverse, with the largest groups comprising enterprise managers (17.5%), professionals or technical staff (16.5%), and self-employed individuals (7.2%). Dietary patterns varied, with meat-based diets being the most commonly reported (31.3%), followed by vegetable-based (21.2%) and mixed diets (15.2%). The majority of participants (78.4%) consumed tap water, and nearly half (46.9%) reported regular use of prenatal supplements during pregnancy. Overall, this cohort represents a relatively young, healthy, and urban population with normal pre-pregnancy BMI, a high rate of uncomplicated full-term deliveries, and sociodemographic characteristics reflective of contemporary Wuhan. These attributes provide a well-characterized and contextually relevant setting for investigating associations between prenatal exposure to POPs and neonatal outcomes.
POP concentration in breast milk samples
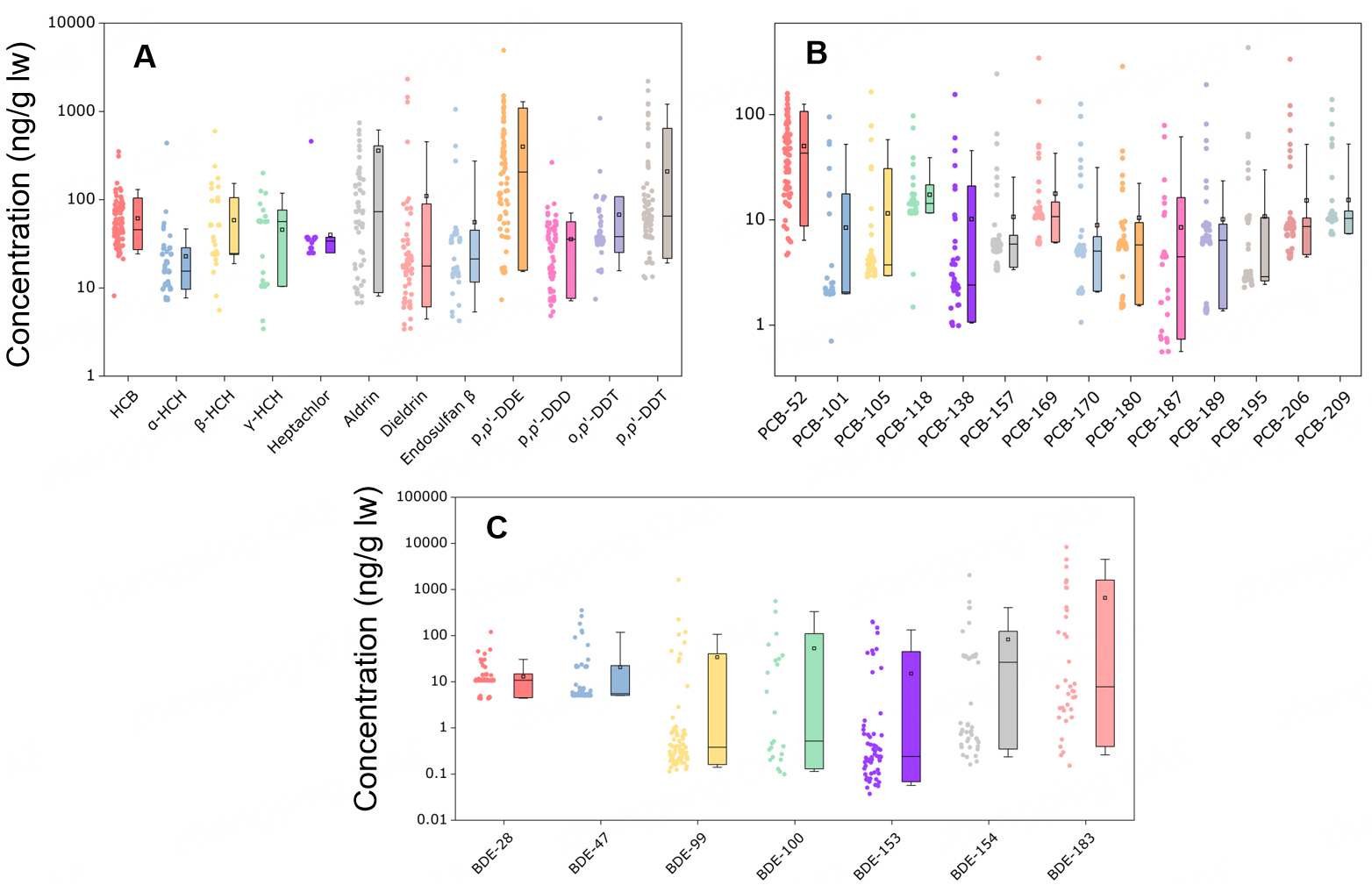
All three major classes of POPs, namely OCPs, PCBs, and PBDEs, were detected in breast milk samples from the study cohort [Figure 1]. Distinct compositional patterns emerged for each class, reflecting Wuhan’s unique environmental and socioeconomic context as a major industrial and agricultural hub, and highlighting the enduring influence of historical usage, local emission sources, and contemporary exposure pathways on maternal body burdens.
Figure 1. Lipid-adjusted concentrations of persistent organic pollutants in breast milk. Shown are organochlorine pesticides (OCPs; A), polychlorinated biphenyls (PCBs; B), and polybrominated diphenyl ethers (PBDEs; C), expressed in nanograms per gram of lipid weight
OCPs
OCPs were universally detected across all 99 samples [Figure 1A]. α-HCH exhibited a 100% detection frequency (DF), while HCB (DF = 92.9%) and DDT (DF = 80%) were also highly prevalent, indicating persistent environmental exposure despite regulatory restrictions. Lipid-adjusted median concentrations (interquartile ranges, IQR) were as follows: 45.75 ng g-1 lw (32.88-75.90 ng g-1 lw) for HCB, 44.40 ng g-1 lw (24.10-118.11 ng g-1 lw) for ∑HCHs, and 350.45 ng g-1 lw (164.27-842.93 ng g-1 lw) for ∑DDTs. ∑HCHs and ∑DDTs accounted for 15.24% and 54.45% of total ∑OCPs, respectively, ranking them as the dominant OCP groups. This profile reflects China’s extensive historical application of technical-grade HCH and DDT in agriculture throughout the mid-to-late 20th century. Despite their official ban [27], the continued detection of these compounds in human tissues is attributable to their exceptional environmental stability, resistance to degradation, and continued cycling through soil, water, and food systems[28].
PCBs
Of the 26 targeted PCB congeners, PCB-189, PCB-195, and PCB-206 were detected in all samples, whereas PCB-18 and PCB-28 were below detection limits [Figure 1B]. Median lipid-adjusted concentrations were 6.39 ng g-1 lw (IQR: 6.16-8.79 ng g-1 lw) for PCB-189, 2.89 ng g-1 lw (IQR: 2.73-10.10 ng g-1 lw) for PCB-195, and 8.65 ng g-1 lw (IQR: 7.20-9.03 ng g-1 lw) for PCB-206. Among quantifiable congeners, PCB-52 and PCB-118 contributed 34.34% and 11.36% of total ∑PCBs, respectively. Notably, PCB-52 exhibited the highest individual concentration (median: 43.29 ng g-1 lw; IQR: 19.54-67.52 ng g-1 lw), identifying it as the predominant PCB congener in this cohort.
The prevalence of medium- and high-chlorinated congeners (e.g., PCB-118, -195, -206) aligns with their greater resistance to metabolic breakdown and propensity for bioaccumulation in adipose tissue. The unexpectedly high levels of the low-chlorinated PCB-52 may be explained by its relatively higher volatility and environmental lability[29], coupled with continuous exposure via inhalation or indoor dust, potentially resulting in an intake rate exceeding its metabolic elimination rate in the body[30].
PBDEs
All seven targeted brominated diphenyl ethers (BDE) congeners were analyzed; BDE-28 and BDE-47 were detected in 100% of samples, whereas BDE-100 exhibited a low DF of 23.2% [Figure 1C]. The highest median concentrations were observed for BDE-154 (26.39 ng g-1 lw; IQR: 0.59-36.29 ng g-1 lw), followed by BDE-28 (10.8 ng g-1 lw; IQR: 10.7-11.1 ng g-1 lw), reflecting very low variability across samples, BDE-47 (5.49 ng g-1 lw; IQR:
Regional comparison of OCP, PCB, and BDE patterns and potential sources
A comparative analysis with global datasets [Supplementary Table 6] reveals region-specific POP concentrations, shaped by historical usage, regulatory timelines, climatic factors influencing environmental fate, and contemporary exposure pathways such as diet and occupation. When considered as a contemporary snapshot, our data show that POP levels in the present cohort are generally lower than those reported in earlier decades both regionally and globally, supporting the long-term effectiveness of regulatory bans. Nevertheless, their continued detection underscores the persistence of environmental reservoirs. For OCPs, median concentrations in Wuhan were generally lower than those reported in Iran[34], Spain[35], Greece[36], Tanzania[37], and France[38], though marginally higher than those in Croatia[39].
These disparities reflect divergent exposure histories. For instance, the markedly lower levels observed in Spain for β-HCH (11 ng g-1 lw vs. 24.61 ng g-1 lw) and heptachlor (3.9 ng g-1 lw vs. 33.93 ng g-1 lw) have been attributed to lower dietary lipid intake, which may limit bioaccumulation[35]. In contrast, elevated ∑DDTs levels reported in Greece (400 ng g-1 lw)[36], Tanzania (135 ng g-1 lw)[37], and France (60.07 ng g-1 lw), compared to Wuhan’s median of 205.91 ng g-1 lw, are linked to historical DDT use for malaria vector control and the agricultural application of dieldrin[38]. Conversely, Croatia’s relatively low POP burden is consistent with its limited historical reliance on synthetic pesticides in small-scale Mediterranean agriculture[39].
For PCBs, Wuhan exhibited substantially higher concentrations of medium- and high-chlorinated congeners (82.78 ng g-1 lw) compared to those reported in Colombia (4.05 ng g-1 lw), Germany (0.85 ng g-1 lw), Hungary (18.64 ng g-1 lw), and Tanzania (1.55 ng g-1 lw). This pattern aligns with the frequent consumption of freshwater fish from the Yangtze River and associated lakes, which are known reservoirs of legacy PCB contamination, thereby facilitating dietary bioaccumulation. Notably, PCB-52 concentrations in Wuhan (43.29 ng g-1 lw) far exceeded those in Colombia (0.072 ng g-1 lw), Germany (0.14 ng g-1 lw), Hungary
Regarding BDEs, Wuhan demonstrated markedly elevated median concentrations across multiple congeners relative to global averages: BDE-28 (10.76 ng g-1 lw vs. Colombia: 0.16 ng g-1 lw[40], Germany: 0.03 ng g-1 lw[41], Sweden: 0.04 ng g-1 lw[42], and the UK: 0.09 ng g-1 lw[43]), BDE-47 (5.49 ng g-1 lw vs. Colombia: 0.5 ng g-1 lw, Germany: 0.2 ng g-1 lw, South Korea: 0.54 ng g-1 lw, Sweden: 0.46 ng g-1 lw, and the UK: 1.92 ng g-1 lw), and BDE-154 (26.39 ng g-1 lw vs. Colombia: 0.02 ng g-1 lw, Germany: 0.007 ng g-1 lw, Sweden: 0.05 ng g-1 lw and, the UK: 0.07 ng g-1 lw). These elevated levels are consistent with Wuhan’s dense industrial landscape, particularly its concentration of electronics manufacturing, automotive, and steel industries, which have historically employed BFRs. Persistent emissions from industrial sources, building materials, and consumer products have likely contributed to long-term environmental accumulation and subsequent human exposure via diet, dust, and air. The relatively high levels of BDE-183, a congener less associated with dietary intake and more frequently linked to inhalation of indoor or ambient particulate matter, further suggest that non-dietary exposure routes, particularly airborne uptake, may play a significant role in maternal body burdens. Collectively, these regional contrasts highlight the significance of local context, encompassing historical chemical use, industrial activity, and lifestyle factors, in shaping human POP exposure profiles. Wuhan’s unique combination of legacy agricultural practices and intensive modern industry creates a complex exposure milieu that warrants targeted biomonitoring and source-control strategies.
Patterns of co-exposure to OCPs, PCBs, and BDEs in Breast milk
Spearman correlation analyses [Supplementary Tables 7-9] revealed prevalent and statistically significant positive correlations (P < 0.05 or P < 0.01) among compounds within each POP class, demonstrating shared sources, environmental fate, and synchronized biological processing in the maternal body.
For OCPs, strong intercorrelations among technical HCH isomers (α-HCH, β-HCH, γ-HCH), as well as between aldrin and its stable metabolite dieldrin, are consistent with the historical use of common commercial formulations[44,45]. Significant correlations across chemically distinct OCP groups, such as α-HCH and endosulfan-β[46] or β-HCH and heptachlor[47], suggest either concurrent agricultural application in the past or co-transport and co-deposition via atmospheric and hydrological pathways. The strong correlation between p,p’-DDT and its persistent metabolite p,p’-DDE indicates extensive post-application environmental degradation, suggesting that current body burdens predominantly derive from historical exposure. Moreover, the consistent positive correlations across different OCP classes (e.g., dieldrin with HCHs) are strongly indicative of their simultaneous mobilization from maternal adipose tissue during lactation-induced lipolysis[48].
For PCBs, significant correlations among congeners likely stem from exposure to legacy industrial mixtures (e.g., Aroclors), in which congeners were manufactured and released into the environment as a unit[49]. These patterns are further shaped by selective biological retention; for example, highly chlorinated ortho-substituted congeners (e.g., PCB-138, -153, -170, -180) exhibit strong intercorrelations due to their enhanced resistance to metabolic breakdown and high affinity for lipid-rich tissues such as tissue[50]. During lactation, hormonal shifts trigger the mobilization of these stored congeners into systemic circulation, followed by partitioning into breast milk via serum lipid transport mechanisms[51]. Conversely, the absence of significant correlations for certain low-chlorinated or non-ortho congeners may reflect more rapid biotransformation, distinct exposure sources, or different kinetic behaviors.
The correlation patterns among BDE congeners closely align with those of commercial BFR formulations. The strong associations among BDE-47, -99, and -100 are characteristic of exposure to the commercial penta-BDE mixture, whereas correlations involving BDE-183, -153, and -154 are indicative of exposure to octa-BDE[52]. These formulations were widely incorporated into polyurethane foams, electronic housings, and textiles, from which they leach into indoor environments over time. Consequently, humans are concurrently exposed via inhalation and ingestion of contaminated dust[53], resulting in integrated internal doses that manifest as correlated congener profiles in biological matrices.
Collectively, the observed correlation patterns across all three POP classes arise from a combination of shared physicochemical properties, namely environmental persistence, lipophilicity, and resistance to biotic and abiotic degradation, which drive common environmental fate, shared exposure pathways, and synchronized pharmacokinetics[54]. Within the maternal body, these compounds co-accumulate in adipose tissue over decades, undergo hormonally mediated co-mobilization during lactation, and are transported to breast milk via circulating lipoproteins[55]. Metabolic interconversion (e.g., DDT to DDE, higher brominated BDEs to lower brominated BDEs) further strengthens statistical associations among related compounds. These enduring correlation structures, detectable decades after regulatory bans, serve as molecular fingerprints of historical exposure to complex chemical mixtures. Their persistence not only attests to the long environmental half-lives of POPs but also underscores a critical public health challenge: even in the absence of ongoing use, legacy pollutants continue to co-occur, co-accumulate, and be transferred to vulnerable populations through natural biological processes such as breastfeeding.
Correlation between maternal sociodemographic factors and POP body burdens
Spearman correlation analyses revealed significant associations between sociodemographic and lifestyle variables and body burdens of OCPs, PCBs, and BDEs [Supplementary Tables 10-12]. These relationships underscore the interplay of physiological dynamics, exposure pathways, and congener-specific toxicokinetics.
An inverse relationship was observed between pre-pregnancy BMI and specific OCPs, suggesting that adiposity modulates the pharmacokinetics of lipophilic contaminants. Lower BMI may reduce sequestration capacity in adipose tissue or alter metabolic clearance, potentially increasing circulating concentrations. These effects may be mediated by BMI-dependent variations in lipid metabolism and endocrine signaling, particularly estrogen-regulated adipose remodeling, which influences storage and mobilization[56].
A key finding is the divergent effect of parity, highlighting the dichotomous association of pregnancy and lactation. The positive correlation with total OCP burden suggests net cumulative retention during successive pregnancies, which is attributable to the high lipophilicity and slow elimination kinetics of these compounds[57]. Adipose tissue acts as a long-term reservoir, with stored OCPs mobilized during postpartum hormonal changes and transferred to the infant via lactation[58,59]. Conversely, the negative correlation between parity and specific higher-chlorinated PCBs (e.g., PCB-170, -206, -209) suggests that lactation plays a significant role as an excretory pathway for these congeners. Prolactin-driven lipolysis mobilizes lipid-soluble POPs from maternal adipose stores into circulation, facilitating their transfer to breast milk and thereby reducing the maternal body burden over successive lactation cycles[60]. This dichotomy demonstrates that lactation can simultaneously act as a depuration mechanism for certain POPs (such as these PCBs) while serving as a vector for the transfer of others (such as OCPs).
Dietary patterns also influenced exposure profiles. A vegetable-rich diet was significantly correlated with higher PCB-187 levels, consistent with the soil persistence and plant uptake of this congener[61], implicating dietary intake of contaminated produce as a key exposure route. Conversely, non-occupational status (e.g., stay-at-home mothers) was correlated with elevated BDE-28 concentrations, demonstrating that the indoor environment is a dominant source. This is consistent with evidence of BDE-28 prevalence in household
Notably, congener-specific effects extended to reproductive outcomes, underscoring the importance of molecular structure. The significant positive correlation between BDE-154, but not its structural isomer BDE-153, and miscarriage history suggests toxicity mediated by specific structural attributes. The ortho-bromination pattern of BDE-154 likely permits a more planar molecular conformation[64], which may enhance its affinity for endocrine receptors and its capacity to induce oxidative stress, thereby disrupting early pregnancy maintenance[65].
Source apportionment of POPs
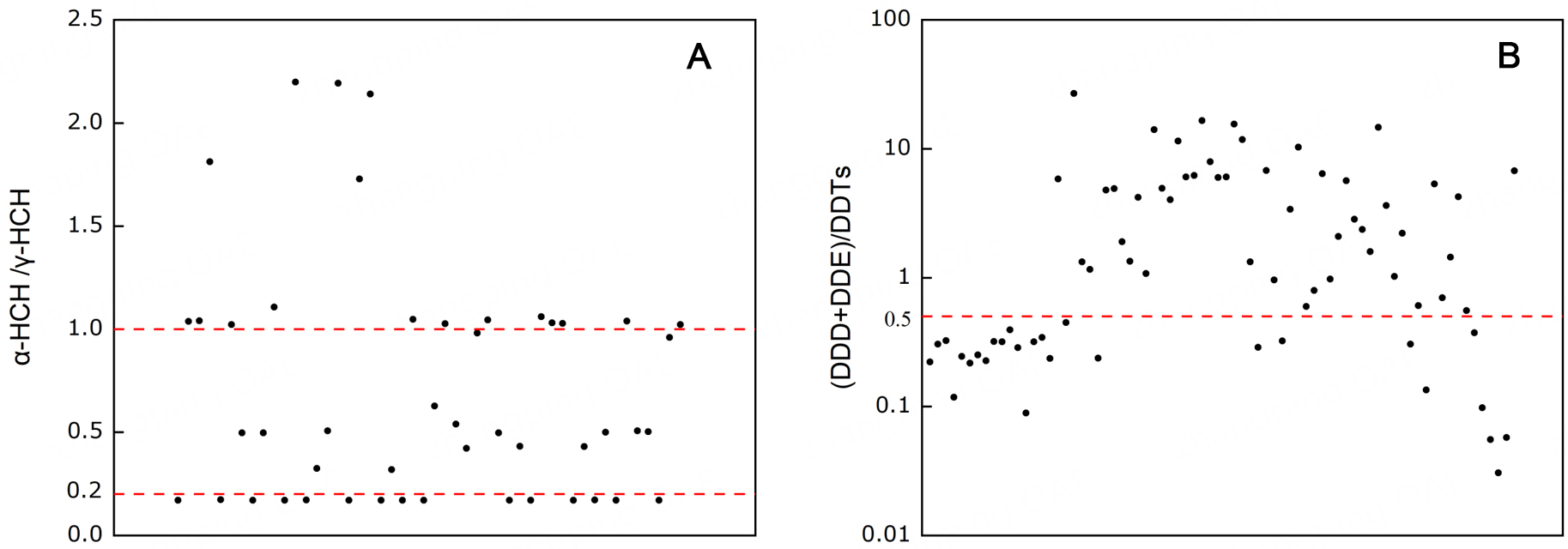
To discern the origins and historical usage patterns of POPs, we analyzed diagnostic isomeric ratios, metabolite profiles, and congener distributions in breast milk samples. For HCH isomers, the α-HCH/γ-HCH ratio was calculated across valid breast milk samples (N = 48). The results, plotted in Figure 2A, show a sample mean ratio of 0.702 (maximum: 2.20), with approximately 80% of values falling within the range of 0.2-1.0. This ratio is a well-established indicator of source type; for instance, values between 0.2 and 1.0 are characteristic of exposure to lindane (technical-grade γ-HCH, > 99% purity), whereas ratios of 4-15 suggest input from technical HCH mixtures (containing 60%-70% of α-HCH) or photochemical isomerization of γ- to α-HCH in the environment[66]. The predominance of low ratios in our cohort strongly indicates that HCH residues in maternal tissues derive primarily from historical agricultural use of lindane, rather than industrial emissions or occupational exposure. This finding points to dietary intake of legacy residues, particularly via contaminated foodstuffs, as the dominant exposure route[67].
Figure 2. Diagnostic ratios of persistent organochlorine compounds in breast milk samples. (A) Ratio of α-hexachlorocyclohexane (α-HCH) to γ-hexachlorocyclohexane (γ-HCH) (N = 48); (B) Ratio of [p,p’-DDE + p,p’-DDD] to total DDTs (N = 74). These ratios provide insight into environmental sources and degradation patterns. DDE: Dichlorodiphenyldichloroethylene; HCH: hexachlorocyclohexanes; DDD: dichlorodiphenyldichloroethane; DDT: dichlorodiphenyltrichloroethane; HCH: hexachlorocyclohexanes.
For DDT and its transformation products, the metabolite ratio (DDE + DDD)/∑DDT was computed for valid samples (n = 74) (Figure 2B; log-scaled axis). The mean value was 3.48, with 65% of samples exceeding the threshold of 0.5. A ratio > 0.5 is widely interpreted as evidence of historical exposure, as DDT undergoes progressive microbial degradation; under aerobic conditions, p,p’-DDE is formed, while under anaerobic conditions, p,p’-DDD is produced[68]. The elevated proportion of samples (65%) exceeding this threshold aligns with China’s 1983 ban on agricultural DDT use and confirms that current body burdens stem from persistent environmental residues rather than recent inputs. Given the compound’s high lipophilicity and resistance to degradation, dietary consumption, especially of animal fats and aquatic products, remains the principal exposure pathway for the general population.
Congener-specific profiles further elucidate the sources of PCBs and PBDEs. PCBs in breast milk were dominated by highly chlorinated congeners (penta- to hepta-chlorinated biphenyls), consistent with both legacy industrial formulations and secondary formation processes. While historical electronics manufacturing in urban centers contributed to PCB releases[69], the observed pattern cannot be fully explained by commercial Aroclor mixtures alone[70]. Notably, the presence of non-Aroclor indicator congeners, which are recognized markers of pyrogenic origin, suggests a substantial contribution from incomplete combustion processes, including waste incineration, industrial heating, and residential biomass burning[71]. This finding implies that atmospheric deposition from ongoing combustion sources remains a relevant exposure route contributing to contemporary maternal PCB body burdens.
For PBDEs, an unexpected congener pattern was observed. For example, BDE-28 (tri-BDE) was present at higher concentrations than BDE-47 (tetra-BDE), despite the latter being the dominant congener in commercial penta-BDE mixtures. This inversion is highly informative, as BDE-28 is only a minor component in original products but a major debromination product of higher-brominated congeners such as BDE-47[72]. The elevated BDE-28/BDE-47 ratio provides compelling evidence of post-emission transformation, whether through environmental microbial degradation or in vivo metabolic debromination[73]. Consequently, the PBDE profile in breast milk reflects a degraded signature rather than direct exposure to primary commercial mixtures.
Finally, the Yangtze River Basin functions as a critical environmental conduit for POPs. Urban and industrial activities release these hydrophobic compounds into the air and soil, where they undergo atmospheric deposition and surface runoff, particularly during rainfall events[74,75]. These processes transport POPs into aquatic ecosystems, where their persistence and lipophilicity drive bioaccumulation in fish and other aquatic organisms. Given that the Yangtze River is a major source of food and water for Wuhan residents, the consumption of contaminated aquatic products is hypothesized to be the dominant pathway linking environmental contamination to human body burdens[76]. Together, these lines of evidence reveal a multifaceted exposure landscape shaped by historical pesticide use, industrial legacies, ongoing pyrogenic emissions, and environmental transformation processes, all of which converge in human tissues decades after regulatory controls were implemented.
Associations between prenatal POP exposure and neonatal birth outcomes
The reported associations are based on the use of breast milk POP levels as biomarkers of cumulative prenatal exposure. Because of their high lipophilicity and long half-lives, these compounds accumulate in maternal adipose tissue and are mobilized during pregnancy and lactation; thus, breast milk levels reflect integrated body burdens and fetal exposure. Correlation analysis revealed a statistically significant inverse association between PCB-170 and infant head circumference (r = -0.234, P < 0.05; Supplementary Table 13), consistent with prior epidemiological findings[77]. One possible explanation for this relationship could involve placental mechanisms. PCB-170 is a known inducer of cytochrome P450 enzymes (e.g., CYP1A1) in placental tissue, which may promote the formation of reactive oxygen species (ROS) and contribute to oxidative stress[78]. Such oxidative stress could affect trophoblast function, potentially including reduced expression of angiogenic factors such as vascular endothelial growth factor (VEGF), thereby perturbing placental vascularization and perfusion. These alterations, if present, might limit the transfer of oxygen and nutrients to the fetus. Head circumference, as a sensitive marker of fetal brain growth, could thus serve as an early indicator of such possible developmental effects[79].
To further elucidate these relationships while accounting for potential confounders (e.g., maternal age, pre-pregnancy BMI, gestational weight gain, and parity), multiple linear regression models were used to assess the associations between summed POP classes (OCPs, PCBs, and BDEs) and neonatal outcomes. Following diagnostic assessment, ∑OCP was excluded from the birth weight model due to influential outliers. The modified models revealed congeners- and class-specific associations with neonatal outcomes. Notably, higher ∑HCH concentrations were associated with increased birth weight, an observation that may appear counterintuitive but is likely driven by β-HCH, the most biologically active HCH isomer and a known environmental endocrine disruptor with estrogenic activity. β-HCH exhibits estrogenic properties that can stimulate adipocyte differentiation, proliferation, and lipid accumulation[80]. Additionally, it has been shown to reduce circulating thyroid hormone levels[81], which may lower the basal metabolic rate and promote energy retention, thereby contributing to increased fetal weight gain.
In contrast, elevated ∑Heptachlor and ∑Endosulfan were significantly associated with reduced birth weight [Supplementary Table 14], indicating adverse effects on fetal growth. ∑Endosulfan may impair metabolic homeostasis by disrupting insulin signaling, particularly the phosphoinositide 3-kinase (PI3K)/protein kinase B (AKT) pathway, leading to peripheral insulin resistance, reduced glucose uptake, and a compensatory shift toward the catabolism of proteins and lipids at the expense of anabolic growth[82]. ∑Heptachlor, meanwhile, targets mitochondrial function by inhibiting electron transport chain complexes, thereby limiting adenosine triphosphate (ATP) production[83]. Concurrently, it downregulates hypothalamic leptin receptor expression, blunting satiety signaling and dysregulating neuroendocrine control of energy allocation[84]. Together, these effects redirect energy toward cellular maintenance and stress responses rather than somatic growth, culminating in restricted fetal weight gain.
Interestingly, the regression analysis also indicated that elevated concentrations of specific BDE congeners, BDE-28 and BDE-99, were associated with increased infant head circumference, with BDE-99 showing a more pronounced effect [Supplementary Table 12]. These findings align with limited prior evidence but contrast with the typical inverse associations observed for many environmental toxicants. A plausible, though speculative, mechanism involves modulation of the thyroid axis, which is critical for neurodevelopment. BDE-99 structurally resembles thyroxine (T4) and may competitively bind to thyroxine-binding globulin (TBG), potentially reducing free T4 levels[85]. The ensuing compensatory increase in thyroid-stimulating hormone (TSH) might stimulate thyroid hyperplasia. More critically, animal studies suggest that maternal exposure to BDE-99 can upregulate the expression of thyroid hormone receptors (TRα and TRβ) in the fetal brain[86]. This upregulation, possibly a compensatory response to mild hypothyroidism, could enhance the sensitivity of brain tissue to available thyroid hormones, thereby promoting neuronal proliferation, glial cell differentiation, and potentially increasing brain volume[87]. However, the long-term functional consequences of this apparent growth require further investigation, as it may represent a dysregulated rather than a beneficial adaptive response.
While these analyses provide compelling evidence of congener-specific associations between POP exposure and birth outcomes, several limitations must be acknowledged. First, exposure was assessed using a single postpartum breast milk sample, which, although reflective of cumulative burden, may not capture critical windows of susceptibility during early gestation. Second, despite statistical adjustment, residual confounding by unmeasured socioeconomic, dietary, or environmental co-exposures cannot be excluded. Future studies employing repeated biomonitoring throughout pregnancy and integrating exposome-wide approaches would strengthen causal inference.
Health risk assessment of oral intake of pops via breastfeeding in the early life stage
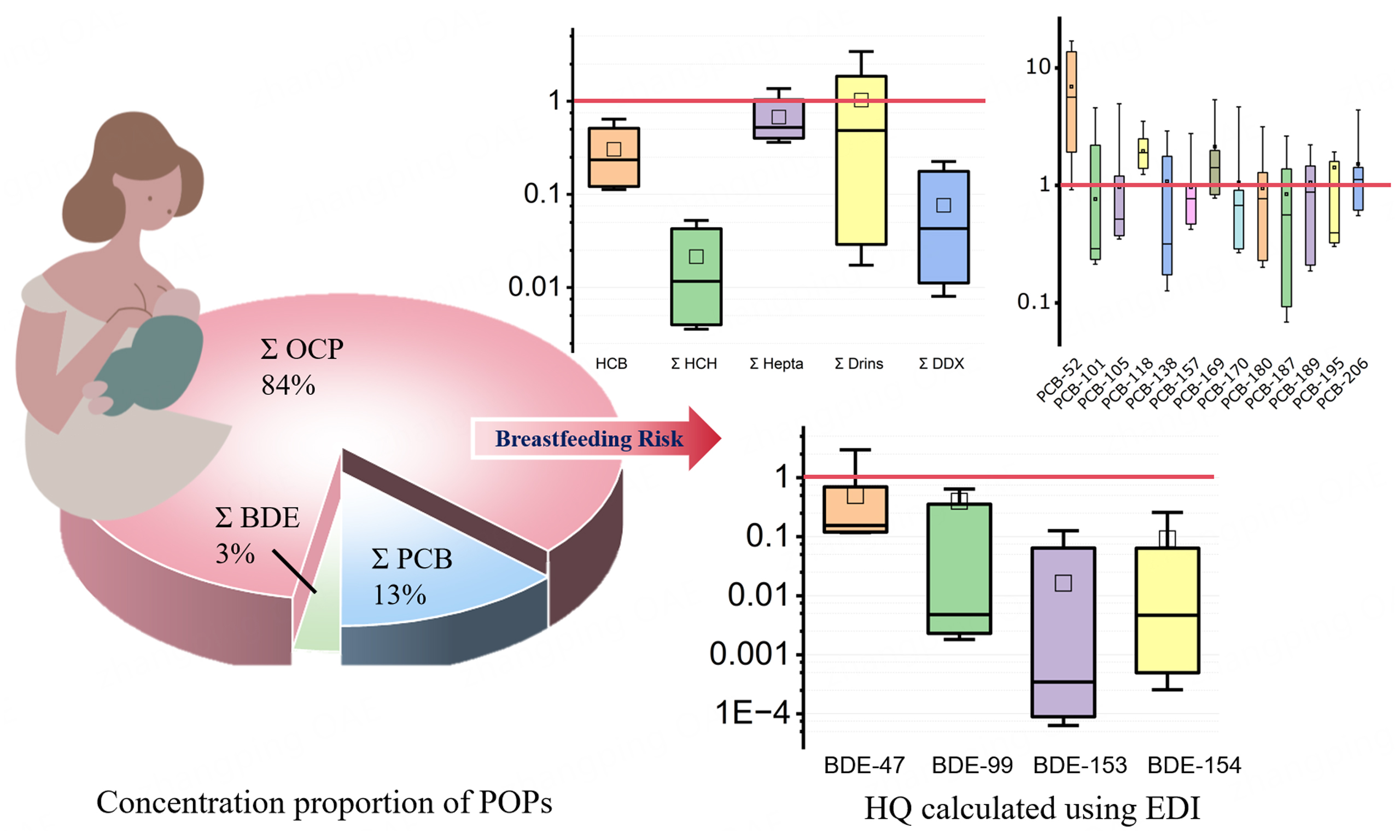
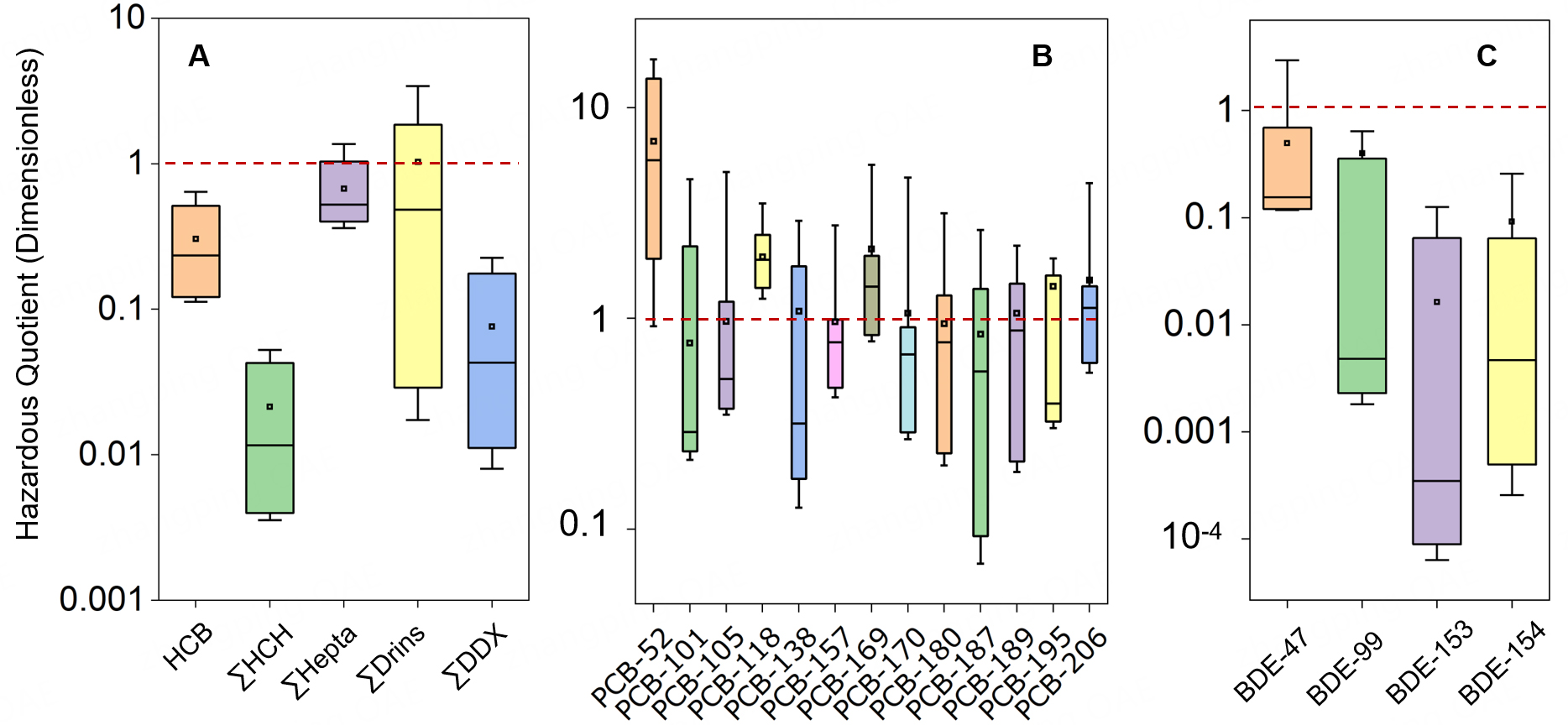
Although global health authorities universally endorse breastfeeding for its irreplaceable nutritional, immunological, and neurodevelopmental benefits, the presence of POPs in breast milk necessitates a quantitative risk assessment to contextualize potential concerns. In this study, the EDI of various POPs for infants was calculated and compared with established health-based guidance levels, including acceptable daily intake (ADI) or reference doses (RfDs). The median EDI values for OCPs, including HCB
Figure 3. Hazard quotients (HQs) for estimated infant exposure to organochlorine pesticides (OCPs; A), polychlorinated biphenyls (PCBs; B), and polybrominated diphenyl ethers (PBDEs; C) via breast milk. An HQ > 1 (red dashed line) indicates a potential health risk. For each boxplot, the box limits represent the 10th and 90th percentiles, the whiskers extend to the 5th and 95th percentiles, the interior line marks the median, and the square denotes the mean. Individual data points are plotted. Concentrations below the LOQ were imputed as (DF * LOQ) for the calculation of EDI and subsequent HQ values. Only compounds with DF% > 30 were presented in Figure 1. HCB: Hexachlorobenzene; LOQ: limit of quantification; DF: detection frequency; HCB: hexachlorobenzene; HCH: hexachlorocyclohexane; Drins: a group of related insecticides (i.e, Aldrin, Dieldrin, Endrin); DDX: a collective term for DDT and its metabolites (e.g., DDE, DDD).
However, a subset of samples indicated elevated exposure levels, warranting specific attention. For ∑Drins, 10% of the samples had an HQ > 1, and for PCBs, 50% exceeded the threshold. This suggests that, although the population-wide risk is low, certain infants may be exposed to higher levels of these specific contaminants. Aldrin and dieldrin (∑Drins) are established neurotoxicants, while PCBs can interfere with thyroid hormone transport and signaling, potentially disrupting neurodevelopment[88].
These findings should be interpreted in light of infant vulnerability. Infants have developing organ systems, an immature blood-brain barrier, and less efficient detoxification pathways, which could theoretically increase susceptibility to chemical exposure[89]. Furthermore, cumulative and interactive effects of co-occurring POPs, many of which share mechanisms of action such as endocrine disruption or oxidative stress, represent a recognized limitation of single-chemical risk assessment[90]. Although individual HQs suggest low risk, the potential for additive or synergistic effects within complex mixtures remains an active area of research.
Notwithstanding these considerations, the quantitative evidence is unequivocal. For the vast majority of infants in this cohort, POP exposures via breastfeeding remain well below established safety thresholds. Crucially, the profound lifelong benefits of breastfeeding, including optimal nutrient delivery, passive immune protection, microbiome establishment, and enhanced cognitive outcomes[91], substantially outweigh the theoretical risks associated with low-level exposure to environmental contaminants. Therefore, public health guidance should continue to strongly promote breastfeeding as the gold standard of infant nutrition. At the same time, the identification of outliers for ∑Drins and PCBs highlights the importance of targeted public health strategies, such as enhanced dietary counseling for women of reproductive age in high-exposure areas, continued biomonitoring of human milk to track temporal trends, and source-reduction initiatives aimed at reducing legacy pollutants in food and environmental media. These measures aim to reduce maternal body burdens at the source, thereby protecting infant health without undermining the irreplaceable advantages of breastfeeding, a balanced approach aligned with the precautionary principle and One Health objectives.
Strengths and limitations
This study has several no Supplementary Table trengths. It provides a comprehensive assessment of a wide spectrum of legacy and emerging POPs in breast milk from a well-defined cohort in Wuhan, China. By integrating high-sensitivity chemical analysis with detailed sociodemographic, clinical, dietary, and reproductive data, the study enabled a nuanced exploration of exposure determinants and their congener-specific associations with precise birth outcomes. The resulting data contribute valuable region-specific biomonitoring evidence to the global understanding of POP trends. Furthermore, the quantitative health risk assessment, based on estimated daily infant intake, demonstrates that exposure levels remain well below established safety thresholds, reinforcing the public health value of breastfeeding.
Several limitations must be acknowledged. First, exposure was assessed from a single postpartum breast milk sample. Although this reflects maternal body burden, it may not capture critical susceptibility windows during early gestation. Second, the study population was drawn exclusively from an urban, industrialized setting, which may limit the generalizability of findings to populations with different exposure profiles. Third, despite statistical adjustment for a wide range of confounders, residual confounding by unmeasured or imperfectly measured factors cannot be excluded. Fourth, the health risk assessment was conducted on a compound-by-compound basis using a deterministic approach. The application of probabilistic methods, such as Monte Carlo simulation (MCS), was considered; such an analysis could provide a more realistic characterization of variability and uncertainty, as advocated in the U.S. Environmental Protection Agency (EPA) Probabilistic Risk Assessment framework[92]. However, conducting a reliable MCS requires robust parameter distributions and specific methodological expertise. While the current dataset may be structured to support such an analysis, its execution was beyond the immediate scope of this study and should be considered a suggested enhancement for future work rather than a mandatory requirement for the present assessment. Finally, the standard risk assessment approach does not account for the potential cumulative or interactive effects of the complex POP mixtures to which infants are exposed, representing a critical consideration for future research.
CONCLUSION
This study provides a comprehensive assessment of POPs in breast milk from a cohort in Wuhan, China. Various legacy contaminants, including OCPs, PCBs, and PBDEs, were detected, with ∑DDTs and PCB 52 being dominant. In the health risk assessment, the EDI of POPs for the majority of breastfed infants remained below established safety thresholds, reaffirming the overall safety of breastfeeding. However, several specific POP congeners showed significant associations with adverse birth outcomes: PCB-170 exhibited a significant negative correlation with infant head circumference, while ∑Heptachlor and ∑Endosulfan were significantly associated with reduced birth weight. In contrast, ∑HCH concentrations were associated with increased birth weight. Future research should prioritize longitudinal biomonitoring across gestation to better capture critical windows of susceptibility, as well as mixture-based risk assessment approaches to evaluate the cumulative developmental impacts of co-exposure to complex POP profiles. Only through such integrated efforts can we safeguard maternal and child health while advancing toward a cleaner, more sustainable future for chemicals.
DECLARATIONS
Acknowledgments
The authors sincerely thank the participants who volunteered to provide breast milk samples, as well as the nurses, practitioners, and medical staff at Wuhan No. 1 Hospital for their assistance with sample collection and storage.
Authors’ contributions
Investigation, formal analysis, data curation, visualization, writing - original draft: Chen, X. Investigation, resources, funding acquisition, writing - review & editing: Wu, X.; Gao, J.
Investigation, data curation: Cheng, H.; Le, X.; Dong, Y.
Methodology, validation, formal analysis: Liu, S.
Writing - original draft, writing - review & editing: Hayat, K.
Supervision, funding acquisition: Liu, W.
Conceptualization, methodology, validation, formal analysis, investigation, data curation, writing - original draft, writing - review & editing, visualization, supervision, funding acquisition: Yin, S.
Availability of data and materials
Data are available from the corresponding author upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the National Natural Science Foundation of China (22276166). XW and JG would like to thank the Natural Science Foundation of Hubei Province (2023AFB641) and the Opening Foundation of Hubei Key Laboratory of Regional Development and Environmental Response (2023(C)001, 2023(C)002).
Conflicts of interest
Yin, S. is a Youth Editorial Board Member of Journal of Environmental Exposure Assessment. He had no involvement in the review or editorial process of this manuscript, including but not limited to reviewer selection, evaluation, or the final decision. The other authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
The sampling procedure and protocols were approved and supervised by the Medical Ethics Committee of Wuhan No. 1 Hospital (WH1H [2015] 6) on November 27, 2015. The experimental protocols were approved by the Research Ethics Committee of Zhejiang Shuren University, and the results of this project were approved for submission as a scientific paper (202501087). Informed consent was obtained from all participating mothers.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Jiang, L.; Gao, W.; Ma, X.; et al. Long-term investigation of the temporal trends and gas/particle partitioning of short- and medium-chain chlorinated paraffins in ambient air of King George Island, Antarctica. Environ. Sci. Technol. 2021, 55, 230-9.
2. Sun, H.; Li, Y.; Hao, Y.; et al. Bioaccumulation and trophic transfer of polybrominated diphenyl ethers and their hydroxylated and methoxylated analogues in polar marine food webs. Environ. Sci. Technol. 2020, 54, 15086-96.
3. Tang, C.; Chen, Z.; Huang, Y.; et al. Occurrence and potential harms of organochlorine pesticides (OCPs) in environment and their removal by periphyton. Crit. Rev. Environ. Sci. Technol. 2023, 53, 1957-81.
4. Gabryszewska, M. How are polychlorinated biphenyls currently being produced, despite the production ban, and do they pose a risk to the environment? Environ. Prot. Nat. Resour. 2022, 33, 29-34.
5. An, J.; Li, S.; Zhong, Y.; et al. The cytotoxic effects of synthetic 6-hydroxylated and 6-methoxylated polybrominated diphenyl ether 47 (BDE47). Environ. Toxicol. 2011, 26, 591-9.
6. Domingo, J. L.; Souza, M. C. O.; Nadal, M.; Barbosa, F. A One Health approach to polybrominated diphenyl ethers (PBDEs): Integrating human, animal, and environmental health perspectives. Chemosphere 2025, 386, 144644.
7. Abafe, O. A.; Harrad, S.; Abdallah, M. A. Novel insights into the dermal bioaccessibility and human exposure to brominated flame retardant additives in microplastics. Environ. Sci. Technol. 2023, 57, 10554-62.
8. He, X.; Wu, R.; Jiang, W.; Tian, Y.; Zhang, J.; Huang, Y. Shanghai birth cohort. prenatal exposure to per- and polyfluoroalkyl substances and childhood executive function and behavioral difficulties at age 7: evidence from the Shanghai birth cohort study. Environ. Int. 2025, 202, 109687.
9. Holme, J. A.; Myhre, O.; Øvrevik, J. Adverse neurodevelopment in children associated with prenatal exposure to fine particulate matter (PM2.5) - possible roles of polycyclic aromatic hydrocarbons (PAHs) and mechanisms involved. Reprod. Toxicol. 2024, 130, 108718.
10. Wu, D.; Li, Y.; Chen, L.; et al. Maternal gestational weight gain and offspring’s neurodevelopmental outcomes: a systematic review and meta-analysis. Neurosci. Biobehav. Rev. 2023, 153, 105360.
11. Xu, Y.; Zhao, X.; Ding, Z.; Jiang, S.; Yu, R. Hidden heroes in breast milk: the dual roles of phospholipids and sphingolipids in infant immunity and brain development. Early. Hum. Dev. 2025, 211, 106409.
12. Acharya, N.; Gautam, B.; Subbiah, S.; Rogge, M. M.; Anderson, T. A.; Gao, W. Polycyclic aromatic hydrocarbons in breast milk of obese vs normal women: Infant exposure and risk assessment. Sci. Total. Environ. 2019, 668, 658-67.
13. Hu, L.; Luo, D.; Wang, L.; et al. Levels and profiles of persistent organic pollutants in breast milk in China and their potential health risks to breastfed infants: a review. Sci. Total. Environ. 2021, 753, 142028.
14. Serreau, R.; Terbeche, Y.; Rigourd, V. Pollutants in breast milk: a scoping review of the most recent data in 2024. Healthcare. 2024, 12, 680.
15. Massart, F.; Gherarducci, G.; Marchi, B.; Saggese, G. Chemical biomarkers of human breast milk pollution. Biomark. Insights. 2008, 3, 159-69.
16. Li, J.; Zhang, X.; Kahil, T.; et al. From regional imbalances to latecomer advantage: phosphorus pollution and economic development in the Yangtze economic belt. Ecological. Indicators. 2025, 173, 113423.
17. Zhu, S.; Jin, Y.; Deng, W.; et al. Determining factors and future trajectory of volatile organic compounds emissions in the Yangtze River Delta region of China. Environ. Impact. Assess. Rev. 2025, 112, 107793.
18. Luo, J.; Xing, X.; Wu, Y.; Zhang, W.; Chen, R. S. Spatio-temporal analysis on built-up land expansion and population growth in the Yangtze River Delta Region, China: from a coordination perspective. Appl. Geogr. 2018, 96, 98-108.
19. De Klerk, L.; Smit, N.; Yohannes, Y.; et al. Persistent organic pollutants in South African coastal ecosystems: Analysing the evileye blaasop Amblyrhynchote honckenii (Bloch, 1785) for PCB and OCP accumulation. Reg. Stud. Mar. Sci. 2025, 89, 104368.
20. Chawla, M. Chapter 7 - Environmental contamination and toxicology of benzene-hexachloride (BHC). In Hazardous chemicals; Academic Press, 2025; pp. 85-103.
21. Yin, S.; Zhang, J.; Guo, F.; et al. Supporting dataset and methods for transplacental transfer of organochlorine pesticides: concentration ratio and chiral properties. Data. Brief. 2019, 25, 104278.
22. Kumar, A.; Dayal, P.; Shukla, G.; Singh, G.; Joseph, P. E. DDT and HCH residue load in mother's breast milk: a survey of lactating mother’s from remote villages in Agra region. Environ. Int. 2006, 32, 248-51.
23. European Medicines Agency, 2011. EMA guideline on bioanalytical method validation. https://www.tga.gov.au/sites/default/files/2023-08/ewp1922172009bioanalytical_method_validationcurrent.pdf (accessed 2026-03-31).
24. Xu, C.; Yin, S.; Tang, M.; Liu, K.; Yang, F.; Liu, W. Environmental exposure to DDT and its metabolites in cord serum: distribution, enantiomeric patterns, and effects on infant birth outcomes. Sci. Total. Environ. 2017, 580, 491-8.
25. WHO. Obesity and overweight. https://www.who.int/news-room/fact-sheets/detail/obesity-and-overweight (accessed 2026-03-30).
26. China, NHC, Growth standard for children under 7 years of age; WS/T 423-2022; National Health Commission of the People’s Republic of China: Beijing, 2022. https://www.nhc.gov.cn/fzs/c100048/202211/5001d7cf57774770a1d49c1df46a291f/files/1733125082501_81468.pdf (accessed 2026-03-30).
27. Zhu, Y.; Liu, H.; Xi, Z.; Cheng, H.; Xu, X. Organochlorine pesticides (DDTs and HCHs) in soils from the outskirts of Beijing, China. Chemosphere 2005, 60, 770-8.
28. Zhang, Y., Qi, S., Xing, X., et al. Chapter 15 - Legacies of organochlorine pesticides (OCPs) in soil of China - a review, and cases in Southwest and Southeast China. In Environmental geochemistry, 2th ed.; Elsevier, 2018; pp 543-65.
29. Chen, L.; Qian, Y.; Jia, Q.; et al. A large geographic-scale characterization of organochlorine pesticides (OCPs) in surface sediments and multiple aquatic foods of inland freshwater aquaculture ponds in China: Co-occurrence, source and risk assessment. Environ. Pollut. 2022, 308, 119716.
30. Chen, C.; Yang, F.; Wu, Q.; et al. Ocean as source or sink for legacy persistent organic pollutants. J. Hazard. Mater. 2025, 486, 136987.
31. Hammel, S. C.; Andersen, H. V.; Knudsen, L. E.; Frederiksen, M. Inhalation and dermal absorption as dominant pathways of PCB exposure for residents of contaminated apartment buildings. Int. J. Hyg. Environ. Health. 2023, 247, 114056.
32. Hou, R.; Lin, L.; Li, H.; et al. Occurrence, bioaccumulation, fate, and risk assessment of novel brominated flame retardants (NBFRs) in aquatic environments - a critical review. Water. Res. 2021, 198, 117168.
33. Rawn, D. F.; Sadler, A. R.; Casey, V. A.; et al. Legacy halogenated flame retardants in Canadian human milk from the maternal-infant research on environmental chemicals study. J. Environ. Expo. Assess. 2024, 3, 16.
34. Hammel, S. C.; Vorkamp, K.; Nielsen, J. B.; Sørensen, L. S.; Knudsen, L. E.; Frederiksen, M. Novel and legacy brominated flame retardants in human breast milk and house dust from Denmark. J. Environ. Expo. Assess. 2024, 3, 8.
35. Shahmoradi, B.; Maleki, A.; Kohzadi, S.; Khoubi, J.; Zandi, S. Levels of organochlorine pesticides in human breast milk in marivan, west of iran. J. Adv. Environ. Health. Res. 2019, 7, 32-7.
36. Sanguos, C. L.; Suárez, O. L.; Martínez-Carballo, E.; Couce, M. L. Postnatal exposure to organic pollutants in maternal milk in north-western Spain. Environ. Pollut. 2023, 318, 120903.
37. Dimitriadou, L.; Malarvannan, G.; Covaci, A.; et al. Levels and profiles of brominated and chlorinated contaminants in human breast milk from Thessaloniki, Greece. Sci. Total. Environ. 2016, 539, 350-8.
38. Müller, M. H. B.; Polder, A.; Brynildsrud, O. B.; et al. Prenatal exposure to persistent organic pollutants in Northern Tanzania and their distribution between breast milk, maternal blood, placenta and cord blood. Environ. Res. 2019, 170, 433-42.
39. Antignac, J. P.; Main, K. M.; Virtanen, H. E.; et al. Country-specific chemical signatures of persistent organic pollutants (POPs) in breast milk of French, Danish and Finnish women. Environ. Pollut. 2016, 218, 728-38.
40. Herceg Romanić, S.; Milićević, T.; Jovanović, G.; et al. Persistent organic pollutants in Croatian breast milk: an overview of pollutant levels and infant health risk assessment from 1976 to the present. Food. Chem. Toxicol. 2023, 179, 113990.
41. Torres-Moreno, A. C.; Mejia-Grau, K.; Puente-DelaCruz, L.; et al. Polybrominated diphenyl ethers (PBDEs), polychlorinated biphenyls (PCBs), organochlorine pesticides (OCPs) in human breast milk from Colombia: a probabilistic risk assessment approach. Chemosphere 2023, 339, 139597.
42. Fromme, H.; Fuchs, V.; Albrecht, M.; et al. Polychlorinated dioxins and dibenzofurans (PCDD/F), polybrominated dioxins and dibenzofurans (PBDD/F), polychlorinated biphenyls (PCB), polybrominated diphenyl ethers (PBDE), and per- and polyfluoroalkyl substances (PFAS) in German breast milk samples (LUPE 8). Sci. Total. Environ. 2022, 825, 154066.
43. Darnerud, P. O.; Lignell, S.; Aune, M.; et al. Time trends of polybrominated diphenylether (PBDE) congeners in serum of Swedish mothers and comparisons to breast milk data. Environ. Res. 2015, 138, 352-60.
44. Bramwell, L.; Fernandes, A.; Rose, M.; Harrad, S.; Pless-Mulloli, T. PBDEs and PBBs in human serum and breast milk from cohabiting UK couples. Chemosphere 2014, 116, 67-74.
45. Amininejad, A. H.; Movassaghghazani, M. Risk assessment and daily intake of aldrin, dieldrin, and dichloro-diphenyl-trichloroethane through milk in Tehran, Iran. J. Food. Compos. Anal. 2025, 143, 107583.
46. Vijgen, J.; de Borst, B.; Weber, R.; Stobiecki, T.; Forter, M. HCH and lindane contaminated sites: European and global need for a permanent solution for a long-time neglected issue. Environ. Pollut. 2019, 248, 696-705.
47. Chen, L.; Qian, Y.; Jia, Q.; et al. A national-scale distribution of organochlorine pesticides (OCPs) in cropland soils and major types of food crops in China: Co-occurrence and associated risks. Sci. Total. Environ. 2023, 861, 160637.
48. Avila, B. S.; Mendoza, D. P.; Ramírez, A.; Peñuela, G. A. Occurrence and distribution of persistent organic pollutants (POPs) in the atmosphere of the Andean city of Medellin, Colombia. Chemosphere 2022, 307, 135648.
49. Gopalan, N. K.; Chenicherry, S. Fate and distribution of organochlorine insecticides (OCIs) in Palakkad soil, India. Sustain. Environ. Res. 2018, 28, 179-85.
50. Fábelová, L.; Wimmerová, S.; Šovčíková, E.; et al. Prenatal and postnatal exposure to PCBs and neurodevelopment of preschoolers living in the PCB-contaminated region. Environ. Res. 2025, 282, 122044.
51. Idowu, I. G.; Megson, D.; Tiktak, G.; Dereviankin, M.; Sandau, C. D. Polychlorinated biphenyl (PCB) half-lives in humans: a systematic review. Chemosphere 2023, 345, 140359.
52. Landrigan, P. J.; Sonawane, B.; Mattison, D.; McCally, M.; Garg, A. Chemical contaminants in breast milk and their impacts on children’s health: an overview. Environ. Health. Perspect. 2002, 110, A313-5.
53. Turner, A. PBDEs in the marine environment: sources, pathways and the role of microplastics. Environ. Pollut. 2022, 301, 118943.
54. Ohoro, C. R.; Adeniji, A. O.; Okoh, A. I.; Okoh, O. O. Polybrominated diphenyl ethers in the environmental systems: a review. J. Environ. Health. Sci. Eng. 2021, 19, 1229-47.
55. Jones, K. C. Persistent organic pollutants (POPs) and related chemicals in the global environment: some personal reflections. Environ. Sci. Technol. 2021, 55, 9400-12.
56. Chen, F.; Yin, S.; Kelly, B. C.; Liu, W. Chlorinated polyfluoroalkyl ether sulfonic acids in matched maternal, cord, and placenta samples: a study of transplacental transfer. Environ. Sci. Technol. 2017, 51, 6387-94.
57. Malik, S.; Chakraborty, D.; Agnihotri, P.; Sharma, A.; Biswas, S. Mitochondrial functioning in Rheumatoid arthritis modulated by estrogen: evidence-based insight into the sex-based influence on mitochondria and disease. Mitochondrion 2024, 76, 101854.
58. Kuang, L.; Hou, Y.; Huang, F.; et al. Pesticide residues in breast milk and the associated risk assessment: a review focused on China. Sci. Total. Environ. 2020, 727, 138412.
59. Zheng, G.; Schreder, E.; Dempsey, J. C.; et al. Per- and polyfluoroalkyl substances (PFAS) in breast milk: concerning trends for current-use PFAS. Environ. Sci. Technol. 2021, 55, 7510-20.
60. Anadón, A., Martínez-larrañaga, M. R., Ares, I., Martínez, M. A. Drugs and chemical contaminants in human breast milk. In Reproductive and developmental toxicology, 3th ed.; Elsevier, 2022; pp 1019-52.
61. Gascon, M.; Verner, M. A.; Guxens, M.; et al. Evaluating the neurotoxic effects of lactational exposure to persistent organic pollutants (POPs) in Spanish children. Neurotoxicology 2013, 34, 9-15.
62. Terzaghi, E.; Zanardini, E.; Morosini, C.; et al. Rhizoremediation half-lives of PCBs: role of congener composition, organic carbon forms, bioavailability, microbial activity, plant species and soil conditions, on the prediction of fate and persistence in soil. Sci. Total. Environ. 2018, 612, 544-60.
63. Zhang, X.; Zhang, W.; Liu, T.; et al. Levels, distribution, sources and children's health risk of PBDEs in household dust from 2010 to 2022 in China. Indoor. Environ. 2025, 2, 100114.
64. Pelletier, M.; Glorennec, P.; Mandin, C.; et al. Chemical-by-chemical and cumulative risk assessment of residential indoor exposure to semivolatile organic compounds in France. Environ. Int. 2018, 117, 22-32.
65. Kodavanti, P. R. S., Loganathan, B. G. Polychlorinated biphenyls, polybrominated biphenyls, and brominated flame retardants. In Biomarkers in toxicology; Elsevier, 2019; pp 501-18.
66. Sehgal, N.; Morello-Frosch, R.; Padula, A. M.; et al. Mixture effects of prenatal exposure to polybrominated diphenyl ethers on urinary oxidative stress biomarkers in the chemicals in our bodies cohort. Am. J. Epidemiol. 2025, 194, 1507-14.
67. Liu, M.; Cheng, S.; Ou, D.; et al. Organochlorine pesticides in surface sediments and suspended particulate matters from the Yangtze estuary, China. Environ. Pollut. 2008, 156, 168-73.
68. Ebsa, G.; Gizaw, B.; Admassie, M.; Degu, T.; Alemu, T. The role and mechanisms of microbes in dichlorodiphenyltrichloroethane (DDT) and its residues bioremediation. Biotechnol. Rep. 2024, 42, e00835.
69. Yu, H.; Lin, T.; Hu, L.; et al. Sources of polychlorinated biphenyls (PCBs) in sediments of the East China marginal seas: role of unintentionally-produced PCBs. Environ. Pollut. 2023, 338, 122707.
70. Jahnke, J. C.; Martinez, A.; Hornbuckle, K. C. Distinguishing aroclor and non-aroclor sources to Chicago air. Sci. Total. Environ. 2022, 823, 153263.
71. Zhai, L.; Zhao, J.; Zhu, Y.; et al. Downregulation of leptin receptor and kisspeptin/GPR54 in the murine hypothalamus contributes to male hypogonadism caused by high-fat diet-induced obesity. Endocrine 2018, 62, 195-206.
72. Da, C.; Wang, R.; Huang, Q.; et al. Sediment records of polybrominated diphenyl ethers (PBDEs) from the Anhui province section of Yangtze River, China. Bull. Environ. Contam. Toxicol. 2021, 106, 334-41.
73. Tokarz JA, 3. R. D.; Ahn, M. Y.; Leng, J.; Filley, T. R.; Nies, L. Reductive debromination of polybrominated diphenyl ethers in anaerobic sediment and a biomimetic system. Environ. Sci. Technol. 2008, 42, 1157-64.
74. Chen, J.; Wang, C.; Pan, Y.; Farzana, S. S.; Tam, N. F. Biochar accelerates microbial reductive debromination of 2,2’,4,4’-tetrabromodiphenyl ether (BDE-47) in anaerobic mangrove sediments. J. Hazard. Mater. 2018, 341, 177-86.
75. Wang, Y.; Feng, Y.; Chen, Y.; et al. Annual flux estimation and source apportionment of PCBs and PBDEs in the middle reach of Yangtze River, China. Sci. Total. Environ. 2023, 885, 163772.
76. Wang, G.; Liu, Y.; Tao, W.; Zhao, X.; Li, X. Reflection of concentrations of polybrominated diphenyl ethers in health risk assessment: A case study in sediments from the metropolitan river, North China. Environ. Pollut. 2019, 247, 80-8.
77. Shim, M.; Lee, J. Y.; Joung, S. K.; et al. Simultaneous determination of 12 novel brominated flame retardants and their potential exposure via the dietary intake of processed foods in Korea. Food. Chem. 2025, 494, 142733.
78. Yuan, T. H.; Tai, C. J.; Tsai, C. H.; et al. Exploring the influence of PCB exposure on neonatal birth outcomes and neurobehavioral development after 15 years of prohibition. Environ. Pollut. 2025, 368, 125761.
79. Wang, Y.; Wang, Q.; Zhou, L.; et al. Metabolomics insights into the prenatal exposure effects of polybrominated diphenyl ethers on neonatal birth outcomes. Sci. Total. Environ. 2022, 836, 155601.
80. Morley, L. C.; Debant, M.; Walker, J. J.; Beech, D. J.; Simpson, N. A. B. Placental blood flow sensing and regulation in fetal growth restriction. Placenta 2021, 113, 23-8.
81. Dewan, P.; Jain, V.; Gupta, P.; Banerjee, B. D. Organochlorine pesticide residues in maternal blood, cord blood, placenta, and breastmilk and their relation to birth size. Chemosphere 2013, 90, 1704-10.
82. Han, B.; Wang, L.; Wang, X.; et al. Association between multipollutant exposure and thyroid hormones in elderly people: a cross-sectional study in China. Environ. Res. 2024, 252, 118781.
83. Park, C. M.; Kim, K. T.; Rhyu, D. Y. Low-concentration exposure to organochlorine pesticides (OCPs) in L6 myotubes and RIN-m5F pancreatic beta cells induces disorders of glucose metabolism. Toxicol. In. Vitro. 2020, 65, 104767.
84. Hong, S.; Kim, J. Y.; Hwang, J.; Shin, K. S.; Kang, S. J. Heptachlor induced mitochondria-mediated cell death via impairing electron transport chain complex III. Biochem. Biophys. Res. Commun. 2013, 437, 632-6.
85. Sheikh, I. A.; Beg, M. A. Structural studies on the endocrine-disrupting role of polybrominated diphenyl ethers (PBDEs) in thyroid diseases. Environ. Sci. Pollut. Res. Int. 2020, 27, 37866-76.
86. Chen, L.; Wang, C.; Cui, C.; et al. Prenatal exposure to polybrominated diphenyl ethers and birth outcomes. Environ. Pollut. 2015, 206, 32-7.
87. Ison, E. K.; Kent-Dennis, C. E.; Fazioli, J.; Mulligan, M. K.; Pham, A.; Pasternak, J. A. Compensatory mechanisms in response to induced hypothyroidism in the late gestation pig fetus†. Biol. Reprod. 2023, 108, 731-43.
88. Sharma, N.; Garg, D.; Deb, R.; Samtani, R. Toxicological profile of organochlorines aldrin and dieldrin: an Indian perspective. Rev. Environ. Health. 2017, 32, 361-72.
89. Goasdoué, K.; Miller, S. M.; Colditz, P. B.; Björkman, S. T. Review: the blood-brain barrier; protecting the developing fetal brain. Placenta 2017, 54, 111-6.
90. Drakvik, E.; Altenburger, R.; Aoki, Y.; et al. Statement on advancing the assessment of chemical mixtures and their risks for human health and the environment. Environ. Int. 2020, 134, 105267.
91. Deoni, S. Dean D, 3. R. D.; Joelson, S.; O'Regan, J.; Schneider, N. Early nutrition influences developmental myelination and cognition in infants and young children. Neuroimage 2018, 178, 649-59.
92. US-EPA - United States Environmental Protection Agency, 2008. Child-specific exposure factors handbook. EPA/600/R-06/096F, 2008. U.S. Environmental Protection Agency, Washington, DC. https://ordspub.epa.gov/ords/eims/eimscomm.getfile?p_download_id=484738 (accessed 2026-03-30).
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.