Synergistic catalytic strategies for multipollutant control
Abstract
The synergistic catalytic removal of multipollutants, such as nitrogen oxides and volatile organic compounds, has emerged as a transformative strategy for air pollution control. It directly tackles the interconnected challenges of PM2.5 and ozone formation. This mini review summarizes recent breakthroughs in this field, encompassing the design of novel catalysts with multifunctional active sites, the elucidation of the kinetic mechanisms underlying synergistic reactions, and the development of anti-poisoning strategies to maintain activity under complex flue gas conditions. We further examine how tailored acid-redox bifunctionality and dynamic active-site cooperation can overcome inherent issues such as temperature-window mismatches and competitive adsorption between pollutant molecules. Finally, by integrating advances in operando characterization and artificial intelligence, we propose a forward-looking roadmap for synergistic catalysis. This roadmap aligns with carbon neutrality goals and highlights the critical role of catalytic innovations in enabling clean and efficient environmental protection.
Keywords
INTRODUCTION
Nitrogen oxides (NOx) and volatile organic compounds (VOCs), primarily emitted from waste incineration, coal-fired plants, and chemical industries, are critical precursors to secondary pollutants such as PM2.5 and ozone[1]. These two classes of pollutants exhibit distinct molecular characteristics and chemical behaviors. NOx (including NO and NO2) possesses oxidizing properties and is typically mitigated via reductive pathways. In contrast, VOCs act as reducing agents and are therefore eliminated through oxidative processes. Conventional end-of-pipe technologies, such as selective catalytic reduction (SCR) for NOx and catalytic oxidation for VOCs, face several intrinsic limitations[2]. The first is a temperature-window mismatch. Commercial V2O5-WO3/TiO2 catalysts for NH3-SCR achieve peak NOx reduction at 300-400 °C, while complete oxidation of VOCs generally requires temperatures above 400 °C[3]. The second is competitive adsorption, where VOC molecules occupy the acid sites essential for activating NH3 in the SCR process, thereby reducing NOx conversion efficiency. The third challenge is the risk of secondary pollution. For instance, chlorinated VOCs (CVOCs) can generate HCl or even dioxins during oxidation, while sulfur-containing VOCs (SVOCs) produce SO2, which poisons SCR catalysts and complicates flue gas treatment. Given these challenges, synergistic abatement has become imperative for effective multipollutant control. Synergistic catalysis, which integrates the removal of both pollutants into a single-unit operation, can reduce energy consumption and capital costs[4,5]. This mini review is structured around three pillars: synergistic catalyst innovation, profound mechanistic insights, and demonstrated robustness under complex conditions.
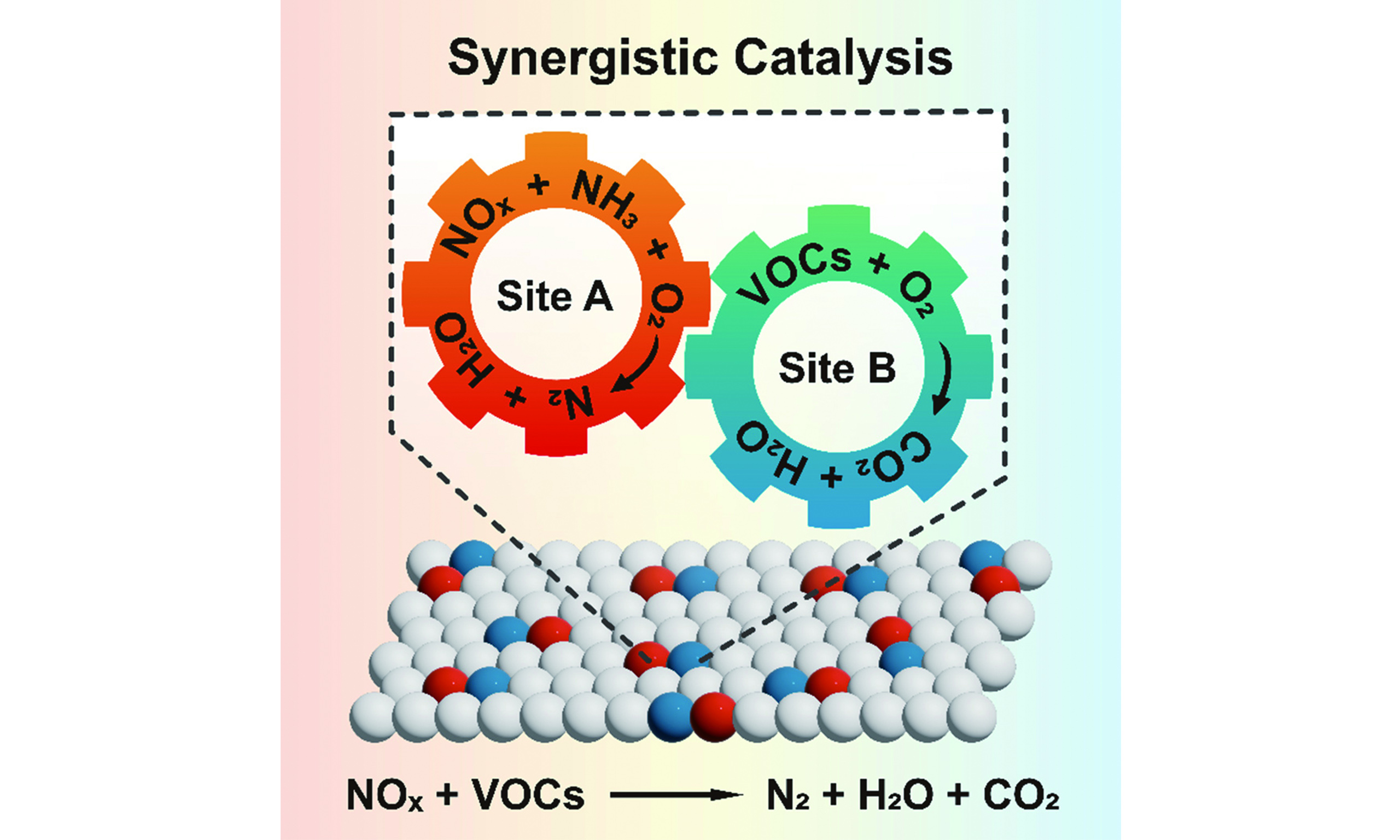
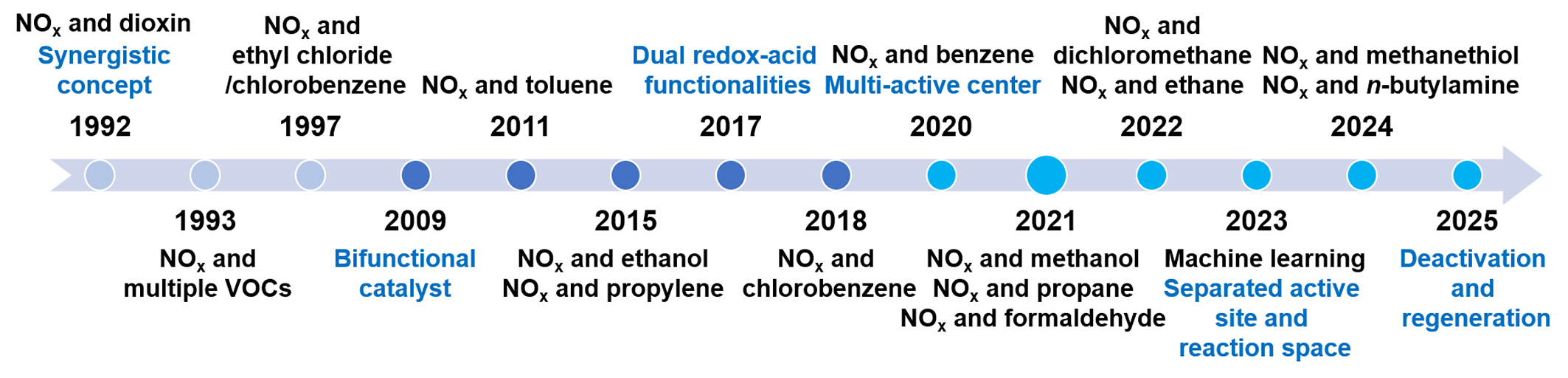
The coexistence of NOx and VOCs in waste incineration flue gas spurred the development of synergistic catalysis. This paradigm aims at the concurrent abatement of diverse pollutants [Figure 1]. The concept was first introduced for dioxin destruction by Carlsson, K. B. in 1992, using an NH3-SCR catalyst in waste incinerators[6]. Early research focused on modifying conventional vanadium-tungsten-titanium (V-W-Ti) SCR catalysts, initially designed for NOx reduction, to simultaneously oxidize dioxins. Notably, Swedish incineration plants demonstrated the feasibility of achieving ultralow dioxin emissions [< 0.1 ng TEQ (toxicity equivalency quantity)/Nm3] via dry scrubbing/fabric filter systems. Concurrently, laboratory studies showed that V2O5-WO3/TiO2 honeycomb catalysts could efficiently decompose dioxins, chlorobenzene (CB), NOx, and odorous compounds at 200-400 °C[7]. Despite these advances, the limitations of vanadium-based catalysts, including toxicity risks and a narrow operational temperature window, prompted the search for alternatives. This first developmental phase (circa 1992-2008) was characterized by empirical performance screening of V2O5-based SCR catalysts under complex multipollutant conditions, with mechanistic understanding remaining limited. A pivotal shift occurred during the second phase (circa 2009-2019), driven by advances in catalyst synthesis[8] and in situ characterization techniques. These advancements enabled the targeted design of multifunctional catalysts and facilitated deeper mechanistic probing[9]. Researchers began employing model VOC molecules (e.g., CB as a dioxin surrogate in waste incineration flue gas[10-12], toluene in coal-fired flue gas[13,14]) to decouple and elucidate synergistic pathways. Catalyst design also diversified beyond traditional V2O5-based catalysts to include manganese-based[10,11,15,16], copper-based[8-14], ceria-based[17], layered aluminosilicates[18], zeolites[19], and their composites[20]. Mechanistically, the importance of tailoring dual redox-acid functionalities was established and evidenced in catalysts such as VOx/TiO2/ZSM5 (Zeolite Socony Mobil-5) for the simultaneous abatement of polychlorinated dibenzo-p-dioxins/dibenzofurans (PCDD/Fs) and NOx[21]. The current period (2020-present) has witnessed exponential growth in synergistic catalysis research, fueled by stringent environmental policies and innovations in catalyst design. Recent studies highlight environmentally benign formulations (for example, atomically dispersed asymmetric Mn-O-Ce sites on montmorillonite[22]) that achieve simultaneous removal efficiency above 90%. This era is defined by atomic-scale mechanistic studies and scalable reactor configurations, heralding a new frontier in cooperative pollutant control.
THE SCOPE OF SYNERGISTIC REACTION IN ENVIRONMENTAL CHEMISTRY
As shown in Figure 2, research on model reactions for synergistic multipollutant control in flue gas currently focuses on two primary systems: the simultaneous elimination of NOx with CB, and that with toluene. In recent years, increased attention has also been directed toward the synergistic removal of NOx with other VOCs.
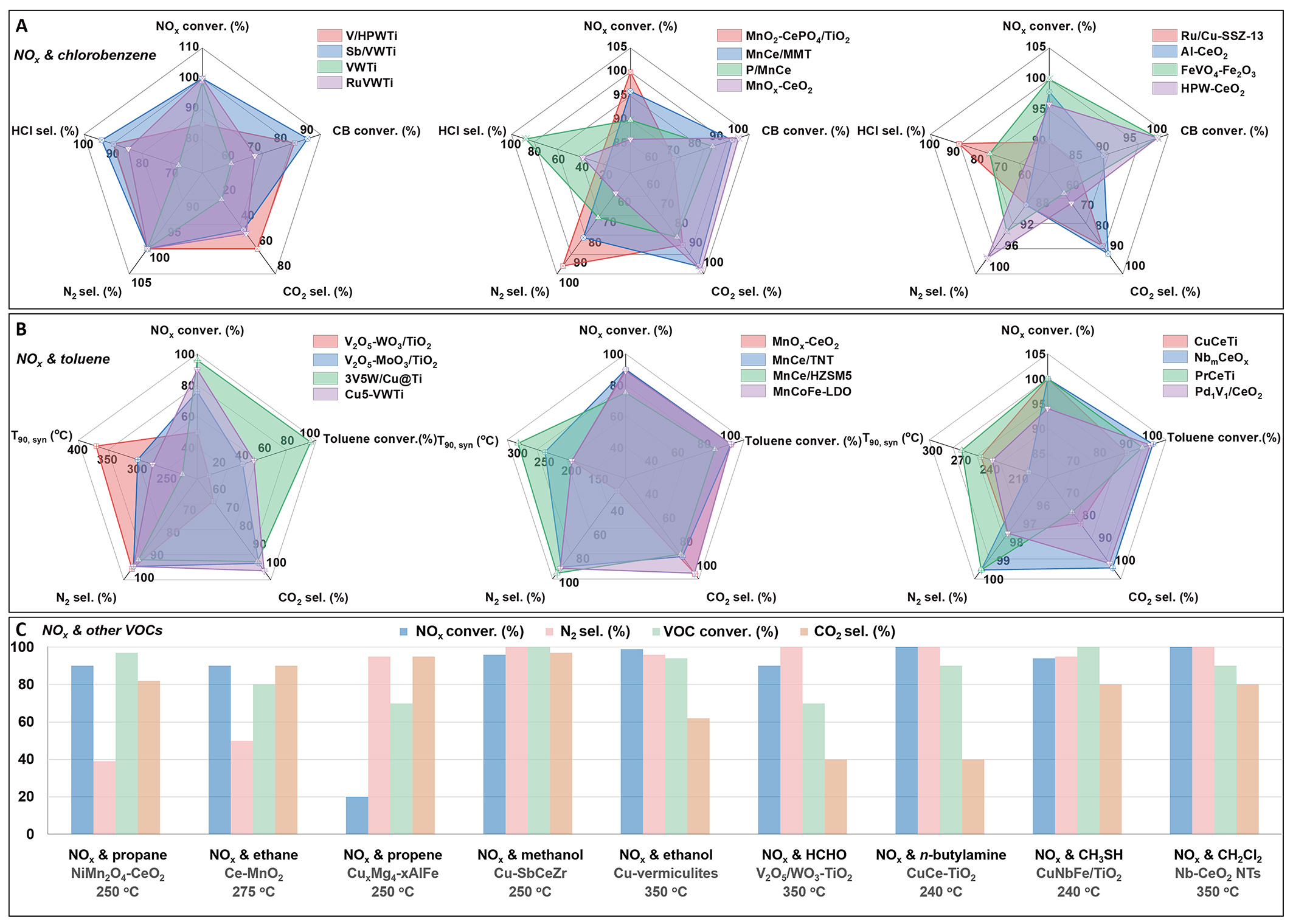
Figure 2. Advances in synergistic catalytic conversion of NOx and different VOCs on typical catalysts with optimized performance. (A) NOx reduction with CB oxidation at 300 °C over typical V-Ti-based[23-26], Mn-Ce-based[10,11,22,27,30,31], and novel catalysts[32-36]; (B) NOx reduction with toluene oxidation at 250 °C over V-Ti-based[46-49], Mn-Ce-based[51-55], and other optimized CeO2-based catalysts[56-59]; (C) NOx reduction with other VOCs in typical catalysts with optimal synergistic catalytic performance under proper conditions[60-68]. NOx: Nitrogen oxides; VOCs: volatile organic compounds; CB: chlorobenzene; TNT: TiO2 nanotubes; HZSM5: hydrogen form of zeolite Socony Mobil-5; LDO: layered double hydroxide.
Synergistic control of NOx and CB
Systematic studies on the synergistic removal of NOx and CVOCs are increasingly conducted, primarily to avoid the high direct biotoxicity of dioxins/furans. The NOx-CB model system originates from the demand for synergistic catalytic purification of NOx and dioxins under waste incineration conditions. CB shares structural similarities with dioxins[23], serves as a precursor in their formation, and is widely adopted as a model molecule for investigating both catalyst development and catalytic mechanisms for the co-removal of NOx and CVOCs. Synergistic catalysts for NOx-CB purification can be classified into three categories [Figure 2A]: V-W-Ti-based, manganese-cerium (Mn-Ce) based, and other novel catalytic systems. Despite their excellent NOx removal performance, conventional V2O5-WO3/TiO2 catalysts suffer from insufficient CB oxidation at typical denitrification temperatures (< 60% conversion at
Synergistic control of NOx and toluene
Toluene is a major precursor for the formation of tropospheric ozone and secondary organic aerosols. The NOx-toluene model system primarily addresses the co-purification of NOx and VOCs in coal-fired power plants and the iron/steel smelting industry[4,46,47]. Synergistic catalysts for NOx-toluene purification can also be classified into three categories [Figure 2B]. Conventional V2O5-WO3 (MoO3)/TiO2 catalysts exhibit limited synergistic performance, with temperature at 90% conversion (T90) temperatures exceeding 300 °C[48,49]. Cu-modified VW/Cu-Ti catalysts with optimized structures demonstrate significantly enhanced activity[4,50]. MnOx-CeO2 catalysts achieve lower operating temperatures (around 200 °C) but also face challenges with low N2 selectivity[51,52]. Structural optimizations, such as the use of TiO2 nanotubes[53], H-ZSM5 composites[54], or layered metal oxides[55], can substantially improve both N2 selectivity and overall conversion efficiency. Notably, modified CeO2 catalysts[56-59], particularly NbmCeOx solid solutions formed by Nb-doped CeO2, exhibit exceptional performance. They achieve a T90 as low as
Synergistic control of NOx and other VOCs
With the growing emphasis on multipollutant control strategies, research attention is expanding to other synergistic catalytic systems beyond NOx-CB and NOx-toluene. These include the co-removal of NOx with various types of VOCs, such as alkanes (e.g., ethane[60], propane[61], and propylene[4,20]), alcohols (e.g., methanol[62,63] and ethanol[18]), aldehydes (e.g., formaldehyde[64]), heteroatom-containing VOCs (e.g., n-butylamine[65] and methanethiol[66,67]), and emerging pollutants (e.g., dichloromethane[4,68]), as summarized in Figure 2C. The general principles underpinning synergistic catalysis aim to simultaneously achieve high conversion and selectivity for multiple pollutants across a broad temperature window.
CURRENT FRONTIERS IN SYNERGISTIC CATALYST DESIGN
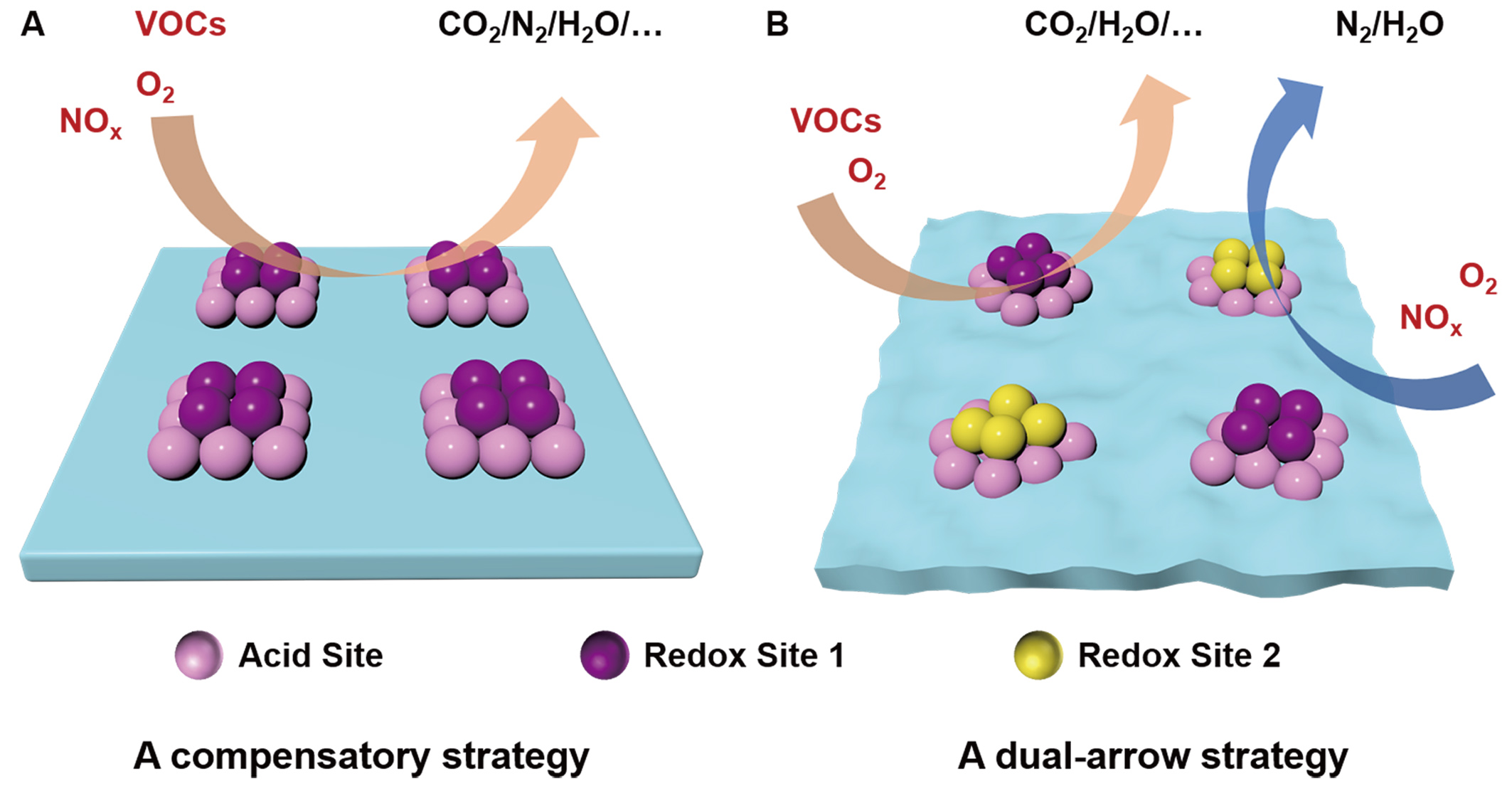
Achieving synergistic catalytic purification of NOx and VOCs hinges on the development of efficient catalysts capable of driving multiple reactions simultaneously. Conventional catalysts are typically designed for a single, specific reaction. In contrast, effective multipollutant control demands a single catalyst system that can concurrently and selectively convert two or more pollutants, with the individual reaction pathways enhancing rather than inhibiting one another. The core design challenge lies in reconciling the disparate, and often seemingly contradictory, requirements of the constituent reactions. For instance, traditional NOx removal via NH3-SCR and VOC purification via catalytic oxidation appear fundamentally opposed. The former requires a balanced combination of relatively strong acidity and moderate redox properties, while the latter necessitates robust redox capabilities. Although both processes involve acid cycles (on acidic sites) and redox cycles (on redox sites), providing a theoretical basis for synergy, their specific demands for the nature and strength of these active sites are inherently asymmetric. The primary challenge, therefore, is to balance these asymmetric demands within a unified catalytic architecture. Consequently, designing high-performance synergistic catalysts requires a strategy that finds common ground between the different reaction requirements while preserving their distinct catalytic functions. Current design philosophies can be broadly categorized into two approaches: the traditional compensatory strategy and the emerging dual-arrow strategy [Figure 3].
Figure 3. Strategies in the synergistic catalyst design. (A) The compensatory strategy; (B) The dual-arrow strategy. NOx: Nitrogen oxides; VOCs: volatile organic compounds.
Traditional strategies for synergistic catalysts design
Traditionally, the predominant strategy for achieving synergy has been to modify the catalytic sites of existing materials to meet the distinct requirements of both the SCR and VOC oxidation reactions[21,26,30-32,34,45,57,69]. Early research on the synergistic conversion of NOx and VOCs primarily focused on modifying commercial V2O5-TiO2 denitration catalysts[7,70-73]. To address the inefficiency in simultaneous conversion under co-feed conditions, various modifications were implemented to balance the necessary acidity and redox properties. These included adjusting vanadium oxide content and geometry[41,45], doping with other metal promoters (e.g., Ce, Fe, Co, Pd, Ru, Cu, Pr)[4,13,26,69,74-79], compositing with zeolites[21] or other oxides, such as Al2O3[80], and acid treatment[25]. This represents the compensatory strategy[Figure 3A], where synergy is achieved by narrowing the functional gap between ideal SCR and VOC oxidation catalysts. For example, V2O5-TiO2-based NH3-SCR catalysts possess inherent oxidative capacity insufficient for efficient VOC oxidation. Modifications such as doping with redox-active metals (e.g., Ce or Cu) enhance oxidation activity and improve VOC conversion[4,5]; however, this often comes at the cost of reduced N2 selectivity in the SCR. Conversely, conventional VOC oxidation catalysts exhibit overly strong redox properties coupled with insufficient acidity for effective NH3-SCR[10,11]. Acidification treatments can boost acidity and moderate redox activity, but this risks lowering VOCs conversion rates and selectivity[31]. Thus, the conventional compensatory approach facilitates synergistic reactions by partially sacrificing the optimal performance of either the SCR or the VOC oxidation function, embodying a trade-off. This strategy has been extensively reviewed for traditional V-Ti and Mn-Ce catalysts designed for NOx and VOC degradation[81].
Emerging strategies for synergistic catalysts design
An alternative and evolving strategy employs dual-functional active sites to drive synergistic reactions, representing a current and future research trend. This approach, termed the dual-arrow strategy [Figure 3B], moves beyond compensatory modifications. It involves first delineating the distinct catalytic property requirements for NH3-SCR and VOC oxidation reactions. Efforts are then focused on the rational design and construction of differentiated active sites within a single catalyst, with each site type tailored to promote one of the target reactions in parallel. A key implementation of this strategy is the construction of three-dimensional hierarchical carriers, such as tubular hierarchically porous, core-shell, or morphologically tailored architectures. Acidic and oxidative active sites with tailored strengths are subsequently engineered onto distinct surfaces or regions of these carriers. This design enables a spatially segregated yet cooperative arrangement of the differentiated active sites. Spatial isolation allows redox-mediated and acid-mediated reactions to proceed concurrently with minimal mutual interference. Consequently, this architecture facilitates efficient and precise synergistic conversion of NOx and VOCs, moving closer to the goal of truly efficient, multipollutant abatement.
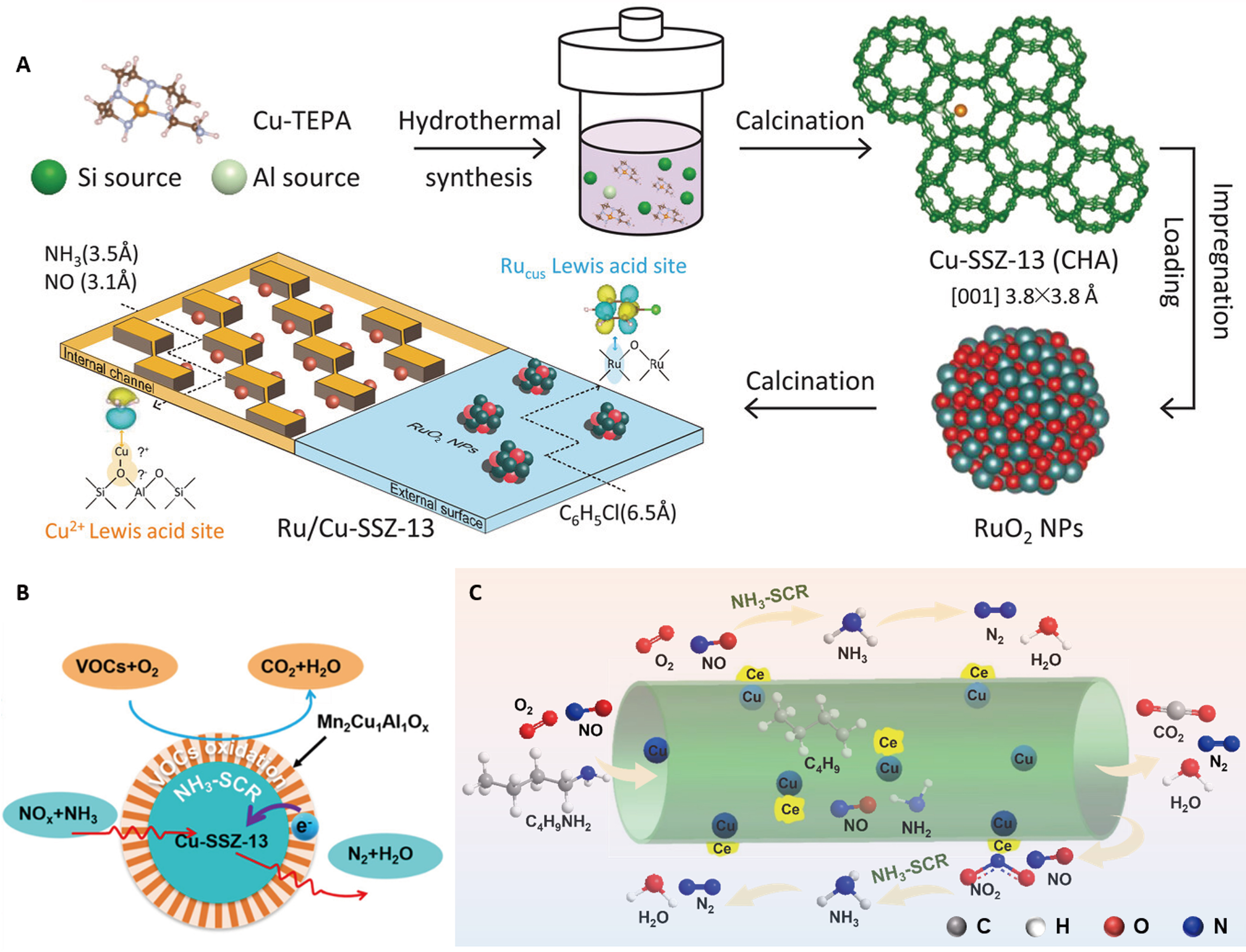
Dual-functional catalysts exemplify the dual-arrow strategy in practice. For instance, in a Cu-Ce/TiO2 catalyst, separated Cu sites selectively oxidize toluene to CO2 (> 95% conversion at 250 °C), while Ce sites facilitate NH3-SCR, achieving N2 selectivity exceeding 95%[59]. The electronic coupling between the Cu2+/Cu+ and Ce4+/Ce3+ redox cycles accelerates oxygen transfer, thereby suppressing the formation of partial oxidation byproducts (e.g., benzaldehyde)[59]. In situ DRIFTS (diffuse reflectance infrared Fourier transform spectroscopy) studies confirm that toluene is adsorbed on Cu sites, whereas NH3 is activated on Ce Lewis acid sites. Ru/Cu-SSZ-13 is another representative dual-functional catalyst[36]. The parent Cu-SSZ-13 zeolite support (Si/Al = 15) was first synthesized via the copper-amine complex templating method. Subsequently, RuO2 nanoparticles were loaded onto the acid-washed support via impregnation, yielding the Ru/Cu-SSZ-13 catalyst. The spatial separation of the dual Ru/Cu Lewis acid sites, with Ru species located externally and Cu sites confined within the zeolite channels, creates isolated active sites and distinct reaction spaces [Figure 4A][36]. This configuration effectively disentangles the competing interactions between the catalyst and different reactants, thereby significantly enhancing selectivity toward low-toxicity products[36]. This principle of spatial and functional segregation is further demonstrated in other systems[28]. For the synergistic catalytic elimination of NOx and CB, a bifunctional Cu-SSZ-13@-Mn2Cu1Al1Ox core-shell catalyst was designed, achieving superior activity for simultaneous pollutant removal [Figure 4B][82]. In another case, the synergistic removal of NOx and n-butylamine was accomplished via spatially separated cooperative sites on a Cu-Ce/TiO2 [Figure 4C][65]. This catalyst was prepared by a conventional precipitation method that exploits the differential binding affinity of Cu and Ce ions toward the inner versus outer surfaces of TiO2 nanotubes. Catalytic oxidation of n-butylamine primarily occurs at the Cu sites located inside the tube, while the NH3-SCR reaction proceeds undisturbed on the outer Ce sites within a compatible temperature window. Similarly, a 3V5W (V2O5 + WO3 + CuO + TiO2 catalyst with 3 wt% V2O5, 5 wt% WO3 and 1 wt% CuO)/Cu@Ti catalyst featuring this reaction site-isolation (RSI) function was successfully synthesized. In this architecture, NH3 is preferentially adsorbed on the outer layer modified with V-W, while toluene is adsorbed on the inner layer loaded with Cu. This RSI design allows NH3 and toluene to react on distinct active sites, substantially reducing undesirable byproduct formation[50].
Figure 4. Advanced strategies of designing synergistic catalysts with dual-functional active sites for parallel reactions. (A) Separated active site and reaction space over Ru/Cu-SSZ-13 for synergistic catalytic elimination of NOx and CB[36]; (B) Bifunctional Cu-SSZ-13@Mn2Cu1Al1Ox core-shell catalyst for the co-removal of NOx and toluene[82]; (C) The CuCe-TiO2 NTs catalyst with spatially separated sites for the synergistic removal of NOx and n-butylamine[65]. Figure 4A is reprinted with permission from Ref.[36], Copyright © 2023 WILEY. Figure 4B is reprinted with permission from Ref.[82], Copyright © 2023 American Chemical Society. Figure 4C is reprinted with permission from Ref.[65], Copyright © 2024 American Chemical Society. Ru/Cu-SSZ-13: Copper-siliceous special zeolite-13 supported Ru; NOx: nitrogen oxides; CB: chlorobenzene; NTs: nanotubes; TEPA: tetraethylenepentamine; CHA: chabazite; NPs: nanoparticles.
Atomic-level dispersion of complex oxides is an emerging frontier in synergistic catalysis. While composite oxides such as V2O5-WO3-TiO2 and MnO2-CeO2 are commonly engineered for multipollutant removal, and strategies such as additive doping have been extensively explored, achieving precise control over the dispersion of active components remains a significant challenge, ranging from bulk phases down to isolated atoms[22]. Recent advances highlight the potential of this approach. For instance, a MnCeOx/MMT-Ti (TiO2 pillared montmorillonite) catalyst featuring atomically dispersed MnCeOx on montmorillonite has been developed. The isolated, asymmetric Mn-O-Ce sites in this architecture provide exceptional capabilities for O2 adsorption and activation, driving the synergistic conversion of CB and NOx[21]. Moreover, this atomically dispersed catalyst exhibits excellent long-term stability even under industrial flue gas conditions containing complex impurities such as heavy metals and moisture. Similarly, a dual single-atom catalyst, Pd1V1/CeO2, has been designed for NOx and toluene co-removal[58]. A key design strategy involved saturating Pd single atoms with ligands from acidic transition metal precursors. This approach successfully stabilizes the coexistence of Pd and V dual single atoms. The resulting structure significantly suppresses lattice oxygen activation, thereby preventing the formation of catalytically inactive nitrate species. Collectively, these studies represent a breakthrough, introducing an innovative paradigm for designing highly efficient synergistic catalysts. This strategy centers on engineering atomically dispersed noble- or transition-metal active sites on complex oxide supports, offering a promising pathway for the simultaneous purification of NOx and VOCs.
Machine learning (ML) holds significant potential for advancing the design of synergistic catalysts. While substantial experimental efforts have been devoted to developing bifunctional catalysts for the simultaneous elimination of NOx and VOCs, a major bottleneck persists. The acidity and redox properties of catalysts still lack quantitative descriptors. This knowledge gap poses a significant challenge, hindering the rational and knowledge-driven design of optimal catalysts. To bridge this gap, a ML strategy informed by domain knowledge has been proposed for the design and discovery of catalysts for the co-removal of NOx and CB[83]. First, symbolic regression (SR) is employed to derive an analytical expression capable of predicting the acidity of metal ions. Second, several ML regression models are developed to evaluate the redox ability, characterized by the standard reduction potential (SRP) of metallic ions. By integrating these dual-property predictions, the framework can recommend promising bifunctional catalyst candidates. This ML-driven methodology provides effective, analytical, and quantifiable guidance for catalyst design. It thus represents a promising avenue for accelerating discovery in the field of synergistic catalysis.
SYNERGISTIC CATALYTIC MECHANISM OF MULTIPOLLUTANT CO-REMOVAL
A thorough comprehension of NOx reduction and VOC oxidation mechanisms is essential for designing rational catalysts and achieving high co-removal efficiency. Achieving optimal performance in these synergistic systems is challenging, primarily due to the intricate interactions between multiple reactants and catalytic active sites[84]. A critical factor is the co-adsorption dynamics of NOx and VOCs on catalytic surfaces, as their interactions can either synergize to enhance overall efficiency or compete for the same active sites, thereby limiting it. The simultaneous progression of two distinct reaction pathways within a shared spatiotemporal framework creates complex synergistic interactions, making mechanistic elucidation significantly more difficult. Current research has identified three dominant interaction scenarios: mutual inhibition, mutual promotion, and asymmetric promotion. The specific effect that a given VOC exerts on the SCR process is governed by multiple factors[5,81], including the type of catalyst, the chemical nature of the VOC, the NO/NH3 ratio, and the reaction intermediates formed. A key mechanistic issue in this multipollutant catalysis is understanding the migration and transformation pathways of carbon and heteroatom (Cl, N, S, etc.) species.
Synergistic catalytic mechanism of NOx and CB co-removal
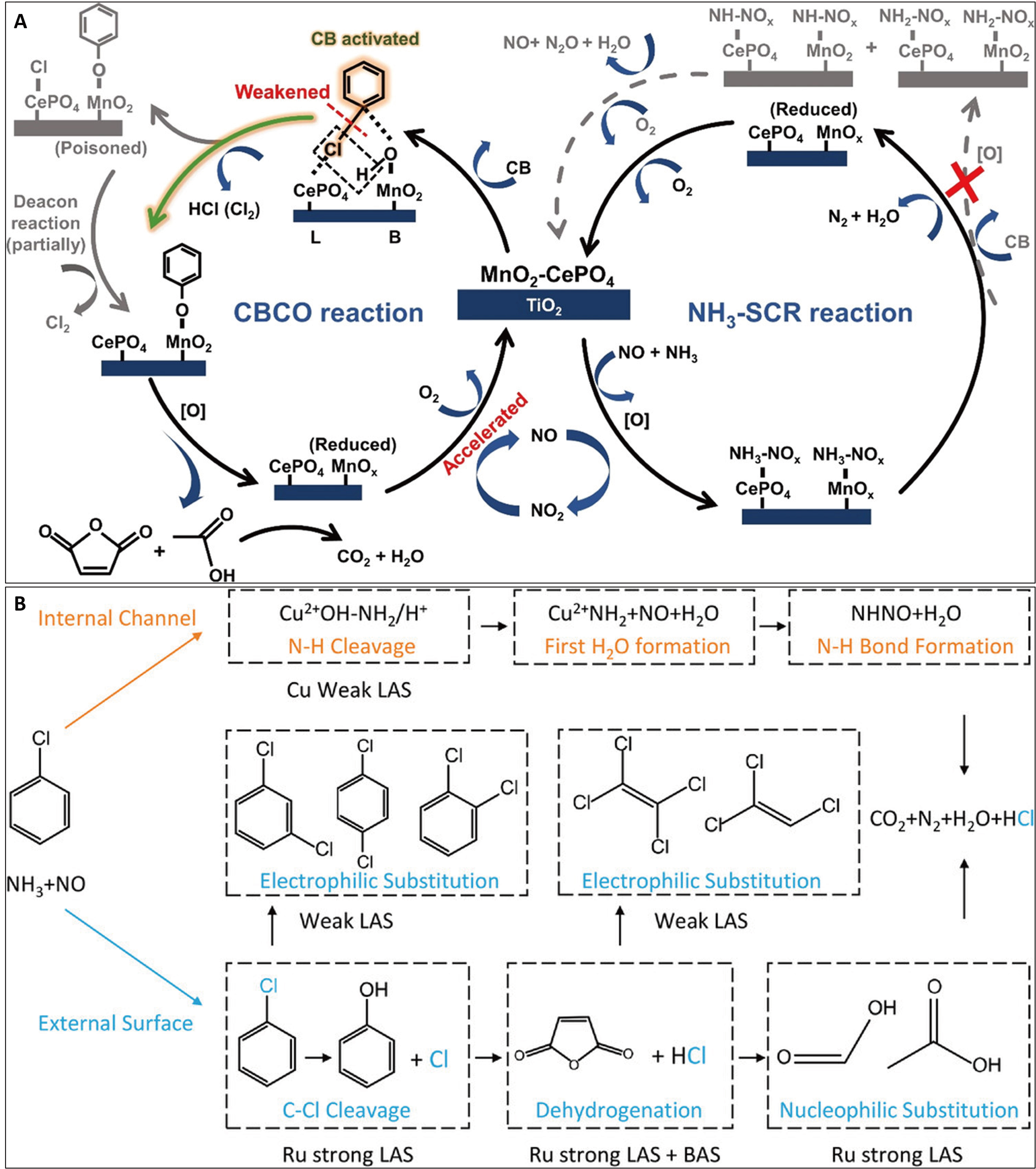
The synergistic reaction mechanism for the co-removal of NOx and CB has been extensively studied. The catalytic oxidation of CB primarily involves the cleavage of C−H and C−Cl bonds. Since the C−Cl bond has a lower bond energy than the C−H bond, it is more susceptible to nucleophilic attack, leading to the dissociative adsorption of Cl- species on the catalytic active sites. For V2O5-based catalysts, vanadium can exist in three structural forms (monomeric, polymeric, and crystalline), regulated by surface coverage density[41,81,85]. CB adsorption primarily occurs on V−OH bonds in monomeric domains and V=O bonds in polymeric domains. Monomeric vanadyl species enhance CB oxidation activity[86]. Achieving a dual redox-acid balance through the proper vanadium structure is practicable. In general, monomeric vanadium (VO4) exhibits strong dichlorination capacity but weak hydrocarbon activation, whereas polymeric/crystalline vanadium shows superior performance in NH3-SCR and aromatic oxidation. However, competitive adsorption between CVOCs and NH3 on polymeric acid sites can reduce CVOC conversion, especially at high temperatures[81]. NO can exert a positive effect on CB oxidation by promoting the reoxidation of the VOx phase via NO2 over VOx/TiO2 catalysts[72,73]. While the SCR influences CB elementary steps, this promotion is often asymmetric. SCR can enhance CB oxidation, leading to increased formation of phenolate intermediates on VOx/TiO2 and V2O5-WO3/TiO2 catalysts[86-88]. Conversely, CB addition typically decreases SCR activity, partly by forming inactive NH4Cl species and inhibiting the formation of key SCR intermediates such as cis-N2O22-. Dissociated Cl- can bind to vanadium, activating adjacent bridging hydroxyl groups on V−OH−Ti bonds and thereby enhancing surface Brønsted acidity. The presence of NO and O2 generally favors deep CB oxidation, while NH3 tends to inhibit it[88]. On V2O5-MoO3/TiO2 catalysts, SCR decreases CB conversion due to the competitive adsorption of NH3 ( a fast process) and CB (a slow process), whereas CB oxidation has a lesser impact on SCR[76,89]. Many modified VOx/TiO2 or V2O5-WO3/TiO2 catalysts exhibit a mutual inhibition effect. For instance, Ru- and Pr-modified V2O5-WO3/TiO2 catalysts show this behavior[26,79]. Ru modification, which stabilizes oxygen vacancies on Ru-O-Ti linkages and accelerates benzoate ring-opening, can slightly decrease SCR performance below 325 °C by altering the energy barriers of critical steps in CB oxidation and byproduct formation[26]. In PdV/TiO2 catalysts, competitive adsorption of NO and NH3 on active sites hinders CB oxidation. However, introducing protonated sulfate (-HSO4) groups can induce a mutual promotion effect[90]. The protonated sulfate promotes the formation of polymeric vanadyl species with a higher oxidation state and d-band center of V. This, via a “V 3d-O 2p-S 3p” electronic network, substantially alleviates competitive adsorption, strengthens Brønsted acid sites, and promotes C−Cl bond cleavage. For most MnO2-CeO2-based catalysts, optimal multipollutant activity is exhibited at 200-300 °C[10,11,16,22,28,31,91]. CB can enhance SCR efficiency by suppressing side reactions such as NH3 over-oxidation, while NO/NH3 promotes CB oxidation via NO2-mediated pathways, as demonstrated on a MnO2-CePO4/TiO2 catalyst [Figure 5A][31]. Introducing CePO4 modulates the acid properties of the MnO2-TiO2 catalyst, constructing compensated acid-redox sites. Under the cooperation of Lewis and Brønsted acid sites and balanced redox ability, CB oxidation is promoted alongside synergistically improved NH3-SCR performance above 240 °C[31]. Phosphate modification provides a novel dual-molecule modulation strategy, improving surface Cl- dissociation and HCl formation[30,31]. A positive effect of CB on SCR at 200 °C is also evidenced on a Mn6Co4Ox catalyst[92]. Overall, on catalysts with a balanced distribution of Lewis acid, Brønsted acid, and redox sites, achieving an acid-redox equilibrium, the NOx reduction and CB oxidation reactions tend to be mutually promotional. This principle has been corroborated in the development of novel catalysts[29,33]. For example, in a Ru/Cu-SSZ-13 catalyst, the spatially confined zeolite environment plays a key role [Figure 5B][36]. The Ru4+ Lewis acid sites on the external surface enhance CB oxidation by weakening the p-π conjugation between the chlorine atom and the benzene ring[36]. Simultaneously, the Cu2+ Lewis acid sites inside the internal channels facilitate the conversion of NOx and NH3 to N2. This configuration effectively prevents mutual interference between the catalytic VOC oxidation and NOx reduction processes, regardless of temperature fluctuations[36].
Figure 5. The synergistic reaction pathways of simultaneously catalytic conversion of NOx and CB. (A) Synergistic mechanism of NOx and CB on MnO2-CePO4 catalyst with regular acid-redox active sites[31]; (B) Synergistic mechanism of NOx and CB on Ru/Cu-SSZ-13 catalyst with dual-functional active sites[36]. Figure 5A is reprinted with permission from Ref.[31], Copyright © 2022 American Chemical Society. Figure 5B is reprinted with permission from Ref.[36], Copyright © 2023 WILEY. NOx: Nitrogen oxides; CB: chlorobenzene; Ru/Cu-SSZ-13: copper-siliceous special zeolite-13 supported Ru; CBCO: chlorobenzene catalytic oxidation; SCR: selective catalytic reduction; LAS: Lewis acid sites; BAS: Brønsted acid sites.
Synergistic catalytic mechanism of NOx and toluene co-removal
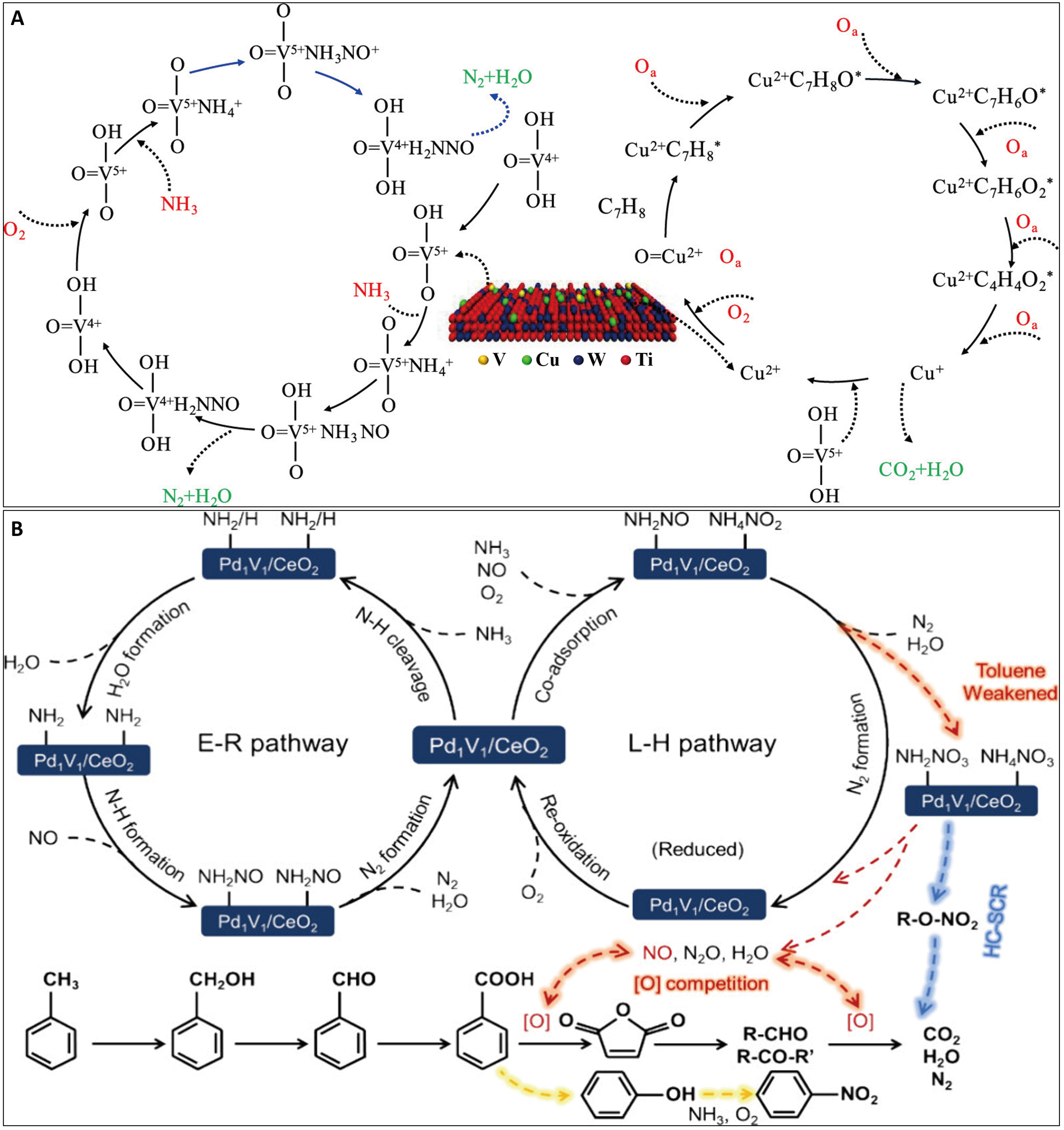
The synergistic catalytic mechanisms for NOx and toluene exhibit distinct differences from those involving chlorinated aromatics such as CB. Unlike CB, toluene has no chlorine atoms in its molecular structure. Therefore, it does not promote the formation of NH4+ species via Cl- adsorption, thereby avoiding the generation of NH4Cl. Furthermore, the interference of toluene in the NH3-SCR reaction differs from that of CB. Toluene oxidation produces easily characterizable ring-opening products and non-ring-opening byproducts. It transforms into benzoate species on active sites, which can lower the activation energy. Meanwhile, NH3 molecules adsorbed on Lewis acid sites can accelerate VOC mineralization while consuming NOx species. On conventional V2O5-based catalysts, NH3 promotes the formation of nitrile intermediates, inhibiting their further decomposition into small molecules. This process typically displays low selectivity and generates numerous by-products[9]. To address this, transition metal modification strategies are commonly employed to enhance synergistic performance. Notably, Cu-modified VWTi (V2O5-WO3-TiO2) catalysts represent an optimized choice [Figure 6A][4]. In this catalyst, toluene is first adsorbed at Cu sites. It is then nucleophilically attacked by surface reactive oxygen species and finally oxidized stepwise to CO2 and H2O. Simultaneously, NH3 molecules are adsorbed at V sites, where it reacts with NO to form an NH3NO intermediate, which ultimately decomposes into N2 and H2O. The cyclic process allows toluene oxidation and NOx reduction to proceed concurrently, enabling their synergistic removal[4]. MnOx-CeO2 catalysts have also been explored for NOx and toluene co-removal. However, even a trace amount of toluene can lead to a significant decrease in SCR activity, which is particularly undesirable at low operating temperatures[51,52]. The adsorption of toluene and its partially oxidized byproducts inhibit the SCR. The co-removal performance can be enhanced by using supports such as TiO2 nanotubes or HZSM-5 (hydrogen form of Zeolite Socony Mobil-5)[53,54], which augment the Lewis acidity and oxygen activation capacity of the MnOx-CeO2 catalyst. Under these conditions, gaseous NOx predominantly reacts with NH3 adsorbed on Lewis acid sites to form N2. Concurrently, the consumption of adsorbed oxygen species during toluene decomposition significantly suppresses undesirable side reactions, including catalytic NH3 oxidation and N2O formation. However, MnOx-CeO2 catalysts are susceptible to deactivation by byproducts such as acetonitrile and benzonitrile, and special attention must be paid to the potential formation of benzamide species[52]. For most Mn-based catalysts, the NH3-SCR and toluene oxidation pathways tend to exhibit mutual inhibition[55,93,94]. Additionally, byproducts formed from the reaction of NH3 with benzaldehyde or benzoate intermediates can inhibit the toluene oxidation activity[54,93]. It is noteworthy that the mutual interference between NH3-SCR and toluene oxidation pathways can be largely circumvented on catalysts with dual-functional active sites[4,50,58,59,82]. In novel catalysts such as the dual single-atom Pd1V1/CeO2 catalyst[58] and CeNbOx composites[57], the SCR and toluene oxidation reaction exhibit a mutually promotional effect. Specifically, in the Pd1V1/CeO2 catalyst, the coexistence of Pd and V as dual single-atom sites enhances reactant adsorption. A synergistic interaction between these dual atoms and surface oxygen suppresses lattice oxygen activation, thereby significantly inhibiting nitrite overoxidation [Figure 6B][58]. Furthermore, the interplay between the active Pd sites and the inert V sites leads to a moderated reduction in the redox property of Pd and a notable increase in that of V[58].
Figure 6. Synergistic reaction pathways of simultaneously catalytic conversion of NOx and toluene. (A) Synergistic mechanism of NOx and toluene on Cu-VWTi catalyst[4]; (B) Synergistic mechanism of NOx and toluene on Pd1V1/CeO2 catalyst[58]. Figure 6A is reprinted with permission from Ref.[4], Copyright © 2022 American Chemical Society. Figure 6B is reprinted with permission from Ref.[58], Copyright © 2024 American Chemical Society. NOx: Nitrogen oxides; VWTi: V2O5-WO3-TiO2.
Synergistic catalytic mechanism of NOx and other VOCs
Compared to the extensively studied co-removal of NOx-CB and NOx-toluene, the mechanistic understanding of synergistic catalysis involving NOx and other VOC classes, such as sulfur- or nitrogen-containing compounds, remains relatively limited. Nevertheless, preliminary insights are emerging from specific model systems. For instance, the synergistic mechanism between NOx and methanethiol over a CuNbFeOx catalyst has been investigated. The presence of multifunctional active sites in this catalyst was found to mitigate sulfur poisoning and reduce the mutual interference between the SCR and methanethiol oxidation pathways[66], thereby enabling co-removal. Similarly, the synergistic mechanism between NOx and ethane on a CeMnO2 catalyst has been examined. In this system, ethane oxidation primarily follows a Mars-van Krevelen (MvK) mechanism, while NH3-SCR proceeds via both Langmuir-Hinshelwood (L-H) and Eley-Rideal (E-R) pathways. This exemplifies a typical dual-functional mechanism with temperature-dependent interactions[60]. In situ spectroscopic techniques have proven invaluable for identifying critical reaction intermediates and elucidating synergistic effects. Advanced characterization techniques continue to provide essential insights for performance optimization. Future studies should prioritize the integrated use of experimental and computational approaches to elucidate the key bottlenecks governing these processes. A deeper exploration of the inhibitory dynamics in multicomponent flue gas remains a significant challenge.
POISONING RESISTANCE AND STABILITY IN REALISTIC ENVIRONMENTS
For VOCs containing heteroatoms such as Cl, N, and S, synergistic catalytic systems face a dual challenge: the potential formation of toxic byproducts and catalyst poisoning. During the oxidation of such VOCs, heteroatoms can be released and subsequently transformed into hazardous substances, such as dioxins from chlorinated compounds or sulfur dioxide (SO2) from sulfur-containing species. Concurrently, catalysts are susceptible to deactivation through mechanisms such as carbon deposition and the surface accumulation of chlorine or sulfur species. Consequently, a pivotal scientific challenge in the co-conversion of NOx-VOCs is the development of synergistic catalysts that simultaneously exhibit robust anti-poisoning stability and high selectivity to minimize the formation of toxic byproducts[95].
Chlorine-poisoning resistance
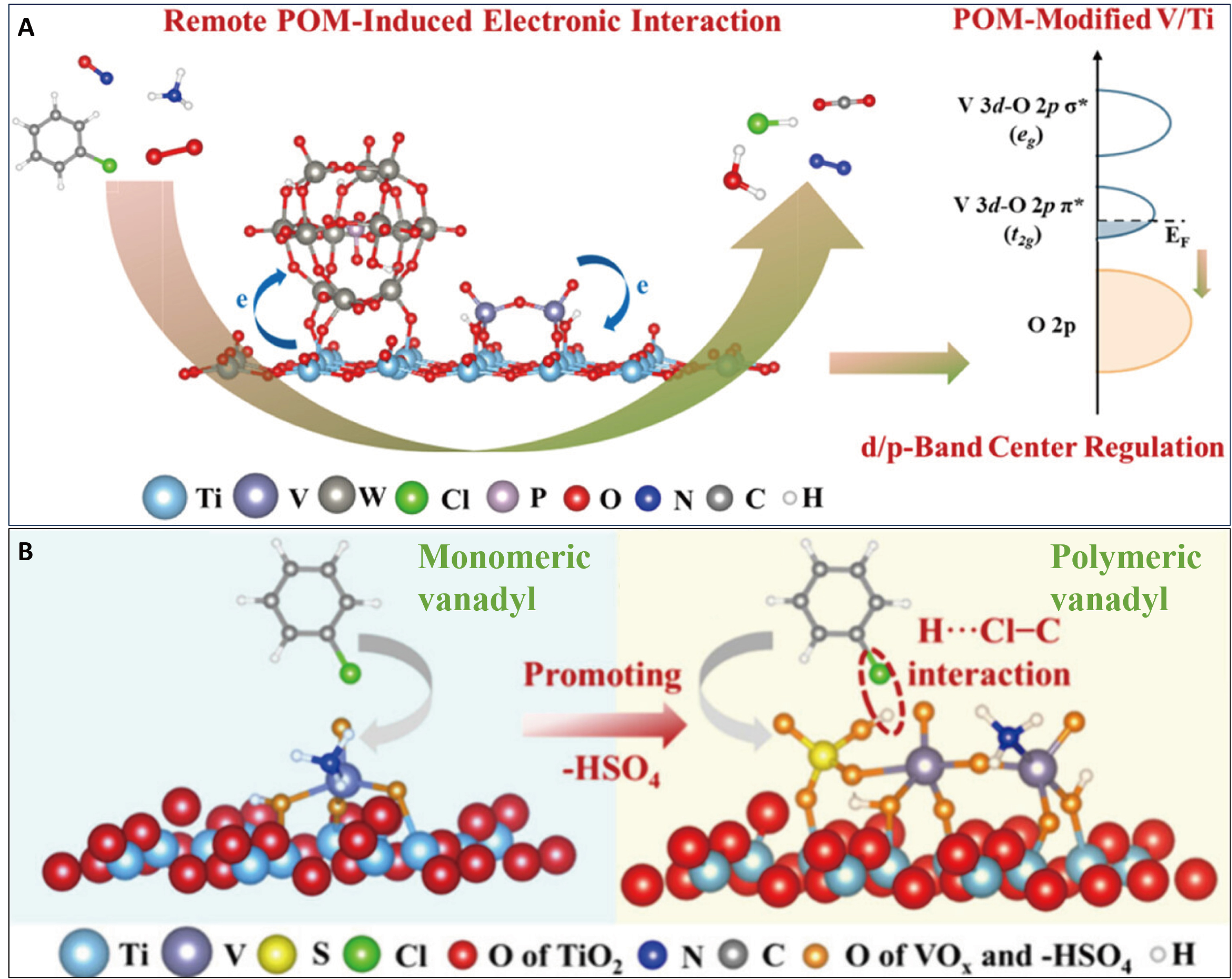
The oxidation of CVOCs is often accompanied by catalyst Cl-poisoning. However, VOx/TiO2 catalysts exhibit superior resistance to this deactivation, as Cl species can be effectively removed via HCl formation, a process facilitated by H2O activation[86]. In contrast, on catalysts with abundant oxygen vacancies, such as certain polymeric vanadyl structures, Cl tends to accumulate through the formation of V−Cl bonds. This can lead to the significant generation of polychlorinated byproducts. A key strategy for mitigating the detrimental effects of chlorinated compounds involves narrowing the band gap of the catalyst and enhancing its electron transfer capability. POM modification has proven to be an effective method for facilitating the synergistic conversion of NOx and CB on supports such as TiO2 and CeO2. This is attributed to the strong electron-withdrawing and Brønsted acid properties of Keggin-type POMs [Figure 7A][25,34]. Through a remote POM-induced electronic interaction, electrons are transferred from the support to the POM clusters. This electronic modulation modifies the nature of the active sites, promoting C−Cl bond cleavage and HCl formation, thereby alleviating Cl poisoning. Furthermore, -HSO4-modified vanadia-based catalysts achieve high turnover efficiency of NOx and CB [Figure 7B][90]. The hydrogen sulfate groups promote the polymerization of vanadyl species and modulate the d-band center of vanadium. Additionally, acting as a proton donor, -HSO4 can directly promote C−Cl bond breaking and HCl production.
Figure 7. The strategies for improving the synergistic catalytic conversion of NOx and VOCs under chlorine-poisoning conditions. (A) The strategy of incorporating functional POMs to prevent the formation of polychlorinated species[25]; (B) A preoccupied anchoring strategy on vanadia-based catalyst by -HSO4 group modification for promoted C−Cl bond activation in NOx and CB synergistic removal[90]. Figure 7A is reprinted with permission from Ref.[25], Copyright © 2025 American Chemical Society. Figure 7B is reprinted with permission from Ref.[90], Copyright © 2024 American Chemical Society. NOx: Nitrogen oxides; VOCs: volatile organic compounds; POMs: polyoxometalates; CB: chlorobenzene.
Metal-poisoning resistance
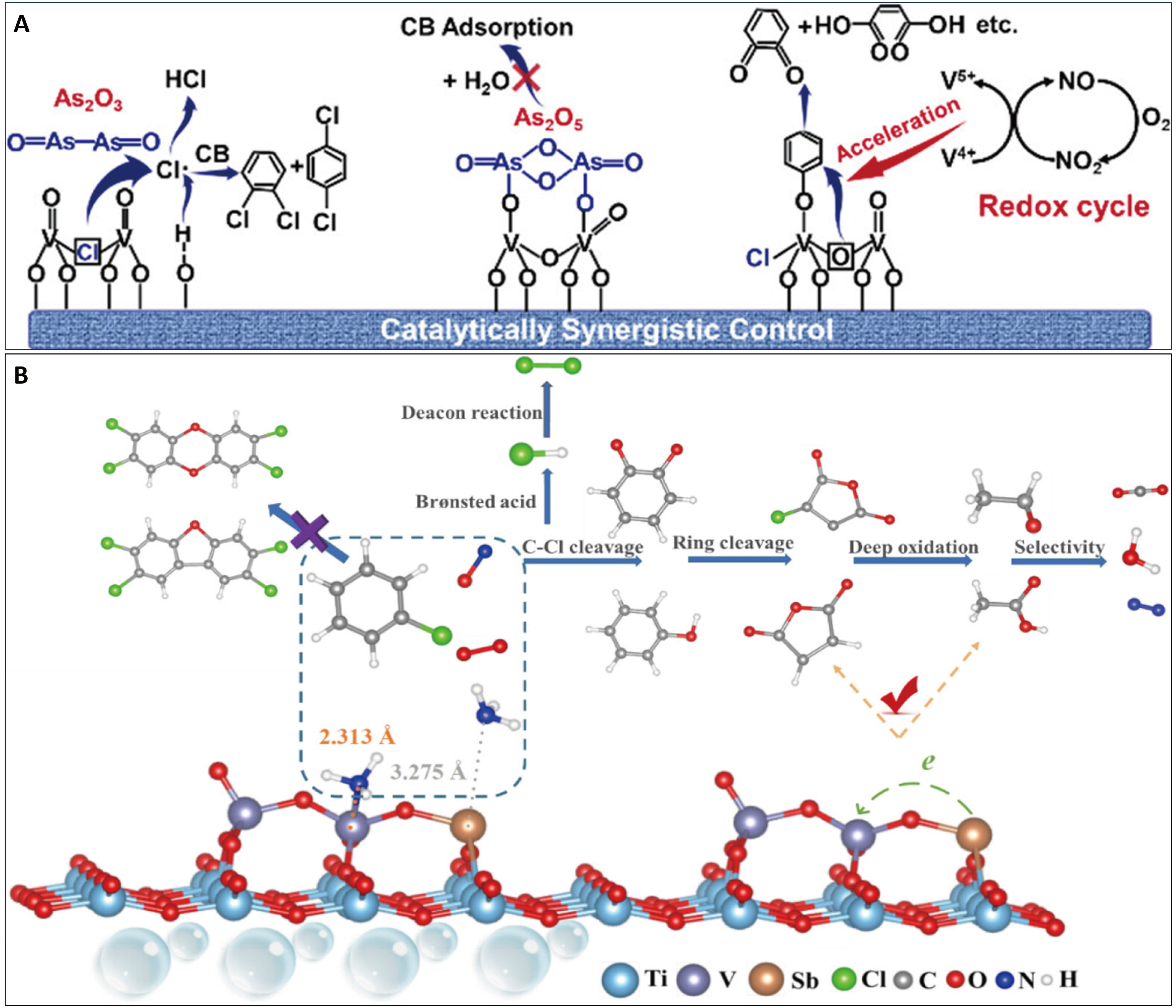
Flue gas often contains metallic impurities, which can lead to catalyst poisoning. The impact of heavy metals such as arsenic (As) and antimony (Sb) on the synergistic catalysis of NOx and CB has been explored [Figure 8]. The deactivating effect of As on V2O5-WO3/TiO2 catalysts has been analyzed[23]. Both As2O3 and As2O5 deactivate the deNOx and CB oxidation reactions by forming As-O-V bands and facilitating H2O adsorption, which blocks active sites [Figure 8A][23]. Molybdenum (Mo) doping can significantly enhance arsenic resistance. It promotes the polymerization of vanadyl species, resulting in a higher concentration of V5+ chemical states. This modification helps protect the vanadium species from arsenic poisoning, thereby enhancing the overall synergistic performance. Conversely, doping antimony (Sb) into V2O5-WO3/TiO2 catalysts serves a dual purpose. It acts as an NH3 adsorption mitigator and a polychlorinated compound inhibitor. This design achieves outstanding selectivity towards HCl and N2, leading to superior catalytic performance [Figure 8B][24]. The formation of V-O-Sb chains on the catalyst surface narrows the band gap of vanadium and modulates the Lewis acid sites. This restructuring suppresses undesirable chlorination reactions, thereby maintaining high selectivity for HCl and N2. These insights provide a novel perspective for subsequent synergistic catalyst design. The strategy involves engineering protective sites to anchor poisoning species. This process can lead to the in situ generation of new and resilient active sites, which effectively promotes the synergistic catalytic elimination of NOx and VOCs while inhibiting catalyst deactivation[24].
Figure 8. The strategies for improving the synergistic catalytic conversion of NOx and VOCs under metal-poisoning conditions. (A) Efficient synergistic control of NOx and chlorinated organics considering the arsenic (As) effect[23]; (B) Anti-poisoning mechanisms of Sb on vanadia-based catalysts for NOx and CB[24]. Figure 8A is reprinted with permission from Ref.[23], Copyright © 2021 American Chemical Society. Figure 8B is reprinted with permission from Ref.[24], Copyright © 2023 American Chemical Society. NOx: Nitrogen oxides; VOCs: volatile organic compounds.
Sulfur-poisoning resistance
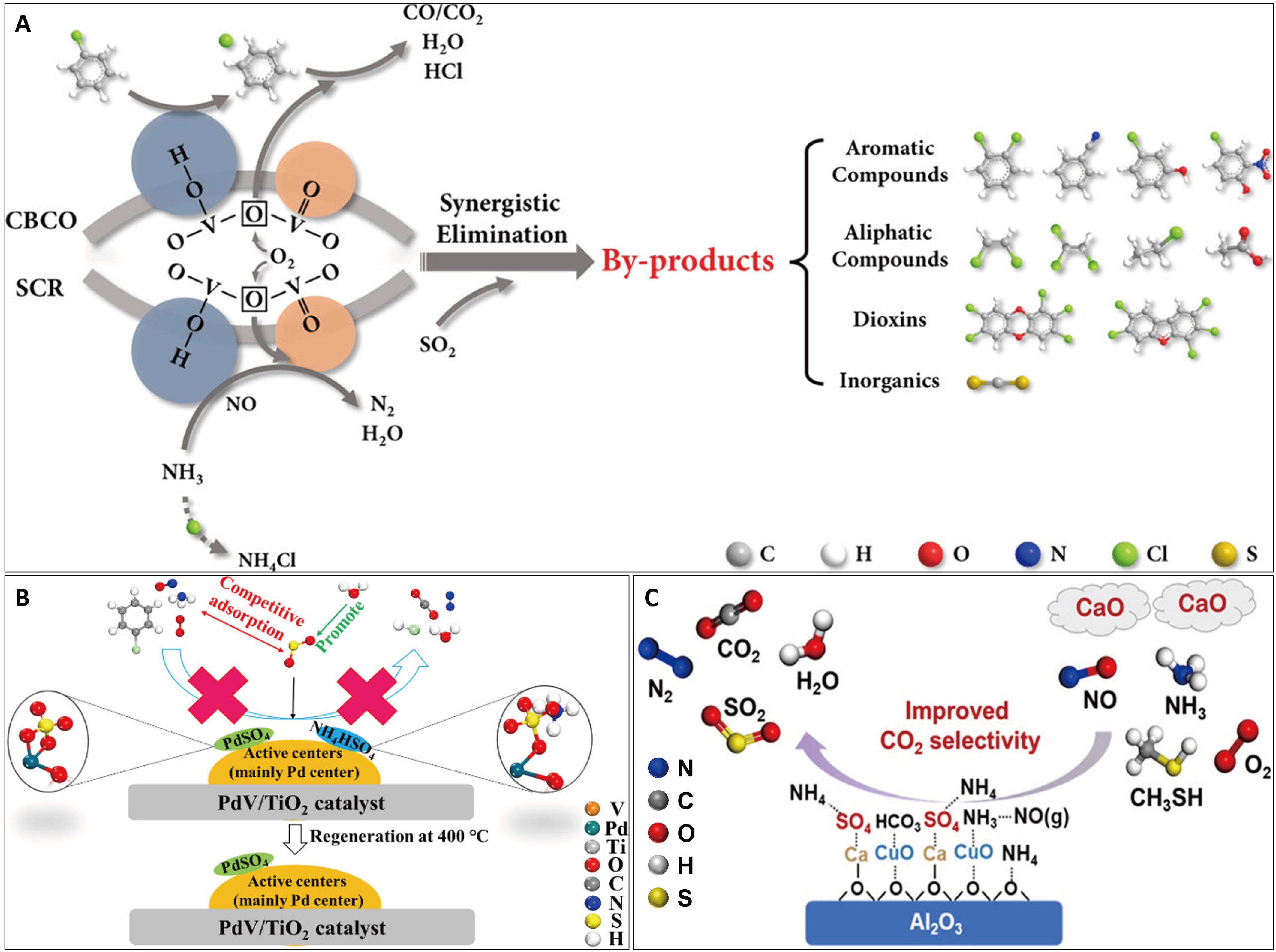
The design of sulfur-tolerant catalysts is imperative due to the presence of H2O and SO2 in flue gas, which induce competitive adsorption and metal sulfation. A fundamental understanding of sulfur poisoning mechanisms is therefore crucial for developing robust, sulfur-resistant catalysts. On a typical V2O5-WO3/TiO2 catalyst used for NOx and CB removal, SO2 was found to promote the generation of polychlorinated byproducts while inhibiting the formation of PCDD/Fs [Figure 9A][87]. A similar poisoning mechanism was elucidated for a NiMn2O4-CeO2 catalyst, where sulfur exposure altered the catalyst structure and active sites distribution[61]. The rapid formation of MnSO4 and Ce(SO4)2 significantly reduced the specific surface area and decreased catalytic capacity. Furthermore, SO2 acts as an inhibitor, suppressing both C=C bond breaking in VOC oxidation and the L-H pathway in denitrification reactions. Building an alkaline oxide protective layer is a proven strategy. For example, incorporating MgO into Mn-Ce-based materials led to the synthesis of a MnOx@CeO2@MgO core-shell catalyst. This architecture exhibited high sulfur resistance and conversion efficiency for NOx and CB, as SO2 primarily reacts with the outer MgO layer, shielding the inner active sites from poisoning[96]. Similarly, a MnOx@CeSnOx core-shell catalyst, where the CeSnOx shell contains Sn4+ ions that act as sacrificial sites for SO2, demonstrated excellent sulfur resistance[97]. Doping with specific additives is another effective approach. The synergistic removal of NOx and CB on a PdV/TiO2 catalyst is severely hindered by sulfur poisoning, primarily due to the irreversible formation of PdSO4 or PdS species that occupy the active Pd centers [Figure 9B][98]. Modifying this catalyst with antimony (Sb) effectively inhibits the deposition of sulfur compounds, thereby protecting the Pd active sites[77]. Guided by Sabatier’s principle, which states that overly weak gas adsorption leads to inefficient reactions, the optimal application of Sb prevents the adsorption of sulfur species, conferring high anti-SO2 poisoning capacity. Furthermore, doping Al2O3 into a V2O5-MoO3/TiO2 (VMo/Ti) catalyst to form Al-VMo/Ti enhances NOx and CB conversion across a wide temperature range. The Lewis acid sites on Al2O3, combined with the modified reducibility of vanadium, broaden the active temperature window while maintaining excellent resistance to SO2 and H2O and minimizing byproduct formation[80]. In addition, for the synergistic elimination of NOx and CH3SH on CuO/Al2O3 catalysts, the introduction of CaO can mitigate sulfur poisoning through an antagonistic effect [Figure 9C][99].
Figure 9. Strategies for improving the synergistic catalytic conversion of NOx and VOCs under sulfur-poisoning conditions. (A) SO2 effect on V2O5-WO3/TiO2 catalyst for synergistic removal of NOx and CB[87]; (B) SO2 poisoning mechanism of the PdV/TiO2 catalyst for NOx and CB synergistic degradation[98]; (C) CaO-induced mitigated sulfur-poisoning on the synergistic catalyst for NOx and CH3SH elimination[99]. Figure 9A is reprinted with permission from Ref.[87], Copyright © 2019 American Chemical Society. Figure 9B is reprinted with permission from Ref.[98], Copyright © 2021 American Chemical Society. Figure 9C is reprinted with permission from Ref.[99], Copyright © 2025 American Chemical Society. NOx: Nitrogen oxides; VOCs: volatile organic compounds; CBCO: chlorobenzene catalytic oxidation; SCR: selective catalytic reduction.
CONCLUSION AND OUTLOOK
Synergistic catalysis for the co-removal of NOx and VOCs has evolved from a fundamental curiosity to a technology nearing commercial reality, driven by increasingly stringent air quality policies and catalytic innovations. In this mini review, we have discussed the synergistic catalytic elimination of NOx and VOCs, critically analyzing how engineered acid-redox bifunctionality and dynamic active site cooperation can overcome inherent challenges such as temperature-window mismatch and competitive adsorption in complex flue gases. However, several critical aspects require further elucidation to advance the rational design and performance of these catalysts. These include the precise engineering of reactive sites, optimal tuning of acid-redox properties, enhancement of poisoning resistance, and resolving the cost-performance dilemma.
Novel catalyst design strategies are urgently needed to address the core challenges of temperature-window mismatch and competitive adsorption. A primary focus is the rational design and construction of multifunctional active sites to minimize competitive interactions between different pollutant molecules. Here, ML shows immense potential for accelerating the discovery of optimal bimetallic pairs tailored for specific NOx-VOCs combinations. Integrating ML with high-throughput experimental screening will create a powerful, efficient pipeline for next-generation catalyst discovery. Furthermore, dynamic active site regulation is emerging as a promising frontier. The goal is to develop smart, self-adapting catalysts capable of switching their redox states in response to fluctuating flue-gas compositions, thereby dynamically mitigating competitive interference. Developing advanced poison-resistance strategies is paramount to ensure the long-term durability and robustness of synergistic catalysts under harsh, realistic industrial flue gas conditions.
A key insight from the reviewed studies is the power of fully utilizing catalyst interfaces to skillfully construct dual or multiple active sites on a single catalyst. This strategy enables the specific and simultaneous conversion of different pollutant molecules within the same spatiotemporal domain, achieving true synergistic multipollutant control. Importantly, this paradigm can be extended beyond traditional pollutants. It holds promise for the synergistic catalysis of traditional and emerging pollutants (for example, NOx with dichloromethane, CH2Cl2) or even greenhouse gases such as CO2. Looking further ahead, the rational design of synergistic sites could pave the way for the hierarchical and intelligent conversion of small molecules, such as CO2, to value-added products. In conclusion, the synergistic catalytic strategy represents a transformative approach with the potential to fundamentally address energy and environmental challenges.
DECLARATIONS
Authors’ contributions
Conceptualization: Chen, A.
Writing - original draft preparation: Chen, A.
Writing - review & editing: Chen, A.; Gao, M.; Wang, F.; Jiang, X.; Qu, W.; Shen, Y.; Palacio, R.
Supervision: Chen, A.
Project administration: Chen, A.
Funding acquisition: Chen, A.; Wang, F.
Investigation: Gao, M.; Wang, F.; Jiang, X.; Shen, Y.; Palacio, R.
Data curation: Gao, M.; Jiang, X.
Visualization: Gao, M.
Validation: Wang, F.; Jiang, X.
Methodology: Qu, W.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
The authors are grateful for the financial support from the National Natural Science Foundation of China (Nos. 22576130, 22276120, and 22506107) and the Science and Technology Commission of Shanghai Municipality (No. 23230713700). They are also grateful to the University of Antioquia (UdeA).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Finlayson-Pitts, B. J.; Pitts JN, J. R. Tropospheric air pollution: ozone, airborne toxics, polycyclic aromatic hydrocarbons, and particles. Science 1997, 276, 1045-52.
2. Han, L.; Cai, S.; Gao, M.; et al. Selective catalytic reduction of NOx with NH3 by using novel catalysts: state of the art and future prospects. Chem. Rev. 2019, 119, 10916-76.
3. Chen, Y.; Liu, X.; Wang, P.; et al. Challenges and perspectives of environmental catalysis for NOx reduction. JACS. Au. 2024, 4, 2767-91.
4. Xiao, G.; Guo, Z.; Lin, B.; Fu, M.; Ye, D.; Hu, Y. Cu-VWT catalysts for synergistic elimination of NOx and volatile organic compounds from coal-fired flue gas. Environ. Sci. Technol. 2022, 56, 10095-104.
5. Wang, D.; Chen, Q.; Zhang, X.; et al. Multipollutant control (MPC) of flue gas from stationary sources using SCR technology: a critical review. Environ. Sci. Technol. 2021, 55, 2743-66.
6. Carlsson, K. B. Dioxin destruction in catalysts for NOx-reduction (SCR-DENOx). Chemosphere 1992, 25, 135-8.
7. Ok, G.; Hanai, Y.; Katou, T. Decomposition of chlorinated dioxins, odourous compounds and NOx from MSW incineraton plant by oxidizing catalyst. Chemosphere 1993, 26, 2167-72.
8. Karthik, M.; Lin, L.; Bai, H. Bifunctional mesoporous Cu-Al-MCM-41 materials for the simultaneous catalytic abatement of NOx and VOCs. Microporous. Mesoporous. Mater. 2009, 117, 153-60.
9. Xiao, G.; Guo, Z.; Li, J.; et al. Insights into the effect of flue gas on synergistic elimination of toluene and NOx over V2O5-MoO3(WO3)/TiO2 catalysts. Chem. Eng. J. 2022, 435, 134914.
10. Gan, L.; Li, K.; Xiong, S.; et al. MnOx-CeO2 catalysts for effective NOx reduction in the presence of chlorobenzene. Catal. Commun. 2018, 117, 1-4.
11. Gan, L.; Shi, W.; Li, K.; Chen, J.; Peng, Y.; Li, J. Synergistic promotion effect between NOx and chlorobenzene removal on MnOx-CeO2 catalyst. ACS. Appl. Mater. Interfaces. 2018, 10, 30426-32.
12. Wang, D.; Chen, J.; Peng, Y.; et al. Dechlorination of chlorobenzene on vanadium-based catalysts for low-temperature SCR. Chem. Commun. 2018, 54, 2032-5.
13. Chen, L.; Liao, Y.; Xin, S.; Song, X.; Liu, G.; Ma, X. Simultaneous removal of NO and volatile organic compounds (VOCs) by Ce/Mo doping-modified selective catalytic reduction (SCR) catalysts in denitrification zone of coal-fired flue gas. Fuel 2020, 262, 116485.
14. Aissat, A.; Courcot, D.; Cousin, R.; Siffert, S. VOCs removal in the presence of NOx on Cs-Cu/ZrO2 catalysts. Catal. Today. 2011, 176, 120-5.
15. Fan, C.; Li, K.; Peng, Y.; et al. Fe-doped α-MnO2 nanorods for the catalytic removal of NOx and chlorobenzene: the relationship between lattice distortion and catalytic redox properties. Phys. Chem. Chem. Phys. 2019, 21, 25880-8.
16. Gan, L.; Wang, Y.; Chen, J.; et al. The synergistic mechanism of NOx and chlorobenzene degradation in municipal solid waste incinerators. Catal. Sci. Technol. 2019, 9, 4286-92.
17. Su, G.; Huang, L.; Liu, S.; Lu, H.; Yang, F.; Zheng, M. The combined disposal of 1,2,4-trichlorobenzene and nitrogen oxides using the synthesized Ce0.2TiAlαOx micro/nanomaterial. Catal. Sci. Technol. 2015, 5, 1041-51.
18. Motak, M. Kuterasiński, Ł.; Da Costa, P.; Samojeden, B. Catalytic activity of layered aluminosilicates for VOC oxidation in the presence of NOx. Comptes. Rendus. Chimie. 2015, 18, 1106-13.
19. Gallastegi-villa, M.; Aranzabal, A.; González-marcos, J.; González-velasco, J. Metal-loaded ZSM5 zeolites for catalytic purification of dioxin/furans and NOx containing exhaust gases from MWI plants: effect of different metal cations. Appl. Catal. B. Environ. 2016, 184, 238-45.
20. Mrad, R.; Cousin, R.; Saliba, N. A.; Tidahy, L.; Siffert, S. Degradation of VOCs and NOx over Mg(Cu)-AlFe mixed oxides derived from hydrotalcite-like compounds. Comptes. Rendus. Chimie. 2015, 18, 351-7.
21. Gallastegi-villa, M.; Aranzabal, A.; González-marcos, J.; González-velasco, J. Tailoring dual redox-acid functionalities in VOx/TiO2/ZSM5 catalyst for simultaneous abatement of PCDD/Fs and NOx from municipal solid waste incineration. Appl. Catal. B. Environ. 2017, 205, 310-8.
22. Wang, M.; Naman, N.; Shen, Y.; et al. Synergistic catalytic removal of NOx and chlorinated aromatics via atomically dispersed asymmetric Mn-O-Ce sites on montmorillonite. Appl. Catal. B. Environ. Energy. 2025, 378, 125594.
23. Long, Y.; Su, Y.; Xue, Y.; Wu, Z.; Weng, X. V2O5-WO3/TiO2 catalyst for efficient synergistic control of NOx and chlorinated organics: insights into the arsenic effect. Environ. Sci. Technol. 2021, 55, 9317-25.
24. Yuan, X.; Peng, Y.; Zhu, X.; et al. Anti-poisoning mechanisms of Sb on vanadia-based catalysts for NOx and chlorobenzene multi-pollutant control. Environ. Sci. Technol. 2023, 57, 10211-20.
25. Yuan, X.; Peng, Y.; Zhu, X.; et al. Remote polyoxometalates modulated the d/p-band center proximity in vanadia-based catalyst for simultaneous elimination of NOx and chlorobenzene. ACS. Catal. 2025, 15, 7470-81.
26. Song, Z.; Peng, Y.; Zhao, X.; et al. Roles of Ru on the V2O5-WO3/TiO2 catalyst for the simultaneous purification of NOx and chlorobenzene: a dechlorination promoter and a redox inductor. ACS. Catal. 2022, 12, 11505-17.
27. Song, Z.; Yu, S.; Liu, H.; et al. Carbon/chlorinate deposition on MnOx-CeO2 catalyst in chlorobenzene combustion: the effect of SCR flue gas. Chem. Eng. J. 2022, 433, 133552.
28. Chu, P.; Zhang, L.; Wei, L.; et al. Disentangling the interaction of metal oxide catalyst-reactant via spatial separation in the synergistic catalytic elimination of NO and chlorobenzene. Appl. Catal. B. Environ. Energy. 2025, 378, 125642.
29. Naman, N.; Wang, M.; Xu, Z.; et al. Synergistic catalytic removal of NOx and chlorobenzene by a combination punch of Lewis and Bronsted acid and redox sites. J. Colloid. Interface. Sci. 2025, 695, 137741.
30. Zhu, X.; Yuan, X.; Song, Z.; Peng, Y.; Li, J. A dual-balance strategy via phosphate modification on MnO2-CeO2 for NOx and chlorobenzene synergistic catalytic control. Appl. Catal. B. Environ. 2024, 342, 123364.
31. Zhang, C.; Zhang, J.; Shen, Y.; et al. Synergistic catalytic elimination of NOx and chlorinated organics: cooperation of acid sites. Environ. Sci. Technol. 2022, 56, 3719-28.
32. Wei, L.; Liu, Y.; Dai, H.; et al. Electronic structure tailoring of Al3+- and Ta5+-doped CeO2 for the synergistic removal of NO and chlorinated organics. Appl. Catal. B. Environ. 2022, 304, 120939.
33. Wang, Z.; Gao, M.; Chen, X.; et al. Boosting synergistic catalytic abatement of NOx and chlorobenzene via bidirectional promotion of Nb within asymmetrical Ce-O-Nb sites. Appl. Catal. A. Gen. 2025, 697, 120223.
34. Yang, H.; Chen, A.; Wang, F.; et al. Phosphotungstic acid as a dechlorination agent collaborates with CeO2 for synergistic catalytic elimination of NOx and chlorobenzene. Environ. Sci. Technol. 2024, 58, 7672-82.
35. Yin, R.; Chen, J.; Mi, J.; et al. Breaking the activity-selectivity trade-off for simultaneous catalytic elimination of nitric oxide and chlorobenzene via FeVO4-Fe2O3 interfacial charge transfer. ACS. Catal. 2022, 12, 3797-806.
36. Wei, L.; Liu, Y.; Cui, S.; et al. Separated active site and reaction space for multi‐pollutant elimination significantly enhancing low toxic product selectivity. Adv. Funct. Mater. 2023, 33, 2306129.
37. Wang, Q.; Huang, X.; Feng, Y.; Zhou, J.; Shi, H.; Jin, J. Interaction mechanism study on simultaneous removal of 1,2-dichlorobenzene and NO over MnOx-CeO2/TiO2 catalysts at low temperatures. Ind. Eng. Chem. Res. 2021, 60, 4820-30.
38. Martín-martín, J.; Sánchez-robles, J.; González-marcos, M.; Aranzabal, A.; González-velasco, J. Effect of preparation procedure and composition of catalysts based on Mn and Ce oxides in the simultaneous removal of NOx and o-DCB. Molecular. Catalysis. 2020, 495, 111152.
39. Wang, Q.; Lin, F.; Zhou, J.; Zhang, J.; Jin, J. Effect of HCl and o-DCBz on NH3-SCR of NO over MnO/TiO2 and MnO-CeO2/TiO2 catalysts. Appl. Catal. A. Gen. 2020, 605, 117801.
40. Wang, Q.; Feng, J.; Peng, Y.; et al. Specialized nanotubular MnO-CeO2/TiO2 composite catalysts for simultaneous low-temperature elimination of nitric oxide and ortho-dichlorobenzene. J. Environ. Chem. Eng. 2025, 13, 117229.
41. Gallastegi-villa, M.; Aranzabal, A.; Boukha, Z.; et al. Role of surface vanadium oxide coverage support on titania for the simultaneous removal of o-dichlorobenzene and NOx from waste incinerator flue gas. Catal. Today. 2015, 254, 2-11.
42. Martín-martín, J.; Gallastegi-villa, M.; González-marcos, M.; Aranzabal, A.; González-velasco, J. Bimodal effect of water on V2O5/TiO2 catalysts with different vanadium species in the simultaneous NO reduction and 1,2-dichlorobenzene oxidation. Chem. Eng. J. 2021, 417, 129013.
43. Wang, J.; Xing, Y.; Su, W.; et al. Promotional effect of Sn additive on the chlorine resistance over SnMnOx/LDO catalysts for synergistic removal of NOx and o-DCB. Catal. Sci. Technol. 2022, 12, 3863-73.
44. Jin, Q.; Shen, Y.; Mei, C.; Zhang, Y.; Zeng, Y. Catalytic removal of NO and dioxins over W-Zr-Ox/Ti-Ce-Mn-Ox from flue gas: performance and mechanism study. Catal. Today. 2022, 388-389, 372-82.
45. Gallastegi-villa, M.; Aranzabal, A.; González-marcos, M.; Markaide-aiastui, B.; González-marcos, J.; González-velasco, J. Effect of vanadia loading on acidic and redox properties of VOx/TiO2 for the simultaneous abatement of PCDD/Fs and NOx. J. Ind. Eng. Chem. 2020, 81, 440-50.
46. Chen, Y.; Chen, Z.; Zhang, C.; et al. Multiple pollutants control of NO, benzene and toluene from coal-fired plant by Mo/Ni impregnated TiO2-based NH3-SCR catalyst: a DFT supported experimental study. Appl. Surf. Sci. 2022, 599, 153986.
47. Lin, B.; Guo, Z.; Li, J.; Xiao, G.; Ye, D.; Hu, Y. V-Cu bimetallic oxide supported catalysts for synergistic removal of toluene and NOx from coal-fired flue gas: the crucial role of support. Chem. Eng. J. 2023, 458, 141443.
48. Liu, H.; Chen, J.; Wang, Y.; et al. Interaction mechanism for simultaneous elimination of nitrogen oxides and toluene over the bifunctional CeO2-TiO2 mixed oxide catalyst. Environ. Sci. Technol. 2022, 56, 4467-76.
49. Gong, Z.; Yang, H.; Li, C.; et al. The simultaneous elimination of NO and VOCs over industrial V-based catalytic filter: Reaction behaviors and catalyst structural evolutions. J. Environ. Chem. Eng. 2024, 12, 111933.
50. Shi, H.; Li, X.; Jiang, J.; Zhu, T.; Liu, X. Reaction site-isolation for simultaneous removal of NO and toluene: a reverse strategy to realize bifunctional catalysis. Appl. Catal. B. Environ. Energy. 2025, 363, 124786.
51. Ye, L.; Lu, P.; Chen, X.; et al. The deactivation mechanism of toluene on MnOx-CeO2 SCR catalyst. Appl. Catal. B. Environ. 2020, 277, 119257.
52. Ye, L.; Lu, P.; Peng, Y.; Li, J.; Huang, H. Impact of NOx and NH3 addition on toluene oxidation over MnOx-CeO2 catalyst. J. Hazard. Mater. 2021, 416, 125939.
53. Ye, L.; Lu, P.; Xianhui, Y.; Huang, H. Boosting simultaneous catalytic removal of NOx and toluene via cooperation of Lewis acid and oxygen vacancies. Appl. Catal. B. Environ. 2023, 331, 122696.
54. Lu, P.; Ye, L.; Yan, X.; et al. Impact of toluene poisoning on MnCe/HZSM-5 SCR catalyst. Chem. Eng. J. 2021, 414, 128838.
55. He, X.; Shen, B.; Gao, J.; Ji, Y.; Wu, M.; Wang, F. Constructing multi-active sites of LDH-derived MnCoFe layered mixed oxide catalysts for simultaneous removal of NO and toluene. Appl. Catal. B. Environ. Energy. 2025, 377, 125496.
56. Mi, J.; Liu, H.; Yang, S.; et al. Dual activation of molecular NO and O2 on a Pr-doped CeO2-TiO2 catalyst for the simultaneous catalytic purification of NOx and toluene. Environ. Sci. Technol. 2025, 59, 11321-9.
57. Li, Y.; Sun, Z.; Zhang, Z.; Niu, X.; Zhu, Y. Enhancing reactive oxygen release-replenishment and Lewis acid acidity by Nb doping in NbmCeOx for simultaneous elimination of NOx and toluene. Appl. Catal. B. Environ. Energy. 2025, 364, 124842.
58. Chu, P.; Zhang, L.; Wang, Z.; et al. Regulation lattice oxygen mobility via dual single atoms for simultaneously enhancing VOC oxidation and NOx reduction. Environ. Sci. Technol. 2024, 58, 17475-84.
59. Guo, Z.; Lin, B.; Huang, Y.; et al. Design of bimetallic catalyst with dual-functional Cu-Ce sites for synergistic NOx and toluene abatement. Appl. Catal. B. Environ. 2024, 342, 123430.
60. Du, S.; Hu, J.; Peng, D.; et al. Simultaneous removal of ethane and NOx over Ce-MnO2 catalysts: insights into dual-functional mechanisms and temperature-dependent interactions. J. Catal. 2025, 450, 116281.
61. Ma, S.; Hou, Y.; Yang, Y.; et al. Mechanism of SO2 effect on the simultaneous removal of NOx and propane over bifunctional NiMn2O4-CeO2 catalysts. J. Hazard. Mater. 2024, 480, 136473.
62. Zhu, Y.; Zhou, F.; Wang, X.; Liu, Y.; Wu, Z. Reaction behaviors of NOx and methanol simultaneous abatement over a ceria-based NH3-SCR catalyst at low-medium temperatures. J. Phys. Chem. C. 2021, 125, 14666-74.
63. Wang, X.; Zhu, Y.; Liu, Y.; Weng, X.; Wu, Z. Tailoring the simultaneous abatement of methanol and NOx on Sb-Ce-Zr catalysts via copper modification. Front. Environ. Sci. Eng. 2022, 16, 130.
64. Elsener, M.; Nuguid, R. J. G.; Kröcher, O.; Ferri, D. HCN production from formaldehyde during the selective catalytic reduction of NOx with NH3 over V2O5/WO3-TiO2. Appl. Catal. B. Environ. 2021, 281, 119462.
65. Yan, L.; Zhu, H.; Liu, X.; et al. Synergistic catalytic removal of NOx and n-butylamine via spatially separated cooperative sites. Environ. Sci. Technol. 2024, 58, 11781-90.
66. Liu, X.; Hu, X.; Zhang, K.; et al. Selective synergistic catalytic elimination of NOx and CH3SH via engineering deep oxidation sites against toxic byproducts formation. Environ. Sci. Technol. 2023, 57, 21470-82.
67. Zhang, H.; Liu, X.; Xiao, H.; et al. Catalytic elimination of NOx and CH3SH over synergistic reaction induced active sites. Chem. Eng. J. 2024, 485, 150003.
68. Ouyang, W.; Zhou, Y.; Fei, X.; Bai, Y.; Wang, H.; Wu, Z. Simultaneous removal of NO and dichloromethane (CH2Cl2) over Nb-loaded cerium nanotubes catalyst. J. Environ. Sci. 2022, 111, 175-84.
69. Chen, L.; Liao, Y.; Chen, Y.; Wu, J.; Ma, X. Performance of Ce-modified V-W-Ti type catalyst on simultaneous control of NO and typical VOCS. Fuel. Process. Technol. 2020, 207, 106483.
70. Jones, J.; Ross, J. R. The development of supported vanadia catalysts for the combined catalytic removal of the oxides of nitrogen and of chlorinated hydrocarbons from flue gases. Catal. Today. 1997, 35, 97-105.
71. Busca, G.; Baldi, M.; Pistarino, C.; et al. Evaluation of V2O5-WO3-TiO2 and alternative SCR catalysts in the abatement of VOCs. Catal. Today. 1999, 53, 525-33.
72. Bertinchamps, F.; Treinen, M.; Blangenois, N.; Mariage, E.; Gaigneaux, E. Positive effect of NO on the performances of VO/TiO-based catalysts in the total oxidation abatement of chlorobenzene. J. Catal. 2005, 230, 493-8.
73. Bertinchamps, F.; Treinen, M.; Eloy, P.; Dos Santos, A.; Mestdagh, M.; Gaigneaux, E. Understanding the activation mechanism induced by NOx on the performances of VOx/TiO2 based catalysts in the total oxidation of chlorinated VOCs. Appl. Catal. B. Environ. 2007, 70, 360-9.
74. Chen, Y.; Liao, Y.; Chen, L.; Chen, Z.; Ma, X. Performance of transition metal (Cu, Fe and Co) modified SCR catalysts for simultaneous removal of NO and volatile organic compounds (VOCs) from coal-fired power plant flue gas. Fuel 2021, 289, 119849.
75. Chen, Z.; Liao, Y.; Chen, Y.; Ma, X. In situ DRIFTS FT-IR and DFT study on Fe-V-W/Ti removal of NOx and VOCs. Environ. Sci. Pollut. Res. Int. 2022, 29, 81571-82.
76. Huang, X.; Liu, Z.; Wang, D.; Peng, Y.; Li, J. The effect of additives and intermediates on vanadia-based catalyst for multi-pollutant control. Catal. Sci. Technol. 2020, 10, 323-6.
77. Li, G.; Ding, S.; Hou, X.; Shen, K.; Zhang, S.; Zhang, Y. Unlocking low-temperature and anti-SO2 poisoning performance of bimetallic PdV/TiO2 catalyst for chlorobenzene/NOx catalytic removal by antimony modification design. Chem. Eng. J. 2023, 457, 141210.
78. Li, G.; Shen, K.; Wang, L.; et al. Synergistic degradation mechanism of chlorobenzene and NOx over the multi-active center catalyst: the role of NO2, Brønsted acidic site, oxygen vacancy. Appl. Catal. B. Environ. 2021, 286, 119865.
79. Xu, P.; Zhu, N.; Hou, L.; Wang, S.; Li, S. Pr-modified vanadia-based catalyst for simultaneous elimination of NO and chlorobenzene. Mol. Catal. 2023, 548, 113430.
80. Yu, S.; Niu, X.; Song, Z.; Huang, X.; Peng, Y.; Li, J. Improvement of Al2O3 on the multi-pollutant control performance of NOx and chlorobenzene in vanadia-based catalysts. Chemosphere 2022, 289, 133156.
81. Chu, P.; Zhang, L.; Wang, Z.; et al. Synergistic catalytic elimination of NO and VOCs: State of the art and open challenges. Surf. Interfaces. 2024, 51, 104718.
82. Li, Z.; Xiao, J.; Gao, Y.; Gui, R.; Wang, Q. Design of bifunctional Cu-SSZ-13@Mn2Cu1Al1Ox core-shell catalyst with superior activity for the simultaneous removal of VOCs and NOx. Environ. Sci. Technol. 2023, 57, 20326-38.
83. Wu, J.; Zhang, J.; Qian, G.; Zhang, T. The design and discovery of catalysts for simultaneous catalysis of chlorobenzene and nitrogen oxides via domain knowledge guided machine learning. Appl. Catal. A. Gen. 2023, 668, 119487.
84. Wang, Y.; Lan, T.; Han, L.; et al. Non-Precious metal catalysts with gradient oxidative dual sites boost bimolecular activation for catalytic oxidation reactions. Angew. Chem. Int. Ed. Engl. 2025, 64, e202506018.
85. Lian, Z.; Wei, J.; Shan, W.; et al. Adsorption-induced active vanadium species facilitate excellent performance in low-temperature catalytic NOx abatement. J. Am. Chem. Soc. 2021, 143, 10454-61.
86. Zhai, S.; Su, Y.; Weng, X.; Li, R.; Wang, H.; Wu, Z. Synergistic elimination of NOx and chlorinated organics over VOx/TiO2 catalysts: a combined experimental and DFT study for exploring vanadate domain effect. Environ. Sci. Technol. 2021, 55, 12862-70.
87. Jiang, W.; Yu, Y.; Bi, F.; Sun, P.; Weng, X.; Wu, Z. Synergistic elimination of NOx and chloroaromatics on a commercial V2O5-WO3/TiO2 catalyst: byproduct analyses and the SO2 effect. Environ. Sci. Technol. 2019, 53, 12657-67.
88. Gao, C.; Yang, G.; Huang, X.; et al. Key intermediates from simultaneous removal of NOx and chlorobenzene over a V2O5-WO3/TiO2 catalyst: a combined experimental and DFT study. Catal. Sci. Technol. 2021, 11, 7260-7.
89. Huang, X.; Wang, D.; Yang, Q.; Peng, Y.; Li, J. Multi-pollutant control (MPC) of NO and chlorobenzene from industrial furnaces using a vanadia-based SCR catalyst. Appl. Catal. B. Environ. 2021, 285, 119835.
90. Yuan, X.; Wang, Y.; Zhu, X.; et al. Promoting C-Cl bond activation via a preoccupied anchoring strategy on vanadia-based catalysts for multi-pollutant control of NOx and chlorinated aromatics. Environ. Sci. Technol. 2024, 58, 16357-67.
91. Yang, B.; Jin, Q.; Huang, Q.; et al. Synergetic catalytic removal of chlorobenzene and NO from waste incineration exhaust over MnNb0.4Ce0.2O catalysts: performance and mechanism study. J. Rare. Earths. 2020, 38, 1178-89.
92. Kang, D.; Zhao, P.; Shi, Q.; et al. Effect of chlorobenzene on the performance of NH3-SCR over Mn6Co4Ox catalyst. Sep. Purif. Technol. 2025, 357, 129901.
93. Li, Z.; Gao, Y.; Wang, Q. The influencing mechanism of NH3 and NOx addition on the catalytic oxidation of toluene over Mn2Cu1Al1Ox catalyst. Journal. of. Cleaner. Production. 2022, 348, 131152.
94. Pan, H.; Chen, Z.; Ma, M.; et al. Mutual inhibition mechanism of simultaneous catalytic removal of NOx and toluene on Mn-based catalysts. J. Colloid. Interface. Sci. 2022, 607, 1189-200.
95. Deng, J.; Hu, X.; Klaver, A. J.; et al. Catalyst deactivation in the abatement of atmospheric pollutants: origin, resistance, and regeneration. Chem. Rev. 2025, 125, 11260-357.
96. Pan, H.; Chen, Z.; Feng, X.; et al. Sulfur-resistant mechanism of MnOx@CeO2@MgO core-shell catalyst in simultaneous removal of NOx and chlorobenzene. Appl. Catal. B. Environ. Energy. 2025, 362, 124761.
97. Zhang, X.; Lin, X.; Xue, Q.; et al. MnOx@CeSnOx with core-shell structure and electronic interaction boosting sulfur tolerance in simultaneous removal of NOx and chlorobenzene. Appl. Catal. B. Environ. Energy. 2026, 381, 125868.
98. Li, G.; Shen, K.; Wu, P.; et al. SO2 poisoning mechanism of the multi-active center catalyst for chlorobenzene and NOx synergistic degradation at dry and humid environments. Environ. Sci. Technol. 2021, 55, 13186-97.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data












Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.