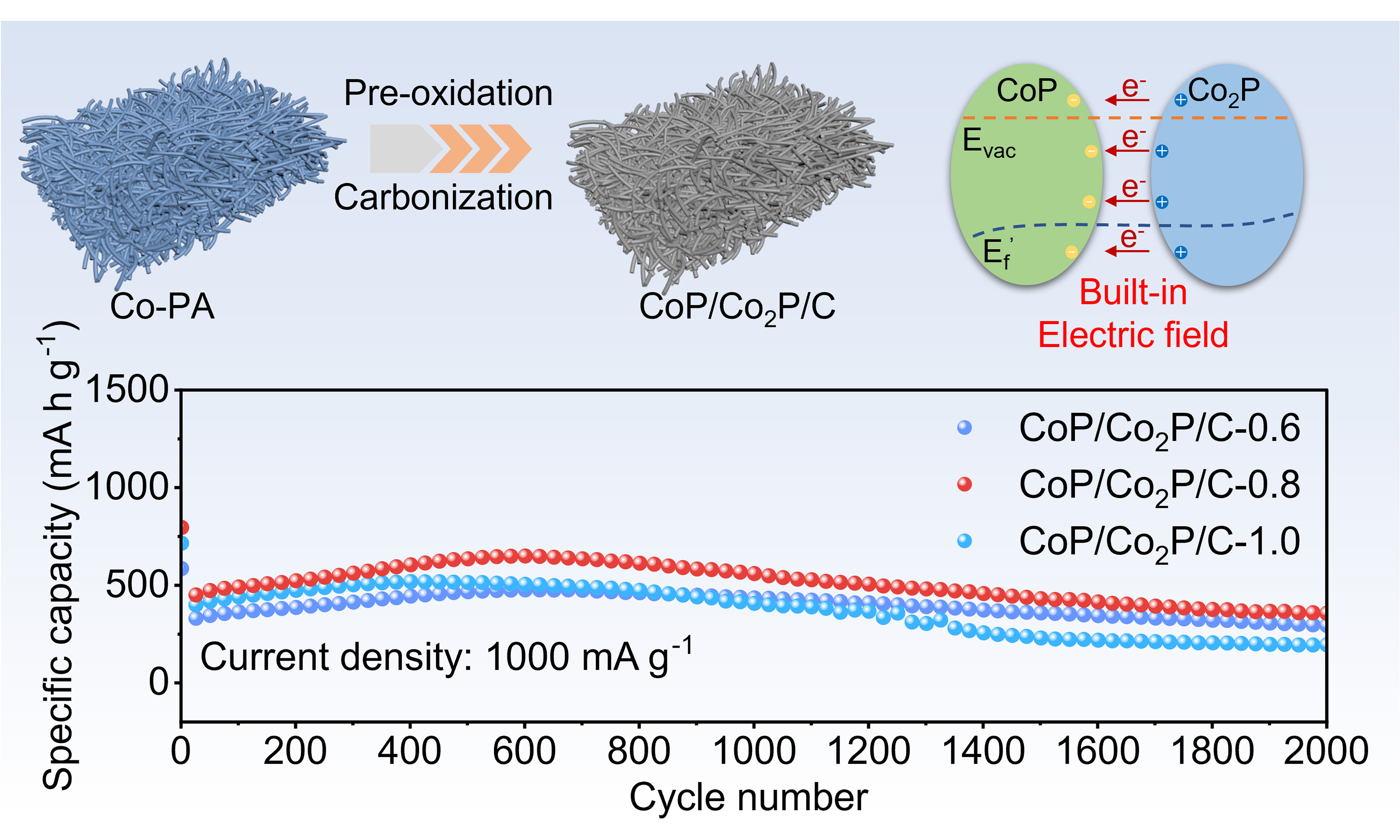
CoP/Co2P heterostructure in N, P co-doped carbon nanofibers enable superior lithium storage via the built-in electric field
Abstract
Transition metal phosphides (TMPs) have garnered significant attention as anode for lithium-ion batteries (LIBs) owing to their high theoretical capacity and moderate Li-intercalation potential. However, TMP still suffer from challenges, including severe volume effects and poor electrical conductivity. Herein, the heterostructure nanofibers anode is synthesized by uniformly distributing CoP/Co2P nanoparticles onto N, P co-doped carbon substrate (CoP/Co2P/C). The built-in electric field generated by the heterostructure enhances electron/ion conductivity, provides additional Li storage sites, thereby optimizing electrochemical performance. The CoP/Co2P/C nanofibers exhibit great cycling stability in applications as LIBs anodes, maintaining the specific capacity above 356 mA h g-1 after 2000 cycles under 1,000 mA g-1. By regulating the ratio of CoP to Co2P, the numbers of heterostructure within the nanofibers were effectively controlled. Based on this, the correlation between heterostructure and electrochemical performance was analyzed. The strategy of constructing heterostructure using the same metal significantly simplified the preparation process for high-performance TMPs anode, providing a viable approach for developing novel anode for LIBs.
Keywords
INTRODUCTION
Solar energy, as the most abundant renewable resource, holds the key to breaking free from fossil fuels, mitigating climate change, and safeguarding energy security. However, its inherent intermittency limits widespread application. Efficient energy storage is crucial for establishing a continuous, stable and dispatchable energy supply network. Owing to the high energy/power density and long cycle lifespan, lithium-ion batteries (LIBs) account for a dominant share of the current energy storage market[1,2]. However, constrained by the lower theoretical specific capacity of graphite anodes (372 mA h g-1), the electrochemical performance of lithium-ion batteries requires further improvement. Therefore, it is imperative to develop anode for LIBs that exhibit high specific capacity and stable cycling performance[3-5].
Transition metal phosphides (TMPs) are considered promising anode, primarily due to their high theoretical specific capacity and suitable voltage plateau[6-9]. The cycling stability and rate performance of TMPs requires improvement. This is due to the poor conductivity and significant volume effect of TMPs[10-14]. Several approaches have been developed to improve the electrochemical performance of TMPs, such as designing nanostructure, compositing with carbon materials, and preparing porous structures. First, the introduction of nanostructures facilitates faster transport of Li+ and electrons by shortening their diffusion distance, and avoid pulverization caused by volume effects and enhance the electrochemical performance of TMPs. Second, designing TMPs as composites with conductive materials can overcome their shortcomings in conductivity[15,16]. In addition, carbon also can keep the anode structurally intact during severe volume changes, thereby extending the cycle life of the electrode. Another effective approach to improve the electrochemical performance of TMPs is design porous structures. The porous structure prevents pulverization of TMPs during charging and discharging, significantly enhancing cycle stability[17]. Porous structure also increases the electrochemical reaction area, accelerating Li+ diffusion at the electrode-electrolyte interface.
In addition to these strategies, introduction of heterostructure also serves as a feasible measure to optimize the electrochemical performance of TMPs. The introduction of heterostructure is able to modify the crystal and electronic structures, generating numerous defects and lattice distortions, and inducing the built-in electric field (BIEF). This provides additional active sites and Li+ transport channels[18]. Additionally, heterostructures can prevent powdering caused by volume effects, thereby enhancing the cycling stability of the anode. Finally, the heterostructure can combine the advantages of different materials to achieve optimal electrochemical performance. For instance, Xiao et al. synthesized the hierarchical hybrid anode [Co(OH)2/Ni2P@N-C] by uniformly dispersing N-doped carbon coated Ni2P nanoparticles on Co(OH)2 nanosheets. This unique hybrid structure enhances electron/ion conductivity, resulting in superior electrochemical performance as an anode material compared to Ni2P@N-C [19]. Li et al. optimized the electrochemical performance of TMPs by the BIEF formed at the FeP/SnP heterointerface[20]. The study found that this BIEF not only optimizes the electrochemical reaction kinetics but also provides additional active sites. When used as the anode for LIBs, the FeP/SnP heterostructure exhibits outstanding electrochemical performance. However, the heterostructure of multi-metal phosphides typically require complex synthesis processes. Constructing such heterostructure using phosphides of the same metal but with different stoichiometric ratios holds promise for significantly simplifying the preparation process while retaining the excellent electrochemical performance. As the typical example of TMPs, cobalt phosphide exhibits multiple thermodynamically stable compounds such as Co2P, CoP, CoP2 and CoP3[7,21-23]. Among these, Co2P belongs to the hexagonal crystal system. It is rich in metallic cobalt-cobalt bonds, thereby exhibiting excellent electronic conductivity; however, its theoretical specific capacity is relatively low. Although CoP, which belongs to the orthorhombic crystal system, possesses the higher theoretical specific capacity, its electrochemical performance is limited by its poor intrinsic conductivity and the severe volume effect during charge/discharge process [21]. Therefore, constructing CoP/Co2P heterostructure is expected to simplify the preparation process and achieve outstanding electrochemical performance.
Herein, CoP/Co2P nanoparticles onto N, P co-doped carbon substrate (CoP/Co2P/C) nanofibers are synthesized via the facile electrospinning and thermal reduction process. By controlling the amounts of Co2+ and phytic acid (PA), the ratio of Co2P to CoP can be altered, thereby obtaining nanofibers with varying heterostructure contents. The difference in work function between CoP and Co2P induces electron transfer at the heterointerface, resulting in the BIEF, which provides the electron/ion transport channels and creates novel active sites[18]. Moreover, the abundant N, P co-doped carbon (NPC) significantly improves the conductivity of the TMPs, alleviates mechanical stress caused by volume changes, thereby enables TMPs anodes with long lifespan and excellent rate performance. When used as anode for LIBs, CoP/Co2P/C nanofibers show great electrochemical performance. At the current density of 1,000 mA g-1, the specific capacity of CoP/Co2P/C remained above 356 mA h g-1 after 2,000 cycles, demonstrating long cycle lifespan and excellent high-current charge/discharge capability. This result shows that the heterostructure composed of TMPs with different stoichiometric ratios of the same metal can combine the advantages of both TMPs, making it a promising anode material for high-performance LIBs. This simple heterostructure preparation method is also expected to be applicable to the design of other anodes.
EXPERIMENTAL
Synthesis of Co-PA nanofibers
First, the PA (0.424 g) was added into dimethylformamide (DMF, 10 mL) and stirred at 80 °C. Next, added polyacrylonitrile (PAN, 0.8 g) and stirred for 4 h. Then, a quantity of CoCl2·6H2O was added and stirred for 12 h, resulting in the electrospinning solution. Finally, electrospinning was performed at 15kV[22]. When the mass of CoCl2·6H2O in the electrospinning solution was 0.4, 0.6, 0.8 and 1.0 g were named Co-PA-n (n is the mass of CoCl2·6H2O).
Synthesis of CoP/Co2P/C nanofibers
Firstly, the Co-PA film was pre-oxidized at 250 °C. Subsequently, carbonize it for 3 h in the Ar atmosphere under 800 °C. The resulting product was named as CoP/Co2P/C-n (n is the mass of CoCl2·6H2O).
Synthesis of CoP/C nanofibers
To synthesize a single-phase CoP, Co-PA-0.4 was prepared using an electrospinning solution containing 0.4 g of CoCl2·6H2O. Following pre-oxidation and carbonization. The resulting product is CoP/C nanofibers.
RESULTS AND DISCUSSION
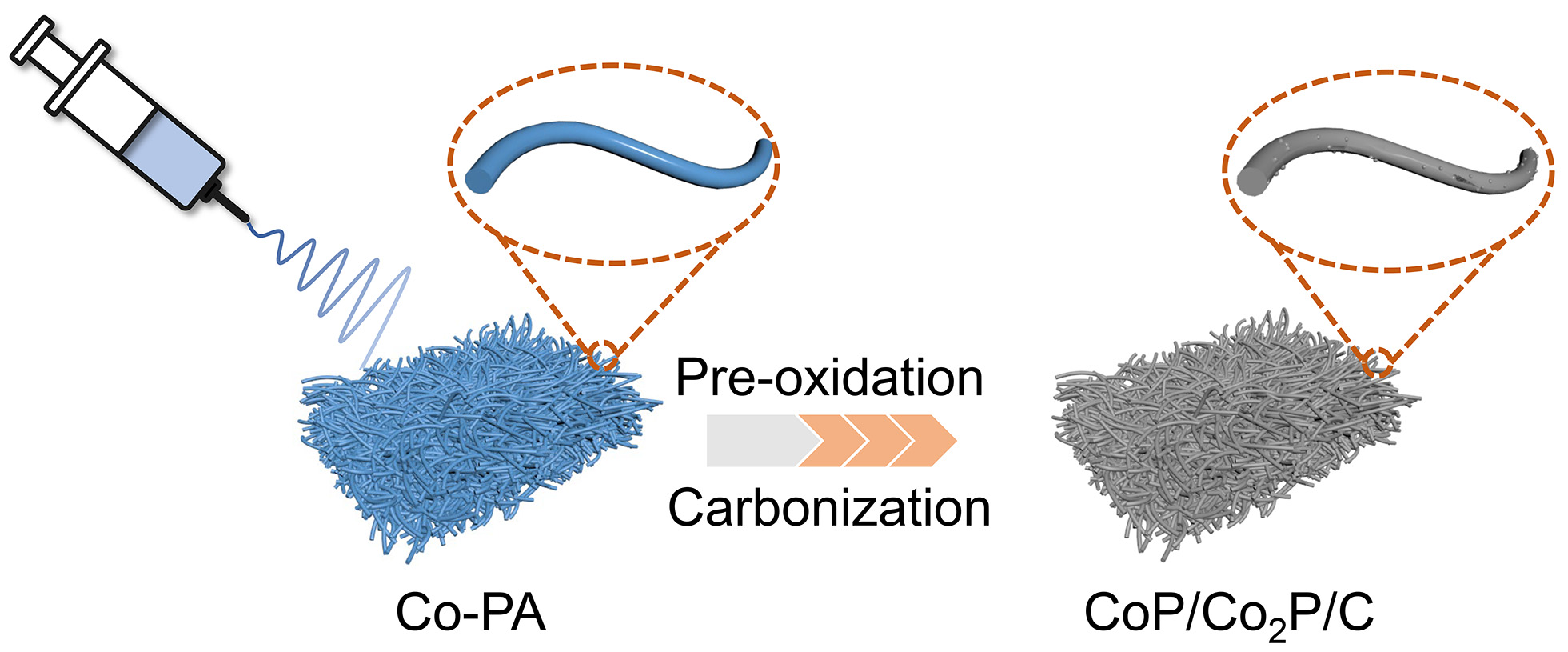
The synthesis process of nanofibers is illustrated in Figure 1. First, the nanofibers film, containing the phosphorus source and CoCl2, is prepared via electrospinning. Subsequently, the precursor undergoes pre-oxidation and carbonization. During this process, -PO4 in PA reacts with Co2+ to form a CoP/Co2P heterostructure. Additionally, the carbon-containing parts of PA and PAN are reduced to carbon substrate doped with N and P. This substrate tightly coats the surface of the CoP/Co2P nanoparticles, ultimately forming CoP/Co2P/C nanofibers.
Figure 1. Schematic of the preparation process of CoP/Co2P/C. PA: Phytic acid; CoP/Co2P/C: CoP/Co2P nanoparticles onto N, P co-doped carbon substrate.
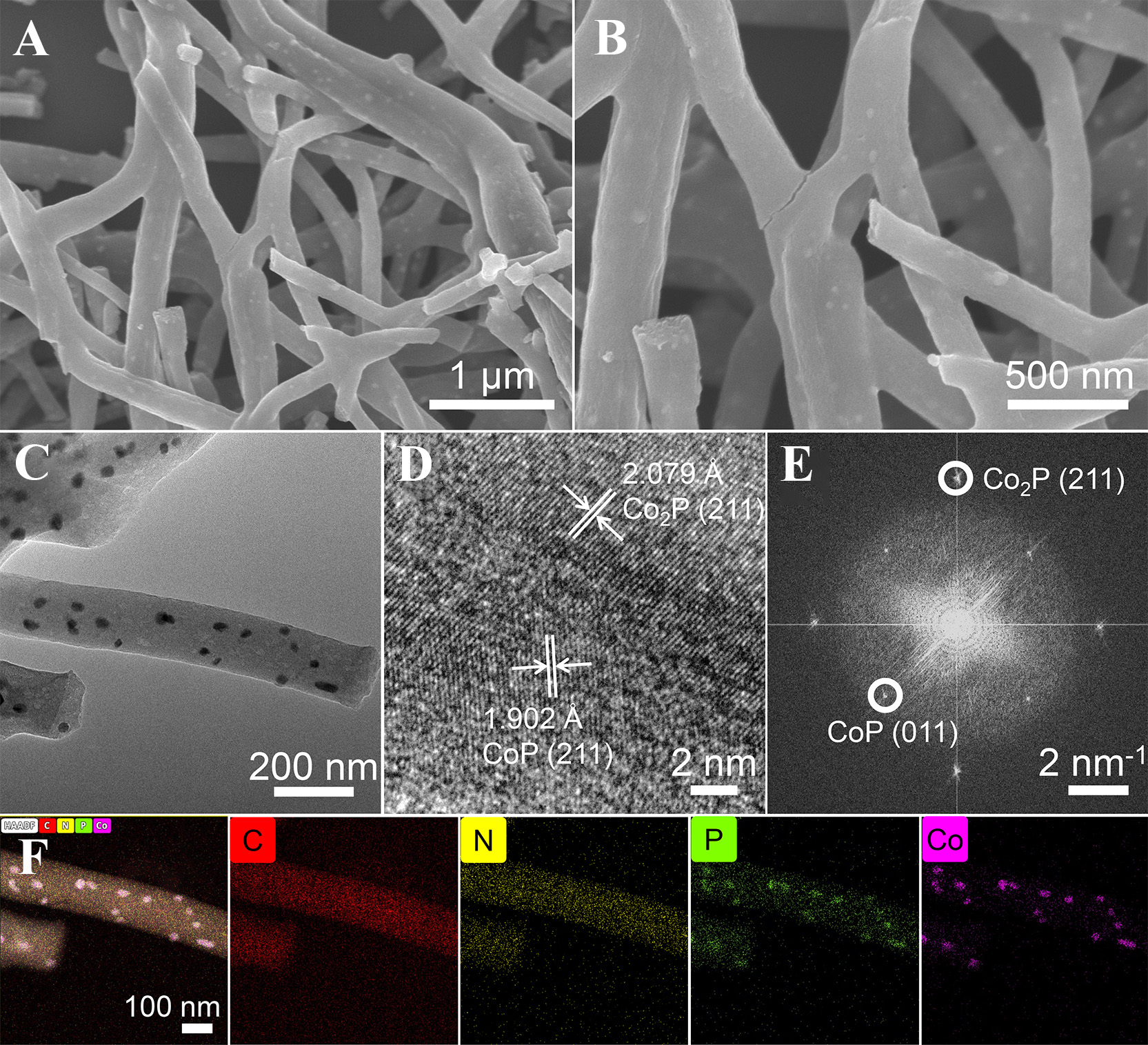
Morphology and surface changes of the nanofibers during the preparation process is analyzed through scanning electron microscopy (SEM) characterization. As displayed in Supplementary Figure 1, the morphology of the precursor is nanofibers, whose diameter is approximately 200 nm. The surface of nanofibers is smooth with no visible pores. To enhance the structural toughness of the nanofibers, they are subsequently subjected to the pre-oxidation treatment. The pre-oxidized samples exhibit the network structure composed of nanofibers [Supplementary Figure 2], which facilitates the formation of the conductive network. Finally, the nanofibers undergo high-temperature treatment to transform into the CoP/Co2P/C. For CoP/Co2P/C-0.8 [Figure 2A and B], it remains interconnected nanofibers. Additionally, there are nanoparticles uniformly distributed on the surface of nanofibers, the diameter of nanoparticles is approximately 20 nm. The small size of nanoparticles effectively relieves the significant volume change during the charge/discharge process, improving cycling stability. Additionally, numerous pores exist on the surface of nanofibers, which is beneficial for electrolyte infiltration and thus accelerates Li+ diffusion at the interface. The morphology of CoP/Co2P/C-0.6 and CoP/Co2P/C-1.0 is similar to CoP/Co2P/C-0.8 [Supplementary Figure 3], also exhibiting nanofibers structures composed of CoP/Co2P nanoparticles and carbon substrate.
Figure 2. (A and B) SEM images; (C) TEM image; (D) HRTEM image; (E) SAED image; and (F) EDS elemental mapping of CoP/Co2P/C-0.8. SEM: Scanning electron microscopy; TEM: transmission electron microscopy HRTEM: high-resolution transmission electron microscopy; SAED: selected area electron diffraction; EDS: energy dispersive X-ray spectroscopy.
The structure of CoP/Co2P/C-0.8 is further characterized by transmission electron microscopy (TEM). As illustrated in the Figure 2C, CoP/Co2P/C-0.8 nanofibers consist of uniformly sized nanoparticles and carbon, and carbon is tightly coated on the surface of the nanoparticles. In the high-resolution transmission electron microscopy (HRTEM) image [Figure 2D] of nanoparticles, distinct lattice fringes can be observed. The lattice fringe with the spacing of 1.902 Å corresponds to the (211) crystal plane of CoP, while the lattice fringe with the spacing of 2.079 Å corresponds to the (211) crystal plane of Co2P. There is the significant interface between the two crystals, indicating the presence of the CoP/Co2P heterostructure within the nanoparticles. Furthermore, the selected area electron diffraction (SAED) of the CoP/Co2P/C-0.8 show diffraction spots belonging to CoP and Co2P [Figure 2E], further confirming that the nanoparticles are composed of CoP and Co2P. The energy dispersive X-ray spectroscopy (EDS) elemental mapping results of CoP/Co2P/C-0.8 are given in Figure 2F. The surfaces of the samples contain C, N, P, and Co elements. Among these, C and N are uniformly dispersed throughout the nanofibers, while Co is concentrated on the nanoparticles. For P, distribution occurs not only on the nanoparticles but also on the carbon. This suggests that the nanofibers are consisted of the CoP/Co2P heterostructure and NPC. Furthermore, the N element content is about 5.21 at% [Supplementary Figure 4]. The higher N doping concentration can introduce abundant defects into the carbon substrate, which contributes to enhancing the conductivity and lithium storage capacity of the nanofibers.
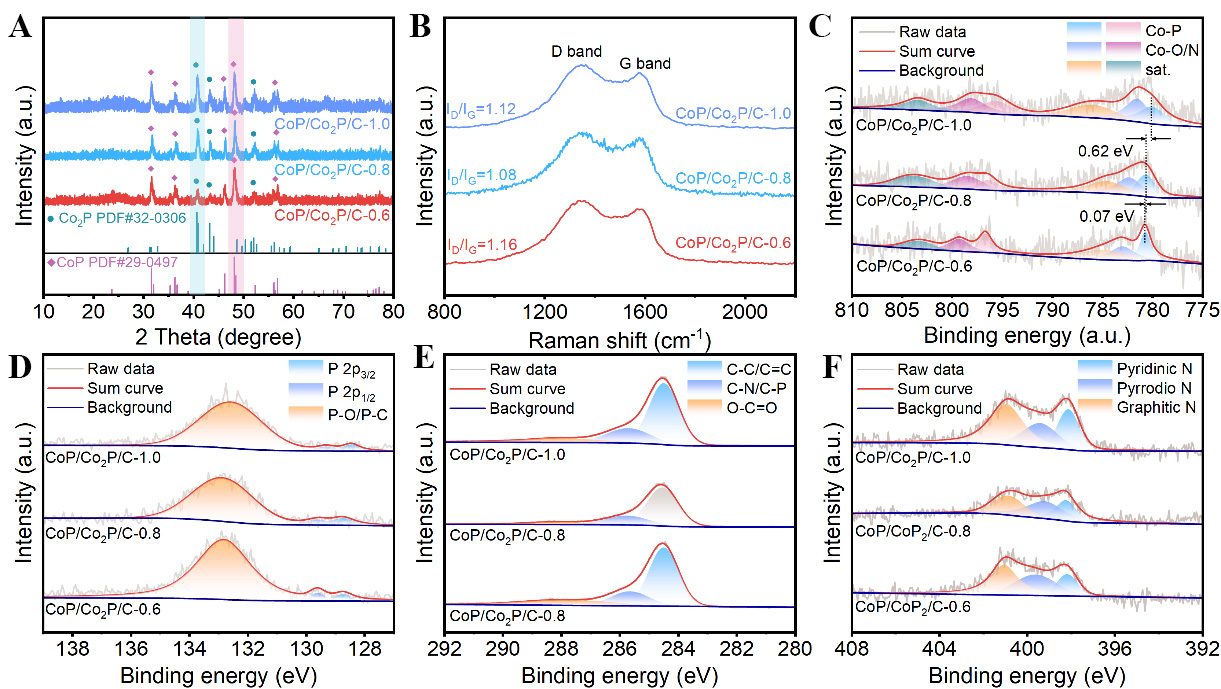
The composition of the nanofibers and the ratio of CoP to Co2P are characterized through X-ray diffraction (XRD) analysis. As shown in the Figure 3A, diffraction peaks belonging to CoP and Co2P are found in the XRD patterns of all three nanofibers. Interestingly, form the CoP/Co2P/C-0.6 to CoP/Co2P/C-1.0, the ratio of the intensity of the (211) crystal plane diffraction peak for Co2P to that of the (211) crystal plane diffraction peak for CoP gradually increases. This indicates that as the ratio of Co2+ to PA increased, the Co2P content in the nanofibers also gradually increased. Furthermore, the XRD patterns of the three nanofibers show no diffraction peaks from other substances, indicating that after the pre-oxidation and reduction processes, Co-PA has been completely converted into CoP/Co2P/C nanofibers without producing any other impurities. In addition, the XRD pattern of CoP/C [Supplementary Figure 5] matches well with the standard CoP pattern, indicating high phase purity without impurity phases. Figure 3B shows the Raman spectra of three kinds of CoP/Co2P/C nanofibers. The two peaks of Raman spectra are belonged to D peaks (belonging to disordered carbon) and G peaks (belonging to graphitic carbon), respectively. The ratio of D peaks to G peaks intensity (ID/IG) values of all CoP/Co2P/C nanofibers fall within the range of 1.08-1.16. This indicates that the NPC of the nanofibers is all abundant in defects[24,25]. These defects will provide a large number of Li+ adsorption sites, thereby enhancing the specific capacity of nanofibers. To analyze the thermal stability of the sample, nanofibers are tested by thermogravimetric analysis in the air [Supplementary Figure 6]. For CoP/Co2P/C-0.8, the mass change is mainly attributed to the desorption of H2O and air, the decomposition of carbon, and the oxidation of CoP/Co2P[21]. Calculations indicate that the content of nanoparticles in CoP/Co2P/C-0.8 ranges between 28.8% and 31.8%. As the content of Co2P in the nanofibers increases, the mass loss of nanofibers at 900 °C is primarily reduced. This is attributed to the increased content of Co2P and decreased carbon content within the nanofibers.
Figure 3. Structures and compositions of CoP/Co2P/C nanofibers: XRD patterns (A); Raman spectra (B); high-resolution XPS survey spectra of Co 2p (C), P 2p (D), C 1s (E), and N 1s (F). CoP/Co2P/C: CoP/Co2P nanoparticles onto N, P co-doped carbon substrate; XRD: X-ray diffraction; XPS: X-ray photoelectron spectroscopy.
X-ray photoelectron spectroscopy (XPS) measurements are performed on the nanofibers to analyze the valence states of chemical elements on their surfaces. As shown in Supplementary Figure 7, the CoP/Co2P/C nanofibers contain Co, P, N and C elements. Figure 3C is the Co 2p high-resolution spectrum for CoP/Co2P/C-0.8. The peaks at 780.69 eV and 796.40 eV represent the Co 2p3/2 and 2p1/2 orbitals of Co-P, respectively. The appearance of these peaks confirms the successful conversion of Co-PA into CoP/Co2P. Additionally, peaks belonging to surface-oxidized cobalt (782 eV and 795 eV) are observed. This is common in metallic TMPs, mainly due to sample oxidation upon exposure to air[26,27]. Importantly, as the increase of Co2P content in CoP/Co2P nanoparticles, the Co-P peak gradually shifts toward lower binding energy. This is attributed to electron transfer occurring at the CoP/Co2P heterointerface, where the resulting BIEF will provide additional Li+ storage sites and fast diffusion channels for Li+. In the P 2p spectrum of CoP/Co2P/C-0.8 [Figure 3D], P-Co peaks are observed with binding energies of 128.74 eV (P 2p3/2) and 129.62 eV (P 2p1/2). The presence of P-Co bonds suggests that after high-temperature reaction, Co2+ react with P from PA to form CoP/Co2P. The peak at 132.88 eV correlates with the P-O/P-C bond. The P-O bond mainly results from the oxidation of CoP/Co2P in air. The presence of P-C bonds indicates that phosphorus has been successfully doped into the carbon, which contribute to the electrical conductivity and lithium storage capacity of the nanofibers[10]. The P 2p high-resolution spectra of CoP/Co2P/C-0.6 and CoP/Co2P/C-1.0 are similar to that of CoP/Co2P/C-0.8. The slight shifts in peak positions are also attributed to electron transfer at the heterointerface. The high-resolution C 1s spectra of CoP/Co2P/C-0.8 are composed of C-C/C=C, C-P/C-N and O-C=O. As shown in Figure 3F, the high-resolution N 1s spectrum exhibits three fitted peaks at 398.24 eV (pyridinic N), 399.22 eV (pyrrolic N), and 400.87 eV (graphitic N). This contributes significantly to enhancing electrical conductivity[28,29].
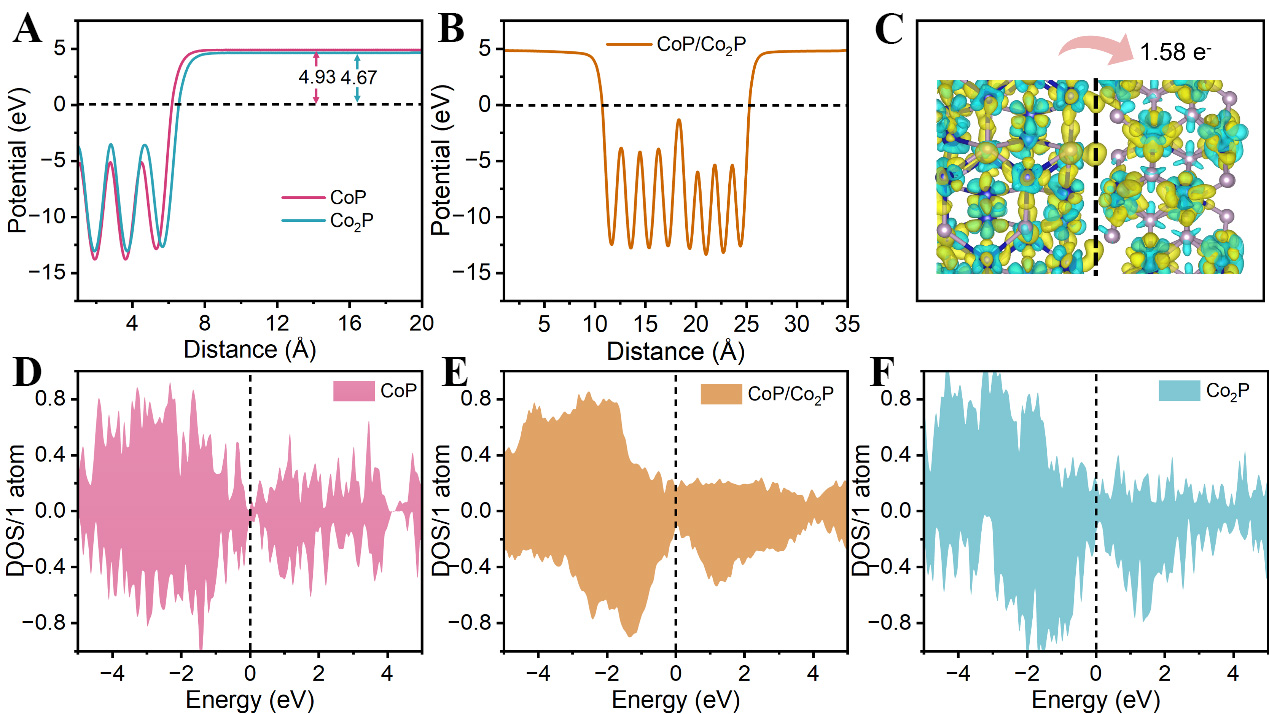
To further analyze the charge transfer direction and quantity in the CoP/Co2P heterostructure, the work function difference between CoP and Co2P is calculated by density functional theory (DFT). Computational details are provided in Supporting information. As shown in Figure 4A, the work function value of CoP is 4.67 eV, which is lower than that of Co2P at 4.93 eV, indicating a significant difference between the materials. In the CoP/Co2P heterostructure model, the work function is further calculated [Figure 4B], which is situated between CoP and Co2P (4.82 eV). This is driven by the difference in the work function of the CoP and Co2P. As demonstrated by the spin charge density differences and Bader charge analysis, it is clear that charge redistribution is evident at the interface of CoP/Co2P [Figure 4C]. This results in a charge transfer of 1.58 eV electrons from Co2P to CoP. The total density of states (TDOS) for CoP, CoP/Co2P heterostructure and Co2P are summarized in Figure 4D and E. The TDOS of CoP/Co2P heterostructure also shows charge reconstruction upon building the heterostructure. Importantly, the occupancy around the Fermi level of the CoP/Co2P heterostructure is comparable to that of Co2P and significantly higher than CoP, indicating that the CoP/Co2P heterostructure combines the great electronic conductivity of Co2P with the high theoretical specific capacity of CoP.
Figure 4. (A and B) The work functions of CoP, Co2P and CoP/Co2P heterostructure; (C) Bader charge and spin charge density differences of CoP/Co2P heterostructure; (D-F) TDOS of CoP, CoP/Co2P heterostructure and Co2P. DOS: Density of states; TDOS: total density of states.
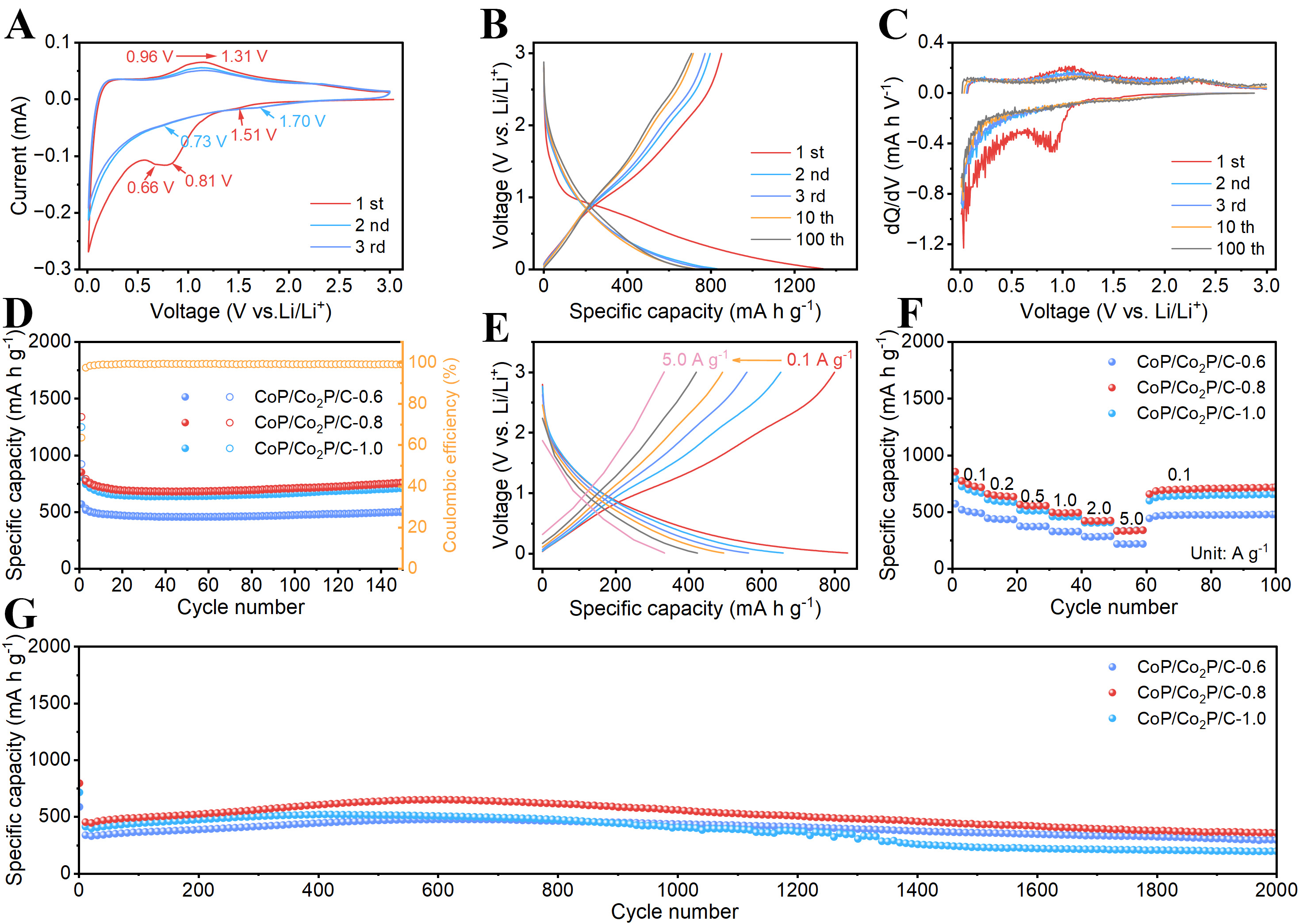
The electrochemical performance of nanofibers as anode for LIBs is measured by assembling half-cells. Figure 5A shows the cyclic voltammetry (CV) curves of CoP/Co2P/C-0.8 between 3.0 V and 0.01 V under 0.1 mV s-1. In the first cathodic scan, the peak at 1.51 V corresponds to the electrochemical reaction of Li insertion into CoP/Co2P to form LixCoP and LixCo2P[23,30]. Subsequently, the peak observed at 0.81 V, which disappears in subsequent cycles, corresponds to the formation of the solid electrolyte interphase (SEI) film[31]. The peak observed at 0.66 V demonstrates the electrochemical reaction of lithiation of LixCoP and LixCo2P into Li3P and Co. During the subsequent charging process, peaks appear between 0.96 and 1.31 V, indicating that Co and Li3P multiple conversion steps to reform CoP/Co2P. During the second charge/discharge cycle, the reduction peaks shifted from 1.51 V and 0.66 V to 1.70 V and 0.73 V, respectively. This shift resulted from the irreversible phase transition, structural reorganization, and changes in interfacial impedance that occurred in CoP/Co2P/C-0.8 during the first cycle. Most importantly, the CV curves in subsequent cycles of CoP/Co2P/C-0.8 show good overlap, indicating excellent electrochemical reversibility and cycling stability. Furthermore, the CV curves of the other three nanofibers are similar to those of CoP/Co2P/C-0.8 [Supplementary Figure 8A, 9A and 10A], suggesting they undergo comparable electrochemical processes during cycling.
Figure 5. Electrochemical performance of CoP/Co2P/C-0.8. (A) CV curves with the scan rate of 0.1 mV s-1; (B) charge/discharge curves with the current density of 100 mA g-1; (C) dQ/dV curves; (D) cycling performance of different nanofibers with the current density of 100 mA g-1; (E) charge/discharge curves with different current densities; (F) rate capabilities and (G) cycling performances under 1,000 mA g-1 of different nanofibers. CoP/Co2P/C: CoP/Co2P nanoparticles onto N, P co-doped carbon substrate; CV: cyclic voltammetry; dQ/dV: differential capacity.
The cycling performance of the nanofibers is evaluated at 100 mA g-1. Supplementary Figure 8B, 9B, 10B and Figure 5B is the galvanostatic charge/discharge curves of nanofibers. The CoP/Co2P/C-0.8 exhibited discharge and charge capacities of 1,339 and 852 mA h g-1 in the first cycle, corresponding to Coulombic efficiency of 63.6%. This loss in the first cycle is mainly due to SEI formation and irreversible electrolyte decomposition and incomplete delithiation induced by electrode structure rearrangement[32-33]. After the initial cycle, the charge/discharge curves remain stable, demonstrating excellent electrochemical reversibility, which is crucial for its cycling stability. Figure 5C shows the differential capacity (dQ/dV) curves of CoP/Co2P/C-0.8. The positions of the redox peaks on this curve are similar to CV curves. Furthermore, after 100 cycles, the position of the redox peaks remained the same, showing that the electrochemical reaction of CoP/Co2P/C-0.8 is reversible. Furthermore, the dQ/dV curves of CoP/C, CoP/Co2P/C-0.6 and CoP/Co2P/C-1.0 are similar to those of CoP/Co2P/C-0.8 [Supplementary Figure 8B, 9B and 10B]. Figure 5D and Supplementary Figure 11A shows the cycling performance of the four nanofibers at 100 mA g-1. After 100 cycles, the specific capacity of the CoP/Co2P/C-0.8 is 785 mA h g-1, significantly exceeding that of the CoP/C (429 mA h g-1), CoP/Co2P/C-0.6 (503 mA h g-1) and CoP/Co2P/C-1.0 (716 mA h g-1). The superior cycling performance of CoP/Co2P/C-0.8 is mainly attributed to its optimal CoP and Co2P ratios and CoP/Co2P heterostructure. The high CoP ratio reduces the conductivity of nanofibers, preventing some active materials from joining electrochemical reactions and causing the decrease in specific capacity. However, the high Co2P ratio reduces the theoretical specific capacity of nanofibers, resulting in lower specific capacity. Furthermore, the optimal CoP and Co2P ratio introduces the appropriate number of heterointerface, thereby creating more Li storage sites. To evaluate the rate performance of the four nanofibers, they are charged and discharged under various current densities. At different currents density, the plateau of the curve is not significantly shifted, and polarization is smaller, indicating that ion/electron transfer of CoP/Co2P/C-0.8 is fast and the delithiation/lithiation process is stable and reversible [Figure 5E]. The rate performance of CoP/Co2P/C nanofibers are shown in Figure 5F and Supplementary Figure 11B. The CoP/Co2P/C-0.8 exhibits superior rate capability, with reversible capacities of 726, 643, 554, 491, 422, and 334 mA h g-1 at increasing current densities from 100 to 5,000 mA g-1, which is higher than that of CoP/C, CoP/Co2P/C-0.6 and CoP/Co2P/C-1.0. Importantly, the specific capacity recovers to 708 mA h g-1 when the current density is restored to 100 mA h g-1, demonstrating excellent rate performance. Then, the CoP/Co2P/C is cycled under 1,000 mA g-1 to evaluate its cycling life. After 2,000 cycles, CoP/Co2P/C-0.8 retained the specific capacity exceeding 356 mAh g-1, demonstrating outstanding cycling performance. Additionally, as shown in Supplementary Figure 12, CoP/Co2P/C-0.8 maintains the nanofibers morphology after many cycles, suggesting that its heterostructure, smaller particle size, and abundant carbon effectively prevent electrode pulverization and agglomeration during cycles. This is a key reason for its outstanding electrochemical performance.
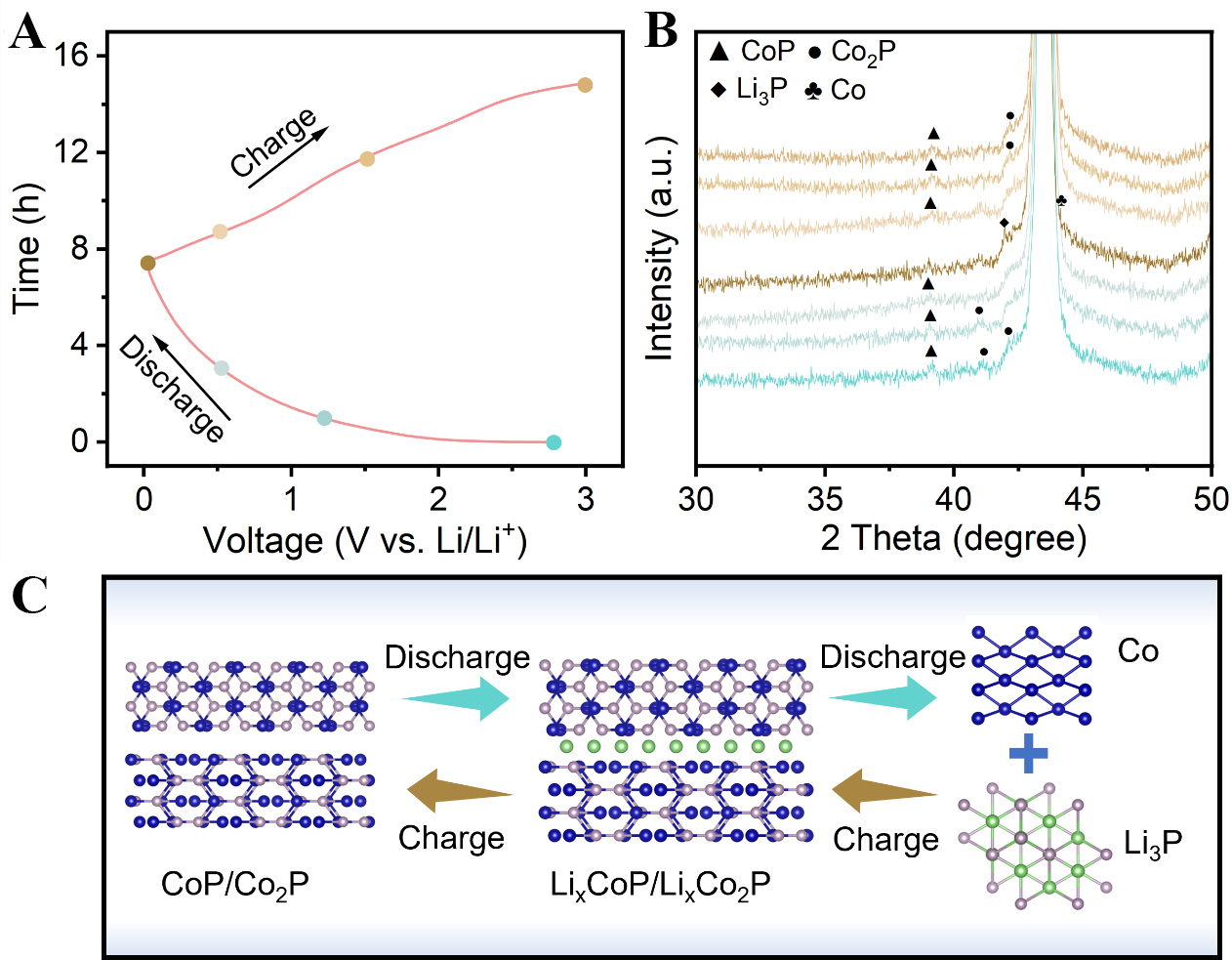
The lithiation mechanism of CoP/Co2P/C-0.8 nanofibers is elucidated by ex situ XRD. Figure 6A and B shown the charge/discharge curves and ex situ XRD analysis results of the electrodes at different voltage states. The diffraction peaks before lithiation correspond to CoP and Co2P. When discharge to 1.2 V, the diffraction peaks shift towards smaller angles, attributable to lattice expansion caused by Li+ insertion into the CoP and Co2P lattices. When discharge continues to 0.5 V, the diffraction peaks shift further and the intensity weakens. Upon complete discharge to 0.01 V, the CoP and Co2P phase disappear, forming Co and Li3P phases. During subsequent charging cycles, the Li3P and Co phases gradually disappear, while the CoP and Co2P phases reform, indicating that the redox reaction is fully reversible. Based on the above analysis, the electrochemical mechanism of CoP is illustrated in Figure 6C. During the discharge stage, Li first inserts into the lattice of CoP and Co2P, and finally forms Co and Li3P. Importantly, the charging and discharging process is highly reversible.
Figure 6. (A) Galvanostatic charge/discharge curves of CoP/Co2P/C-0.8; (B) Ex-situ XRD patterns under different cycled states; (C) Schematic diagram of lithium storage mechanism of CoP/Co2P/C nanofibers during the charge/discharge processes. CoP/Co2P/C: CoP/Co2P nanoparticles onto N, P co-doped carbon substrate; XRD: X-ray diffraction.
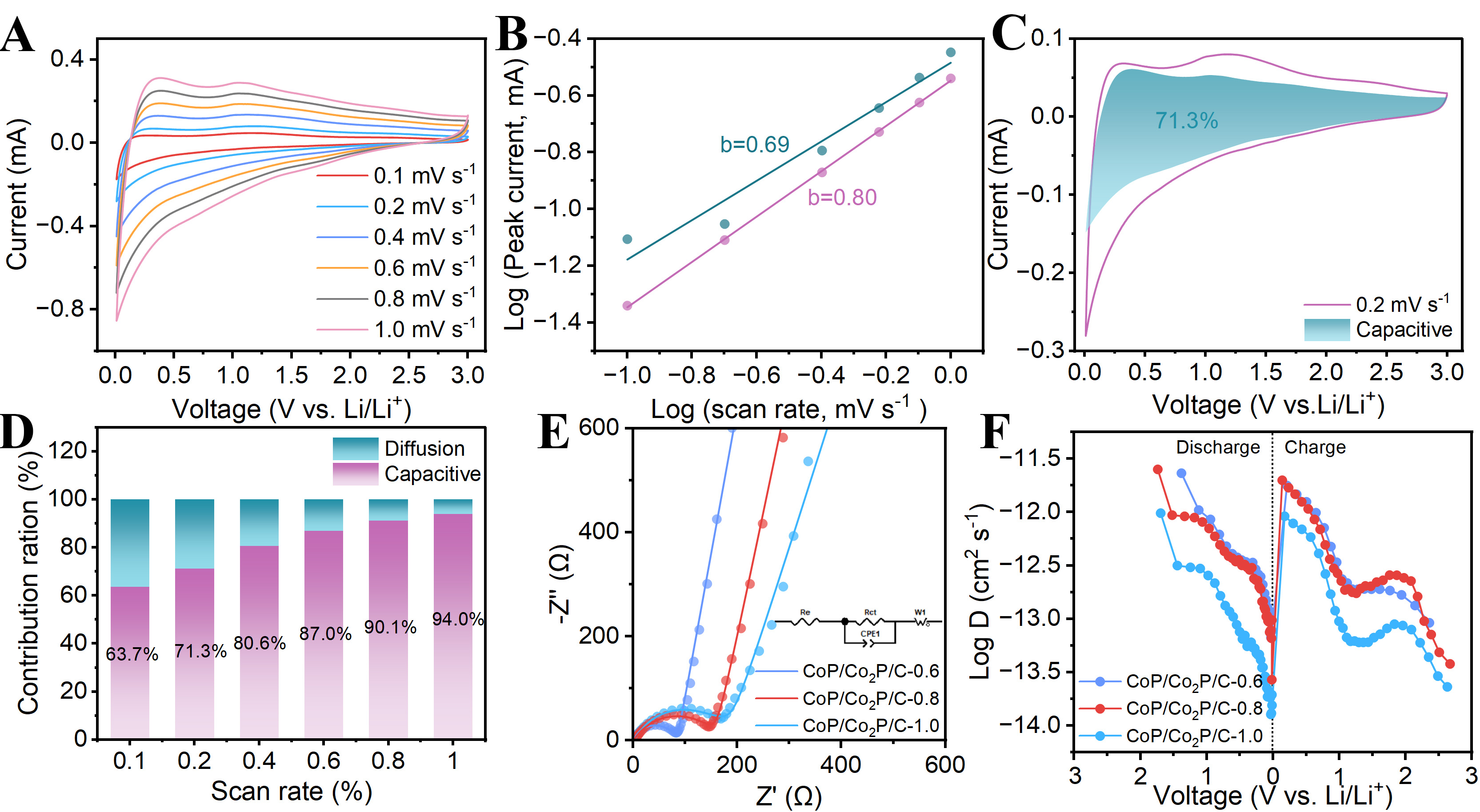
To analyze the reasons for the differences in electrochemical performance of nanofibers, CV tests are performed on them at different scan rates. Supplementary Figure 13A, 14A, 15A and Figure 7A shows the CV curves of CoP/C and CoP/Co2P/C nanofibers. For the CoP/Co2P/C-0.8, the redox peaks are not disappeared with increasing of scan rate, only slightly shifted. This indicates that CoP/Co2P/C-0.8 exhibits excellent ionic/electronic conductivity and fast Li+ storage kinetics. Additionally, the lithium storage mechanism of nanofibers is analyzed using the formula: i = avb. Here, v denotes the scan rate, i denotes the peak current, a and b are constants. If b = 0.5, it indicates diffusion-controlled electrochemical reactions; b = 1 indicates capacitive-controlled lithium storage[31,34]. For CoP/C and CoP/Co2P/C nanofibers [Supplementary Figure 13B, 14B, 15B and Figure 7B], their b values all fall between 0.5 and 1, suggesting that Li+ storage processes is co-controlled by diffusion and capacitive. To calculate the ration of pseudo-capacitive contribution to the capacity, the formula: i = k1v + k2v1/2 is employed for analysis. Here, k1v represents the current contributed by pseudo-capacitance, while k2v1/2 denotes the current contributed by diffusion-controlled. At 0.2 mV s-1, pseudo-capacitance contributes 71.3% of the capacity of CoP/Co2P/C-0.8 [Figure 7C], which is exceeding than that of CoP/C, CoP/Co2P/C-0.6 and CoP/Co2P/C-1.0 [Supplementary Figure 13C, 14C and 15C]. Moreover, for CoP/Co2P/C-0.8 [Figure 7D] the pseudo-capacitive contribution progressively increased with rising scan rates, ultimately reaching 94%. This value is higher than CoP/C, CoP/Co2P/C-0.6 and CoP/Co2P/C-1.0. [Supplementary Figure 13D, 14D and 15D]. This indicates that charge separation induced by BIEF can enhance the pseudo-capacitive contribution, thereby increasing lithium-ion diffusion rates and Li+ storage sites. Figure 7E is the electrochemical impedance spectroscopy (EIS) spectrum of three kinds of CoP/Co2P nanofibers before cycling. After fitting, the charge transfer resistance (Rct) of the three CoP/Co2P/C nanofibers are 71.63,139.3, and 152.2 Ω, respectively. As carbon content increases, the value of Rct gradually rises, indicating that the conductive network formed by NPC optimizes the conductivity of nanofibers. Additionally, the impedance spectrum slopes of CoP/Co2P/C-0.6 and CoP/Co2P/C-0.8 in the low-frequency region are steeper than that of CoP/Co2P/C-1.0, indicating superior Li+ diffusion performance. This is primarily attributed to the moderate ratio of CoP to Co2P in the nanofibers, which results in the formation of abundant heterointerface. These heterointerfaces provide rapid diffusion channels for Li+. The Li+ diffusion coefficient (DLi+) is determined using the galvanostatic intermittent titration technique (GITT), with complete measurement conditions and the calculation process detailed in the Supplementary Materials [Supplementary Figure 16]. As demonstrated in Figure 7F, among the three nanofibers, CoP/Co2P/C-0.6 and CoP/Co2P/C-0.8 exhibit higher DLi+. This is attributed to their optimized CoP/Co2P ratio, which promotes the formation of more heterointerface. The BIEF generated by these heterointerface provides the rapid diffusion channels for Li⁺, which is the key reason for the outstanding rate performance exhibited by CoP/Co2P/C-0.8.
Figure 7. Electrochemical kinetic behavior analysis of CoP/Co2P/C-0.8. (A) CV curve as the scan rate increases from 0.1 to 1.0 mV s-1; (B)b values; (C) pseudo-capacitive contribution at 0.2 mV s-1; (D) the values of pseudo-capacitive contribution at different scan rates; (E) EIS curves and (F) Li+ diffusion coefficients of different nanofibers. CoP/Co2P/C: CoP/Co2P nanoparticles onto N, P co-doped carbon substrate; EIS: electrochemical impedance spectroscopy.
CONCLUSIONS
In summary, nanofibers containing the CoP/Co2P heterostructure and NPC are effectively prepared using electrospinning and reduction approaches. By controlling the ratio of Co2+ to PA in the electrospinning solution, the radio of CoP and Co2P as well as the number of heterointerface can be regulated. Owing to its novel heterogeneous structure, the CoP/Co2P/C displays long cycle lifespan and rapid charge/discharge capability for use as the anode in LIBs. First, the NPC optimizes electronic conductivity and avoid pulverization caused by volume effects and enhance cycle stability. Second, the optimal ratio of CoP and Co2P successfully balances high capacity with high conductivity. Finally, the strong coupling between CoP and Co2P within the nanofibers generates abundant defects and BIEF, which creates active sites and Li+ diffusion channels, thereby enhancing electrochemical performance of nanofibers. This work demonstrates that introducing an appropriate number of CoP/Co2P heterostructure can effectively enhance their electrochemical performance, which is expected to be applicable in the design of other high-performance anodes.
DECLARATIONS
Authors’ contributions
Conceived the idea, conducted part of the experiments, and wrote the manuscript: Wang, X.
Carried out electrochemical performance measurements: Yue, C.
Synthesized the sample: Wang, W.
Verified the data and revised the manuscript: Yao, F.; Zhu, W.
Characterized the samples: Li, J.
Supervised the project: Nie, P.; Chang, L.
All authors discussed the results and contributed to the revision and finalization of the manuscript.
Availability of data and materials
Some results of supporting the study are presented in the Supplementary Materials.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by the Natural Science Foundation of Jilin Province (YDZJ202301ZYTS296), National Natural Science Foundation of China (52501281), the Research Program on Science and Technology from the Education Department of Jilin Province (JJKH20240558KJ).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Huang, X. Y.; Zhao, C. Z.; Kong, W. J.; et al. Tailoring polymer electrolyte solvation for 600 Wh kg-1 lithium batteries. Nature 2025, 646, 343-50.
2. Wu, D.; Liu, Q.; Zhu, Q.; et al. Fast-charging li-ion battery enabled by an acetonitrile-based electrolyte. ACS. Energy. Lett. 2025, 10, 4911-8.
3. Xiao, Z.; Wu, H.; Quan, L.; et al. Micro-sized CVD-derived Si-C anodes: challenges, strategies, and prospects for next-generation high-energy lithium-ion batteries. Energy. Environ. Sci. 2025, 18, 4037-52.
4. Sharma, G. K.; Elkins, J.; Puthirath, A. B.; et al. Binder-free MoO2-MoO3 nanoarrays as high-performance anodes for Li-Ion batteries. Small 2025, 21, e2500361.
5. Li, Z.; Zhang, Z.; Liao, H.; Zheng, Y.; Gao, Y. Anode materials for proton batteries: progress and prospects. ACS. Nano. 2025, 19, 12680-709.
6. Liu, Y.; Hu, Q.; Shi, Q.; et al. Green synthesis of Cu3P to achieve low-temperature and high initial coulombic efficiency sodium ion storage. Adv. Energy. Mater. 2025, 15, 2500723.
7. Lan, X.; Li, Z.; Zeng, Y.; Han, C.; Peng, J.; Cheng, H. M. Phosphorus‐based anodes for fast‐charging alkali metal ion batteries. EcoMat 2024, 6, e12452.
8. Li, W.; Li, Y.; Wang, J.; et al. Generic synthesis of high-entropy phosphides for fast and stable Li-ion storage. Energy. Environ. Sci. 2024, 17, 5387-98.
9. Li, W.; Wang, J. H.; Yang, L.; et al. High-entropy engineering of cubic SiP with metallic conductivity for fast and durable Li-ion batteries. Adv. Mater. 2024, 36, e2314054.
10. Zhu, H.; Li, W.; Yang, K.; Zhang, L.; Sun, L. One-step thermal synthesis of FeP nanoparticles highly-dispersed into a porous NPPCG framework for enhanced lithium storage. J.. Energy. Storage. 2025, 112, 115595.
11. Zhou, Y.; Wang, Y.; Zhang, Y.; et al. Abundant Cu3P/Co2P/CoP@NC heterostructures boost charge transfer toward fast and durable sodium storage. Carbon. Energy. 2025, 7, e721.
12. Zeng, H.; Liu, R.; Wu, Q.; et al. Naturalized bioreactor assisted fabrication of ferrous(II) phosphate nanodots decorated in carbon for “fast-charging” lithium-ion batteries. Adv. Funct. Mater. 2025, 35, 2503614.
13. Liu, J.; Li, B.; Mu, P.; et al. Fabricating 3D network for FeP@MXene toward stable and high-capacity lithium-ion storage. Small. Methods. 2025, 9, e2500185.
14. Fan, Z.; Wang, J.; Wu, Y.; Zhang, P. Advanced high-entropy materials for high-quality energy storage and conversion. Energy. Storage. Mater. 2025, 74, 103954.
15. Shi, H.; Zhang, Y.; Li, X.; et al. Ordered design of N-doped carbon fiber hybrid bilayer skeleton for controlled bottom-up Na plating/stripping toward anode-less sodium metal batteries. Chem. Eng. J. 2025, 519, 165444.
16. Yuan, F.; Li, Z.; Wang, Q.; et al. sp2 configuration coupled vacancy rich carbon enables excellent low potential potassium storage. Energy. Storage. Mater. 2025, 76, 104119.
17. Islam, T.; Bayat, S.; Wright, M. A.; et al. Porous and amorphous MnxMo3S13 chalcogel electrode for high-capacity conversion-based lithium-ion batteries. J. Am. Chem. Soc. 2025, 147, 7400-10.
18. Zhang, M.; Xiao, H.; Liu, Y.; et al. Interfacial build-in electric field unlocking hetero-architectured CoO‐Co3O4 nanotubes toward high-performance Li‐S batteries. Adv. Funct. Mater. 2025, 36, e20708.
19. Xiao, S.; Chen, Y.; Zhou, X.; et al. Hierarchical phosphide-based hybrid anodes for high-performance lithium-ion batteries. Nano. Lett. 2025, 25, 3532-40.
20. Li, J.; Li, C.; Hu, Y.; et al. Long-lasting lithium-ion batteries enabled by advanced anode design of a hydrangea-like FeP/SnP@C heterostructure. ACS. Appl. Mater. Interfaces. 2025, 17, 12085-94.
21. Hao, Z.; Liu, D.; Zuo, X.; Yu, H.; You, T.; Zhang, Y. Built‐In Electric Field Induced Uniform Li Deposition via Construction of CoP/Co2P Heterojunction in 3D Carbon Nanofiber Networks. Adv. Funct. Mater. 2024, 35, 2415251.
22. An, Y.; Pei, Z.; Luan, D.; Lou, X. W. D. Foldable anode-free sodium batteries enabled by N,P-codoped carbon macroporous fibers incorporated with CoP nanoparticles. Sci. Adv. 2025, 11, eadv2007.
23. Li, S.; Xu, Y.; Zhang, X.; et al. CoP Quantum dots embedded in carbon polyhedra through Co-P-C bonding enabling high-energy lithium-ion capacitors. Adv. Funct. Mater. 2024, 34, 2314870.
24. Zhang, H.; Pang, M.; Yang, M.; et al. Constructing three-dimensional porous SnS2/rGO as superior-rate and long-life anodes for lithium-ion batteries. Langmuir 2025, 41, 876-84.
25. Wang, X.; Xu, Y. J.; Wu, X. S.; Ma, M. W.; Tian, Y.; Su, Z. M. Nitrogen-doped Co3O4@porous carbon derived from metal-organic frameworks for high-performance lithium ion batteries. Langmuir 2025, 41, 10782-8.
26. Liu, L.; Xie, H.; Zheng, Y.; et al. Multicomponent Anodes Based on Amorphous ZnP2 for Fast‐Charging/Discharging Lithium‐Ion Batteries. Advanced. Energy. Materials. 2024, 15, 2404900.
27. Lu, Y.; Kang, Q.; Dong, F.; et al. Metalloid phosphorus induces tunable defect engineering in high entropy oxide toward advanced lithium-ion batteries. Adv. Funct. Mater. 2024, 35, 2413782.
28. Li, X.; Guan, G.; Tong, S.; et al. Two birds with one stone: Bimetallic ZnCo2S4 polyhedral nanoparticles decorated porous N-doped carbon nanofiber membranes for free-standing flexible anodes and microwave absorption. J. Colloid. Interface. Sci. 2025, 678, 1031-42.
29. Chen, B.; Cao, T.; Yu, Y.; et al. Rational design of yolk-shell Fe7S8@C-N for high rate and long cycle Li-Ion batteries. Nano. Lett. 2025, 25, 10279-86.
30. Tan, W.; Liu, Z.; Wu, Q.; et al. Flexible free-standing Fe-CoP-NAs/CC nanoarrays for high-performance full lithium-ion batteries. J. Colloid. Interface. Sci. 2024, 667, 441-9.
31. Yu, J.; He, Y.; Li, J.; et al. In-situ rooting biconical-nanorods-like Co-doped FeP @carbon architectures toward enhanced lithium storage performance. Chem. Eng. J. 2023, 477, 146996.
32. Liu, X.; Yu, Y.; Li, K.; et al. Intergrating hollow multishelled structure and high entropy engineering toward enhanced mechano-electrochemical properties in lithium battery. Adv. Mater. 2024, 36, e2312583.
33. Ou, H.; Huang, M.; Li, P.; et al. Tailoring and understanding the lithium storage performance of triple-doped cobalt phosphide composites. J. Colloid. Interface. Sci. 2024, 672, 753-64.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.