Fully inorganic LLZO-based composite cathodes: the impact of Ga substitution on compatibility with cathode active materials during co-sintering
Abstract
In order to make garnet-based all-solid-state batteries (ASSBs) attractive for industrial applications, their rate capability has to be significantly improved. Recently, cubic Li6.4Ga0.2La3Zr2O12 (LLZO:Ga) was found to have the highest total ionic conductivity of any oxide solid-state electrolyte by far, reaching up to 2 × 10-3 S/cm at room temperature. Since the rate performance of composite cathodes is directly linked to their ionic conductivity, LLZO:Ga is an ideal solid-state electrolyte for high-performance ASSBs. However, careful material selection is required for the fabrication of such ceramic composite cathodes at elevated temperatures in order to avoid incompatibility issues that could lead to low electrochemical performance. We therefore systematically studied the co-sintering behavior of cubic LLZO:Ga in combination with common cathode active materials, including LiCoO2 (LCO), LiNi1/3Mn1/3Co1/3O2 (NCM111), and LiNi0.8Mn0.1Co0.1O2 (NCM811). The analyses were performed using X-ray diffraction, Raman spectroscopy, scanning electron microscopy, and transmission electron microscopy. The experimental conditions were chosen to enable a direct comparison with our previous study on
Keywords
INTRODUCTION
All-solid-state lithium batteries are considered to be promising candidates for next-generation energy storage systems. They offer solutions to several challenges of current lithium-ion battery (LIB) technology, primarily by replacing the flammable organic electrolyte with a solid-state electrolyte (SSE). The SSE can prevent unwanted chemical reactions, resulting in excellent long-term stability. Additionally, their high Li-ion transfer number and low polarization can lead to high power densities. Oxide SSEs enable the use of elemental lithium as an anode, leading to a significant increase in the energy density of all-solid-state batteries (ASSBs) compared to conventional LIBs. At the same time, oxide-based ASSBs offer unsurpassed safety at the cell level, enabling the safe use of lithium-metal anodes even when the formation of dendrites cannot be prevented completely[1-7]. Nevertheless, a large variety of solid electrolytes - such as halides, sulfides, oxides, and polymers - are being investigated, with each individual class exhibiting its own strengths and benefits. To date, no individual solid electrolyte has been able to fulfill all the requirements of an “ideal” SSE. Solid polymer electrolytes offer flexibility and good interfacial contact with the electrodes, but suffer from low ionic conductivities (< 10-4 S/cm) and inferior thermal and electrochemical stability. In contrast, inorganic ceramic electrolytes (ICEs) exhibit higher ionic conductivities (10-3-10-2 S/cm), a broad electrochemical window, and high mechanical strength, but offer poor interfacial contact with the electrodes[7-11]. Among the ICEs, garnet-type Li7La3Zr2O12 (LLZO) has attracted considerable attention due to its relatively high ionic conductivity (up to 2 × 10-3 S/cm at room temperature), processability in air, non-flammability, broad electrochemical stability window, and high reduction stability when used in combination with lithium metal anodes[11-16]. For high-performance ASSBs, which have a thick composite cathode, the SSE must provide a large contact area with the cathode active material (CAM) and percolating pathways with high total ionic conductivity similar to that of a liquid electrolyte penetrating the porous cathode and wetting CAM particles in conventional cells[17]. The fabrication of LLZO-based composite cathodes always requires a heat treatment (sintering) step at temperatures of around 1,000 °C to achieve proper interface bonding between the CAM and SSE particles, as well as the densification of the electrode[18]. These high sintering temperatures often result in material compatibility issues due to the interdiffusion of cations across the LLZO/CAM interface and/or the formation of undesired secondary phases at the interface. Among the various CAMs used in conventional LIBs, LiCoO2 (LCO) is the only one that is thermodynamically stable in combination with LLZO at temperatures of up to 1,085 °C, while other CAMs - such as LiMn2O4 (LMO), LiFePO4 (LFP), LiNixCoyMnzO2 (NCM), and LiNixCoyAlzO2 (NCA) - react at significantly lower temperatures between 400 and 800 °C[13,17,19-23]. LCO has therefore been the CAM of choice for the few reported fully inorganic garnet-type ASSBs[4,24-28] and composite cathodes[29,30] fabricated by co-sintering. However, LCO has a relatively low specific capacity (140 mAh/g) due to an irreversible phase change below a Li content of x = 0.5, which limits the energy density of LCO-based ASSBs[31,32]. For higher energy densities, it is necessary to incorporate higher capacity CAMs such as Ni-rich NCM with capacities
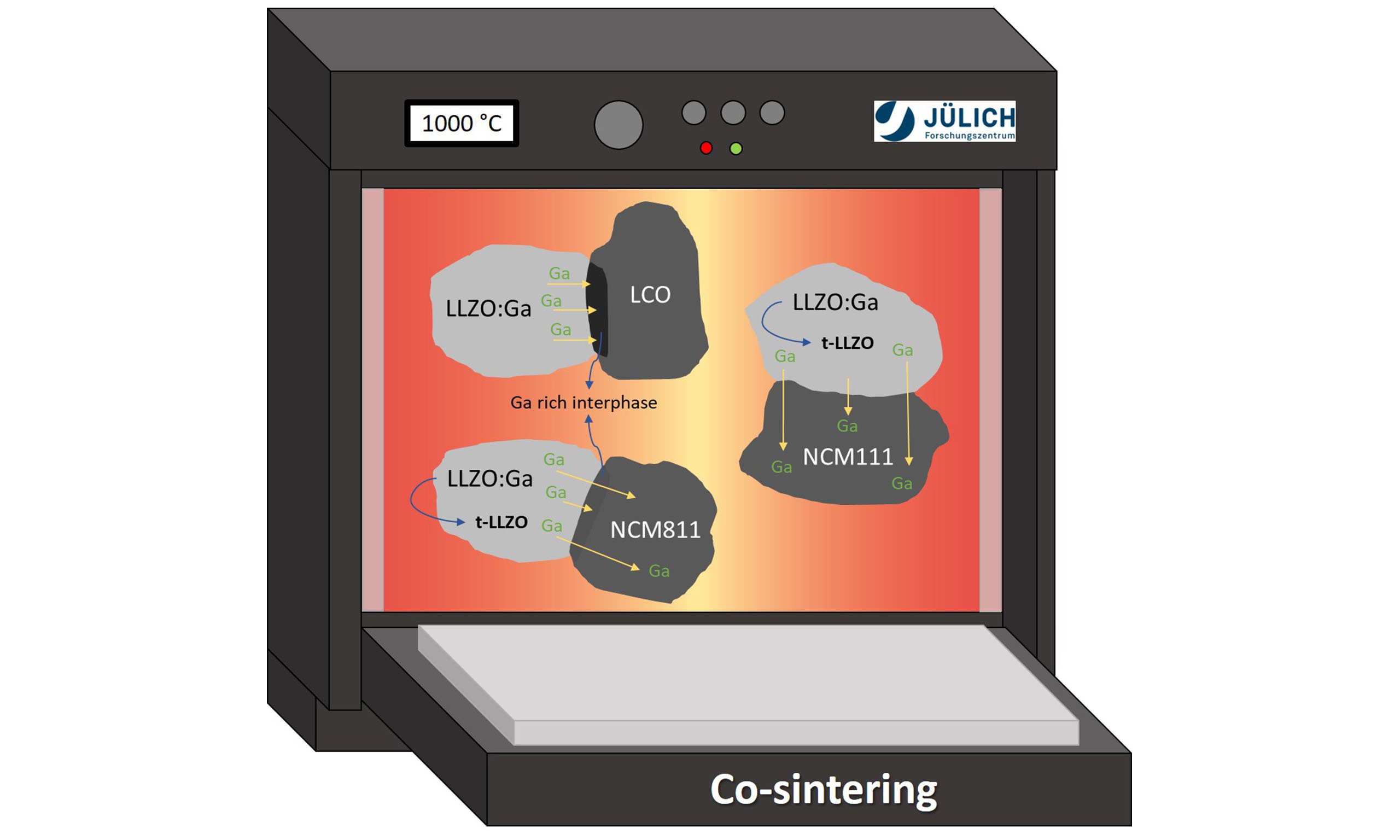
In this work, we built on our earlier investigations into the thermal compatibility of different CAMs and LLZO[13,21], using a similar experimental approach, but extended the study to a different LLZO composition with higher ionic conductivity. While we previously only investigated cubic LLZO:Ta as the SSE, we now examine the thermodynamic stability between cubic LLZO:Ga and three different layered CAMs-LCO, LiNi1/3Mn1/3Co1/3O2 (NCM111), and NCM811 - during co-sintering at elevated temperatures of up to
EXPERIMENTAL
The Ga-substituted LLZO (nominal composition: LLZO:Ga) was synthesized via a solid-state reaction (SSR), as previously described[38]. The starting materials LiOH∙H2O (AppliChem GmbH, Darmstadt, Germany, 99%), La2O3 (Merck Chemicals GmbH, Darmstadt, Germany, 99.9%, 10 h pre-dried at 900 °C), ZrO2 (Treibacher Industrie AG, Althofen, Austria, 99.7%), and Ga2O3 (Alfa Aesar, Darmstadt, Germany, 99.995%) were weighed in a stoichiometric ratio and homogenized using an automatic mortar grinder (RM 200, Retsch GmbH, Haan, Germany). Pellets were pressed from the homogenized powder (uniaxial,
For the thermal compatibility tests, the commercial CAMs-LCO, NCM111, or NCM811 - (MTI Corporation, Richmond, CA, USA, battery grade) were mixed with LLZO:Ga at a weight ratio of 1:1 and homogenized using a mortar and pestle. The CAM powders were used as received (D50:~10 µm), while sintered and ground LLZO:Ga powder (D50: ~6 µm) was used. The powder mixtures were pelletized by pressing with a force of 38 kN and a pressing die with a 13 mm diameter (resulting pellet height: ~1.7 mm). The evolution of secondary phases during heating was investigated by in situ high temperature XRD (HT-XRD) using an Empyrean diffractometer (Malvern Panalytical Ltd, Malvern, UK) with Cu-Kα radiation. In situ measurements were performed in air for isotherms at the following selected temperatures (°C): 25, 100, 200, 300, 350, 400, 450, 500, 550, 600, 650, 700, 750, 800, 900, and 1,000. The heating rate applied to reach the next temperature was 5 K min-1. Data were collected over a 2θ range of 10°-80°, with a step width of 0.026° and an accumulation time of 200 s using a 255-channel PIXcel detector, resulting in a holding time of roughly
Further phase analysis was performed by Raman spectroscopy using an inViaTM (Renishaw plc, Wotton-under-Edge, UK) Raman microscope with a 532 nm laser (~2.5 mW) and a 2,400 L mm-1 grating. The second set of sintered pellets was analyzed here. The spectra were collected as a map with a step size of
For microstructural investigations and secondary phase detection and visualization, scanning electron microscopy (SEM) was performed using a Zeiss Gemini SEM 450 (Carl Zeiss Microscopy Deutschland GmbH, Oberkochen, Germany) equipped with an Ultim® Max 170 energy-dispersive X-ray spectroscopy (EDS) detector (Oxford Instruments plc, Abingdon, UK). Prior to analysis, the co-sintered pellet samples were embedded in EpoFix epoxy (Struers GmbH, Willich, Germany) and mirror polished. To gain a more detailed understanding of the CAM/LLZO:Ga interface, EDS was performed in a scanning transmission electron microscopy (STEM) with a Bruker Nano XFlash Detector 5060 (Bruker Corporation, Billerica, MA, USA) and the data were evaluated with Esprit 2.3 software. The measurements were carried out in a double Cs-corrected JEOL JEM-2200FS STEM (JEOL Ltd., Akishima, Tokyo, Japan) with an acceleration voltage of 200 kV and a convergence angle of 22.6 mrad. As STEM analysis requires electron-transparent samples (thickness < 100 nm), lamella preparation and Xe-ion beam thinning were carried out in advance using the HeliosTM 5 Hydra CX DualBeam (Thermo Fisher Scientific Inc., Waltham, MA, USA). The samples were transferred in an inert gas atmosphere from an argon-filled glovebox (O2 < 0.1 ppm, H2O < 1 ppm) to the Helios Hydra and back using the Hydra’s inert transfer system (CleanConnect). The samples were then loaded into a vacuum transfer TEM holder (Melbuild Corporation, Fukuoka, Japan) to ensure transfer to the microscope without air contamination.
RESULTS AND DISCUSSION
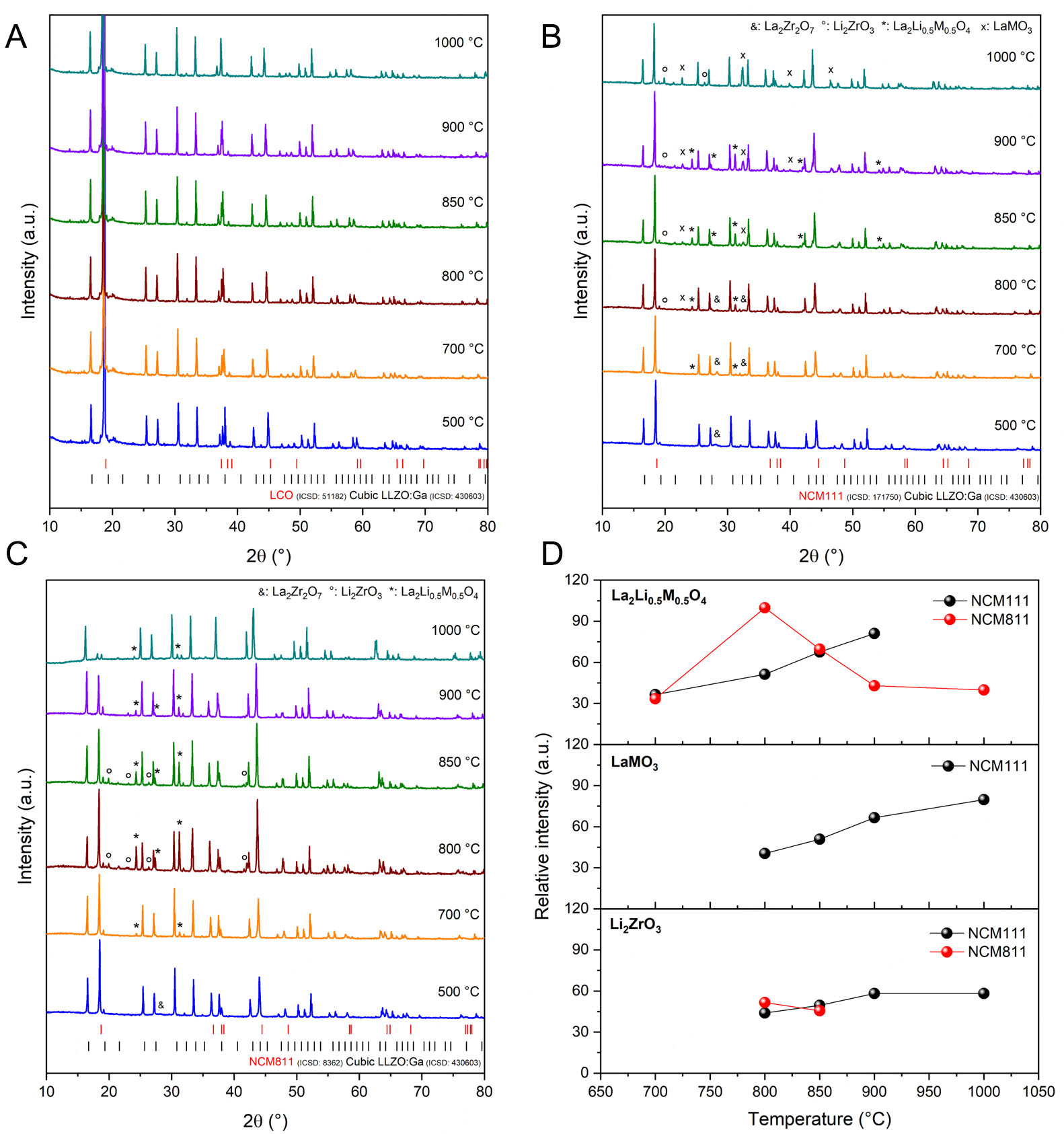
We first investigated the thermodynamic stability of LLZO:Ga in combination with different CAMs (LCO, NCM111, and NCM811) during co-sintering based on XRD. This approach allowed us to determine the onset temperature of secondary phase formation. To scan a wide temperature range on a single sample, in situ high-temperature XRD (HT-XRD) measurements were performed between room temperature and
Figure 1. In situ HT-XRD patterns for 1:1 mixtures of cubic LLZO:Ga with (A) LCO, (B) NCM111, and (C) NCM811. Only selected temperatures are shown. Diffractions attributed to the pyrochlore or secondary phases are labeled accordingly. Due to thermal expansion at elevated temperatures, the Bragg peak positions are shifted slightly in comparison to the reference pattern for LCO, NCM111, NCM811, and cubic LLZO:Ga. (D) Relative intensities of the secondary phase reflections of Li2ZrO3 (110) at ~19.9° 2θ, La2(M0.5Li0.5)O4 reflection (131) at ~31.3° 2θ, and
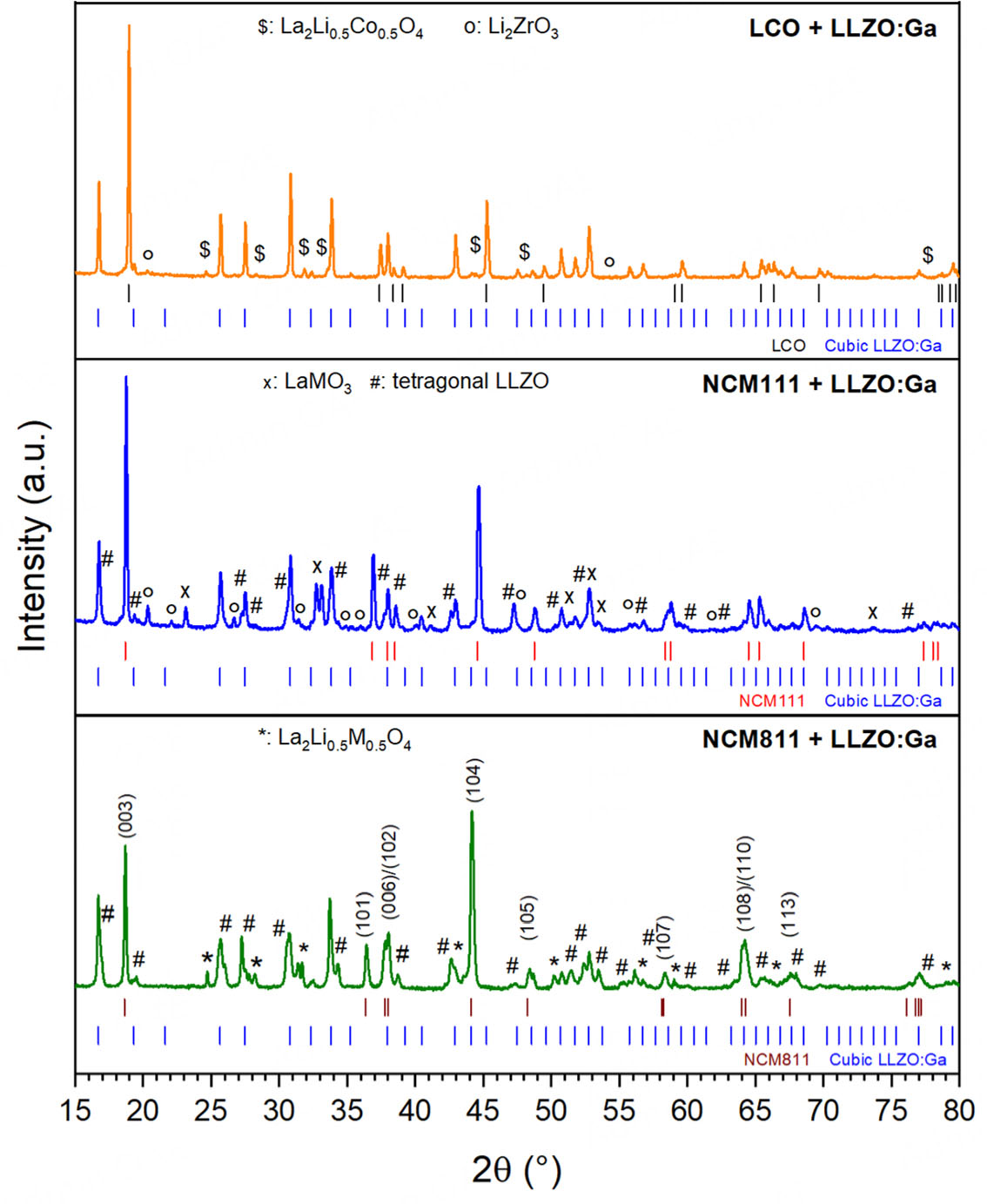
In situ HT-XRD is a suitable method for qualitatively studying secondary phase formation during the co-sintering of CAM and LLZO:Ga over a wide temperature range. However, this method only measures the surface of the pellet. To obtain more reliable and precise results that include the bulk of the composite, the same CAM + LLZO:Ga mixtures used for the HT-XRD investigations were sintered at 1,000 °C for 1 h in a conventional muffle furnace. They were then ground and analyzed as a powder by XRD. Figure 2 shows the powder diffractograms of the co-sintered CAM + LLZO:Ga mixtures. Qualitative and quantitative phase analysis was performed based on Rietveld refinement. The refined patterns are shown in the
Figure 2. Powder XRD patterns of the different CAM + LLZO:Ga mixtures obtained after co-sintering at 1,000 °C using Cu kα1/2 radiation. All intensities are normalized to the LLZO:Ga reflection (024) at ~30.8 °2θ. (M = Ni, Co, Mn).
The above-discussed HT-XRD experiments, which mainly analyzed the surface of the composite pellets, led to the conclusion that the LCO + LLZO:Ga mixture is stable during co-sintering. However, a more detailed analysis of the mixture by powder XRD revealed 2 wt.% La2(Co0.5Li0.5)O4 and 3 wt.% Li2ZrO3 as secondary phases after co-sintering at 1,000 °C. The maximum co-sintering temperature without side phase formation is therefore below 1,000 °C for LCO. LCO and cubic LLZO:Ga remain the main phases; however, a second cubic LLZO:Ga phase with a slightly larger lattice parameter was found compared to the starting material [Supplementary Tables 2 and 3]. This second cubic phase can be assigned to protonated LLZO:Ga, which forms during handling in an ambient atmosphere, as previously reported[38].
For the NCM111-containing composite, undesired secondary phases formed during co-sintering include
Changes in lattice parameters during co-sintering were analyzed based on the a and c parameters, which were calculated from the powder XRD pattern [see Supplementary Tables 2 and 3]. For LCO + LLZO:Ga, the lattice parameters did not change significantly within the error range. In the case of the NCM111-containing mixture, the NCM111 lattice parameters remained similar to those of pristine material, while slightly smaller lattice parameters were observed for cubic LLZO. In contrast to NCM111, the lattice parameters a and c of NCM811 increased significantly, while the lattice parameter a of cubic LLZO remained almost unchanged. The increased lattice parameters for NCM811 after heat treatment in the presence of LLZO:Ga are most likely explained by the doping of the NCM host structure with elements from the SSE, such as Zr, La, or Ga.
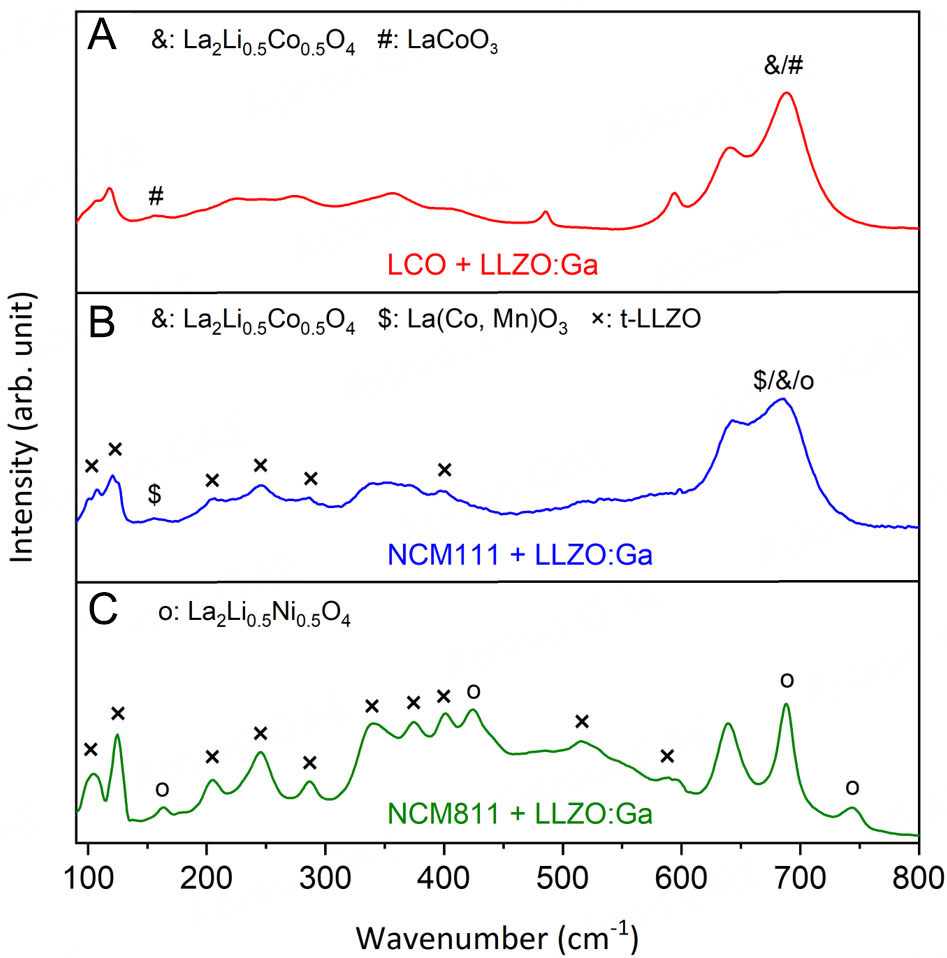
In addition to XRD, Raman spectroscopy was used to analyze the composites, as it typically has a lower detection limit for side phases compared to XRD. The Raman spectra of the co-sintered CAM + LLZO:Ga mixtures are shown in Figure 3. By comparing the obtained Raman spectrum for the LCO + LLZO:Ga composite [Figure 3A] with the reference Raman spectra of cubic LLZO:Ga[47] and LCO[48], the observed signals can be assigned to the following vibration modes: La cation vibration at 106 cm-1 (T2g) and 120 cm-1 (Eg); broad O-bending modes at 226 cm-1 (T2g) and 275 cm-1 (A1g); Li vibrational modes at 357 cm-1 (T2g) and 406 cm-1 (Eg or T2g); Zr-O bond stretching at 641 cm-1 (A1g) for LLZO:Ga and O-Co-O bending at 486 cm-1 (Eg); and Co-O stretching vibrations at 595 cm-1 (A1g) for LCO. These results confirm the presence of both LCO and cubic LLZO:Ga after co-sintering. However, the two additional bands found at 157 and
Figure 3. Averaged Raman spectra of (A) LCO, (B) NCM111, and (C) NCM811 + LLZO:Ga pellets after co-sintering at 1,000 °C. The signals belonging to secondary phases are labeled accordingly.
In contrast to LCO + LLZO:Ga, where only the cubic LLZO:Ga phase is present after co-sintering at
In summary, the XRD and Raman spectroscopy results confirm that cubic LLZO and LCO remained the main phases after the co-sintering of the LCO + LLZO:Ga mixture at 1,000 °C. In total, only around 5 wt.% of side phases (La2(Co0.5Li0.5)O4, LaCoO3, and Li2ZrO3) were identified based on Rietveld refinement
Summary of qualitative and quantitative phase analysis of CAM + LLZO mixtures co-sintered for 1 h at 1,000 °C, based on X-ray diffraction with Rietveld refinement and Raman spectroscopy. Results for LLZO:Ta are from ref.[21]
| LLZO:Ta[21] | LLZO:Ga | |||
| Secondary phase | wt.% | Secondary phase | wt.% | |
| LCO | * | * | Li2ZrO3 La2Li0.5Co0.5O4 LaCoO3 | 3 2 - |
| NCM111 | La2Li0.5M0.5O4 LaMO3 | 1 0 | La2Li0.5M0.5O4 Li2ZrO3 LaMO3 t-LLZO | 0 5 9 8 |
| NCM811 | La2Li0.5M0.5O4 Li0.5M0.25Zr0.25O | 3 10 | La2Li0.5M0.5O4 t-LLZO | 3 23 |
Combining LLZO:Ga with CAMs such as NCM, where Co is partially substituted by Ni and Mn, leads to reduced thermodynamic stability, which is in agreement with previous compatibility studies on NCM + LLZO:Ta[13,19,21]. For NCM111 + LLZO:Ga, a total of 22 wt.% secondary phases (LaMO3 (M = Ni, Co, Mn), Li2ZrO3, and t-LLZO) were identified, while for the Ni-rich NCM811 mixture, an even higher amount of
The t-LLZO phase was detected as a detrimental side phase that forms during the co-sintering of NCM + LLZO:Ga, indicating a more intense degradation of the SSE in the case of Ga substitution. For NCM811 in particular, t-LLZO, which exhibits much lower ionic conductivity compared to the cubic phase, forms with a significantly higher weight percentage than the other secondary phases. In general, the tetragonal phase of the garnet increases the ohmic resistance and thus the overpotential during battery cell operation, particularly when located along the CAM/LLZO interface. The possible impact of the other secondary phases on electrochemical performance remains unclear and requires further investigation in follow-up studies.
A phase transition from the cubic to the tetragonal phase during co-sintering was not observed in our previous study on LLZO:Ta-based mixtures, highlighting one of the main issues of using LLZO:Ga instead of LLZO:Ta. Surprisingly, t-LLZO was not observed in the LCO + LLZO:Ga sample, although literature reports that t-LLZO can form at the interface between LLZO and LCO during low-temperature annealing at
Figure 4 shows the cross-sectional SEM images taken with backscattered electrons (BSEs), which visualize the material (Z) contrast, along with the corresponding layered images derived from EDS mappings. The single EDS mappings are available in the supporting information for all of the investigated samples [Supplementary Figures 6-8]. Four regions with different material contrasts can be distinguished for the LCO + LLZO:Ga composite [Figure 4A and B]. In addition to the LLZO (orange) and LCO (blue) main phases, the overlay of the EDS mappings [Figure 4B] and further EDS point analyses [Supplementary Figure 9] confirm the formation of Zr-rich (purple) and La-rich (yellow) secondary phases, which appear as light grey and brighter regions, respectively, in the BSE image. Due to the relatively large excitation volume during EDS analysis and the relatively small regions of side phases, it is not possible to quantify elements or make a valid statement about the secondary phase composition based on EDS. Nevertheless, the SEM/EDS findings support the XRD and Raman spectroscopy results.
Figure 4. Cross-sectional, backscattered electron (BSE) SEM images and the corresponding layered images of the single EDS mappings of La (yellow), Zr (purple), Co (blue), and O (grey) for (A and B) LCO + LLZO:Ga, (C and D) NCM111 + LLZO:Ga, and (E and F) NCM811 + LLZO:Ga after co-sintering at
For the co-sintered NCM111 + LLZO:Ga mixture, SEM combined with EDS shows very similar side phase formation [Figure 4C and D, Supplementary Figure 10], as was reported for the LCO-containing sample. However, the number of secondary-phase particles is strongly increased compared to the LCO-containing sample. This is consistent with the significantly higher weight fractions of secondary phases found for NCM111 + LLZO:Ga by XRD and Rietveld refinement. Only three regions with different material contrasts could be distinguished for the NCM811 + LLZO:Ga composite [Figure 4E and F], indicating the formation of only one secondary phase with a brighter Z contrast. The EDS spectrum taken from this region, which appears much brighter in the BSE-SEM images and yellow in the EDS mapping, indicates that this particle predominantly contains the elements O, Ni, and La (Li cannot be detected by the EDS detector) [Supplementary Figure 11]. Considering the Raman spectroscopy and XRD results, the bright region observed for the NCM811-containing sample is most likely La2Li0.5Ni0.5O4, which mainly forms as well-defined, separated particles.
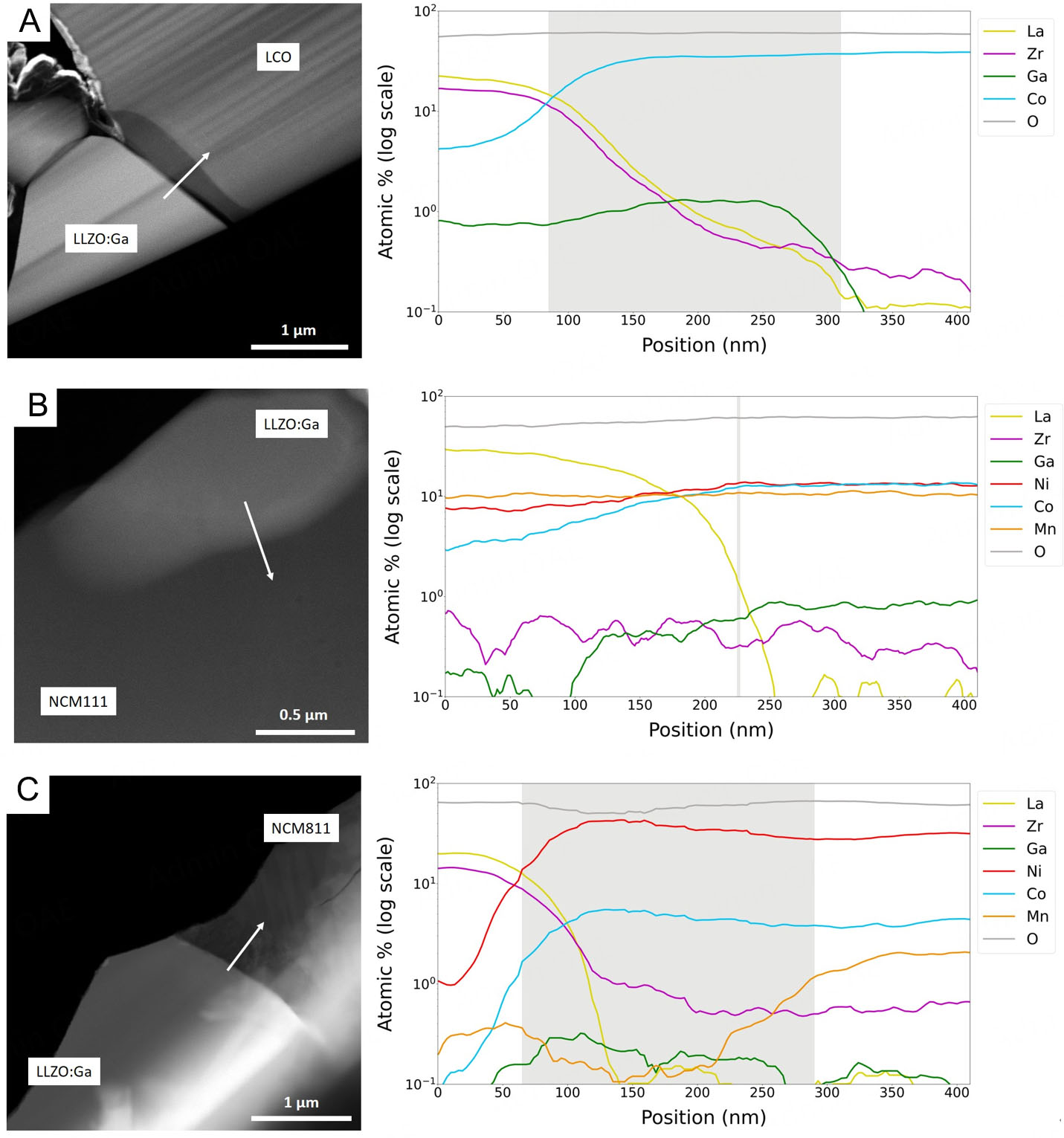
None of the characterization methods used thus far have provided reliable insights into the behavior of the element Ga during co-sintering. The Ga mappings [Supplementary Figures 6-8] suggest that Ga is present in both the LLZO and CAM phases after co-sintering at 1,000 °C. However, the overall Ga content is very low and close to the detection limit of the EDS. A more detailed study of Ga distribution after co-sintering, as well as an analysis of the CAM/LLZO:Ga interface, was therefore performed by high-angle annular dark-field (HAADF) imaging and EDS within a STEM. The HAADF-STEM image and EDS analysis of the LCO/LLZO:Ga interface in Figure 5A clearly reveal that a Ga-enriched layer with a different Z contrast (dark grey) formed between the LCO and LLZO:Ga grains (see EDS mapping in Supplementary Figure 12). In this region, the EDS line scan shows a decrease in the La and Zr signals, as well as an initially increasing and subsequently constant Co signal. Diffusion of the elements Zr and La into the LCO structure is only visible at very low concentrations. Furthermore, Ga could not be detected in the LCO bulk.
Figure 5. HAADF-STEM image (left) and EDS line scan (right) across the interface marked by the arrow for (A) LCO + LLZO:Ga, (B) NCM111 + LLZO:Ga, and (C) NCM811 + LLZO:Ga co-sintered at 1,000 °C. The light grey area in the EDS line scan represents the dark layer at the LCO/LLZO:Ga interface visible in the HAADF image. Further EDS mappings of these samples are available in the
A different situation was observed at the NCM111/LLZO:Ga interface after co-sintering at 1,000 °C
In the case of NCM811 + LLZO:Ga, the HAADF-STEM image [Figure 5C] shows a layer with a different Z contrast at the interface, similar to that observed in the LCO+LLZO:Ga sample, although less pronounced. EDS analysis reveals Mn depletion and Ga enrichment in NCM811 at the interface with LLZO:Ga. In contrast to NCM111, only minor diffusion of Ni, Mn, and Co into the LLZO grain is observed in the Ni-rich, NCM811-containing sample [Supplementary Figure 14]. In the case of NCM811, the elements Zr and Ga are also present in the cathode material particles, albeit in very low amounts. La diffusion was not observed for any of the CAM/LLZO:Ga composites. It should be noted that the NCM811 + LLZO:Ga sample also contains Ga-rich phases that do not exhibit significant high concentrations of other transition metals. In contrast to LCO + LLZO:Ga, however, these phases are not located at the CAM/LLZO interface.
The TEM investigations reveal that the dopant Ga, which stabilizes the cubic LLZO structure, is highly mobile during co-sintering with CAMs. The location of Ga after co-sintering strongly depends on the CAM composition in the composite. In the case of LCO, only a Ga-rich, Co-containing interphase forms between the LLZO:Ga and LCO grains; severe Ga diffusion into the LCO bulk does not occur. For the NCM111- and NCM811-containing mixtures, intensive Ga diffusion occurs into the adjacent NCM grain. The few TEM studies on such composite cathodes in the literature used other LLZO compositions, such as LLZO:Ta or Li6.25Al0.25La3Zr2O12 (LLZO:Al). These studies did not report any Ta or Al diffusion into the CAM grains, nor segregation of LLZO dopants at the CAM/LLZO interface during co-sintering/annealing up to 1,050 °C. Additionally, no phase transition from cubic to tetragonal garnet was reported for their material composition at the investigated co-sintering temperatures. The highly ion-conductive cubic LLZO phase was thus largely maintained after firing[20,28]. Only Park et al.[56] observed the formation of t-LLZO when synthesizing and annealing LCO on top of an LLZO layer at 700 °C. They confirmed the partial phase transition at the LLZO/LCO interface by TEM and attributed it to the destabilization of the cubic garnet framework caused by Al leaching from the LLZO and its diffusion into the LCO phase[56]. Based on the interpretation of the work of Park et al.[56] by Ren et al.[55], the tetragonal phase was caused by LCO leaching Al out of the cubic garnet phase. In their own work,
A thermodynamically favored Ga incorporation into the CAM host structure would explain an additional driving force for the Ga loss from LLZO and the formation of t-LLZO. Ga-doped LCO[57] and Ga-doped, Ni-rich NCM[58] are well-known in the literature due to their improved electrochemical performance through Ga doping. Ga-doped CAMs are characterized by their high Ga-oxygen bond energies within the layered structure, as evidenced by experimental findings and ab initio calculations[57,58]. These higher bond energies are a possible driving force for the incorporation of Ga from the cubic LLZO into the layered structure of the CAMs. Our TEM findings indicate that Ga loss from the LLZO lattice and its subsequent incorporation into the NCM lattice is more preferential than into the LCO lattice. This is underlined by the XRD results, which show a high quantity of t-LLZO and enlarged lattice parameters, especially for NCM811 + LLZO:Ga. This is consistent with the findings of Jamil et al.[58] for Ga-doped, Ni-rich NCM. For the NCM811 sample investigated here, Ga doping is the most likely reason for the lattice parameter change after co-sintering, since no significant diffusion of other transition metals was observed by TEM/EDS. The incorporation of Ga3+ into the NCM lattice induces positive charges that can be compensated by a partial reduction of Ni3+ to Ni2+, which can occupy the Li sites and result in increased cation disorder, as determined by Rietveld refinement for NCM811 + LLZO:Ga after co-sintering. For LCO + LLZO:Ga, Ga loss from LLZO, its incorporation into the layered structure, and the formation of t-LLZO were found to be inhibited, since only a Ga-enriched interphase was observed. It is assumed that a small amount of t-LLZO was formed, below the detection limit of the characterization methods used here. It was not possible to distinguish LLZO:Ga from t-LLZO in STEM diffraction mode due to the presence of numerous diffraction spots that could not be unambiguously assigned, likely caused by the coexistence of multiple phases. Based on the present study, the exact reason for the distinct behavior of the LCO/LLZO:Ga composite remains unclear. However, it seems that the Ga-enriched, Co-depleted phase formed at the LLZO/LCO interface blocks further Ga diffusion into the LCO bulk. This allows the cubic LLZO phase to mostly remain intact during co-sintering. Nevertheless, such an interphase can cause very high interfacial resistance, which limits the electrochemical performance of a battery cell.
Based on our study, LCO - which is known to be very stable in contact with LLZO:Ta - is also the most promising CAM that can potentially be co-sintered with LLZO:Ga to fabricate dense composite cathodes for integration into ASSBs. We therefore fabricated fully inorganic cells based on the recipe[4] developed in our group using LCO but LLZO:Ga as the catholyte and solid separator, respectively. The electrochemical performance of the resulting cells was strongly limited due to very high total cell resistance (> 2,000 Ω), resulting in huge ohmic losses during cycling and a discharge capacity of only 42 mAh/g (0.4 mAh/cm2) in the first cycle [Supplementary Figure 15]. The observed Ga-rich interphase is one explanation for the high cell resistance. In addition, cobalt diffusion into the LLZO separator[59] was much more severe and challenging for the Ga-substituted LLZO system. Analysis of the failure mechanism of the LLZO:Ga-based full cells, as well as a detailed study of the transition metal diffusion into the LLZO:Ga separator during half-cell manufacturing by co-sintering, will be addressed in a comprehensive follow-up study.
CONCLUSION
The thermodynamic stability between the garnet-type SSE LLZO:Ga and three different CAMs - LCO, NCM111, and NCM811 - was investigated in detail and compared to LLZO:Ta. Regardless of the CAM used, the cubic LLZO phase and the initial CAM phase were largely preserved after co-sintering at temperatures up to 1,000 °C. However, various secondary phases were observed close to the CAM/LLZO interface, with their nature, amount, and reaction onset temperature strongly dependent on the chemical composition of the CAM. Four secondary phases - Li2ZrO3, La2Li0.5M0.5O4 (M = Ni, Co, Mn), a perovskite phase LaMO3 (M = Ni, Co, Mn), and t-LLZO - were identified based on XRD and Raman spectroscopy. For LCO + LLZO:Ga, only around
In conclusion, our previous and present studies together demonstrate that both the SSE and CAM composition have a significant impact on chemical stability during the high-temperature fabrication steps required for ASSB manufacturing. Interestingly, the choice of dopant for the LLZO SSE was found to have a more significant impact on material compatibility. LLZO:Ga exhibits the highest ionic conductivity of all the oxide solid-state Li-ion conductors. However, this advantage comes at the price of decreased interface stability due to dopant leaching. The observed phase transformation from cubic to tetragonal must therefore be suppressed during component manufacturing, as the tetragonal phase has significantly lower total ionic conductivity than LLZO:Ga. Therefore, in order to successfully integrate highly conductive LLZO:Ga into co-sintered composite cathodes, strategies must be developed to stabilize the Ga dopant in the LLZO structure during co-sintering at temperatures around
DECLARATIONS
Acknowledgments
The authors would like to thank Christian Dellen (evaluation of Raman spectra), Christian Schwab (calculation of Li-concentrations), Volker Bader (heat treatments), and Philipp Hecker (heat treatments and lab assistance) for their contributions.
Authors’ contributions
Performed the co-sintering experiments, analyzed the data, and wrote the original draft of the manuscript with input from all authors: Roitzheim, C.
Conducted HT-XRD measurements and performed the Rietveld refinements: Sohn, Y. J.
Carried out the TEM investigations: Hueppe, F.; Demuth, T.; Volz, K.
Performed the Raman analysis: Scheld, W. S.
Acquired the SEM images and performed the EDS analysis: Sebold, D.
The manuscript was revised and edited: Roitzheim, C.; Sohn, Y. J.; Hueppe, F.; Demuth, T.; Scheld, W. S.; Collette, Y.; Sebold, D.; Finsterbusch, M.; Fattakhova-Rohlfing, D.; Guillon, O.
Conceived the study, provided the resources, and supervised the work: Finsterbusch, M.; Guillon, O.; Fattakhova-Rohlfing, D.
All authors approved the final version of the manuscript.
Availability of data and materials
The data supporting the findings of this study (including detailed representations of SEM/EDS and TEM/EDS analyses, Raman spectroscopy data, and results of the Rietveld refinement) can be found in the Supplementary Materials published with this article.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
The authors are grateful for financial support provided by the German Federal Ministry of Research, Technology, and Space (Bundesministerium für Forschung, Technologie und Raumfahrt, BMFTR) under grant numbers 13XP0434A (FB2-Oxid), 03XP0433D (FB2-Char), and 13XP0510A (CatSE2), as well as by the German Research Foundation (Deutsche Forschungsgemeinschaft, DFG) under grant number 429409150 (project E-TIP).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Noh, H.; Youn, S.; Yoon, C. S.; Sun, Y. Comparison of the structural and electrochemical properties of layered Li[NixCoyMnz]O2
2. Yao, X.; Huang, B.; Yin, J.; et al. All-solid-state lithium batteries with inorganic solid electrolytes: Review of fundamental science. Chinese. Phys. B. 2016, 25, 018802.
3. Sakuda, A. Favorable composite electrodes for all-solid-state batteries. J. Ceram. Soc. Jpn. 2018, 126, 675-83.
4. Finsterbusch, M.; Danner, T.; Tsai, C. L.; Uhlenbruck, S.; Latz, A.; Guillon, O. High capacity garnet-based all-solid-state lithium batteries: fabrication and 3D-microstructure resolved modeling. ACS. Appl. Mater. Interfaces. 2018, 10, 22329-39.
6. Janek, J.; Zeier, W. G. Challenges in speeding up solid-state battery development. Nat. Energy. 2023, 8, 230-40.
7. Schmaltz, T.; Hartmann, F.; Wicke, T.; Weymann, L.; Neef, C.; Janek, J. A roadmap for solid-state batteries. Adv. Energy. Mater. 2023, 13, 2301886.
8. Thangadurai, V.; Narayanan, S.; Pinzaru, D. Garnet-type solid-state fast Li ion conductors for Li batteries: critical review. Chem. Soc. Rev. 2014, 43, 4714-27.
9. Sand, S. C.; Rupp, J. L. M.; Yildiz, B. A critical review on Li-ion transport, chemistry and structure of ceramic-polymer composite electrolytes for solid state batteries. Chem. Soc. Rev. 2025, 54, 178-200.
10. Xue, Z.; He, D.; Xie, X. Poly(ethylene oxide)-based electrolytes for lithium-ion batteries. J. Mater. Chem. A. 2015, 3, 19218-53.
11. Li, S.; Zhang, S. Q.; Shen, L.; et al. Progress and perspective of ceramic/polymer composite solid electrolytes for lithium batteries. Adv. Sci. 2020, 7, 1903088.
12. Chen, L.; Li, Y.; Li, S.; Fan, L.; Nan, C.; Goodenough, J. B. PEO/garnet composite electrolytes for solid-state lithium batteries: From “ceramic-in-polymer” to “polymer-in-ceramic”. Nano. Energy. 2018, 46, 176-84.
13. Roitzheim, C.; Sohn, Y. J.; Kuo, L.; et al. All-solid-state Li batteries with NCM-garnet-based composite cathodes: the impact of NCM composition on material compatibility. ACS. Appl. Energy. Mater. 2022, 5, 6913-26.
14. Murugan, R.; Thangadurai, V.; Weppner, W. Fast lithium ion conduction in garnet-type Li7La3Zr2O12. Angew. Chem. Int. Ed. 2007, 46, 7778-81.
15. Buschmann, H.; Dölle, J.; Berendts, S.; et al. Structure and dynamics of the fast lithium ion conductor “Li7La3Zr2O12”. Phys. Chem. Chem. Phys. 2011, 13, 19378-92.
16. Buannic, L.; Orayech, B.; López, Del. Amo. J.; et al. Dual substitution strategy to enhance Li+ ionic conductivity in Li7La3Zr2O12 solid electrolyte. Chem. Mater. 2017, 29, 1769-78.
17. Ren, Y.; Danner, T.; Moy, A.; et al. Oxide‐based solid‐state batteries: a perspective on composite cathode architecture. Adv. Energy. Mater. 2022, 13, 2201939.
18. Xiao, Y.; Wang, Y.; Bo, S.; Kim, J. C.; Miara, L. J.; Ceder, G. Understanding interface stability in solid-state batteries. Nat. Rev. Mater. 2019, 5, 105-26.
19. Ren, Y.; Liu, T.; Shen, Y.; Lin, Y.; Nan, C. Chemical compatibility between garnet-like solid state electrolyte Li6.75La3Zr1.75Ta0.25O12 and major commercial lithium battery cathode materials. J. Materiomics. 2016, 2, 256-64.
20. Demuth, T.; Fuchs, T.; Walther, F.; et al. Influence of the sintering temperature on LLZO-NCM cathode composites for solid-state batteries studied by transmission electron microscopy. Matter 2023, 6, 2324-39.
21. Bauer, A.; Roitzheim, C.; Lobe, S.; et al. Impact of Ni-Mn-Co-Al-based cathode material composition on the sintering with garnet solid electrolytes for all-solid-state batteries. Chem. Mater. 2023, 35, 8958-68.
22. Wakasugi, J.; Munakata, H.; Kanamura, K. Thermal stability of various cathode materials against Li6.25Al0.25La3Zr2O12 electrolyte. Electrochemistry 2017, 85, 77-81.
23. Miara, L. J.; Richards, W. D.; Wang, Y. E.; Ceder, G. First-principles studies on cation dopants and electrolyte|cathode interphases for lithium garnets. Chem. Mater. 2015, 27, 4040-7.
24. Han, S.; Kil, D.; Lee, S.; et al. A full oxide-based solid-state lithium battery and its unexpected cathode degradation mechanism. ACS. Energy. Lett. 2023, 8, 4794-805.
25. Han, F.; Yue, J.; Chen, C.; et al. Interphase engineering enabled all-ceramic lithium battery. Joule 2018, 2, 497-508.
26. Ohta, S.; Komagata, S.; Seki, J.; Saeki, T.; Morishita, S.; Asaoka, T. All-solid-state lithium ion battery using garnet-type oxide and Li3BO3 solid electrolytes fabricated by screen-printing. J. Power. Sources. 2013, 238, 53-6.
27. Liu, T.; Ren, Y.; Shen, Y.; Zhao, S.; Lin, Y.; Nan, C. Achieving high capacity in bulk-type solid-state lithium ion battery based on Li6.75La3Zr1.75Ta0.25O12 electrolyte: interfacial resistance. J. Power. Sources. 2016, 324, 349-57.
28. Ihrig, M.; Finsterbusch, M.; Laptev, A. M.; et al. Study of LiCoO2/Li7La3Zr2O12:Ta interface degradation in all-solid-state lithium batteries. ACS. Appl. Mater. Interfaces. 2022, 14, 11288-99.
29. Zhao, H.; Mo, H.; Mao, P.; Ran, R.; Zhou, W.; Liao, K. Tape-casting fabrication techniques for garnet-based membranes in solid-state lithium-metal batteries: a comprehensive review. ACS. Appl. Mater. Interfaces. 2024, 16, 68772-93.
30. Weinmann, S.; Gobena, H.; Quincke, L.; et al. Stabilizing interfaces of all‐ceramic composite cathodes for Li‐garnet batteries. Adv. Energy. Mater. 2025, 15, 2502280.
31. Li, J.; Lin, C.; Weng, M.; et al. Structural origin of the high-voltage instability of lithium cobalt oxide. Nat. Nanotechnol. 2021, 16, 599-605.
32. Oh, P.; Yun, J.; Choi, J. H.; et al. New ion substitution method to enhance electrochemical reversibility of Co‐rich layered materials for Li-ion batteries. Adv. Energy. Mater. 2022, 13, 2202237.
33. Yin, X.; Li, D.; Hao, L.; et al. A high-energy all-solid-state lithium metal battery with "single-crystal" lithium-rich layered oxides. Chem. Commun. 2023, 59, 639-42.
34. Wang, D.; Sun, Q.; Luo, J.; et al. Mitigating the interfacial degradation in cathodes for high-performance oxide-based solid-state lithium batteries. ACS. Appl. Mater. Interfaces. 2019, 11, 4954-61.
35. Hayashi, N.; Watanabe, K. Reaction suppression between a high-Ni cathode material (NMC622) and Li7La3Zr2O12 on co-sintering for manufacturing bulk-type all-solid-state batteries: a new method and its mechanism. Adv. Sci. 2025, 12, e12219.
36. Ma, Z.; Labriola, G.; Salazar, K. A.; Mi, C. C.; Kong, L. Thermal stability and electrochemical behavior of commercial polycrystalline and single-crystalline cathodes integrated with cubic Li6.4La3Zr1.4Ta0.6O12 for all-solid-state lithium batteries. J. Mater. Chem. A. 2025, 13, 26647-59.
37. Kim, Y.; Waluyo, I.; Hunt, A.; Yildiz, B. Avoiding CO2 Improves thermal stability at the interface of Li7La3Zr2O12 electrolyte with layered oxide cathodes. Adv. Energy. Mater. 2022, 12, 2102741.
38. Schwab, C.; Häuschen, G.; Mann, M.; et al. Towards economic processing of high performance garnets - case study on zero Li excess Ga-substituted LLZO. J. Mater. Chem. A. 2023, 11, 5670-80.
39. Qin, S.; Zhu, X.; Jiang, Y.; Ling, M.; Hu, Z.; Zhu, J. Growth of self-textured Ga3+-substituted Li7La3Zr2O12 ceramics by solid state reaction and their significant enhancement in ionic conductivity. Appl. Phys. Lett. 2018, 112, 113901.
40. Degen, T.; Sadki, M.; Bron, E.; König, U.; Nénert, G. The HighScore suite. Powder. Diffr. 2014, 29 Suppl, S13-8.
41. Bruker AXS. Topas V4: General profile and structure analysis software for powder diffraction data. Karlsruhe, Germany; 2008.
42. Larraz, G.; Orera, A.; Sanjuán, M. L. Cubic phases of garnet-type Li7La3Zr2O12: the role of hydration. J. Mater. Chem. A. 2013, 1, 11419.
43. Matsui, M.; Sakamoto, K.; Takahashi, K.; et al. Phase transformation of the garnet structured lithium ion conductor: Li7La3Zr2O12. Solid. State. Ion. 2014, 262, 155-9.
44. Choi, Y. Effects of cation mixing on the electrochemical lithium intercalation reaction into porous Li1-δNi1-yCoyO2 electrodes. Solid. State. Ion. 1996, 89, 43-52.
45. Hua, W.; Schwarz, B.; Knapp, M.; et al. (De)Lithiation mechanism of hierarchically layered LiNi1/3Co1/3Mn1/3O2 cathodes during high-voltage cycling. J. Electrochem. Soc. 2018, 166, A5025-32.
46. Sun, G.; Yin, X.; Yang, W.; et al. The effect of cation mixing controlled by thermal treatment duration on the electrochemical stability of lithium transition-metal oxides. Phys. Chem. Chem. Phys. 2017, 19, 29886-94.
47. Scheld, W. S.; Collette, Y.; Schwab, C.; et al. Ga-ion migration during co-sintering of heterogeneous Ta- and Ga-substituted LLZO solid-state electrolytes. J. Eur. Ceram. Soc. 2025, 45, 116936.
48. Gross, T.; Hess, C. Raman diagnostics of LiCoO2 electrodes for lithium-ion batteries. J. Power. Sources. 2014, 256, 220-5.
49. Yuan, K.; Jin, X.; Xu, C.; et al. Fabrication of dense and porous Li2ZrO3 nanofibers with electrospinning method. Appl. Phys. A. 2018, 124, 403.
50. Tietz, F.; Wegener, T.; Gerhards, M.; Giarola, M.; Mariotto, G. Synthesis and Raman micro-spectroscopy investigation of Li7La3Zr2O12. Solid. State. Ion. 2013, 230, 77-82.
51. Flores, E.; Novák, P.; Aschauer, U.; Berg, E. J. Cation ordering and redox chemistry of layered Ni-rich LixNi1-2yCoyMnyO2: an operando Raman spectroscopy study. Chem. Mater. 2019, 32, 186-94.
52. Gnezdilov, V.; Fomin, V.; Yeremenko, A. V.; et al. Low-temperature mixed spin state of Co3+ in LaCoO3 evidenced from Jahn-Teller lattice distortions. Low. Temp. Phys. 2006, 32, 162-8.
53. Abrashev, M. V.; Litvinchuk, A. P.; Iliev, M. N.; et al. Comparative study of optical phonons in the rhombohedrally distorted perovskitesLaAlO3 and LaMnO3. Phys. Rev. B. 1999, 59, 4146-53.
54. Chaban, N.; Weber, M.; Pignard, S.; Kreisel, J. Phonon Raman scattering of perovskite LaNiO3 thin films. Appl. Phys. Lett. 2010, 97, 031915.
55. Ren, Y.; Wachsman, E. D. All solid-state Li/LLZO/LCO battery enabled by alumina interfacial coating. J. Electrochem. Soc. 2022, 169, 040529.
56. Park, K.; Yu, B.; Jung, J.; et al. Electrochemical nature of the cathode interface for a solid-state lithium-ion battery: interface between LiCoO2 and garnet-Li7La3Zr2O12. Chem. Mater. 2016, 28, 8051-9.
57. Sun, W.; Shi, W.; Yang, J.; et al. Multi-element synergistic doping enhances high-voltage performance of LiCoO2 via stabilizing internal and surface structures. Electrochim. Acta. 2024, 504, 144927.
58. Jamil, S.; Yue, L.; Li, C.; et al. Significance of gallium doping for high Ni, low Co/Mn layered oxide cathode material. Chem. Eng. J. 2022, 441, 135821.
59. Vardar, G.; Bowman, W. J.; Lu, Q.; et al. Structure, chemistry, and charge transfer resistance of the interface between Li7La3Zr2O12 electrolyte and LiCoO2 cathode. Chem. Mater. 2018, 30, 6259-76.
60. Bitzer, M.; Van, Gestel. T.; Uhlenbruck, S.; Hans-peter-buchkremer,
61. Rebohle, L.; Prucnal, S.; Skorupa, W. A review of thermal processing in the subsecond range: semiconductors and beyond. Semicond. Sci. Technol. 2016, 31, 103001.
62. Ping, W.; Wang, C.; Wang, R.; et al. Printable, high-performance solid-state electrolyte films. Sci. Adv. 2020, 6, eabc8641.
63. Ramos, E.; Browar, A.; Roehling, J.; Ye, J. CO2 laser sintering of garnet-type solid-state electrolytes. ACS. Energy. Lett. 2022, 7, 3392-400.
64. Acord, K. A.; Dupuy, A. D.; Scipioni, Bertoli. U.; et al. Morphology, microstructure, and phase states in selective laser sintered lithium ion battery cathodes. J. Mater. Proc. Technol. 2021, 288, 116827.
65. Zhu, H.; Liu, J. Emerging applications of spark plasma sintering in all solid-state lithium-ion batteries and beyond. J. Power. Sources. 2018, 391, 10-25.
66. Li, L.; Andrews, J.; Mitchell, R.; Button, D.; Sinclair, D. C.; Reaney, I. M. Aqueous cold sintering of Li-based compounds. ACS. Appl. Mater. Interfaces. 2023, 15, 20228-39.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data



















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.