Three dimensional porous triptycene network with 12-connectivity of tröger’s base for iodine capture
Abstract
Developing new porous polymers with higher connectivity can improve their pore structure and increase functional group density, thereby enhancing their performance. Herein, we used tröger’s base (TB) groups as linkers to synthesize a novel 12-connected porous triptycene network (12-TB-PTN). The obtained 12-TB-PTN displayed high iodine capture capacity (515 wt%) and a relatively rapid adsorption rate (1.37 g·h-1) because of the high proportion of TB units in the porous polymer. This strategy has a great scientific importance for the development and preparation of rapid and efficient adsorbents for iodine vapor and other toxic pollutants.
Keywords
INTRODUCTION
Nuclear energy, revered for its high quality and cleanliness, is vital for boosting the nation’s economy, healthcare, and military capabilities. However, nuclear waste disposal and nuclear accidents often release large amounts of chemical and radioactive iodine pollutants (129I and 131I). Due to the strong solubility of iodine, these pollutants infiltrate the ecosystem via environmental migration, leading to severe threats to both environment and human health[1]. In addition to nuclear facilities, iodine is utilized in the treatment of thyroid cancer, resulting in the discharge of radioactive wastewater by numerous medical research institutions. Thus, it is imperative to develop novel materials with enhanced adsorptive capabilities and swift adsorption rates to efficiently secure iodine[2-7]. To date, numerous materials have been developed for iodine adsorption[8-14]. Microporous organic polymers (MOPs) are notable for their abundant microporous structure, robust physical and chemical stability, and customizable surface chemistry, making them effective in adsorbing toxic pollutants[15-18].
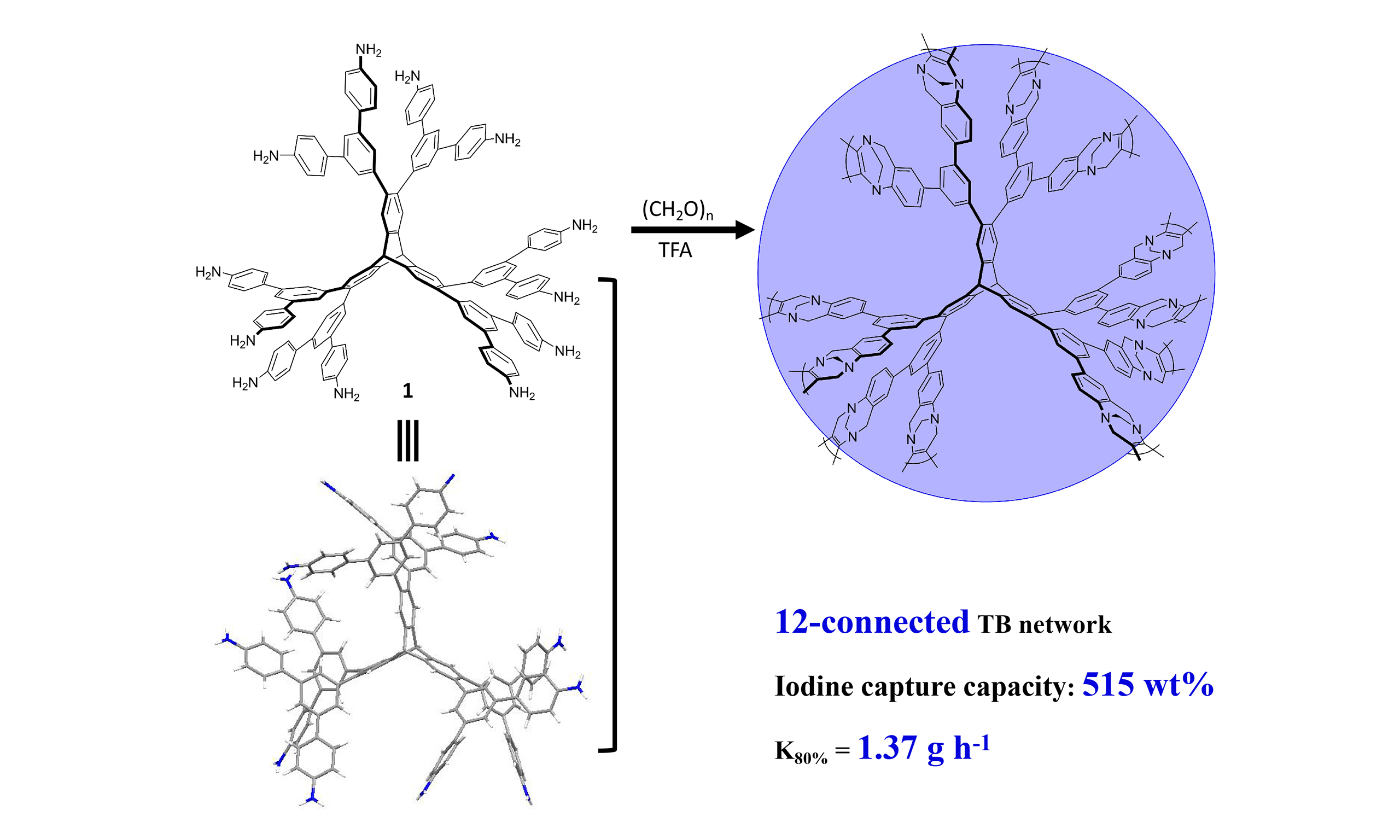
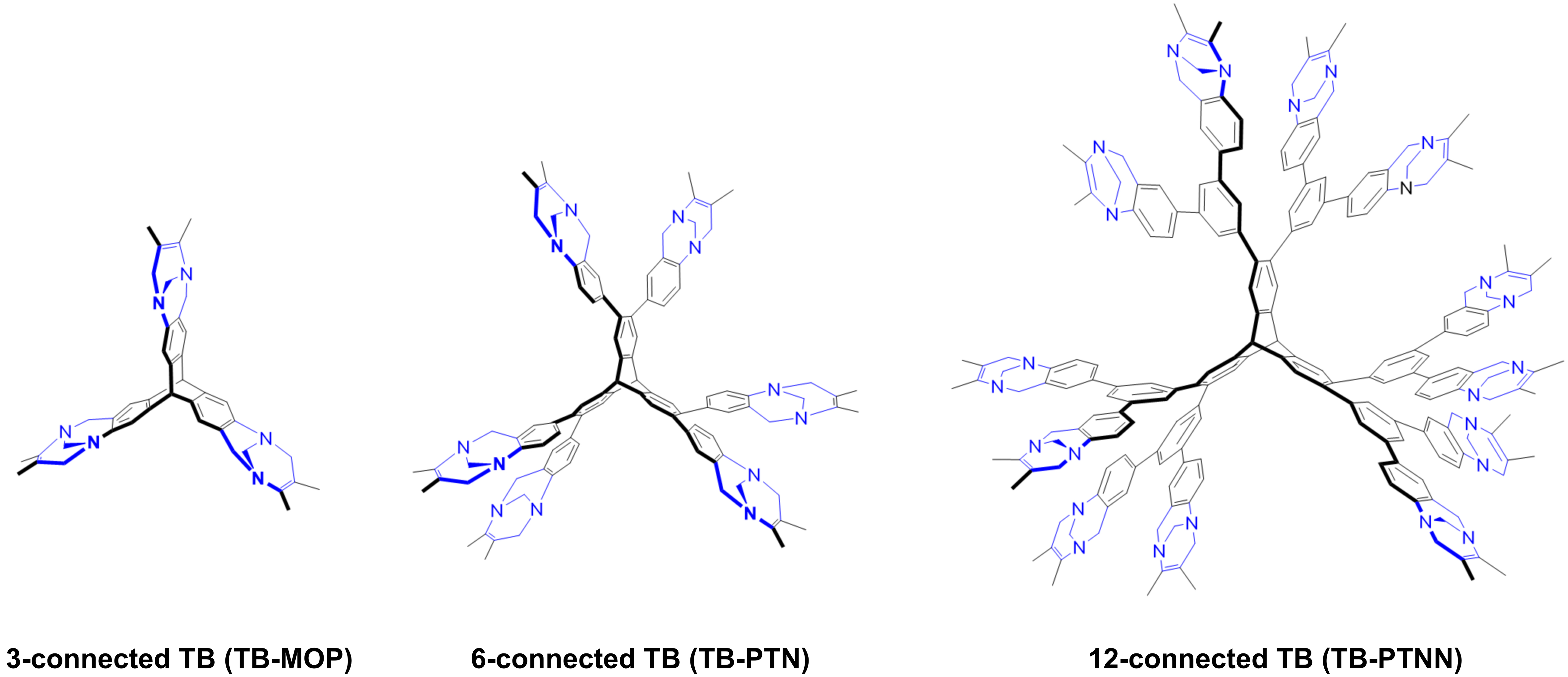
Nowadays, more porous organic materials are being developed by regulating specific pore environments, including coordination sites and Lewis acid-base interactions, etc., to enhance their iodine capture ability[19,20]. Triptycene polymers, bearing specially designed nitrogen-containing units, have attracted significant attention, this is largely due to their distinctive three-dimensional structures and permanent pore structures. This type of material exhibits excellent properties and broad application prospects in fields such as adsorption and separation[21-23]. Among the most promising nitrogen-containing heterocyclic derivatives, tröger’s bases (TB) stand out due to their versatile properties. Notably, TB-derived polymers, which contain two nitrogen atoms per TB unit, provide abundant adsorption sites and strong interactions for iodine capture. Zhu et al. reported a three-dimensional TB-derived porous polymer (TB-MOP), which boasted a high surface area and commendable thermal stability for efficient CO2 capture (17.8 wt%, at 273 K)[24]. Recently, our group[23] reported a “six-connected” porous triptycene network (TB-PTN) utilizing TB units as linkers, exhibiting high adsorption capacities for both CO2 (22.3 wt%, 273 K) and iodine vapor (240 wt%). In fact, the enhanced connectivity among building blocks is crucial for integrating more TB units and improving porosity. It is worth exploring whether the iodine adsorption performance can be improved by increasing the proportion of TB units. Inspired by this, we designed a novel three dimensional porous triptycene network (12-TB-PTN) with a 12-connected structure derived from TB for iodine capture, as illustrated in Figure 1. As we had anticipated, 12-TB-PTN exhibits superior thermal stability and excellent iodine capture capacity (515 wt%) with a relatively fast uptake rate.
EXPERIMENTAL
Synthesis and general procedures
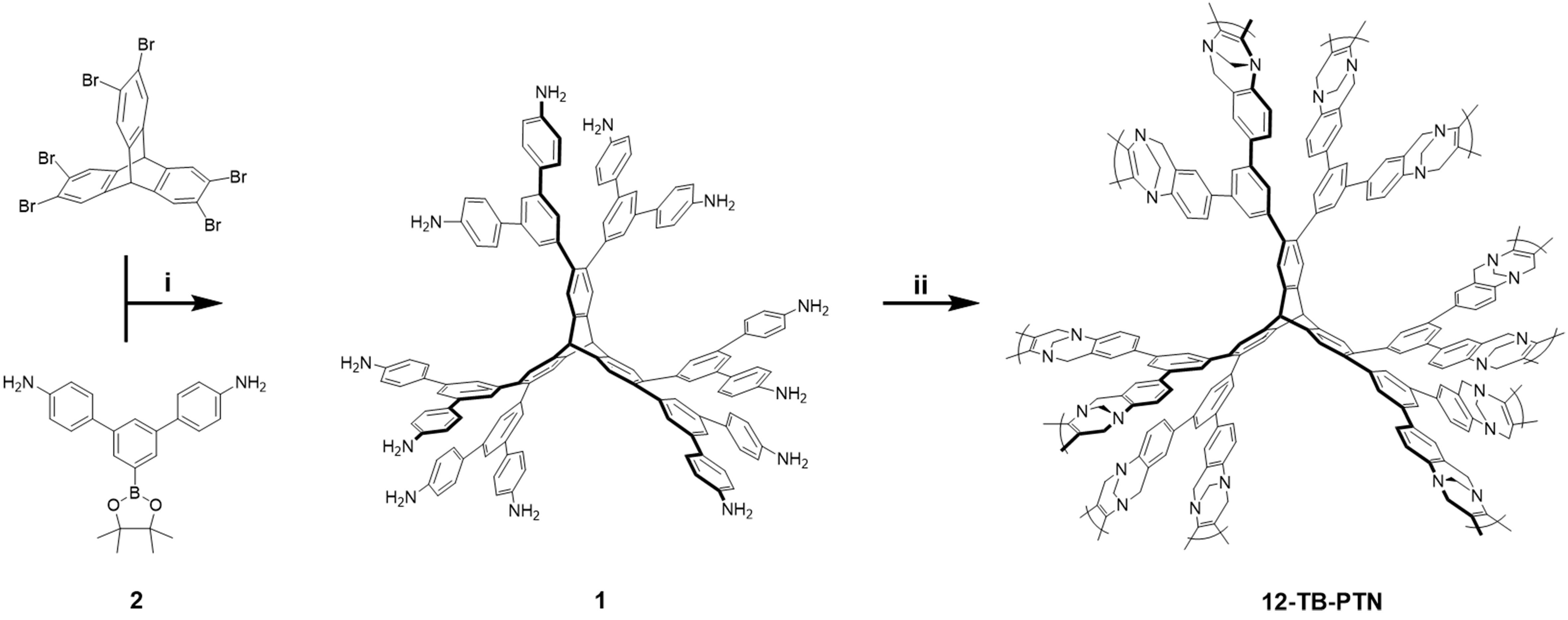
Reagents and conditions detailed: (i) Pd(PPh3)4, K2CO3, in THF and H2O at 75 °C; (ii) TFA, (HCHO)n, room temperature for 3 days. More detailed information, including the sources of the reagents and materials, the manufacturer, model, and country of origin for the experimental instruments were presented in the Supplementary Section 1.1 (Materials) and 1.2 (Characterization).
Synthesis of compound 1
The synthesis of compound 2 was detailed in the Supplementary Section 2. Compound 2 (930 mg,
Synthesis of 12-TB-PTN
As illustrated in Scheme 1, 12-TB-PTN was synthesized according to our previously published procedure[23]. Compound 1 (400 mg, 0.222 mmol) and paraformaldehyde (800 mg, 26.7 mmol) were mixed in trifluoroacetic acid (TFA, 30 mL), and stirred at 200 r·min-1 under 20 °C for 4 days under an argon environment. The resulting mixture was gradually introduced to an ammonia solution and stirred vigorously for 4 h The solid product was collected via filtration, followed by washing with deionized water, methanol, dichloromethane, and dimethyl sulfoxide. The final product, 12-TB-PTN, was dried under a vacuum, acquiring 440 mg of a light brown solid with a 96% yield, and was observed to be insoluble in standard organic solvents.
RESULTS AND DISCUSSION
Characterization of monomer 1
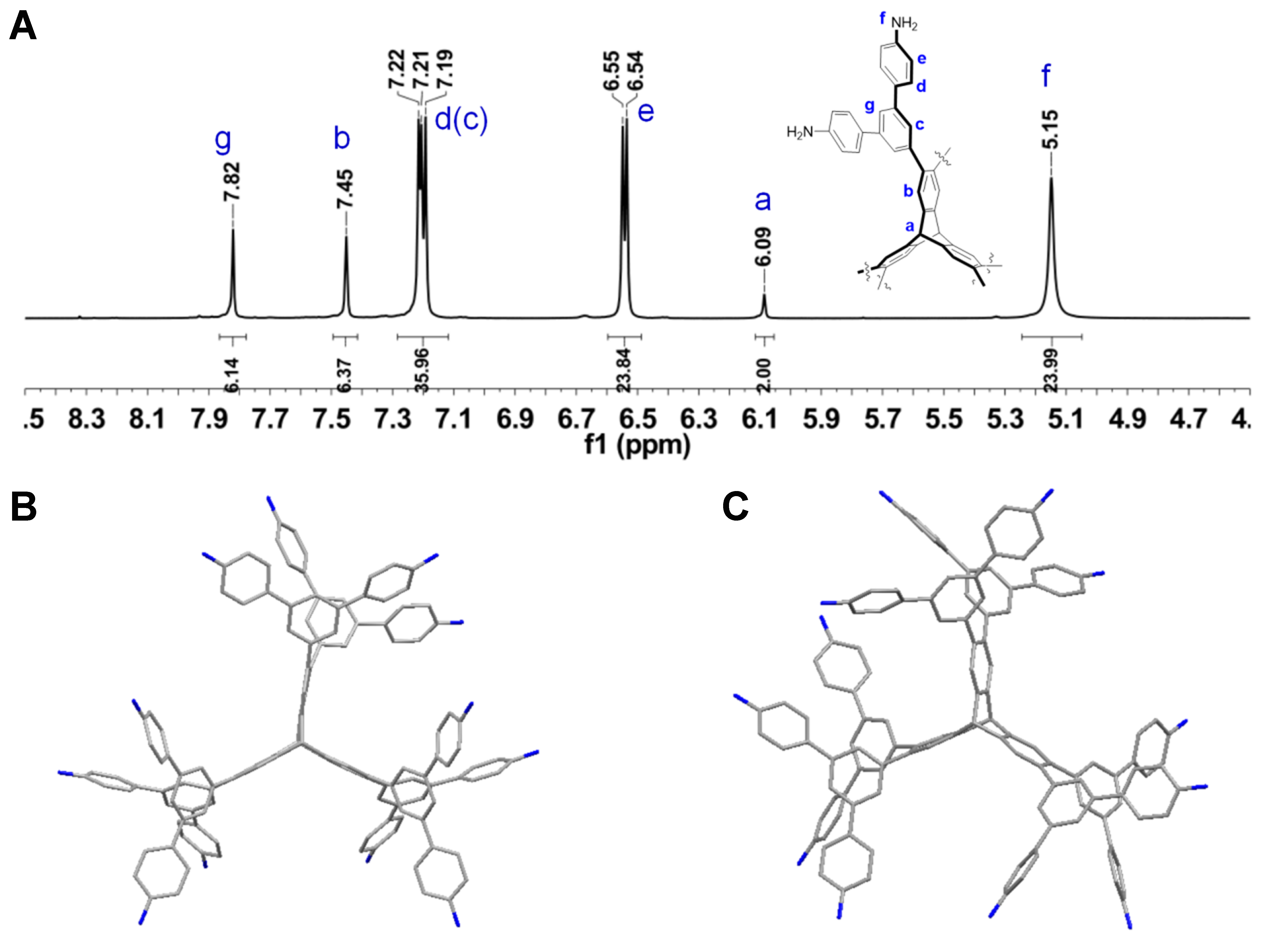
The synthesis of monomer 1 was carried out through a three-step palladium catalyzed coupling reaction in an overall yield of 42%, as verified by Fourier-transform infrared spectroscopy (FT-IR), 1H and 13C NMR spectra. Furthermore, it was found that the electrospray ionization mass spectra (ESI-MS) of compound 1 revealed the peak at m/z 902.9116 for [(M+2H+)/2]2+, which was consistent with its structure [Figure 2A and Supplementary Figures 1-3]. Fortunately, we obtained suitable single crystals of compound 1 for X-ray diffraction (XRD) by gradually introducing isopropyl ether into an acetonitrile solution of 1. As shown in Figure 2B and C, compared to the 6-connected TB monomer’s triclinic crystal system and P-1 space group, monomer 1 was characterized by a monoclinic crystal system and P21/n space group. As a result of the rigidity of the triptycene skeleton and the steric hindrance of the two adjacent 3,5-disubstituted aniline units, 1 exhibited a propeller-like and non-planar conformation [Supplementary Figure 4].
Characterization of 12-TB-PTN
Solid-state 13C cross-polarization magic-angle spinning (CP/MAS) NMR and FT-IR spectroscopic analyses disclosed the successful generation of 12-TB-PTN. As illustrated in Supplementary Figure 5, the signals at approximately 54, 59, 67, 116, 127 and 147 ppm were attributed to the varied carbon environments of the TB units formed within the 12-TB-PTN network, and then the peak at 142 ppm was ascribed to the carbon atoms within the substituted benzene rings. Within the FT-IR spectral analysis [Supplementary Figure 6], the new vibration bands (near 2,943, 2,895, 2,805, and 1,493 cm-1) corresponded to the feature vibrations of
Gas sorption analysis of 12-TB-PTN
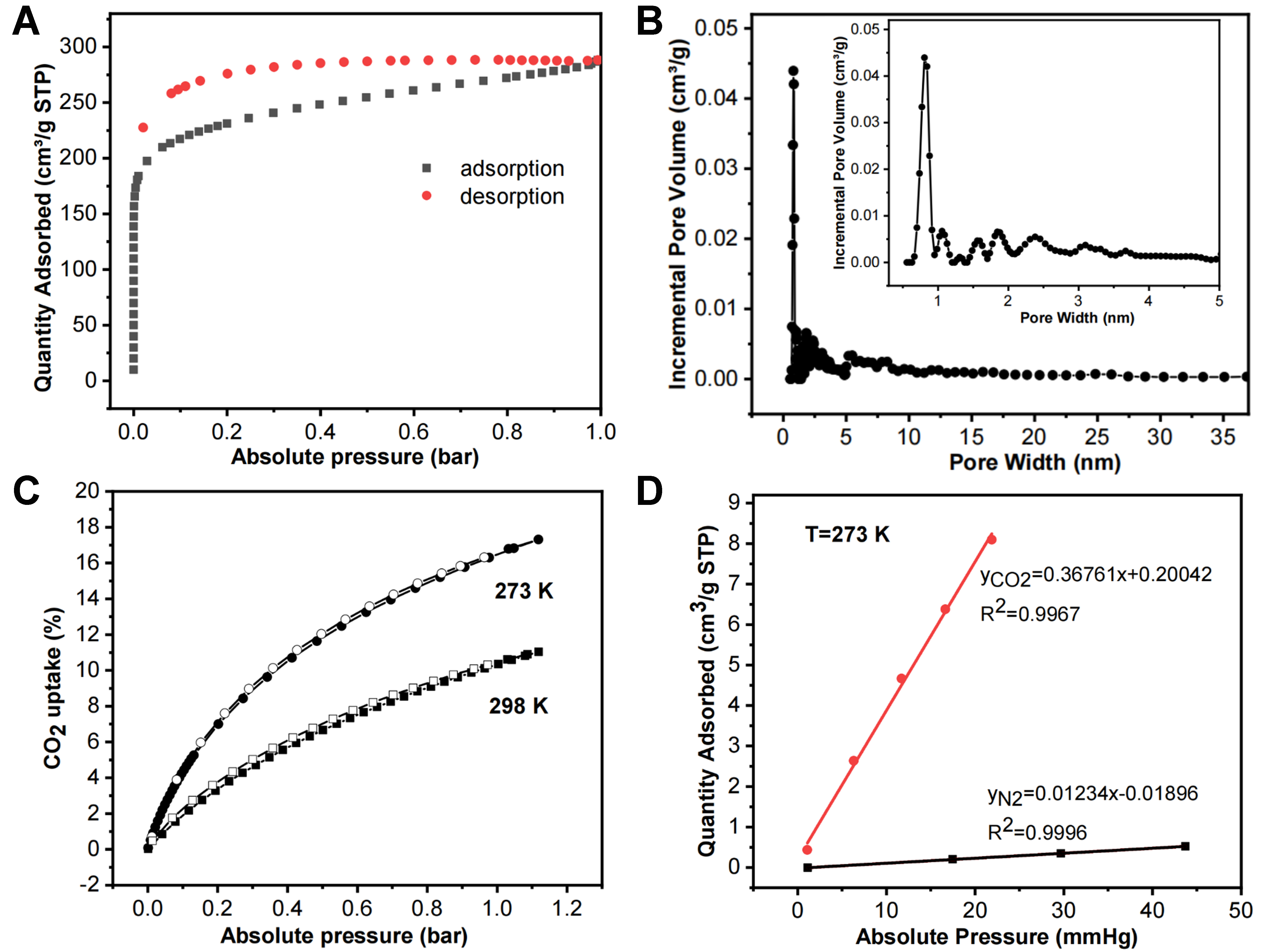
The porosity characteristics of 12-TB-PTN were characterized through a standard typical N2 adsorption-desorption isotherm at 77 K. Figure 3A illustrated that 12-TB-PTN displayed the characteristic type I adsorption behavior, with a steep increase in nitrogen gas uptake at a very low relative pressure (P/P0 < 0.001), indicative of a permanent microporous structure. The observed hysteresis was attributed to the irreversible adsorption of gas molecules within the pores, which was presumably a result of the polymer network’s swelling in the presence of nitrogen at 77 K. The nonlocal density functional theory (NLDFT) was utilized to calculate the pore size distributions (PSD) of 12-TB-PTN. Figure 3B indicated that the primary pore size within the polymer was 0.81 nm, confirming the presence of a primary micropore structure along with some mesopores. The calculated micropore volume (Vmicro) was determined to be 0.23 cm3·g-1, and the total pore volume (Vtotal), estimated from the single point measurement at P/P0 = 0.99, was 0.44 cm3·g-1. The ratio of Vmicro to Vtotal, approximately 0.50, further indicated the mesoporous characteristics of 12-TB-PTN. The mesoporous nature was attributed to the intrinsic porosity and the enhanced network expansion characteristic of the 3D rigid building blocks. The BET (Brunauer-Emmett-Teller) surface area for 12-TB-PTN was determined to be 862 ± 3 m2·g-1, surpassing the 3-connected TB-MOP at 694 m2·g-1, yet below the 6-connected TB-PTN with an area of 1,528 m2·g-1. This observation could potentially be related to the crystalline structure of the monomer. In monomer 1, the significant steric hindrance between the two adjacent 3,5-disubstituted aniline groups on the triptycene skeleton may promote a certain degree of close packing during polymerization and assembly. This could lead to the formation of entangled pore channels within the 12-TB-PTN network, thereby affecting the polymer network’s porosity and BET surface area to some extent.
Figure 3. Structure characterization of 12-TB-PTN. (A) Nitrogen adsorption-desorption isotherms at 77 K; (B) Computed PSD; (C) CO2 adsorption/desorption curves of 12-TB-PTN tested at 273 and 298 K; (D) Calculation of initial slopes from isotherms at 273 K. 12-TB-PTN: 12-Connected tröger’s base porous triptycene network; PSD: pore size distributions.
After establishing the pore property, the CO2 adsorption capabilities of 12-TB-PTN were scrutinized at temperatures of 273 and 298 K. Figure 3C revealed that 12-TB-PTN exhibited a CO2 uptake of 16.8 wt% at 1.0 bar and 273 K. This performance was nearly equivalent to the 3-connected TB-derived TB-MOP, which showed a 17.8 wt% uptake, but it was somewhat less than the 6-connected TB-derived TB-PTN, recording a 22.3 wt% uptake under the same conditions. In addition, 12-TB-PTN exhibited reversible CO2 adsorption, with negligible hysteresis observed between the adsorption and desorption isotherms, suggesting that CO2 was physisorbed in a fully reversible manner. The isosteric enthalpy of adsorption (Qst) for 12-TB-PTN with respect to CO2, derived from the adsorption isotherms using the Clausius-Clapeyron equation at 273 and
Iodine vapor capture
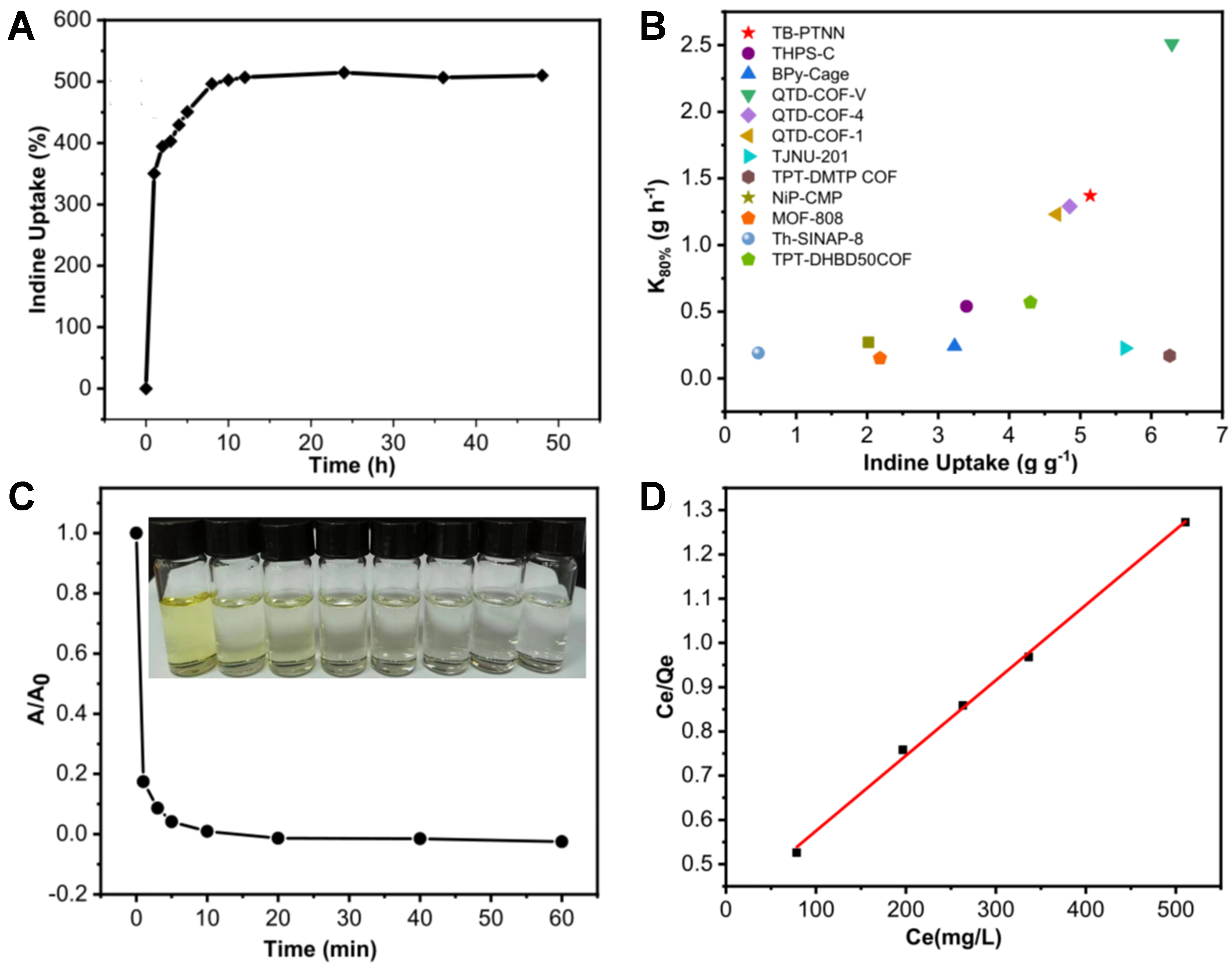
Inspired by the outstanding porous property for 12-TB-PTN, we explored the potential of 12-TB-PTN to serve as an adsorbent for iodine vapor capture. In alignment with standard protocols for nuclear fuel reprocessing, the 12-TB-PTN powder was introduced to an ample supply of iodine vapor at 75 °C and under atmospheric pressure within a sealed glass vial. As shown in Figure 4A, the adsorption capacities rose almost linearly within the initial 3 h, accounting for about 80% of the total adsorption potential. Equilibrium was nearly attained after 10 h, with no additional changes detected beyond 48 h, indicating that the adsorption had reached its limit, the measured saturated adsorption capacity of 12-TB-PTN was 515 wt%, which was considered a high value among materials reported for iodine adsorption. At the same time, we conducted a comparative evaluation of the adsorption performance of various iodine adsorption materials based on K80% (K80% = 80% of the saturated adsorption capacity/adsorption time, in g·h-1) more scientifically and reasonably[6]. From Figure 4B, it can be seen that the K80% value of 12-TB-PTN was relatively high, at
Figure 4. (A) Gravimetric iodine vapor uptake by 12-TB-PTN over time at 75 °C and ambient pressure; (B) Comparison of saturated adsorption capacity and K80% values for various porous materials, with “red star” denoting the 12-TB-PTN in this work; (C) Adsorption kinetics of I3- onto 12-TB-PTN, with accompanying solution images displayed as insets; (D) Langmuir isotherm analysis for I3- adsorption on 12-TB-PTN. 12-TB-PTN: 12-Connected tröger’s base porous triptycene network.
In order to comprehend the mechanism behind iodine adsorption, a suite of analytical methods, encompassing FT-IR, Raman spectroscopy (RM), and X-ray photoelectron spectroscopy (XPS), were meticulously applied to both the pristine 12-TB-PTN and the iodine-saturated I2@12-TB-PTN samples. Supplementary Figure 12A demonstrated that the FT-IR spectra of 12-TB-PTN exhibited noticeable changes following iodine adsorption. The distinctive peaks around 1,207 cm-1, corresponding to the C–N stretching vibration, and between 2,800 and 3,000 cm-1, assigned to the –CH2– bonds in the TB units, showed a reduction in intensity upon iodine adsorption, but recovered fully after desorption. In the RM spectra, it was observed that post-adsorption of iodine by 12-TB-PTN, the signature peaks corresponding to I5- manifested at around 109 and 168 cm-1, markedly different from the 178 cm-1 peak characteristic of unbound iodine molecules, as depicted in Supplementary Figure 12B. This discrepancy suggested a charge transfer interaction between the adsorbent and iodine[6]. In addition, the binding energy shifts observed in the XPS spectra of I2@12-TB-PTN indeed suggested the formation of I5-. As shown in Supplementary Figure 12C, after iodine adsorption, the binding energies of I3d5/2 and I3d3/2 for 12-TB-PTN post-iodine adsorption were observed at around 619.6 and 631.0 eV, respectively. These values exhibited a significant shift compared to the characteristic energies of 618.6 and 630.1 eV for elemental iodine, which can be ascribed to the formation of I5-.
The reversible iodine adsorption experiments, conducted in ethanol as presented in Supplementary Figure 13, revealed that the color of the solution intensified over time, signifying the release of iodine from the polymer networks. The iodine-loaded 12-TB-PTN underwent further purification through Soxhlet extraction with methanol for a duration of 2 days, followed by drying under vacuum. The regenerated 12-TB-PTN was subjected to subsequent cycles by re-exposure to iodine vapor. As depicted in Supplementary Figure 12D, it was observed that 12-TB-PTN retained 95% of its iodine uptake capacity after five consecutive cycles, thereby confirming its outstanding stability and recyclability for the enrichment of iodine. Furthermore, the FT-IR analysis depicted in Supplementary Figure 14 revealed no significant spectral alterations in the recovered 12-TB-PTN, thereby confirming the structural stability of 12-TB-PTN throughout the iodine enrichment process.
Iodine capture in aqueous solution
Nuclear wastewater discharge has always been a topic of great concern, as the radioactive iodine present in nuclear wastewater poses incalculable impacts on the ecological environment and human health. In the aqueous environment, iodine predominantly exists as I3- (I2 is stabilized by an excess of KI via the triiodide equilibrium I2 + I- ⇋ I3-). Hence, it is of significant value to investigate the adsorptive capacity of 12-TB-PTN for capturing iodine from aqueous solutions. In practice, 12-TB-PTN powder was immersed in an I3- aqueous solution with a concentration of 0.2 mmol/L, and the ultraviolet-visible (UV-vis) absorption spectra were obtained by analyzing the supernatant at various time intervals. As shown in Figure 4C, the aqueous solution became nearly colorless within 5 min, indicating that over 95% of I2 was rapidly removed from the water. To further explore adsorption behavior of 12-TB-PTN for I3-, the equilibrium data were subjected to analysis using the Langmuir and Freundlich isotherm models. Figure 4D demonstrated that the adsorption of 12-TB-PTN for I3- more closely aligns with the Langmuir isotherm model, as indicated by the high coefficient of determination (RL2 = 0.995). The adsorption isotherm revealed that the maximum adsorption capacity of 12-TB-PTN for I3- was 631 mg·g-1, which was notably higher compared to certain porous organic polymers (POPs)[26] and inorganic porous materials[27] (Supplementary Table 3, based on Langmuir adsorption isotherm, not titration or gravimetric analysis).
CONCLUSIONS
We have successfully designed and synthesized a new 3D “12-connected” MOP, 12-TB-PTN, by incorporating 3,5-disubstituted aniline phenyl into the traditional triptycene scaffold through a facile three-step Suzuki-Miyaura coupling reaction, then through high-yield polymerization protocol. Proof by facts, the increase in the number of TB units connected in 12-TB-PTN effectively enhances its iodine adsorption capacity and rate, while also regulating its thermal stability and overall adsorption performance. This strategy has significant scientific importance for the development and preparation of rapid and efficient adsorbents for iodine vapor and other toxic pollutants.
DECLARATIONS
Authors’ contributions
Experiment performing, data analysis, results discussion and manuscript drafting: Liu, N.
Data acquisition: Hu, J.
Performed manuscript revision, data acquisition, as well as provided administrative, technical, and material support: Liu, N.; Zhang, C.; Tan, B.; Bu, J.
Availability of data and materials
The raw data supporting the findings of this study are available within this Article and its Supplementary Materials. Further data is available from the corresponding authors upon reasonable request.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
The authors gratefully acknowledge the support from the National Natural Science Foundation of China (22275062 and 22031010), the Analytical and Testing Center of Huazhong University of Science and Technology and the Research Core Facilities for Life Science (HUST) for related analysis.
Conflicts of interest
Tan, B. is the Guest Editor of the Special Issue “Recent Research Progress and Application of Porous Organic Materials”. Tan, B. was not involved in any steps of editorial processing, notably including reviewers’ selection, manuscript handling and decision making. The other authors declare that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Ewing, R. C.; von Hippel, F. N. Energy. Nuclear waste management in the United States - starting over. Science 2009, 325, 151-2.
2. Li, B.; Qiu, W.; Yap, G. P. A.; Dory, Y. L.; Claverie, J. P. Hydrogen-bonded organic frameworks based on endless-stacked amides for iodine capture and detection. Adv. Funct. Mater. 2024, 34, 2311964.
3. Wang, C.; Wang, Y.; Ge, R.; et al. A 3D covalent organic framework with exceptionally high iodine capture capability. Chemistry 2018, 24, 585-9.
4. Huang, Y.; Yu, Z.; Zhang, Q.; Luo, F. Constructing quinazolinone-anchored electron-rich covalent organic frameworks by photocatalytic reductive cyclization for idealizing iodine capture. Sci. China. Mater. 2023, 66, 2339-45.
5. Luo, D.; He, Y.; Tian, J.; Sessler, J. L.; Chi, X. Reversible iodine capture by nonporous adaptive crystals of a bipyridine cage. J. Am. Chem. Soc. 2022, 144, 113-7.
6. Guo, X.; Li, Y.; Zhang, M.; et al. Colyliform crystalline 2D covalent organic frameworks (COFs) with quasi-3D topologies for rapid I2 adsorption. Angew. Chem. Int. Ed. Engl. 2020, 59, 22697-705.
7. Kurisingal, J. F.; Yun, H.; Hong, C. S. Porous organic materials for iodine adsorption. J. Hazard. Mater. 2023, 458, 131835.
8. Xie, Y.; Pan, T.; Lei, Q.; et al. Ionic functionalization of multivariate covalent organic frameworks to achieve an exceptionally high iodine-capture capacity. Angew. Chem. Int. Ed. Engl. 2021, 60, 22432-40.
9. Lv, J.; Li, W.; Li, J.; et al. A triptycene-based 2D MOF with vertically extended structure for improving the electrocatalytic performance of CO2 to methane. Angew. Chem. Int. Ed. Engl. 2023, 62, e202217958.
10. Chen, P.; He, X.; Pang, M.; Dong, X.; Zhao, S.; Zhang, W. Iodine capture using Zr-based metal-organic frameworks (Zr-MOFs): adsorption performance and mechanism. ACS. Appl. Mater. Interfaces. 2020, 12, 20429-39.
11. Li, Z. J.; Yue, Z.; Ju, Y.; et al. Ultrastable thorium metal-organic frameworks for efficient iodine adsorption. Inorg. Chem. 2020, 59, 4435-42.
12. Liu, C.; Li, W.; Liu, Y.; et al. Porous organic cages for efficient gas selective separation and iodine capture. Chem. Eng. J. 2022, 428, 131129.
13. Wang, R.; Zhao, J.; Fang, Q.; Qiu, S. Advancements and applications of three-dimensional covalent organic frameworks. Chem. Synth. 2024, 4, 29.
14. Cui, J.; Yang, J.; Sun, J. Organic cage-based frameworks: from synthesis to applications. Chem. Synth. 2024, 4, 30.
15. Zhang, S.; Fang, N.; Ji, X.; et al. Dispersive 2D triptycene-based crystalline polymers: influence of regioisomerism on crystallinity and morphology. JACS. Au. 2022, 2, 1638-50.
16. Alam, A.; Mishra, S.; Hassan, A.; et al. Triptycene-based and schiff-base-linked porous networks: efficient gas uptake, high CO2/N2 selectivity, and excellent antiproliferative activity. ACS. Omega. 2020, 5, 4250-60.
17. Hassan, A.; Goswami, S.; Alam, A.; Bera, R.; Das, N. Triptycene based and nitrogen rich hyper cross linked polymers (TNHCPs) as efficient CO2 and iodine adsorbent. Sep. Purif. Technol. 2021, 257, 117923.
18. Alam, A.; Bera, R.; Ansari, M.; Hassan, A.; Das, N. Triptycene-based and amine-linked nanoporous networks for efficient CO2 capture and separation. Front. Energy. Res. 2019, 7, 141.
19. Krishna, R. Describing the diffusion of guest molecules inside porous structures. J. Phys. Chem. C. 2009, 113, 19756-81.
20. Yan, Z.; Yuan, Y.; Tian, Y.; Zhang, D.; Zhu, G. Highly efficient enrichment of volatile iodine by charged porous aromatic frameworks with three sorption sites. Angew. Chem. Int. Ed. Engl. 2015, 54, 12733-7.
21. Zhang, Q.; Zhai, T.; Wang, Z.; et al. Hyperporous carbon from triptycene-based hypercrosslinked polymer for iodine capture. Adv. Mater. Inter. 2019, 6, 1900249.
22. Rabbani, M. G.; Reich, T. E.; Kassab, R. M.; Jackson, K. T.; El-Kaderi, H. M. High CO2 uptake and selectivity by triptycene-derived benzimidazole-linked polymers. Chem. Commun. 2012, 48, 1141-3.
23. Liu, N.; Ma, H.; Sun, R.; Zhang, Q. P.; Tan, B.; Zhang, C. Porous triptycene network based on tröger’s base for CO2 capture and iodine enrichment. ACS. Appl. Mater. Interfaces. 2023, 15, 30402-8.
24. Zhu, X.; Do-Thanh, C. L.; Murdock, C. R.; et al. Efficient CO2 capture by a 3D porous polymer derived from tröger’s base. ACS. Macro. Lett. 2013, 2, 660-3.
25. Hu, Y.; Jiang, Y.; Li, J.; et al. New-generation anion-pillared metal–organic frameworks with customized cages for highly efficient CO2 capture. Adv. Funct. Mater. 2023, 33, 2213915.
26. Huang, M.; Yang, L.; Li, X.; Chang, G. An indole-derived porous organic polymer for the efficient visual colorimetric capture of iodine in aqueous media via the synergistic effects of cation-π and electrostatic forces. Chem. Commun. 2020, 56, 1401-4.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Special Topic
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.