Dimetal and duplex heteroatoms co-doped graphene aerogel in electrolytic CO2 reduction to CO in aqueous electrolyte
Abstract
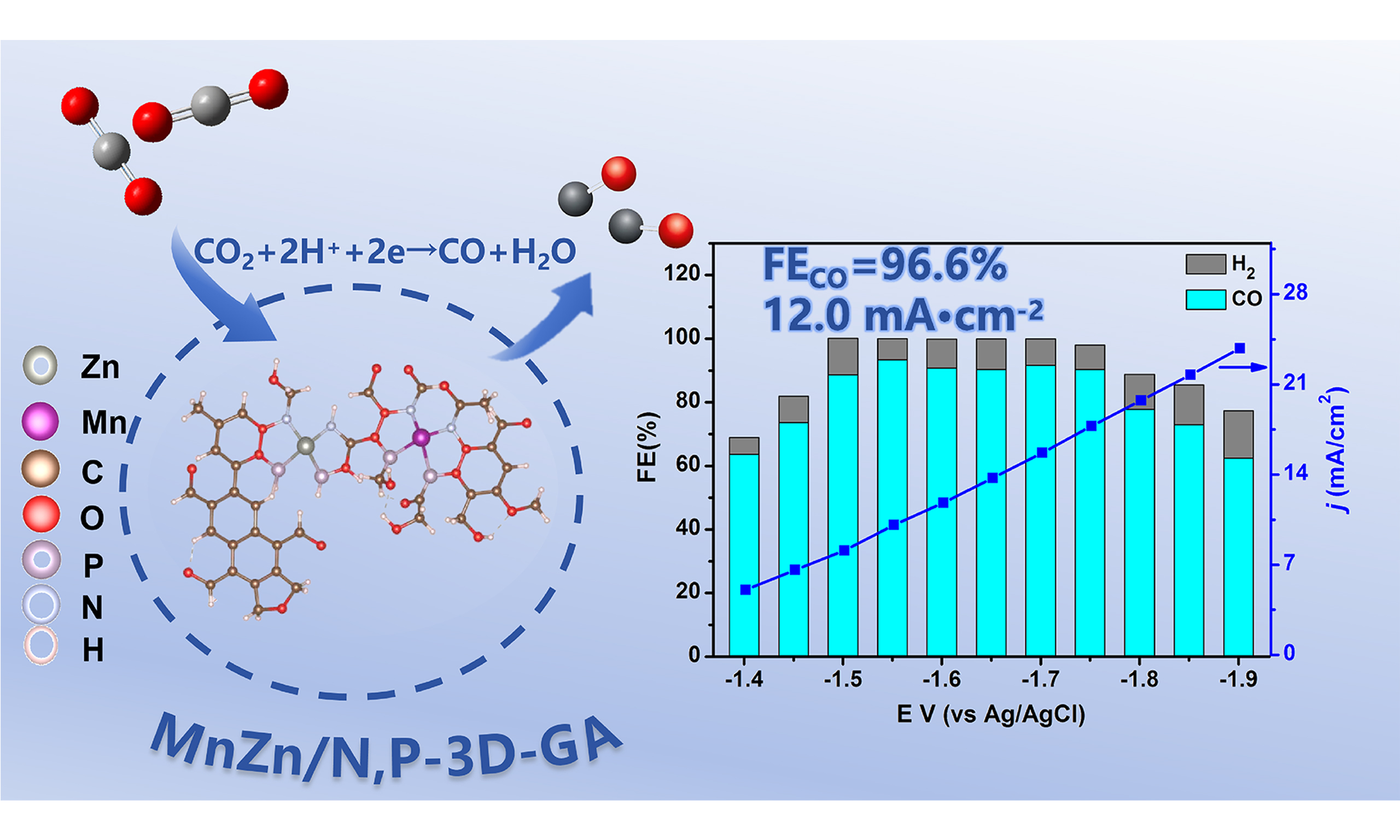
CO2 electrochemical reduction (CO2ER) is a promising alternative for the conversion of CO2 into green and value-added chemicals. Among these chemicals, CO is an important product and platform molecule of the CO2ER, which is always used to produce various chemicals such as methanol, aldehydes, and synthesis gas (H2/CO). Developing efficient catalytic systems for the CO2-to-CO, especially via the electrolytic chemical way, is highly important. In the present work, a N and P co-doped MnZn-bimetal supported 3D graphene aerogel (GA) catalyst, denoted as MnZn/N,P-3D-GA, was prepared and used for the CO2ER to produce CO, which delivered high performance in the reaction. When the potential was -0.92 V [vs. reversible hydrogen electrode (RHE)], the Faradaic efficiency of CO (FECO) reached 96.6% and the current density was 12.0 mA·cm-2. In addition, at the potential range from -0.97 to
Keywords
INTRODUCTION
Utilization of CO2 to produce valuable chemicals is highly important in recent years[1]. Among the CO2-based valuable downstream chemicals, CO is an ideal product in CO2 electrochemical reduction (CO2ER), which is primarily utilized for the production of fundamental chemicals such as methanol, ethanol, fatty aldehydes, and for the liquid hydrocarbons including gasoline and diesel via the Fischer-Tropsch process[2-4]. To date, noble metals such as Au[5], Pd[6], Ag[7], etc. and transition metals including Ni[8], Zn[9], Mn[10], Cu[11], etc. have been used to prepare CO in CO2ER and showed excellent catalytic performance in the reaction. Nonetheless, the metal active components exhibit shortcomings such as being easy to agglomerate and uneven dispersion at elevated temperatures, which further impedes electron transport and diminishes the catalyst’s activity. Therefore, selecting a suitable carrier to improve the dispersion of active species and constructing an interaction force between carrier and active center to improve the stability of the active species is an important approach to preparing efficient catalysts[12,13].
In recent years, carbon materials containing carbon nanotubes (CNTs) and graphene have emerged as significant catalyst carriers, primarily owing to their surface area, excellent electrical conductivity, and cost-effectiveness. Among them, the two-dimensional (2D) graphene material, in which Csp2 is closely packed into a single-layer 2D honeycomb lattice structure, has a high specific surface area, good chemical stability, high conductivity, and special structure. Thus, the material is one of the ideal electrocatalytic materials. However, it tends to irreversibly stack during application, leading to severe agglomeration, which not only leads to a significant reduction in its specific surface area and mass transfer ability but also results in a decrease in its electroactive sites and overall catalytic activity[14]. Therefore, to improve the structural stability of graphene and maintain its catalytic activity, metal nanoparticles are usually inserted between graphene sheets to prevent its accumulation[15,16]. For instance, Gu et al. successfully designed and synthesized a graphdiyne/graphene (GDY/G) heterostructure as a 2D conductive material, which was then combined with cobalt phthalocyanine (CoPc) to achieve the catalytic CO2ER reaction with a Faradaic efficiency of CO (FECO) of 96%[17]. Rogers et al. embed gold nanoparticles (Au NPs) into a graphene nanoribbon (GNR) matrix to obtain Au NPs/GNR composite catalyst, which could achieve CO2ER reaction with FECO greater than 90%[18]. Although the utilization of metal nanoparticle decoration and graphene to fabricate metal nanoparticle-graphene composite catalysts has shown potential in enhancing the activity of graphene in CO2ER reactions, the preparation of catalysts possessing low overpotential, high selectivity, and long-term stability remains challenging.
In general, the interconnected pore structure of three-dimensional (3D) graphene aerogel (GA) can offer a larger number of reactive sites and enhanced electron transport rates, facilitating the doping of heteroatoms such as nitrogen (N), phosphorus (P), boron (B), and others[19-22]. Incorporating heteroatoms into GA to create surface defects can effectively enhance its catalytic activity in the CO2ER reaction. Indeed, N-doped GA can modify the electronic properties of the carbon lattice, resulting in the generation of numerous active sites on the material surface, further enhancing the conversion of CO2 in the CO2ER reaction[23,24]. Zhang
In the present work, an N and P co-doped 3D-GA material was synthesized via a hydrothermal method, and subsequently combined with MnZn bimetals to create an MnZn/N,P-3D-GA catalyst. The catalyst could achieve the CO2ER reaction to CO, especially with Faradaic efficiency for CO (FECO) of 96.6% at a current density of 12.0 mA·cm-2, in a 0.1 mol/L KCl solution at -0.92 V [vs. reversible hydrogen electrode (RHE)]. It also maintained good stability for a duration of up to 70 h. Additionally, we compiled a collection of Mn-, Zn-based catalysts and other bimetal-doped GAs for the electrocatalytic reduction of CO2 to CO
EXPERIMENTAL
Materials
Manganese (II) acetate tetrahydrate (C4H6MnO4·4H2O, > 99.99%), zinc acetate dihydrate (C4H6O4Zn, > 99.99%), phytic acid (C6H18O24P6, 70% in water), and D-(+)-glucose (C6H12O6, > 99.5%) were purchased from Shanghai Macleans Biochemical Technology Co., Ltd. Ammonia (NH3·H2O, 25%-28%, Sinopharm Chemical Reagent Co., Ltd.), and GA (Scientific Compass Co.) were obtained from corresponding commercial resources. Toray carbon paper (CP, TGP-H-60, Micono Technology Co., Ltd.) and Nafion solution (5.0 wt%, Sigma-Aldrich Chemical Reagent Co., Ltd.) were also purchased from given commercial providers. CO2 (99.999%) and Ar (99.999%) were provided by Xi’an Teda Cryogenic Equipment Co., Ltd.
Synthesis of MnZn/N,P-3D-GA
Typically, GA (40 mg) was dispersed in 20 mL ultrapure water and sonicated for 1 h (denoted as A solution). Manganese (II) acetate tetrahydrate (10 mg), zinc acetate dihydrate (9 mg), and D-(+)-glucose
Electrochemical measurements
Specifically, the MnZn/N,P-3D-GA catalyst (5 mg) was dispersed in 800 μL isopropanol, and then 30 μL of Nafion solution (5 wt%) was added and the mixture was sonicated for 0.5 h. Then, the suspension was slowly dropped onto a 1 × 1 cm2 CP substrate. According to the weight of the CP before and after the titration, the loading amount of the catalyst was determined to be 0.93 mg/cm2.
All electrochemical measurements were carried out on a CHI660E electrochemical workstation (Chenhua, Ltd., Shanghai). A three-electrode cell configuration was employed with a working electrode of CP of 1 ×
Before the test, Ar or CO2 gas was purged into the cathode chamber for 40 min, and then the linear sweep voltammetry (LSV) test was conducted starting from 0 to -2 V (vs. Ag/AgCl). It is worth noting that Ag/AgCl is used as the reference electrode in the measurement process, which will eventually convert the potential (vs. Ag/AgCl) into RHE[31]:
Electrochemical active surface area (ECSA) can affect the catalytic activity of the catalyst. However, the ECSA of the catalyst cannot be obtained directly. Since the double-layer capacitance (Cdl) value is proportional to the ECSA value, the latter can be determined by measuring the Cdl value of the catalyst. The cyclic voltammetry (CV) curves measured within the potential range of non-redox processes can be used to determine the electrochemical Cdl[32]. The catalyst was tested by CV at different scanning rates from -0.64 to -0.165 (V, vs. RHE) to obtain the relationship between Δj (ja-jc) and scan rate. The ja and jc are anode current density and cathode current density, respectively. In the CV tests, the scan rates were 20, 40, 60, 80, 100, 120, 140, 160, and 180 mV·s-1, respectively. The Tafel curve is expressed as η = blogj + a, where η is the overpotential, j is the current density, b represents the Tafel slope, and a is the intercept of the current density axis[32]. Electrochemical impedance spectroscopy (EIS) was tested at open circuit voltage (OCP) over a frequency range of 10-2-105 Hz. The obtained data was fitted by Zview software (Scribner Associates 4.0, USA).
Product detection
The gaseous product was measured with a gas chromatograph equipped with flame ionization (FID) and thermal conductivity (TCD) detectors (FuLi 9790, Zhejiang FuLi Chromatography Co., Ltd., China). The liquid products were analyzed by 1H nuclear magnetic resonance (1H NMR, Bruker Avance III 600 HD spectrometer, Germany). Prior to the test, 900 μL of electrolyte solution was mixed with 100 μL internal standard solution. The internal standard solution was prepared by mixing 14 mL of D2O with 10 μL DMSO-d6 (99.9%), yielding a final DMSO concentration of 10 mM in D2O. For each product, the standard curve was estabilished according to the relative peak area ratio.
Materials characterization
A scanning electron microscope (SEM, MAIA3 TESCAN Brno, Czech Republic) was used for morphology and structure characterization. The transmission electron microscope (TEM) image of the catalyst was measured with a TF20 microscope (Thermo Fisher Scientific, Eindhoven, Netherlands) at 200 kV working voltage. X-ray photoelectron spectroscopy (XPS) spectra (RigakuD/Max 2500, Rigaku Corporation, Tokyo, Japan) of the catalysts were also obtained on the conditions of operating voltage (40 kV) and operating current
Density functional theory calculations
Density functional theory (DFT) calculations were performed using the VASP code. Perdew–Burke–Ernzerhof (PBE) functional within generalized gradient approximation (GGA)[33,34] was adopted to handle the exchange-correlation, while the projector augmented-wave pseudopotential (PAW)[35] was employed with a kinetic energy cut-off of 500 eV to describe the expansion of the electronic eigenfunctions. The vacuum thickness was set to 25 Å to minimize interlayer interactions. The Brillouin-zone integration used a Γ-centered 5 × 5 × 1 Monkhorst–Pack k-point. All atomic positions were fully relaxed until the force reached tolerance and energy of 0.03 eV/Å and 1 × 10-5 eV, respectively. The dispersion-corrected DFT-D method was used to take into account the long-range interactions[36].
The Gibbs free energy change (ΔG) was calculated using the computational hydrogen electrode (CHE) model[36], as determined by
Where ΔE represents the reaction energy obtained by the total energy difference between the reactant and product molecules adsorbed on the catalyst surface, and ΔS is the change in entropy for each reaction. ΔZPE is the zero-point energy correction to the Gibbs free energy. T stands for room temperature (298.15 K).
RESULTS AND DISCUSSION
Catalyst characterization
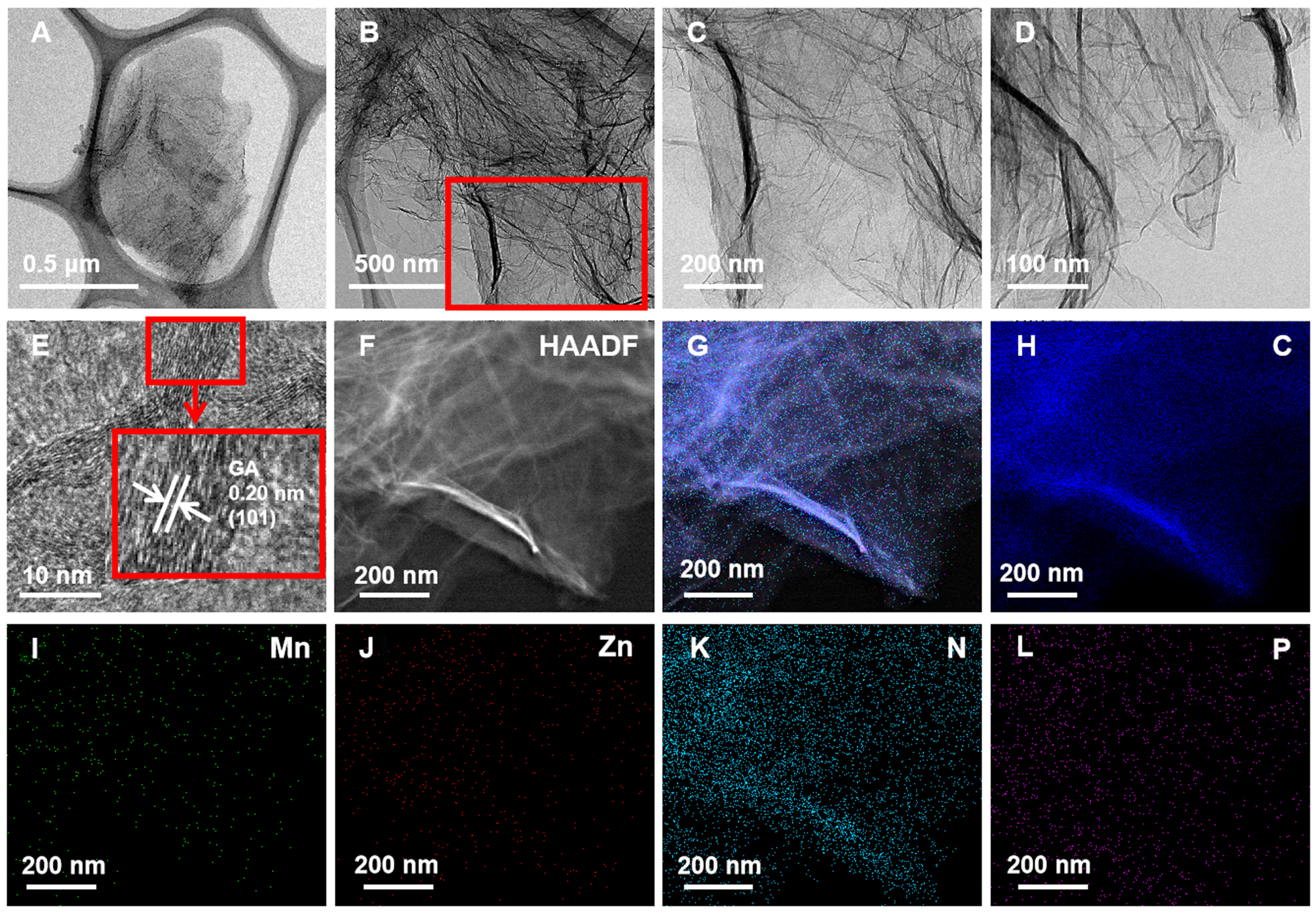
The MnZn/N,P-3D-GA catalyst was synthesized by a hydrothermal method, in which ammonia was the N source, phytic acid was the P source, and GA was the carbon substrate, respectively, and the details are given in supporting information. The preparation processes are illustrated in Supplementary Scheme 1. The Mn and Zn loadings were detected to be 15% and 1.2% via the ICP-MS tests. The morphology and microstructure of the MnZn/N,P-3D-GA catalyst were characterized by SEM and TEM. The MnZn/N,P-3D-GA sample has a lamellar morphology, which is consistent with the lamellar structure of GA
Figure 1. (A-D) TEM images, (E) HRTEM image, (F) HAADF image, and (G-L) EDS elemental mappings of MnZn/N,P-3D-GA. TEM: Transmission electron microscope; HRTEM: high-resolution transmission electron microscopy; HAADF: high-angle annular dark-field imaging; EDS: energy dispersive X-ray spectroscopy; 3D: three-dimensional; GA: graphene aerogel.
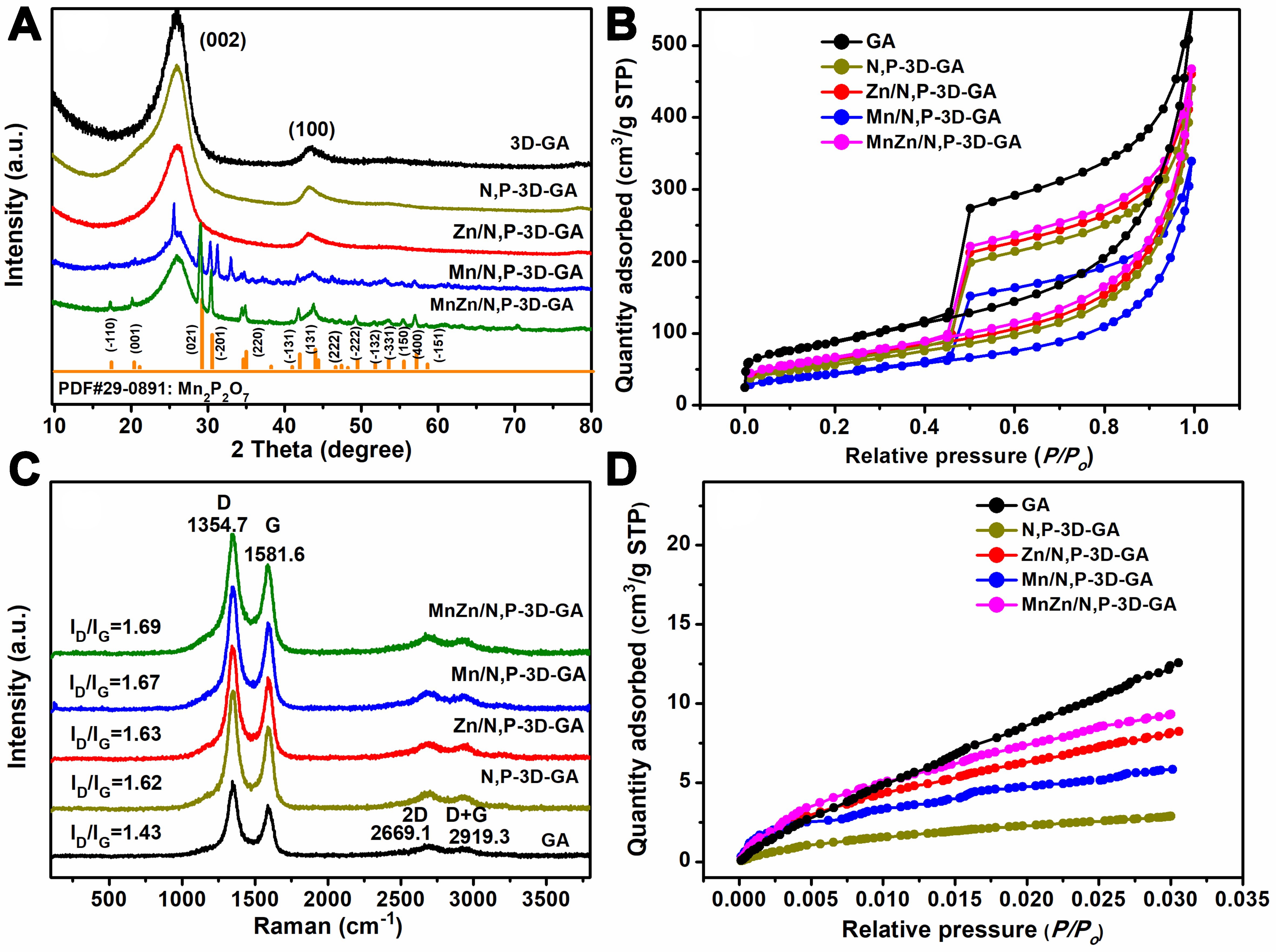
The XRD patterns of catalysts were investigated, with the results given in Figure 2A. The N,P-3D-GA catalyst showed diffraction peaks at 25.6 and 43.4°, which were attributed to the characteristic diffraction peaks of the (002) and (100) crystal planes of GA, indicating that the heteroatom modification did not change the crystal form of GA[37-40]. The MnZn/N,P-3D-GA catalyst was prepared by introducing Zn and Mn metals into N,P-3D-GA. The catalyst showed multiple strong diffraction peaks at 17.6, 20.1, 28.8, 30.6, 42.1, 43.5, 46.6, 47.5, 48.3, 49.1, 51.8, 53.6, 55.3, 56.9, and 58.8°, corresponding to (-110), (001), (021), (-201), (-131), (131), (-202), (002), (041), (-222), (-132), (-331), (150), (400), and (-151) crystal planes of Mn2P2O7 (PDF# 29-0891)[41]. When only Mn is introduced into the N,P-3D-GA, the resulting catalysts Mn/N,P-3D-GA and MnZn/N,P-3D-GA are identical, both containing peaks of Mn2P2O7, indicating that the Mn element has been successfully introduced into the MnZn/N,P-3D-GA catalyst. The incorporation of Zn element could increase the crystallinity of the diffraction peaks of the Mn2P2O7, indicating that the presence of Zn is beneficial for the formation of Mn2P2O7 component. It is worth noting that although the Mn2P2O7 species was detected by XRD tests, the highly dispersed Mn and P species still exist, and this was confirmed by the element mappings in Figure 1I and L. No new phase related to Mn and Zn metals was found in the XRD patterns. The results showed that Mn and Zn were successfully doped into the catalyst.
Figure 2. (A) XRD patterns, (B) N2 adsorption/desorption isotherm curves, (C) Raman spectra, and (D) CO2 adsorption curves of GA, N,P-3D-GA, Zn/N,P-3D-GA, Mn/N,P-3D-GA, and MnZn/N,P-3D-GA. XRD: X-ray diffraction; 3D: three-dimensional; GA: graphene aerogel.
N2 adsorption/desorption isotherms are given in Figure 2B. From the results, GA, N,P-3D-GA, Zn/N,P-3D-GA, Mn/N,P-3D-GA, and MnZn/N,P-3D-GA all exhibit type IV adsorption isotherms, indicating that these samples are all mesoporous structures[42]. Subsequently, the specific surface area and aperture (Dp) of the samples were calculated using the BET and Barrett–Joyner–Halenda (BJH) methods, respectively. As shown in Supplementary Table 2, the surface area (SBET), Dp, and pore volume of the N,P-3D-GA sample were
Figure 2C shows the Raman spectra of GA, N,P-3D-GA, Zn/N,P-3D-GA, Mn/N,P-3D-GA and MnZn/N,P-3D-GA. The outcomes indicated that these samples exhibit distinct characteristic absorption peaks at approximately 1,354, 1,582, 2,669, and 2,919 cm-1, which could be attributed to amorphous carbon (D), graphitized carbon (G), 2D peak, and D + G peak, respectively[45]. The D band is associated with defects in GA, and a higher D peak indicates a higher degree of material defects. The ID/IG ratio could be applied as an index for the material defects. Generally, an ID/IG ratio below 1 suggests that the catalyst has fewer defects and a more perfect structure, and may have lower activity in catalytic reactions. Conversely, an ID/IG ratio between 1 and 2 is often deemed ideal, as it signifies a suitable level of defects that can increase the number of reaction sites and thereby enhance catalytic efficiency. The catalyst within this range typically demonstrates favorable electrochemical performance in catalytic processes. However, an ID/IG ratio exceeding 2 indicates an abundance of defects and an imperfect structure, which could potentially lead to a decline in conductivity[46]. In our study, the ID/IG value of the MnZn/N,P-3D-GA catalyst was 1.69, which is higher than that of GA (1.43), N,P-3D-GA (1.62), Zn/N,P-3D-GA (1.63), and Mn/N,P-3D-GA (1.67), respectively. The results showed that the ID/IG values of the catalysts GA, N,P-3D-GA, Zn/N,P-3D-GA, Mn/N,P-3D-GA, and MnZn/N,P-3D-GA fall within the range of 1 to 2. MnZn/N,P-3D-GA has a higher degree of defects, indicating that the addition of MnZn bimetallic enhances the extent of carbon defects, improves the catalyst’s conductivity, and promotes electron transfer.
Figure 2D shows the CO2 adsorption isotherms over different catalysts. The order of the CO2 adsorption amount of the catalysts is GA > MnZn/N,P-3D-GA > Zn/N,P-3D-GA > Mn/N,P-3D-GA > N,P-3D-GA. For comparison, the MnZn/N,P-3D-GA catalyst exhibits a high capacity for adsorbing CO2, and the adsorption capacity of a catalyst is often related to the number of active sites on the catalyst surface, thereby demonstrating outstanding catalytic performances in subsequent CO2ER performance testing.
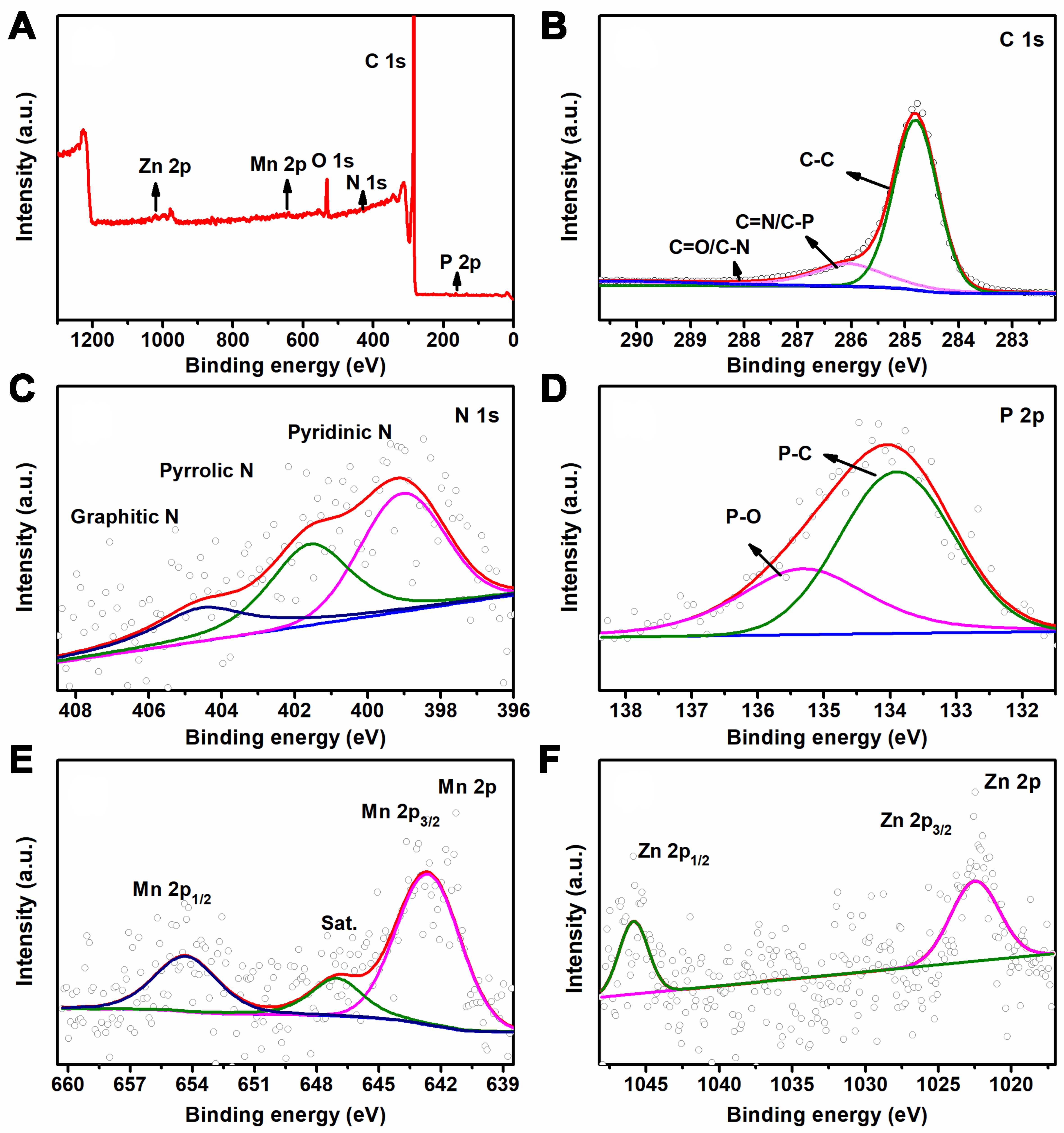
XPS was further carried out to study the chemical composition of the MnZn/N,P-3D-GA catalyst [Figure 3]. The surface of the catalyst comprised six elements: C, O, N, P, Zn, and Mn. The results obtained agree with the analysis performed using energy dispersive X-ray spectroscopy (EDS) element mapping tests [Figure 1G-L]. In the C 1s spectra, the peak could be deconvoluted into three peaks at 284.8, 286.1, and 289.8 eV assigned to C–C, C=N/C–P and C=O/
Catalytic performances
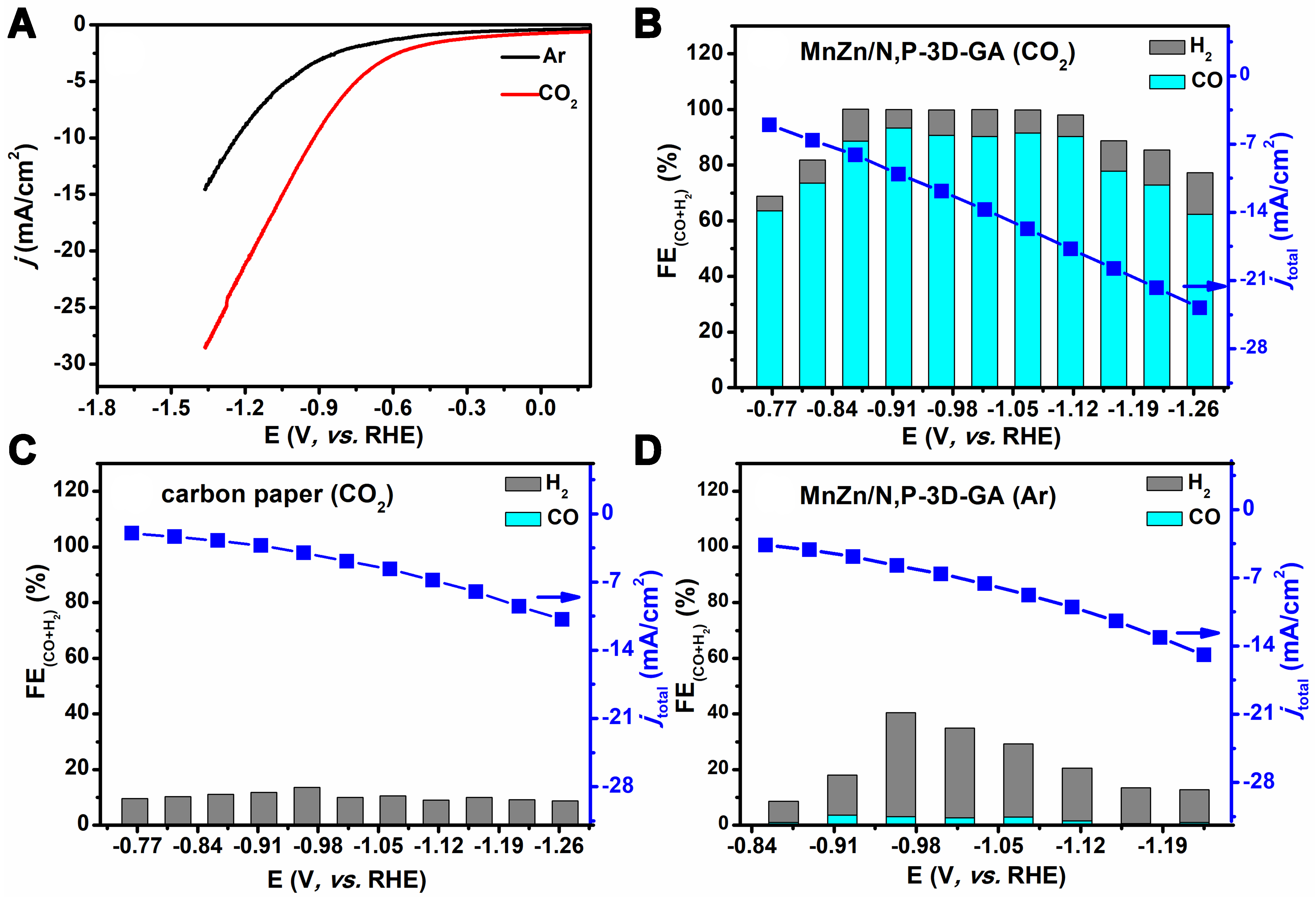
To investigate the electrocatalytic activity of CO2ER, MnZn/N,P-3D-GA was directly used as the working electrode, Ag/AgCl electrode as the reference electrode, and Pt mesh as the auxiliary electrode for electrochemical testing in an H-type electrolytic cell. Initially, the CO2ER performance over the MnZn/N,P-3D-GA catalyst was evaluated by LSV, as shown in Figure 4A. The results indicated that the catalyst exhibited a higher current density in the CO2-saturated electrolyte compared to the Ar-saturated electrolyte, indicating that it has CO2ER activity. Subsequently, the current density and the FE distribution of the product obtained over the catalyst in the CO2ER reaction were recorded in the potential range from -0.77 to -1.27 V (vs. RHE). The gaseous product is analyzed quantitatively using gas chromatography, while the liquid product is analyzed qualitatively and quantitatively using 1H NMR, and detailed information can be found in the supporting information. The results indicated that the gaseous products were mainly H2 and CO, and no liquid products were detected [Supplementary Figures 3 and 4]. Figure 4B showed that CO was the main product at the initial potential of -0.77 V (vs. RHE), and the FECO obtained in the CO2ER reaction was as high as 61.7%. When the potential was increased, the catalyst showed a trend of increasing and then decreasing in FECO. However, the catalyst still managed to achieve higher FECO levels compared to FEH2. In addition, the FECO reached more than 90% in the potential range from -0.92 to -1.12 V (vs. RHE), indicating that the MnZn/N,P-3D-GA catalyst had a wide electrochemical window. When the potential was -0.92 V (vs. RHE), the catalyst achieved the highest FECO of 96.6% and a current density of
Figure 4. (A) LSV curves of MnZn/N,P-3D-GA in CO2- and Ar- saturated 0.1 M KCl electrolytes; Catalytic performances of CO2ER over MnZn/N,P-3D-GA (B) and carbon paper (C) in CO2-saturated 0.1 M KCl electrolyte and over the MnZn/N,P-3D-GA catalyst in Ar-saturated 0.1 M KCl electrolyte (D). LSV: Linear sweep voltammetry; 3D: three-dimensional; GA: graphene aerogel; CO2ER: CO2 electrochemical reduction.
To verify that the CO obtained in the reaction process was generated from CO2, the current densities and product distribution obtained over the CP and the MnZn/N,P-3D-GA catalyst in Ar-saturated electrolyte were recorded and the results are shown in Figure 4C and D. It was discovered that only H2 was detected and the FEH2 was less than 10%, indicating that the MnZn/N,P-3D-GA catalyst plays a critical role in the reaction and the obtained CO in CO2ER was indeed derived from CO2. Figure 4D showed that in the potential range from -0.77 to -1.27 V (vs. RHE), the product obtained over the MnZn/N,P-3D-GA catalyst had only a small amount of CO with less than 5% FECO, indicating that the carbon in the product was generated from the catalytic CO2ER reaction. However, the relatively low total FE generated in CP and MnZn/N,P-3D-GA in the CO2ER is mainly due to the inability to detect dissolved gas products, double-layer charging, the excess heat, and energy consumption caused by the lower current density and larger overpotential.
It is widely recognized that ions in the electrolyte can coordinate with the electrode surface, influencing product selectivity in the CO2ER reaction. Therefore, selecting a suitable electrolyte is significant for optimizing the reaction. We selected K-salt solutions with different anions as an electrolyte to investigate the electrocatalytic activity over the MnZn/N,P-3D-GA catalyst at -0.92 V (vs. RHE), and the results are presented in Supplementary Figure 5A. The MnZn/N,P-3D-GA catalyst achieved the reaction with FECO following the order of KCl > KOH > KHCO3 > K2SO4 > K2CO3. The comparison results revealed that in an electrolyte with an equal concentration of Cl- but different cations [Supplementary Figure 5B], the MnZn/N,P-3D-GA catalyst achieved the synthesis of CO with FECO following the order of KCl > CsCl > NaCl > LiCl. LSV testing shows that the current densities of MnZn/N,P-3D-GA in KCl are significantly higher than those in KOH, KHCO3, K2SO4, and K2CO3 [Supplementary Figure 6A]. Thus, among these electrolytes, the catalyst achieved the highest FECO and current density in 0.1 mol/L KCl electrolyte. This can be attributed to the fact that Cl- does not affect the pH of the solution. However, other tested electrolytes exhibited low catalytic performance, as they provide a distinct local environment at the catalyst surface, particularly with regard to pH changes[27,50]. Supplementary Figure 6B shows that the order of current density follows CsCl > KCl > NaCl > LiCl. Due to the repulsive force of hydrated cations, the surface charge density and electric field are reduced. Since the hydrated cations of Cs+ and K+ have smaller radii, the repulsive force near the electrode is smaller, resulting in higher cation concentrations. This results in greater current density and a stronger interfacial electric field, driving the adsorption of CO2[51]. However, because the HER performance is more severe under higher current densities, we choose K+ as the cation in the electrolyte. Supplementary Figures 7 and 8 indicate that after eight hours of stability testing, the current density of MnZn/N,P-3D-GA remains relatively constant in different electrolytes. Given the decrease in FECO obtained in other electrolytes is relatively fast, we choose KCl solution as the electrolyte.
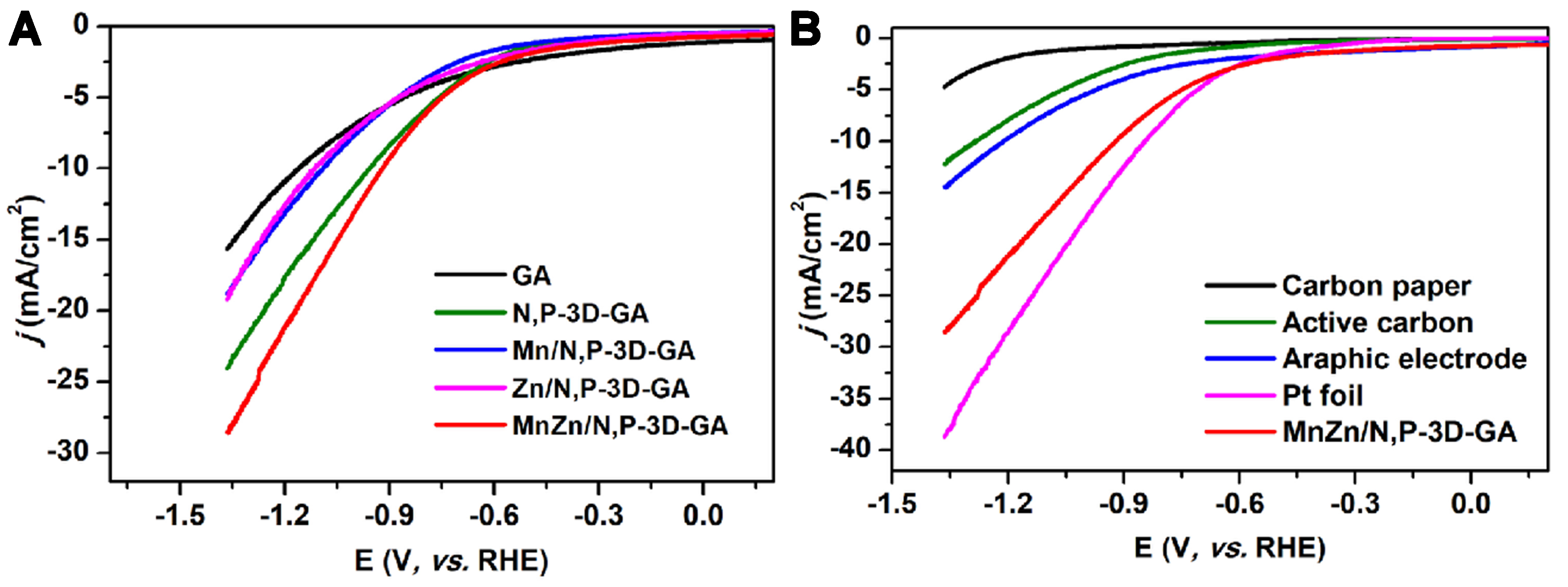
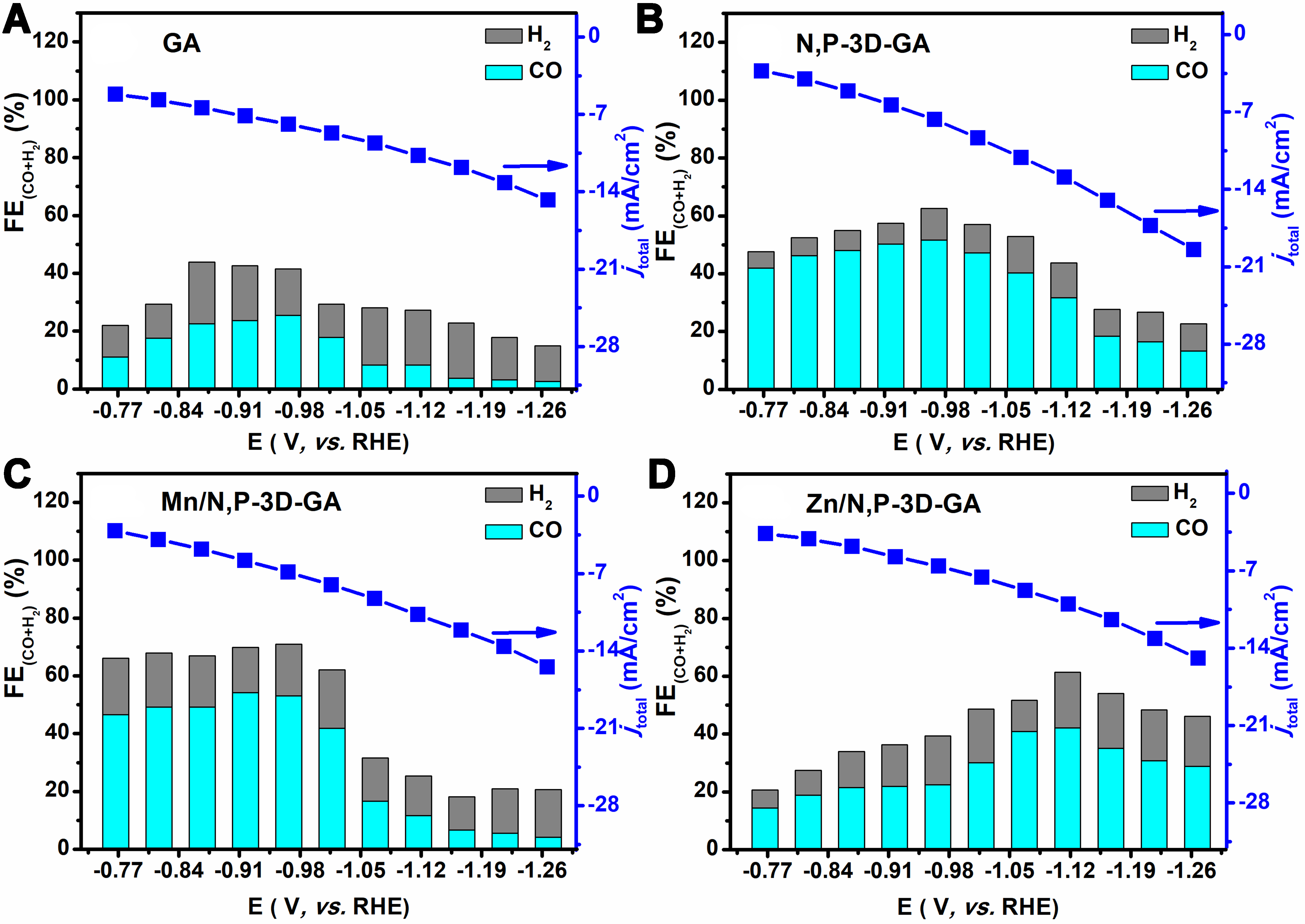
To verify the important role of MnZn bimetal and N, P co-doping on the catalyst, the catalytic performances over the GA, N,P-3D-GA, Mn/N,P-3D-GA, Zn/N,P-3D-GA, and MnZn/N,P-3D-GA catalysts were tested. The LSV curves obtained over these five samples at different potentials are shown in Figure 5A. The results showed that the current density of the catalyst decreases sequentially following the order of MnZn/N,P-3D-GA > N,P-3D-GA > Mn/N,P-3D-GA ≈ Zn/N,P-3D-GA > GA, indicating that MnZn bimetal and N, P co-doping played crucial roles in enhancing the catalytic activity. When compared to commercial electrodes, MnZn/N,P-3D-GA exhibited a larger current density than commercial graphene, CPs, and activated carbon powders, aside from the Pt foil, as shown in Figure 5B. In addition, the FE distribution obtained in the CO2ER reaction over these catalysts was also investigated [Figure 6]. The results showed that the pure GA produced a small amount of H2 and CO (less than 50%) in the potential range from -0.77 to -1.27 V (vs. RHE), indicating that GA had a certain activity for catalyzing CO2ER reaction [Figure 6A]. When GA was doped with N and P, the overall FE, particularly FECO, was significantly increased, indicating that the N and P modification of GA could increase the active sites on the surface of GA, thereby improving the selectivity of the catalyst to CO [Figure 6B]. When Mn was introduced into N,P-3D-GA, the overall FE achieved was comparable to that of N,P-3D-GA. However, the FEH2 value was enhanced, and there was a significant decrease in the production of catalyst products at high potentials. This suggests that the incorporation of Mn atoms could promote the HER [Figure 6C]. Additionally, the introduction of Zn into N,P-3D-GA resulted in a decrease in FECO in the low potential range from -0.77 to
Figure 5. LSV curves obtained over (A) different catalysts and (B) different commercial electrodes in CO2-saturated 0.1 mol/L KCl electrolyte. LSV: Linear sweep voltammetry.
Figure 6. FEs and current densities obtained over (A) GA, (B) N,P-3D-GA, (C) Mn/N,P-3D-GA, and (D) Zn/N,P-3D-GA in a CO2-saturated 0.1 mol/L KCl. FEs: Faradaic efficiencies; GA: graphene aerogel; 3D: three-dimensional.
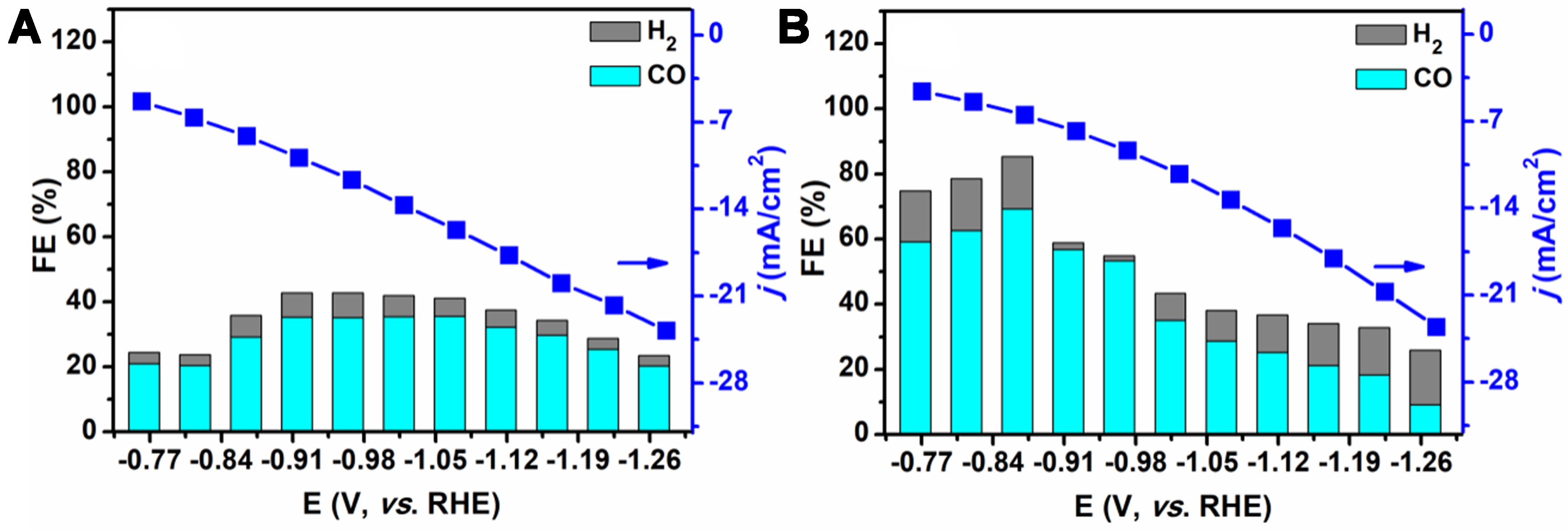
Subsequently, the catalytic activity of MnZn/P-3D-GA was further investigated with the introduction of single heteroatom doping. The findings from this investigation are presented in Figure 7A. When only P was doped, the FECO obtained over the MnZn/P-3D-GA catalyst in the potential range from -0.77 to -1.23 V (vs. RHE) was always higher than FEH2, but the total FE was lower than 50%, indicating that N doping had an important effect on the overall catalytic activity. When only N was doped, the FECO obtained in the potential range from -0.77 to -0.92 V (vs. RHE) was always higher than FEH2, and the total FE was higher than 60%. However, FECO decreased rapidly at high potential, indicating that the doping of P contributed to the enhancement of CO2ER activity in the high potential range [Figure 7B]. These control experiments showed that dual effects of MnZn bimetal and N, P modification can significantly improve the catalytic activity of the MnZn/N,P-3D-GA catalyst in the potential range from -0.77 to -1.27 V (vs. RHE), promoting the formation of CO during the reaction. Additionally, the FECO obtained over the GA, N,P-3D-GA, Mn/N,P-3D-GA, and Zn/N,P-3D-GA catalysts was much lower than that of MnZn/N,P-3D-GA, indicating that the components in the catalyst could indeed promote the CO2ER.
Figure 7. FEs and current densities over catalysts of (A) MnZn/P-3D-GA and (B) MnZn/N-3D-GA in a CO2-saturated 0.1 mol/L KCl. FEs: Faradaic efficiencies; 3D: three-dimensional; GA: graphene aerogel.
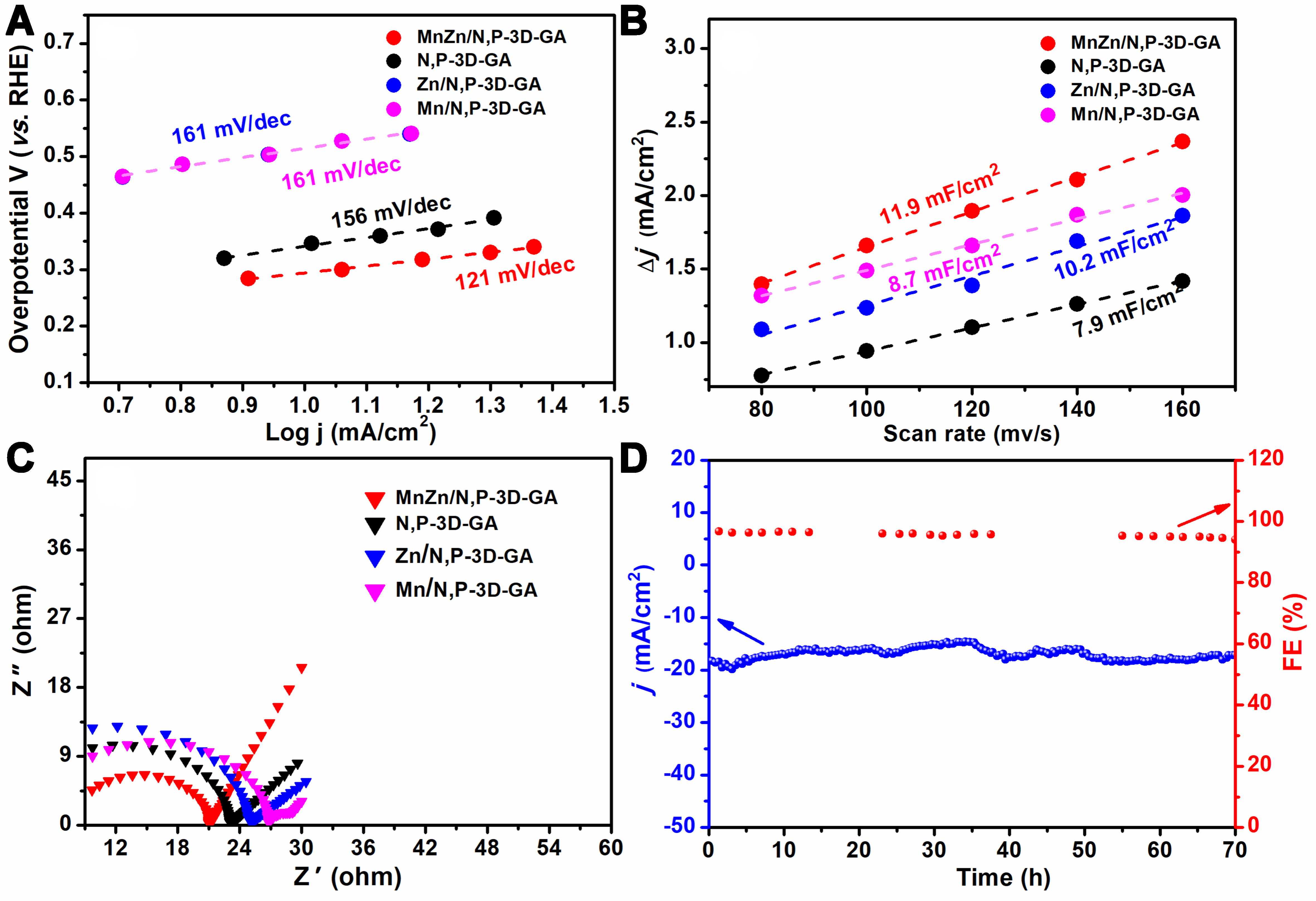
The intrinsic reaction kinetic properties of N,P-3D-GA, Mn/N,P-3D-GA, Zn/N,P-3D-GA and MnZn/N,P-3D-GA were evaluated by Tafel slope, and the results are shown in Figure 8A. The order of Tafel slope values of tested samples followed Mn/N,P-3D-GA ≈ Zn/N,P-3D-GA > N,P-3D-GA > MnZn/N,P-3D-GA. Among them, the MnZn/N,P-3D-GA sample exhibited the lowest value of 121 mV/dec, which is close to the theoretical value (118 mV/dec) of initial electron transfer to CO2 to form surface adsorption, indicating that the CO2 activation process may be the rate-determining step of the CO2ER reaction[53]. At the same time, the ECSA values of N,P-3D-GA, MnZn/N,P-3D-GA, Zn/N,P-3D-GA, and Mn/N,P-3D-GA catalysts were also investigated, and the results are shown in Figure 8B. The values of N,P-3D-GA, MnZn/N,P-3D-GA, Zn/N,P-3D-GA, and Mn/N,P-3D-GA were 7.9, 11.9, 10.2, and 8.7 mF/cm2, respectively. Among them, the ECSA of MnZn/N,P-3D-GA was the largest, indicating that the catalyst has more active sites and facilitates the CO2ER reaction[54,55]. Subsequently, EIS was performed on N,P-3D-GA, MnZn/N,P-3D-GA, Zn/N,P-3D-GA, and Mn/N,P-3D-GA, and the results are shown in Figure 8C. The diameters of the semicircles of the tested catalysts followed the order of Mn/N,P-3D-GA (18.25 Ω) > Zn/N,P-3D-GA
Figure 8. (A) Tafel plots, (B) the ECSA, and (C) EIS of the N,P-3D-GA, MnZn/N,P-3D-GA, Zn/N,P-3D-GA and Mn/N,P-3D-GA; and (D) the stability of MnZn/N,P-3D-GA at -0.92 V (vs. RHE). ECSA: Electrochemical active surface area; EIS: electrochemical impedance spectroscopy; 3D: three-dimensional; GA: graphene aerogel; RHE: reversible hydrogen electrode.
Reaction mechanism
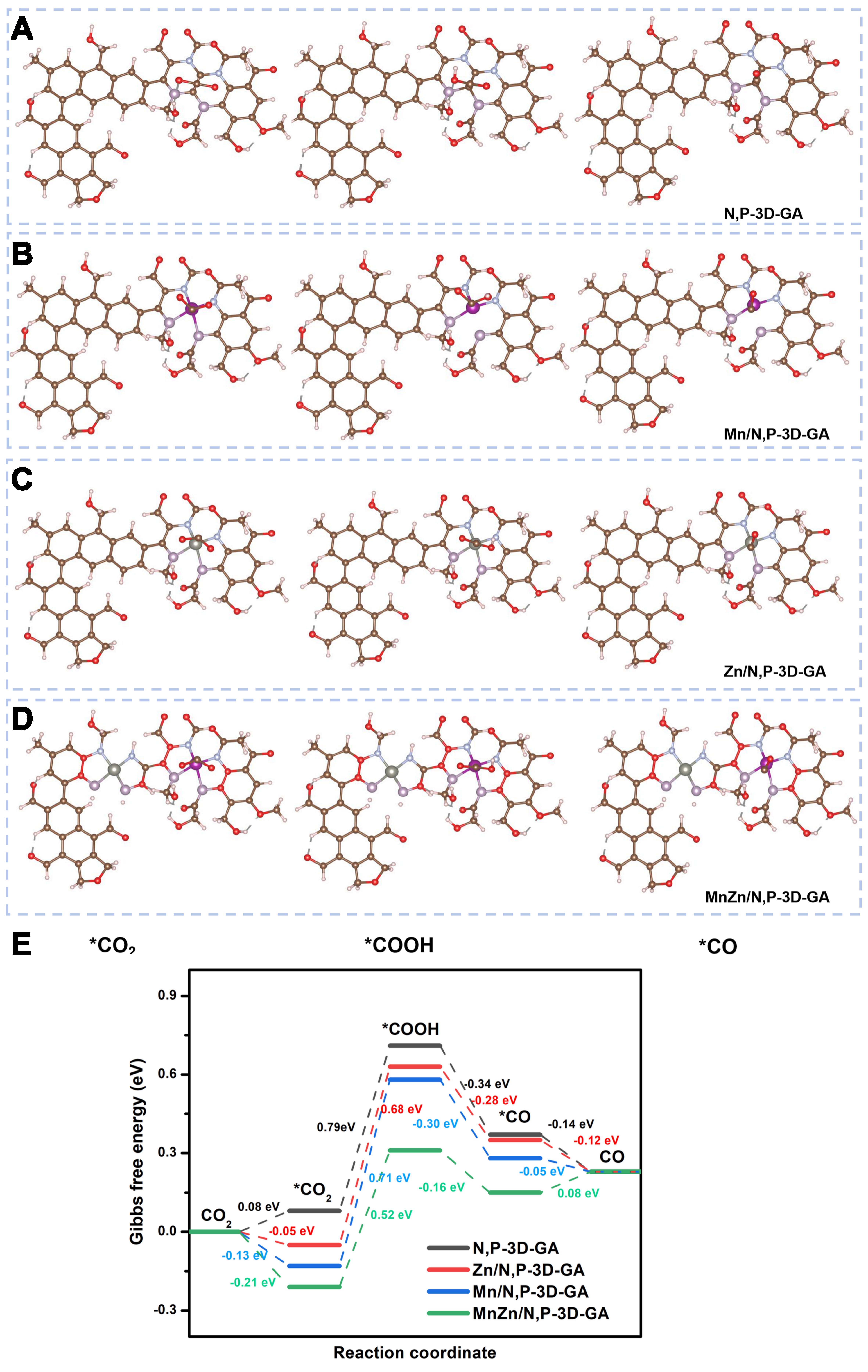
To confirm the mechanism, DFT calculations were conducted to determine the adsorption free energies of reaction intermediates. By utilizing these adsorption energies, we can calculate the reaction energies for each elementary step and the overall reaction at the limiting electrochemical potential of the catalytic surface[59]. The optimized structures were established for N,P-3D-GA, Mn/N,P-3D-GA, Zn/N,P-3D-GA, and MnZn/N,P-3D-GA, as shown in Supplementary Figure 14, and activation processes of CO2 on them via *CO2,
Figure 9. Activation processes of CO2 and intermediates (A-D) and the potential energy curves of CO2ER (E) over N,P/3D-GA (A), Mn/N,P-3D-GA (B), Zn/N,P-3D-GA (C), and MnZn/N,P-3D-GA (D), and MnZn/N,P-3D-GA (D), and Gibbs free energy of CO2ER (E). CO2ER: CO2 electrochemical reduction; 3D: three-dimensional; GA: graphene aerogel.
According to the potential energy curves[62-65], we analyzed the adsorption and activation processes of CO2 over N,P-3D-GA, Mn/N,P-3D-GA, Zn/N,P-3D-GA, and MnZn/N,P-3D-GA, respectively. In the absence of Mn and Zn elements, the energy barrier for CO2 adsorption over N,P-3D-GA is 0.08 eV. In comparison, the energy barriers for CO2 adsorption over Mn/N,P-3D-GA and Zn/N,P-3D-GA are -0.13 and -0.05 eV, respectively. When Mn and Zn elements are co-doped into N,P-3D-GA, i.e., MnZn/N,P-3D-GA, with an energy barrier of -0.21 eV. These results suggest that MnZn/N,P-3D-GA has significant adsorption and activation capabilities, which can overcome the challenges of CO2 adsorption and activation under harsh conditions. The *CO2 to *COOH intermediate in the CO2ER process requires a significant energy barrier. The energy barriers for N,P-3D-GA, Mn/N,P-3D-GA, Zn/N,P-3D-GA, and MnZn/N,P-3D-GA are 0.79, 0.71, 0.68, and 0.52 eV, respectively. This step is the rate-determining step for CO2ER. These values indicated that the formation of the *COOH intermediate requires a higher energy for N,P co-doped GA without a metal loading, suggesting that the Mn and Zn loadings could reduce the energy barriers for converting *CO2 to *COOH species. The results suggested that the metal probably provides an active site for activating CO2, which was confirmed by the configuration in Figure 9A-D. On the other hand, the energy barrier at this step is lower for Zn/N,P-3D-GA compared to Mn/N,P-3D-GA, indicating that Zn incorporation is more favorable for CO production. Moreover, upon introduction of both Mn and Zn, the energy barrier requirement is minimized, leading to facilitated CO2 reduction. The energy barriers for converting *COOH to *CO over N,P-3D-GA, Mn/N,P-3D-GA, Zn/N,P-3D-GA, and MnZn/N,P-3D-GA are -0.34, -0.30, -0.28, and -0.16 eV, respectively. These values indicated that the formation of *CO intermediate releases the highest amount of thermal energy when only N and P were doped into GA. Upon both Mn and Zn introduction, the least amount of thermal energy is released. This further suggests that the combined effects of bimetallic MnZn and N,P modification are more favorable for catalyzing the reduction of CO2 to CO.
CONCLUSIONS
In the present work, bimetallic MnZn loaded on an N,P co-doped 3D GA catalyst, denoted as MnZn/N,P-3D-GA, was synthesized through a hydrothermal method. The catalyst was subsequently utilized to catalyze the CO2ER reaction for the efficient production of CO. The catalyst demonstrated a mesoporous structure and a sizable BET-specific surface area, thereby enhancing the mass transfer capabilities of CO2, intermediates, products, and electrolyte ions. Consequently, it facilitated the CO2ER reaction with the highest FECO conversion efficiency of 96.6% and a current density of 12.0 mA·cm-2, and demonstrated excellent stability for up to 70 h. Besides, within the potential range from -0.97 to -1.12 V (vs. RHE), the catalyst delivered FECO exceeding 90%, illustrating its broad electrochemical window. DFT calculations confirmed that the MnZn/N,P-3D-GA catalyst is primarily involved in *CO2, *COOH, and *CO intermediates during the CO2ER. Additionally, the DFT calculations suggest that CO2 preferentially adsorbs onto the Mn element. Notably, when compared to N,P-3D-GA, Mn/N,P-3D-GA, and Zn/N,P-3D-GA catalysts, the dual modification of MnZn and N,P significantly lowers the energy barrier for *COOH intermediate adsorption. This modification thereby facilitates the efficient conversion of CO2 to CO. The catalytic performance of this catalyst originates from the synergistic effect between the MnZn, N, P, and GA components.
DECLARATIONS
Authors’ contributions
Data curation, writing - original draft: Cao, H. H.
Conceptualization, methodology, writing - original draft, investigation, supervision: He, Z. H.
Data curation: Guo, P. P.
Formal analysis, conceptualization: Tian, Y.
Investigation, data curation: Wang, X.
Data curation, software: Wang, K.
Conceptualization, visualization: Wang, W.
Conceptualization, methodology: Wang, H.
Funding acquisition, supervision, writing - review and editing: Liu, Z. T.
Availability of data and materials
The raw data supporting the findings of this study are available within this Article and its Supplementary Materials. Further data is available from the corresponding authors upon reasonable request.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI tool ChatGPT (version 3.5, released 2022-11-30) was used solely for language editing. The tool did not influence the study design, data collection, analysis, interpratation, or the scientific content of the work. All authors take full responsibility for the accuracy, integrity, and final content of the manuscript.
Financial support and sponsorship
The authors gratefully acknowledge the financial support from the National Natural Science Foundation of China (No. 22078182; No. 22278261) and the Key Project of the Education Department of Shaanxi Province (No. 21JY005).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
Supplementary Materials
REFERENCES
1. Zhu, Z. J.; He, Z. H.; Tian, Y.; et al. Mass-transfer enhancement in the CO2 oxidative dehydrogenation of propane over gan supported on zeolite nanosheets with a short b-axis and hierarchical pores. ACS. Catal. 2024, 14, 10376-91.
2. Wang, C.; Wang, X.; Ren, H.; et al. Combining Fe nanoparticles and pyrrole-type Fe-N4 sites on less-oxygenated carbon supports for electrochemical CO2 reduction. Nat. Commun. 2023, 14, 5108.
3. Tan, X.; Sun, X.; Han, B. Ionic liquid-based electrolytes for CO2 electroreduction and CO2 electroorganic transformation. Natl. Sci. Rev. 2022, 9, nwab022.
4. Zhang, X.; Yan, X.; Chen, P.; et al. Selective and efficient CO2 electroreduction to formate on copper electrodes modified by cationic gemini surfactants. Angew. Chem. Int. Ed. Engl. 2024, 63, e202315822.
5. Mezzavilla, S.; Horch, S.; Stephens, I. E. L.; Seger, B.; Chorkendorff, I. Structure sensitivity in the electrocatalytic reduction of CO2 with gold catalysts. Angew. Chem. Int. Ed. Engl. 2019, 58, 3774-8.
6. He, Q.; Lee, J. H.; Liu, D.; et al. Accelerating CO2 electroreduction to CO over Pd single-atom catalyst. Adv. Funct. Mater. 2020, 30, 2000407.
7. Ma, M.; Liu, K.; Shen, J.; Kas, R.; Smith, W. A. In situ fabrication and reactivation of highly selective and stable Ag catalysts for electrochemical CO2 conversion. ACS. Energy. Lett. 2018, 3, 1301-6.
8. Zheng, T.; Jiang, K.; Ta, N.; et al. Large-scale and highly selective CO2 electrocatalytic reduction on nickel single-atom catalyst. Joule 2019, 3, 265-78.
9. Luo, W.; Zhang, J.; Li, M.; Züttel, A. Boosting CO production in electrocatalytic CO2 reduction on highly porous Zn catalysts. ACS. Catal. 2019, 9, 3783-91.
10. Rosser, T. E.; Windle, C. D.; Reisner, E. Electrocatalytic and solar-driven CO2 reduction to CO with a molecular manganese catalyst immobilized on mesoporous TiO2. Angew. Chem. Int. Ed. Engl. 2016, 55, 7388-92.
11. Guo, P.; He, Z.; Cao, H.; et al. Efficient synthesis of syngas from CO2 electrochemical reduction over a dual functional FexC@CNT/N-MXene catalyst. Appl. Catal. B. Environ. Energy. 2024, 347, 123786.
12. Jiang, J.; Xi, C.; Zhou, S.; Chen, X.; Wei, Y.; Han, S. Fe7S8 coupled with VS4 heterogeneous interface engineering driven by FeV bimetallic MOFs: an efficient all-pH and durable hydrogen evolution. J. Colloid. Interface. Sci. 2024, 674, 913-24.
13. Pan, Y.; Abazari, R.; Tahir, B.; et al. Iron-based metal–organic frameworks and their derived materials for photocatalytic and photoelectrocatalytic reactions. Coord. Chem. Rev. 2024, 499, 215538.
14. Han, P.; Yu, X.; Yuan, D.; et al. Defective graphene for electrocatalytic CO2 reduction. J. Colloid. Interface. Sci. 2019, 534, 332-7.
15. Han, C.; Chen, Z. Study on the synergism of thermal transport and electrochemical of PEMFC based on N, P co-doped graphene substrate electrode. Energy 2021, 214, 118808.
16. Lai, Y.; Chen, W.; Zhang, Z.; Qu, Y.; Gan, Y.; Li, J. Fe/Fe3C decorated 3-D porous nitrogen-doped graphene as a cathode material for rechargeable Li–O2 batteries. Electrochim. Acta. 2016, 191, 733-42.
17. Gu, H.; Zhong, L.; Shi, G.; et al. Graphdiyne/graphene heterostructure: a universal 2D scaffold anchoring monodispersed transition-metal phthalocyanines for selective and durable CO2 electroreduction. J. Am. Chem. Soc. 2021, 143, 8679-88.
18. Rogers, C.; Perkins, W. S.; Veber, G.; Williams, T. E.; Cloke, R. R.; Fischer, F. R. Synergistic enhancement of electrocatalytic CO2 reduction with gold nanoparticles embedded in functional graphene nanoribbon composite electrodes. J. Am. Chem. Soc. 2017, 139, 4052-61.
19. Zhang, B.; Chen, S.; Wulan, B.; Zhang, J. Surface modification of SnO2 nanosheets via ultrathin N-doped carbon layers for improving CO2 electrocatalytic reduction. Chem. Eng. J. 2021, 421, 130003.
20. Li, M.; Yan, C.; Ramachandran, R.; et al. Non-peripheral octamethyl-substituted cobalt phthalocyanine nanorods supported on N-doped reduced graphene oxide achieve efficient electrocatalytic CO2 reduction to CO. Chem. Eng. J. 2022, 430, 133050.
21. Wang, M.; Zhang, B.; Ding, J.; et al. Three-dimensional nitrogen-doped graphene aerogel-supported MnO nanoparticles as efficient electrocatalysts for CO2 reduction to CO. ACS. Sustain. Chem. Eng. 2020, 8, 4983-94.
22. Yu, X.; Han, P.; Wei, Z.; et al. Boron-doped graphene for electrocatalytic N2 reduction. Joule 2018, 2, 1610-22.
23. Büchele, S.; Martín, A. J.; Mitchell, S.; et al. Structure sensitivity and evolution of nickel-bearing nitrogen-doped carbons in the electrochemical reduction of CO2. ACS. Catal. 2020, 10, 3444-54.
24. Zhou, W.; Shen, H.; Wang, Q.; Onoe, J.; Kawazoe, Y.; Jena, P. N-doped peanut-shaped carbon nanotubes for efficient CO2 electrocatalytic reduction. Carbon 2019, 152, 241-6.
25. Zhang, L.; Geng, B.; Gao, Y.; et al. FeNi alloys encapsulated with N-doped porous carbon nanotubes as highly efficient and durable CO2 reduction electrocatalyst. Chem. Eng. J. 2024, 481, 148086.
26. Subhash Kanase, R.; Mulualem Zewdie, G.; Arunachalam, M.; et al. Surface engineering of ZnO electrocatalyst by N doping towards electrochemical CO2 reduction. J. Energy. Chem. 2024, 88, 71-81.
27. Pan, F.; Li, B.; Deng, W.; et al. Promoting electrocatalytic CO2 reduction on nitrogen-doped carbon with sulfur addition. Appl. Catal. B. Environ. 2019, 252, 240-9.
28. An, C.; Wu, S.; Huang, R.; et al. Cooperated catalytic mechanism of atomically dispersed binary Cu-N4 and Zn-N4 in N-doped carbon materials for promoting electrocatalytic CO2 reduction to CH4. Chem. Eng. J. 2023, 471, 144618.
29. Li, Z.; Yuan, Y.; Qu, G.; et al. Preparation of phosphorus-doped Cu-based catalysts by electrodeposition modulates *CHxO adsorption to facilitate electrocatalytic reduction of CO2 to CH4. Appl. Catal. B. Environ. Energy. 2025, 360, 124525.
30. Zhao, Z.; Zibarev, A. V.; Wang, H.; Hao, J.; Luo, L. Towards efficient CO2RR electrocatalysts: a study of structure and properties of M–N–E active moieties embedded in a biphenylene framework (M = Mn, Fe, Co, Ni, Cu; E = C, B). J. Mater. Chem. A. 2024, 12, 32471-9.
31. Jouny, M.; Lv, J. J.; Cheng, T.; et al. Formation of carbon-nitrogen bonds in carbon monoxide electrolysis. Nat. Chem.2019, 11, 846-51.
32. Jiang, J.; Sun, R.; Huang, X.; et al. In-situ derived Mo-doped NiCoP and MXene to form Mott-Schottky heterojunction with tunable surface electron density to promote overall water splitting. Compos. Part. B. Eng. 2023, 263, 110834.
33. Kresse, G.; Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 1996, 6, 15-50.
34. Perdew, J. P.; Burke, K.; Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 1996, 77, 3865-8.
35. Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B. Condens. Matter. 1994, 50, 17953-79.
36. Grimme, S. Semiempirical GGA-type density functional constructed with a long-range dispersion correction. J. Comput. Chem. 2006, 27, 1787-99.
37. Wang, T.; Wang, L. X.; Wu, D. L.; Xia, W.; Jia, D. Z. Interaction between nitrogen and sulfur in co-doped graphene and synergetic effect in supercapacitor. Sci. Rep. 2015, 5, 9591.
38. Lv, H.; Yao, Y.; Yuan, M.; et al. Functional nanoporous graphene superlattice. Nat. Commun. 2024, 15, 1295.
39. Zhou, W.; Ding, J.; Hua, J.; et al. Layer-polarized ferromagnetism in rhombohedral multilayer graphene. Nat. Commun. 2024, 15, 2597.
40. Wu, Y.; An, C.; Guo, Y.; et al. Highly aligned graphene aerogels for multifunctional composites. Nanomicro. Lett. 2024, 16, 118.
41. Boonchom, B.; Baitahe, R. Synthesis and characterization of nanocrystalline manganese pyrophosphate Mn2P2O7. Mater. Lett. 2009, 63, 2218-20.
42. Fu, X.; Choi, J. Y.; Zamani, P.; et al. Co-N decorated hierarchically porous graphene aerogel for efficient oxygen reduction reaction in acid. ACS. Appl. Mater. Interfaces. 2016, 8, 6488-95.
43. Yang, L.; Wang, D.; Lv, Y.; Cao, D. Nitrogen-doped graphitic carbons with encapsulated CoNi bimetallic nanoparticles as bifunctional electrocatalysts for rechargeable Zn–Air batteries. Carbon 2019, 144, 8-14.
44. Ren, J.; Ying, Y.; Liu, Y.; Li, W.; Yuan, Z. Charge redistribution caused by sulfur doping of bimetal FeCo phosphides supported on heteroatoms-doped graphene for Zn-air batteries with stable cycling. J. Energy. Chem. 2022, 71, 619-30.
45. Candotto Carniel, F.; Fortuna, L.; Zanelli, D.; et al. Graphene environmental biodegradation: wood degrading and saprotrophic fungi oxidize few-layer graphene. J. Hazard. Mater. 2021, 414, 125553.
46. Cui, X.; Pan, Z.; Zhang, L.; Peng, H.; Zheng, G. Selective etching of nitrogen-doped carbon by steam for enhanced electrochemical CO2 reduction. Adv. Energy. Mater. 2017, 7, 1701456.
47. Chen, C.; Sun, X.; Yan, X.; et al. Boosting CO2 electroreduction on N,P-Co-doped carbon aerogels. Angew. Chem. Int. Ed. Engl. 2020, 59, 11123-9.
48. Tang, Z.; Tao, Y.; Wang, K.; et al. Lattice Mn2+ doped CdSe/CdS quantum dots for high-performance photoelectrochemical hydrogen evolution. Nano. Energy. 2023, 113, 108533.
49. Yang, Y.; Tang, Y.; Fang, G.; et al. Li+ intercalated V2O5·nH2O with enlarged layer spacing and fast ion diffusion as an aqueous zinc-ion battery cathode. Energy. Environ. Sci. 2018, 11, 3157-62.
50. He, Z.; Li, C.; Yang, S.; et al. Electrocatalytic CO2 reduction to ethylene over CuOx boosting CO2 adsorption by lanthanide neodymium. Catal. Sci. Technol. 2023, 13, 6675-84.
51. Yoo, J. M.; Ingenmey, J.; Salanne, M.; Lukatskaya, M. R. Anion effect in electrochemical CO2 reduction: from spectators to orchestrators. J. Am. Chem. Soc. 2024, 146, 31768-77.
52. Gao, X.; Chen, J.; Che, H.; Ao, Y.; Wang, P. Rationally constructing of a novel composite photocatalyst with multi charge transfer channels for highly efficient sulfamethoxazole elimination: mechanism, degradation pathway and DFT calculation. Chem. Eng. J. 2021, 426, 131585.
53. Yang, H. B.; Hung, S.; Liu, S.; et al. Atomically dispersed Ni(I) as the active site for electrochemical CO2 reduction. Nat. Energy. 2018, 3, 140-7.
54. Li, C. W.; Ciston, J.; Kanan, M. W. Electroreduction of carbon monoxide to liquid fuel on oxide-derived nanocrystalline copper. Nature 2014, 508, 504-7.
55. Guo, P. P.; He, Z. H.; Yang, S. Y.; et al. Electrocatalytic CO2 reduction to ethylene over ZrO2/Cu-Cu2O catalysts in aqueous electrolytes. Green. Chem. 2022, 24, 1527-33.
56. Li, X.; Bi, W.; Chen, M.; et al. Exclusive Ni-N4 sites realize near-unity CO selectivity for electrochemical CO2 reduction. J. Am. Chem. Soc. 2017, 139, 14889-92.
57. Zhang, Z.; Ma, C.; Tu, Y.; et al. Multiscale carbon foam confining single iron atoms for efficient electrocatalytic CO2 reduction to CO. Nano. Res. 2019, 12, 2313-7.
58. Shen, Y.; Liu, T.; Li, R.; et al. In situ electrochemical reconstruction of Sr2Fe1.45Ir0.05Mo0.5O6-δ perovskite cathode for CO2 electrolysis in solid oxide electrolysis cells. Natl. Sci. Rev. 2023, 10, nwad078.
59. Yi, J.; Xie, R.; Xie, Z.; et al. Highly selective CO2 electroreduction to CH4 by in situ generated Cu2O single-type sites on a conductive MOF: stabilizing key intermediates with hydrogen bonding. Angew. Chem. Int. Ed. 2020, 132, 23849-56.
60. Hao, Y.; Hu, F.; Zhu, S.; et al. MXene-regulated metal-oxide interfaces with modified intermediate configurations realizing nearly 100% CO2 electrocatalytic conversion. Angew. Chem. Int. Ed. Engl. 2023, 62, e202304179.
61. Kang, M. P. L.; Kolb, M. J.; Calle-Vallejo, F.; Yeo, B. S. The role of undercoordinated sites on zinc electrodes for CO2 reduction to CO. Adv. Funct. Mater. 2022, 32, 2111597.
62. Cao, H.; He, Z.; Tian, Y.; et al. Highly selective electrocatalytic reduction of CO2 to ethane over a petal-like Zn(OH)2/Cu2+1O/Cu foam catalyst at low overpotentials. J. Mater. Chem. A. 2024, 12, 13510-9.
63. Han, Y.; An, S.; Zhan, X.; et al. Electrocatalytic reduction of CO2 to CO with almost 100% Faradaic efficiency using oxygen-vacancy enriched two-dimensional MgO. CCS. Chem. 2024, 6, 1477-86.
64. Li, X.; Chen, Y.; Zhan, X.; et al. Strategies for enhancing electrochemical CO2 reduction to multi-carbon fuels on copper. Innov. Mater. 2023, 1, 100014.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data

















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.