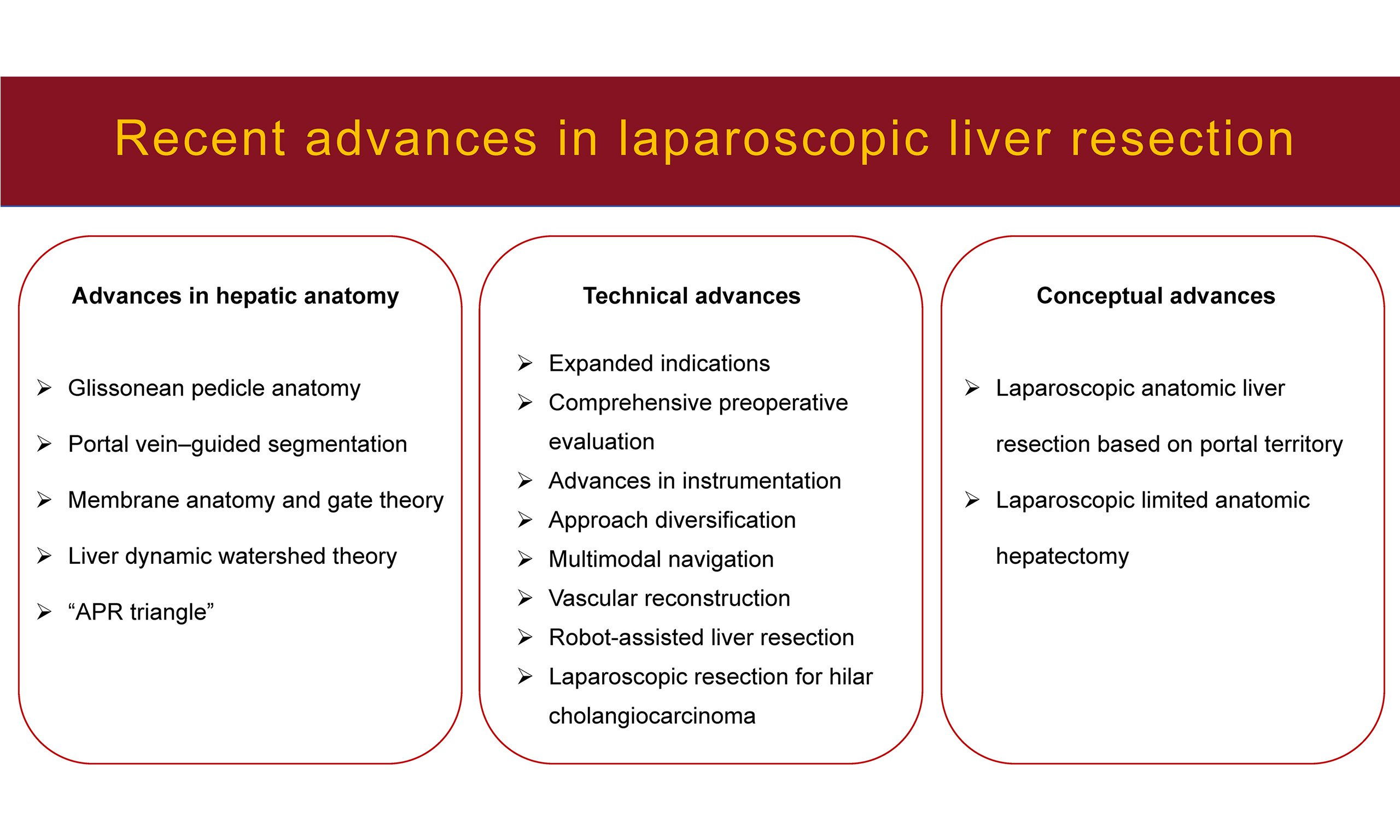
Recent advances in laparoscopic liver resection
Abstract
Since the first laparoscopic liver resection (LLR) was reported in 1991, both surgical techniques and conceptual frameworks have developed rapidly worldwide. Progress in hepatic anatomy - from the classical Couinaud segmentation to refined subsegmental and dynamic portal territory systems - has provided a stronger foundation for precision liver surgery. The “gate theory” and its application in LLR have further enhanced the safety and reproducibility of anatomic dissections. Technically, indications for LLR continue to expand, preoperative evaluation has become more precise, and there has been rapid development in multimodal intraoperative navigation, vascular reconstruction techniques, and robot-assisted surgery. Conceptually, emerging strategies such as anatomic resection based on portal territory and limited anatomic resection are becoming key directions for precision liver surgery. Overall, LLR is advancing toward standardization, precision, and individualization, and is increasingly recognized as a mainstream approach for hepatic resection.
Keywords
INTRODUCTION
Since the first report of laparoscopic liver resection (LLR) in 1991, this technique has gained widespread attention and adoption worldwide. Advances in hepatic anatomy, surgical instrumentation, and minimally invasive techniques have substantially expanded the indications for LLR. This article reviews the recent advances in LLR, focusing on anatomical refinement, technical evolution, and emerging surgical concepts.
ADVANCES IN HEPATIC ANATOMY
Refined anatomical understanding constitutes the theoretical foundation of precision liver surgery. Takasaki et al. established the theoretical and technical foundation of anatomic liver resection through the Glissonean pedicle transection method, with “pedicle anatomy” as its central principle[1,2]. He emphasized that the Glissonean pedicles enter the liver as a unified structure wrapped by Glisson’s capsule, whereas anatomical variations of the hepatic artery and bile duct mainly occur under the hilar plate, rendering dissection in this area unsafe[1,2]. In contrast, transection of the Glissonean pedicles above the hilar plate allows reliable control of segmental or sectional inflow without compromising the remnant liver[1,2].
Building on this Glissonean pedicle approach framework, Cho et al. further advanced hepatic anatomy by redefining liver segmentation according to portal inflow territories in conjunction with hepatic venous drainage patterns[3,4]. In particular, they provided a detailed yet simplified analysis of the right Glissonean pedicle and systematically clarified the venous outflow anatomy of the liver, thereby facilitating a more intuitive and practical anatomical understanding for LLR[3,4].
The membrane anatomy concept represents a further refinement of this pedicle-oriented strategy. Sugioka et al. introduced four anatomical landmarks and the “gate theory” for the extrahepatic Glissonean approach based on Laennec’s capsule[5]. The key significance of the “gate theory” lies in its role as a continuation and standardization of extrahepatic Glissonean pedicle control, providing clear anatomical entry points for safe and reproducible dissection[5]. Under magnified laparoscopic visualization, the potential space between Laennec’s capsule, the portal plate system, and the Glissonean sheaths can be accurately identified and safely dissected, enabling extrahepatic pedicle control without compromising the hepatic parenchyma. A multicenter cohort study involving our center demonstrated that the Laennec approach offers significant intraoperative advantages including shorter operative times for hepatic pedicle isolation, liver mobilization, hepatic vein exposure, and parenchymal transection over conventional techniques in both laparoscopic and robotic procedures, supporting its adoption as a standardized strategy for minimally invasive anatomic liver resection[6].
In addition, Liu et al. proposed the liver dynamic watershed theory, which redefines hepatic vascular distribution and blood flow by transforming the conventional static, trunk-based description into a dynamic, watershed-based paradigm that better reflects functional perfusion patterns[7]. Nevertheless, several critical issues related to this novel theory remain to be fully elucidated, including the mechanisms governing dynamic blood flow redistribution between hepatic watersheds, the objective criteria for assessing blood flow compensation in corresponding watersheds, and the optimal strategies for applying this theory to further enhance the precision of liver resection.
Chen et al. identified the “APR triangle”, a relatively avascular zone observed from the caudal laparoscopic view defined by the right anterior hepatic pedicle (AP), the right posterior hepatic pedicle (PP), and the right hepatic vein (RHV)[8]. This anatomical window offers a safe and efficient pathway for Glissonean pedicle dissection during laparoscopic right liver surgery[8]. While the APR triangle represents a valuable anatomical landmark for simplifying and securing laparoscopic right hepatic pedicle dissection, current evidence remains preliminary and largely descriptive, thus requiring large‑scale, long‑term clinical studies to validate its generalizability in patients. Collectively, these advances in hepatic anatomy underpin the ongoing transition of LLR toward safer, more precise, and more standardized practice.
TECHNICAL ADVANCES IN LLR
Expansion of LLR indications
With the development of precision and minimally invasive liver surgery, along with continuous improvements in surgical instruments and operative technologies, the indications for LLR have progressively expanded. LLR is now increasingly applied to major hepatectomies involving technically demanding segments, as well as to complex procedures such as laparoscopic associating liver partition and portal vein ligation for staged hepatectomy (ALPPS) and laparoscopic living-donor hepatectomy, where it demonstrates pronounced minimally invasive advantages. Nevertheless, these expanded indications should be restricted to high-volume centers with substantial laparoscopic experience.
More comprehensive preoperative evaluation
Beyond conventional ultrasonography and computed tomography (CT), high-resolution CT–based three-dimensional visualization enables precise delineation of third-order and higher-order portal vein branches. This facilitates accurate mapping of tumor-bearing portal territories, identification of critical structures requiring preservation or transection, and assurance of adequate future liver remnant (FLR). Although the indocyanine green (ICG) retention rate at 15 min reflects global hepatic functional reserve, it does not offer a segment-specific functional assessment. Wang et al. reported that the uptake rate of hepatocyte-specific magnetic resonance imaging (MRI) contrast agents such as gadolinium ethoxybenzyl diethylenetriamine pentaacetic acid (Gd-EOB-DTPA) can be used to evaluate segmental liver function[9]. When combined with volumetric assessment, this method allows preoperative prediction of functional FLR and the risk of postoperative liver failure[9].
Advances in instruments and equipment
With continuous technological progress, the range of devices available for parenchymal transection and hemostasis in LLR has expanded considerably. In addition to conventional energy devices such as electrocautery and ultrasonic scalpel, specialized instruments such as the Cavitron Ultrasonic Surgical Aspirator (CUSA) and LigaSure Vessel Sealing System have become widely utilized. For hemostasis, in addition to monopolar and bipolar electrocautery, BiClamp and Aquamantys® system can also be employed. Sui et al. reported that the Aquamantys® system significantly reduced intraoperative blood loss (P = 0.005) and transfusion rates (P = 0.036), and provided improved local recurrence–free survival compared with conventional techniques (P = 0.011)[10]. Surgeons should select transection and hemostatic tools based on device availability, institutional experience, and patient-specific factors such as the degree of cirrhosis. Recently, several manufacturers have introduced integrated 4K/3D/fluorescence laparoscopic imaging systems. These systems provide the high resolution and color fidelity of 4K imaging, the enhanced depth perception of 3D visualization, and real-time ICG fluorescence navigation. Their adoption is expected to further enhance the precision, safety, and overall efficiency of LLR.
Diversification of surgical approaches
Compared with open liver resection, LLR offers greater flexibility in approaches, allowing surgeons to operate via ventral, dorsal, cranial, or caudal routes. In addition to these classifications, surgical approaches can also be categorized into parenchyma-first and Glissonean approaches, depending on whether key structures such as the Glissonean pedicles are dissected before parenchymal transection. Surgeons may select the optimal approach based on the operative requirements, individual preference, and technical expertise, ensuring efficient and safe access to target anatomical structures. Importantly, laparoscopic in situ liver resection by anterior approach avoids excessive mobilization and rotation of the liver, thereby reducing the risk of tumor cell dissemination and aligning with the “no-touch” principle in oncologic surgery[11]. In addition to the conventional transperitoneal approach, our group has innovatively explored retroperitoneal access and single-port transthoracic approaches for minimally invasive liver resection[12,13]. These approaches provide safe and effective alternatives for lesions located in the right posterior sector or diaphragmatic dome, particularly in patients with anticipated severe intra-abdominal adhesions.
Multimodal intraoperative navigation
Because laparoscopy lacks direct tactile feedback, intraoperative ultrasonography is indispensable for comprehensive hepatic assessment, lesion localization, and real-time adjustment of the transection plane. Moreover, intraoperative ICG fluorescence imaging facilitates tumor identification, detection of small or occult lesions, guidance of the transection plane, and evaluation for postoperative bile leakage[14]. Furthermore, based on preoperative three-dimensional visualization and portal territory analysis, augmented-reality (AR) navigation allows superimposition of three-dimensional anatomical models onto the laparoscopic image, displaying transparent virtual representations of lesions, inflow/outflow territories, and critical vascular structures. Oh et al. demonstrated that the AR system achieved a mean registration time of 2.4 ± 1.7 min and an overall average accuracy of 93.8% ± 4.9%[15]. Current evidence also indicates that AR can enhance the efficiency and safety of hepatic resections, and may also contribute to improved oncological outcomes and reduced surgical risk[16]. However, soft-tissue mobility and deformation of the liver remain major challenges for maintaining accurate registration[17,18]. To address these challenges, Une et al. have developed deep learning–based models capable of rapidly detecting hepatic vascular structures, thereby enabling high-precision, real-time navigation during LLR[19]. Collectively, while intraoperative ultrasonography, three-dimensional visualization, and ICG fluorescence have become indispensable components of contemporary LLR, AR- and artificial intelligence (AI)-assisted navigation remain promising but technically immature, with unresolved challenges related to registration accuracy and liver deformation.
Laparoscopic vascular reconstruction
With the expanding indications for LLR, vascular reconstruction has been increasingly applied in minimally invasive liver surgery. Vascular reconstruction should be considered when unexpected intraoperative vascular injuries cannot be controlled by coagulation, when tumors involve major vascular structures, or when venous outflow reconstruction is required[20,21]. Primary repair or end-to-end anastomosis should be the first choice. When direct reconstruction is not feasible, patch angioplasty or the use of artificial grafts may be necessary. From a practical standpoint, these procedures should be attempted only by surgeons with advanced laparoscopic expertise and adequate experience in open vascular reconstruction. Importantly, the selection of the reconstruction strategy should be individualized according to patient-specific conditions. Given the superior depth perception and spatial visualization afforded by stereoscopic imaging, three-dimensional laparoscopy is considered a preferable platform for complex intracorporeal suturing and vascular reconstruction.
Robot-assisted liver resection
Since the first robot-assisted liver resection (RLR) was reported in 2003, robotic surgical systems have gained increasing adoption owing to their articulated and stable robotic arms, natural three-dimensional visualization, and the advantage of a steady third arm that enhances traction and exposure. Collectively, these features enhance operative stability, precision, and safety during liver resection. Multiple studies have demonstrated that robot-assisted liver resection (RLR) achieves comparable outcomes to conventional LLR, with potential advantages including reduced intraoperative blood loss and lower conversion rates to open surgery[22-24]. However, a balanced comparison must also acknowledge the well-established advantages of LLR. As a mature minimally invasive technique, LLR offers lower overall medical costs, broader accessibility across general medical centers, and a well-established global training and dissemination framework. In contrast, RLR remains associated with substantially higher costs, and high-quality randomized controlled trials with long-term oncological outcomes are still lacking. Consequently, the widespread adoption of RLR remains limited. Nevertheless, should these limitations be adequately addressed, RLR holds considerable promise for future development. In parallel with efforts to generate robust clinical evidence and reduce costs, technological innovations are also being introduced to overcome intrinsic limitations of robotic systems. To address the inherent lack of haptic feedback in robotic systems, Xue et al. recently developed an intelligent suture termed “sliputure”, in which the sliding knot releases automatically once a preset tension threshold is reached, thereby offering a surrogate form of tactile feedback[25].
Laparoscopic radical resection for hilar cholangiocarcinoma
In recent years, advances in laparoscopic techniques and instruments have enabled the gradual implementation of laparoscopic radical resection for hilar cholangiocarcinoma (HCCA) in selected high-volume centers[26]. With continued investigation and accumulated operative experience, the safety and feasibility of this approach have been further validated; however, high-quality evidence regarding its long-term oncological outcomes remains lacking. In clinical practice, careful attention must be paid to patient selection, including precise preoperative assessment of the relationship between the tumor, biliary confluence, and major vascular structures. Surgeons should progress in a stepwise manner from less complex to more challenging cases while strictly adhering to the oncologic principles and technical standards established for open surgery. Real-world studies have demonstrated that laparoscopic surgery achieves both short-term outcomes and long-term survival comparable to those of open surgery, and may be applied across all Bismuth–Corlette classifications of HCCA[27]. We believe that laparoscopic radical resection for HCCA will become more precise and truly minimally invasive. This will be driven by the increasing dissemination of laparoscopic and robot-assisted surgery, the integration of multimodal intraoperative navigation, improved capabilities in vascular resection and reconstruction, and ongoing advances in systemic therapy. Meanwhile, as surgical volume accumulates and technical refinement continues, the long-term oncological outcomes of this approach still require validation through large-scale, multicenter comparative studies.
Future progress is likely to be characterized by three key directions: (1) function-guided hepatectomy based on quantitative assessment of segmental liver function rather than solely anatomical demarcation; (2) real-time adaptive navigation integrating AI, deformable organ modeling, and automated vascular recognition; and (3) increased procedural standardization supported by structured training pathways, and objective benchmarking of outcomes. Collectively, the convergence of high-resolution imaging, computational modeling, and intelligent surgical systems may support a progressive transition toward more data-informed and technology-assisted liver resection strategies, with the potential to improve oncological precision while preserving functional liver parenchyma.
CONCEPTUAL ADVANCES IN LLR
Laparoscopic anatomic liver resection based on portal territory
Compared with classical anatomic liver resection, laparoscopic anatomic liver resection based on portal territory emphasizes resection along the tumor-bearing portal territory rather than traditional segmental boundaries[28]. This approach involves prioritizing dissection at the root of the Glissonean pedicle, using intraoperative ICG fluorescence to delineate the tumor-bearing portal territory, and identifying key interterritorial veins to guide precise parenchymal transection[29]. Emerging evidence suggests that this territory-oriented approach may enhance oncological radicality by ensuring complete clearance of potential portal venous tumor spread while preserving maximal functional liver parenchyma. However, it remains technically demanding, particularly in the laparoscopic setting, where precise hilar dissection and complex vascular exposure require advanced expertise. Standardization of procedural steps, objective assessment of learning curves, and validation through high-quality comparative studies are still needed before widespread implementation can be recommended. Looking forward, the integration of three-dimensional reconstruction, augmented reality-assisted navigation, and fluorescence-guided imaging is expected to further refine portal territory-guided resection. With continued technological evolution and accumulation of robust clinical data, this approach may represent an important step toward precision liver surgery.
Laparoscopic limited anatomic hepatectomy
Ho et al. first introduced the concept of laparoscopic limited anatomic hepatectomy (LLAH) for the treatment of hepatocellular carcinoma[30]. The core principle of LLAH is to preserve maximal functional liver parenchyma while still achieving anatomic resection, thereby improving surgical safety[30]. Theoretically, this approach offers dual advantages in both perioperative outcomes and oncologic control. Berardi et al. reported that with precise preoperative planning and standardized operative techniques, LLAH yields blood loss comparable to that of lobectomy or segmentectomy, without significant differences in R0 resection rates or margin width[31]. Nevertheless, high-quality prospective studies remain necessary to determine its perioperative benefits and long-term oncologic outcomes.
We believe that both surgical concepts warrant further development and thoughtful dissemination, as they represent highly refined techniques that merit inclusion in the surgical armamentarium. Nevertheless, the operative approach for each individual patient should ultimately be guided by surgeon expertise and patient safety. Innovation and technical advancement must not compromise patient benefit. Accordingly, these techniques should be applied judiciously and selectively, grounded in sound clinical judgment rather than an indiscriminate pursuit of technical novelty, and their application should be based on rational appraisal and evidence-informed selection rather than generalized assumptions.
CONCLUSION
Over the past three decades, LLR has undergone a comprehensive evolution in instrumentation, techniques and concepts. Advances in precision hepatic anatomy, optimization of surgical devices and approaches, and the development of multimodal intraoperative navigation and robot-assisted platforms have progressively expanded the indications for LLR. Meanwhile, emerging concepts such as anatomic liver resection based on portal territory and limited anatomic resection have accelerated the transition toward precision liver surgery. Looking ahead, with continued progress in systemic therapies, integration of AI–assisted navigation, and broader adoption of advanced minimally invasive technologies and concepts, LLR is poised to advance further toward greater standardization, precision, and individualization, ultimately becoming a mainstream approach for hepatic resection.
DECLARATIONS
Authors’ contributions
Made substantial contributions to the conception and design of the study: Liu L
Contributing to writing the manuscript: Zhang W
Performed revisions and provided administrative support: Liu L
Served as supervising author: Liu L
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
National Natural Science Foundation of China (Grant No. 82373232), Clinical and Translational Research Project of Anhui Province (202204295107020025).
Conflicts of interest
Liu L serves as the Editor-in-Chief of Mini-invasive Surgery. Liu L was not involved in any part of the editorial processing of this manuscript, including reviewer selection, manuscript handling, or decision-making. Zhang W declares that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Takasaki K, Kobayashi S, Tanaka S, Saito A, Yamamoto M, Hanyu F. Highly anatomically systematized hepatic resection with Glissonean sheath code transection at the hepatic hilus. Int Surg. 1990;75:73-7.
2. Takasaki K. Glissonean pedicle transection method for hepatic resection: a new concept of liver segmentation. J Hepatobiliary Pancreat Surg. 1998;5:286-91.
3. Cho A, Okazumi S, Miyazawa Y, et al. Proposal for a reclassification of liver based anatomy on portal ramifications. Am J Surg. 2005;189:195-9.
4. Cho A, Okazumi S, Makino H, et al. Anterior fissure of the right liver - the third door of the liver. J Hepatobiliary Pancreat Surg. 2004;11:390-6.
5. Sugioka A, Kato Y, Tanahashi Y. Systematic extrahepatic Glissonean pedicle isolation for anatomical liver resection based on Laennec’s capsule: proposal of a novel comprehensive surgical anatomy of the liver. J Hepatobiliary Pancreat Sci. 2017;24:17-23.
6. Li B, Yin D, Zhang Q, et al. Laennec approach for anatomical liver resection assisted by laparoscopy or robotics: a multicenter cohort study. Int J Surg. 2025;111:1929-38.
7. Liu R, Wang Y, Zhang XP. Revisiting human liver anatomy: dynamic watershed theory. Hepatobiliary Surg Nutr. 2021;10:139-41.
8. Chen J, Zhang Z, Zhou R, Wang A, Cao J, Chen Y. The APR triangle: a practical zone in the Glissonean approach to laparoscopic anatomical right hepatectomy. ILIVER. 2022;1:176-80.
9. Wang Y, Zhang L, Ning J, et al. Preoperative remnant liver function evaluation using a routine clinical dynamic Gd-EOB-DTPA-enhanced MRI protocol in patients with hepatocellular carcinoma. Ann Surg Oncol. 2021;28:3672-82.
10. Sui MH, Wang HG, Chen MY, et al. Assessment of the effect of the Aquamantys® system on local recurrence after hepatectomy for hepatocellular carcinoma through propensity score matching. Clin Transl Oncol. 2019;21:1634-43.
11. Cubisino A, Peugeot L, Lesurtel M. Robotic right hepatectomy via an anterior approach: technical considerations for managing large hepatocellular carcinomas. Updates Surg. 2026;78:261-5.
12. Cai W, Wang J, Yin D, et al. Retroperitoneal laparoscopic hepatectomy for a subcapsular hepatocellular carcinoma in segment VI (Video). Ann Surg Oncol. 2023;30:5450-1.
13. Wang J, Zhang W, Mei X, Liu L. Single port video-assisted thoracoscopic transdiaphragmatic hepatectomy for liver tumor (Video). Asian J Surg. 2024;48:1752-3.
14. Zhou J, Tan Z, Sun B, Leng Y, Liu S. Application of indocyanine green fluorescence imaging in hepatobiliary surgery. Int J Surg. 2024;110:7948-61.
15. Oh MY, Yoon KC, Hyeon S, et al. Navigating the future of 3D laparoscopic liver surgeries: visualization of internal anatomy on laparoscopic images with augmented reality. Surg Laparosc Endosc Percutan Tech. 2024;34:459-65.
16. Dai J, Qi W, Qiu Z, Li C. The application and prospection of augmented reality in hepato-pancreato-biliary surgery. Biosci Trends. 2023;17:193-202.
17. Gholizadeh M, Bakhshali MA, Mazlooman SR, et al. Minimally invasive and invasive liver surgery based on augmented reality training: a review of the literature. J Robot Surg. 2023;17:753-63.
18. Chatzikomnitsa P, Gkaitatzi AD, Papakonstantinou M, et al. The role of artificial intelligence and 3D printing in minimally invasive liver surgery. Mini-invasive Surg. 2025;9:8.
19. Une N, Kobayashi S, Kitaguchi D, et al. Intraoperative artificial intelligence system identifying liver vessels in laparoscopic liver resection: a retrospective experimental study. Surg Endosc. 2024;38:1088-95.
20. Chang YJ, Siow TF, Lin TC, Chen KH. Laparoscopic extended left hepatectomy with middle hepatic vein reconstruction for intrahepatic cholangiocarcinoma with a video vignette. Asian J Surg. 2022;45:941-2.
21. Gao J, Zheng J, Zhu Z, et al. Laparoscopic orthotopic right hemihepatectomy by anterior approach combined with inferior vena cava thrombectomy. Ann Surg Oncol. 2022;29:5548-9.
22. D’Silva M, Han HS, Liu R, et al.; International Robotic and Laparoscopic Liver Resection Study Group Investigators. Limited liver resections in the posterosuperior segments: international multicentre propensity score-matched and coarsened exact-matched analysis comparing the laparoscopic and robotic approaches. Br J Surg. 2022;109:1140-9.
23. Sucandy I, Rayman S, Lai EC, et al.; International Robotic, Laparoscopic Liver Resection Study Group Investigators. Robotic versus laparoscopic left and extended left hepatectomy: an international multicenter study propensity score-matched analysis. Ann Surg Oncol. 2022;29:8398-406.
24. Panettieri E, Chirban AM, Hansen B, Vega EA, Conrad C. Robotic vs. laparoscopic resection for hepatocellular carcinoma. Mini-invasive Surg. 2023;7:13.
25. Xue Y, Cao J, Feng T, et al. Slipknot-gauged mechanical transmission and robotic operation. Nature. 2025;647:889-96.
26. Ratti F, Fiorentini G, Cipriani F, Catena M, Paganelli M, Aldrighetti L. Perihilar cholangiocarcinoma: are we ready to step towards minimally invasiveness? Updates Surg. 2020;72:423-33.
27. Qin T, Wang M, Zhang H, et al.; Minimally Invasive Pancreas Treatment Group in the Pancreatic Disease Branch of China’s International Exchange and Promotion Association for Medicine and Healthcare. The long-term outcome of laparoscopic resection for perihilar cholangiocarcinoma compared with the open approach: a real-world multicentric analysis. Ann Surg Oncol. 2023;30:1366-78.
28. Gotohda N, Cherqui D, Geller DA, et al. Expert Consensus Guidelines: how to safely perform minimally invasive anatomic liver resection. J Hepatobiliary Pancreat Sci. 2022;29:16-32.
29. Liang X, Zheng J, Xu J, et al. Laparoscopic anatomical portal territory hepatectomy using Glissonean pedicle approach (Takasaki approach) with indocyanine green fluorescence negative staining: how I do it. HPB. 2021;23:1392-9.
30. Ho CM, Wakabayashi G, Nitta H, et al. Total laparoscopic limited anatomical resection for centrally located hepatocellular carcinoma in cirrhotic liver. Surg Endosc. 2013;27:1820-5.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data
















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.