Endothelial progenitor cells in vascular remodeling
Abstract
The integrity of the vascular endothelium is fundamental to its barrier function, maintaining vascular homeostasis and microenvironmental stability, and serves as a prerequisite for preventing certain vascular diseases. After vascular integrity is compromised, endothelial progenitor cells (EPCs), a diverse population of progenitor cells with the capacity to develop into endothelial cells, can reconstruct blood vessels. This occurs primarily through two mechanisms: (1) direct integration into existing vessels for repair; and (2) paracrine secretion of proangiogenic factors to promote EPC mobilization and migration, regulate immune functions, and inhibit endothelial hyperplasia. This review aims to elucidate the mechanisms by which EPCs participate in vascular remodeling, and to discuss the latest advances in clinical translation strategies such as cell therapy, EPC-derived exosome therapy, and EPC functional modulation, as well as the current challenges in standardization and clinical application.
Keywords
INTRODUCTION
The vascular endothelium, which constitutes the inner lining of blood vessels, is integral to the filtration of bodily fluids and the regulation of blood flow and vascular tone. To maintain cardiovascular homeostasis, the endothelium can undergo dilation or contraction in response to hormonal, metabolic, and other blood-borne signals. It contributes to thrombosis prevention under normal physiological conditions through several mechanisms, including anticoagulation, antiplatelet activity, and fibrinolysis. However, endothelial dysfunction occurs when endothelial tissue is damaged or when there are significant alterations in the vascular microenvironment. This dysfunction is associated with various diseases, such as atherosclerosis, diabetes, hypertension, and renal failure, and serves as a fundamental pathological mechanism underlying these conditions[1]. Consequently, vascular remodeling is necessary for the treatment of such diseases.
Traditional theories propose that damaged endothelial cells (ECs) are replenished by adjacent mature ECs. However, recent research indicates that endothelial progenitor cells (EPCs) can either directly incorporate into existing blood vessels by differentiating into ECs or enhance their mobilization, migration, differentiation, and secretion activities via paracrine factors to facilitate vascular repair. Since the initial discovery and isolation of EPCs by Asahara et al.[2] in 1997, extensive research has been conducted to explore these cells in detail. EPCs are now classified into more precise subtypes, moving beyond the previously vague definitions, and the specific functions of each subtype are becoming increasingly well-defined. For instance, EPCs can be categorized according to their origins into bone marrow-derived EPCs (BM-EPCs), circulating EPCs (cEPCs), and resident EPCs (rEPCs). These classifications are also associated with distinct functional characteristics. Numerous peer-reviewed studies have demonstrated that EPCs possess immunomodulatory, anti-thrombotic, and anti-endothelial hyperplasia properties, in addition to their role in promoting vascular remodeling through integration.
Consequently, the augmentation of the quantity of EPCs and the enhancement of their functional capabilities constitute pressing challenges that demand immediate scholarly and clinical focus. While innovative methodologies - including EPC capture stents, intracardiac administration of CD34+ progenitor cells, and progenitor cell-derived exosomes - have shown encouraging outcomes in preclinical animal models, comprehensive clinical trials are imperative to ascertain their safety and efficacy for therapeutic application in humans.
This review synthesizes current knowledge on EPC biological characteristics, their core mechanisms in vascular remodeling, therapeutic applications in various diseases, and clinical translation strategies. By addressing these topics, we aim to provide insights into optimizing EPC-based therapies and advancing their clinical utility for vascular remodeling-related diseases.
BIOLOGICAL CHARACTERISTICS OF ENDOTHELIAL PROGENITOR CELLS
Discovery and identification of endothelial progenitor cells
In 1997, Asahara et al.[2] identified hematopoietic stem cells in peripheral blood that share markers with vascular progenitor cells, indicating the existence of cells with the potential to differentiate into ECs. They successfully isolated CD34+ and Fetal liver kinase 1 (FLK-1+) EPCs from peripheral blood utilizing magnetic bead sorting techniques. Subsequent studies have also isolated and characterized BM-EPCs[3]; however, there remains a lack of consensus within the academic community regarding the bone marrow origin of these cells[4]. Beyond peripheral blood, EPCs have been identified in umbilical cord blood[5], adipose tissue[6], and the peripheral vascular wall[7,8]. Nevertheless, due to definitional ambiguities, the lack of standardized isolation and cultivation protocols, and the heterogeneity of sources, EPCs isolated in various studies frequently display differing surface markers. As of now, EPCs lack a precise definition. They are generally characterized as circulating cells with the potential to differentiate into ECs, contribute to postnatal angiogenesis, and facilitate endothelial repair at sites of vascular injury[9]. EPCs derived from umbilical cord blood (UCB-EPCs) exhibit superior growth, functionality, and reduced immunogenicity[10] compared to those sourced from peripheral blood or adipose tissue.
The identification of EPCs is based on their surface antigens, with proposed marker combinations[10] including CD34+, CD31+, CD133+, VEGFR2+ (vascular endothelial growth factor receptor 2), CD144+, CD146+, CD45-/+, CD14+, among others. However, a universally accepted molecular signature has yet to be established.
Recent advancements in single-cell RNA sequencing (scRNA-seq) have shown that EPCs uniquely overexpress bone morphogenetic proteins BMP2 and BMP4, as well as ephrinB2, compared with human umbilical vein endothelial cells (HUVECs) and adult ECs from adipose and skin tissues. This distinct transcriptional profile[11] may enhance the isolation and cultivation of EPCs from peripheral blood.
EPC isolation and culture are fundamental to investigating their biological properties
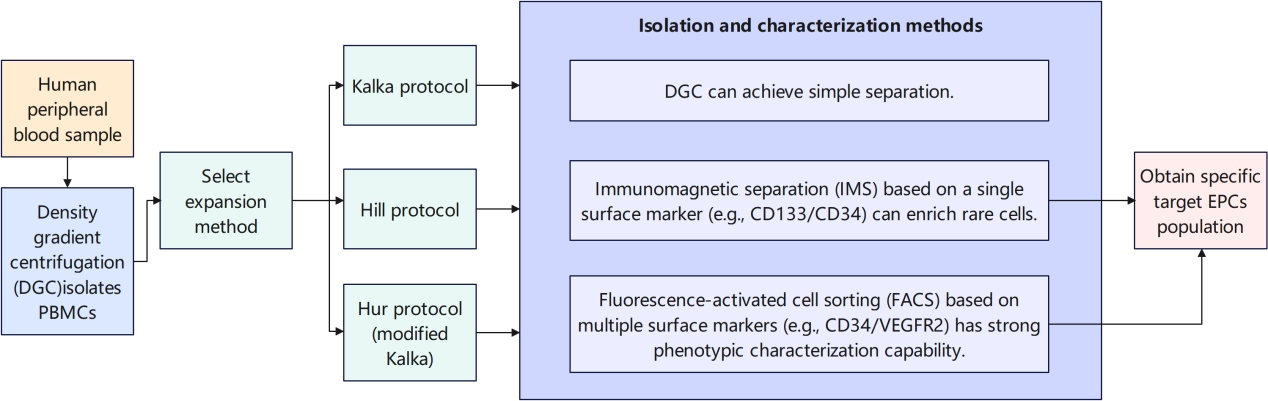
Owing to the limited availability of human EPCs in the bloodstream, researchers have developed in vitro expansion systems. These protocols typically commence with the isolation of peripheral blood mononuclear cells (PBMCs), which serve as the foundation for EPC expansion. PBMCs are predominantly isolated through density gradient centrifugation (DGC), although specific procedural details may vary depending on the tissue source.
Following the isolation of PBMCs, three distinct expansion methodologies are available. One such technique, pioneered by Kalka et al.[12], involves the plating of cells on fibronectin-coated dishes. Non-adherent cells are removed by washing with phosphate-buffered saline (PBS) after four days, followed by periodic medium changes until days seven to ten.
Alternatively, if PBMCs are pre-plated for 48 hours to deplete mature ECs before washing, the non-adherent cells can subsequently be collected and replated with medium changes every three days. Colonies of outgrowth cells and core cell clusters[13], known as Colony-Forming Unit-Hill (CFU-Hill), are then observed after seven days.
The third method, developed by Hur et al.[14], represents a modification of Kalka's protocol. This approach facilitated the identification of two distinct populations of EPCs, classified as “early” and “late” EPCs, a categorization that has been widely adopted in subsequent research. Late EPCs are typically obtained after two weeks of culture, as indicated in Table 1 .
Comparison of three protocols for in vitro expansion of EPCs
| Characteristics | Protocol by Chopra et al.[10] | Protocol by Abdelgawad et al.[11] | Protocol by Kalka et al.[12] |
| Protocol summary | Adherence selection based on culture medium | Sequential adherence selection based on timing | Modification of the Kalka protocol to distinguish different cell populations |
| Initial step | Isolated PBMCs are plated directly | Isolated PBMCs are pre-plated for 48 hours to deplete adherent cells | Isolated PBMCs are plated directly (same as Kalka) |
| Culture medium | EBM-2 + EGM-2 MV SingleQuots (containing 5% FBS, VEGF, FGF-2, EGF, IGF-1, ascorbic acid) | Media 199 + 20% FBS + penicillin+streptomycin | Same as Kalka (EBM-2 + EGM-2 MV SingleQuots) |
| Key step | Removal of non-adherent cells by PBS washing on day 4 | Collection of non-adherent cells from the pre-plating step and re-plating them | Initiating medium changes on day 6 (then every 3 days) |
| Culture days | 7-10 days | 7 days | 2 weeks |
| Final product | Adherent cell population | CFU-Hill colonies (with a central core of rounded cells and surrounding spindle-shaped cells) | Early EPCs Late EPCs |
To further sort and identify EPCs from expanded cell cultures, three primary methods are employed: DGC, immunomagnetic separation (IMS), and fluorescence-activated cell sorting (FACS)[1,12-16]. DGC is a less precise method for isolating mononuclear cells from blood. In contrast, IMS and FACS provide more accurate separation of EPCs based on surface markers. IMS has the advantage of recovering rare cells that may be missed by FACS and allows for morphological analysis, although it targets only one antibody. Each method presents distinct trade-offs, and there is no consensus on the optimal protocol for EPC isolation[17]. Despite the potential of hybrid techniques, the lack of standardized surface markers for EPCs continues to impede clinical translation [Figure 1].
Classification of EPCs
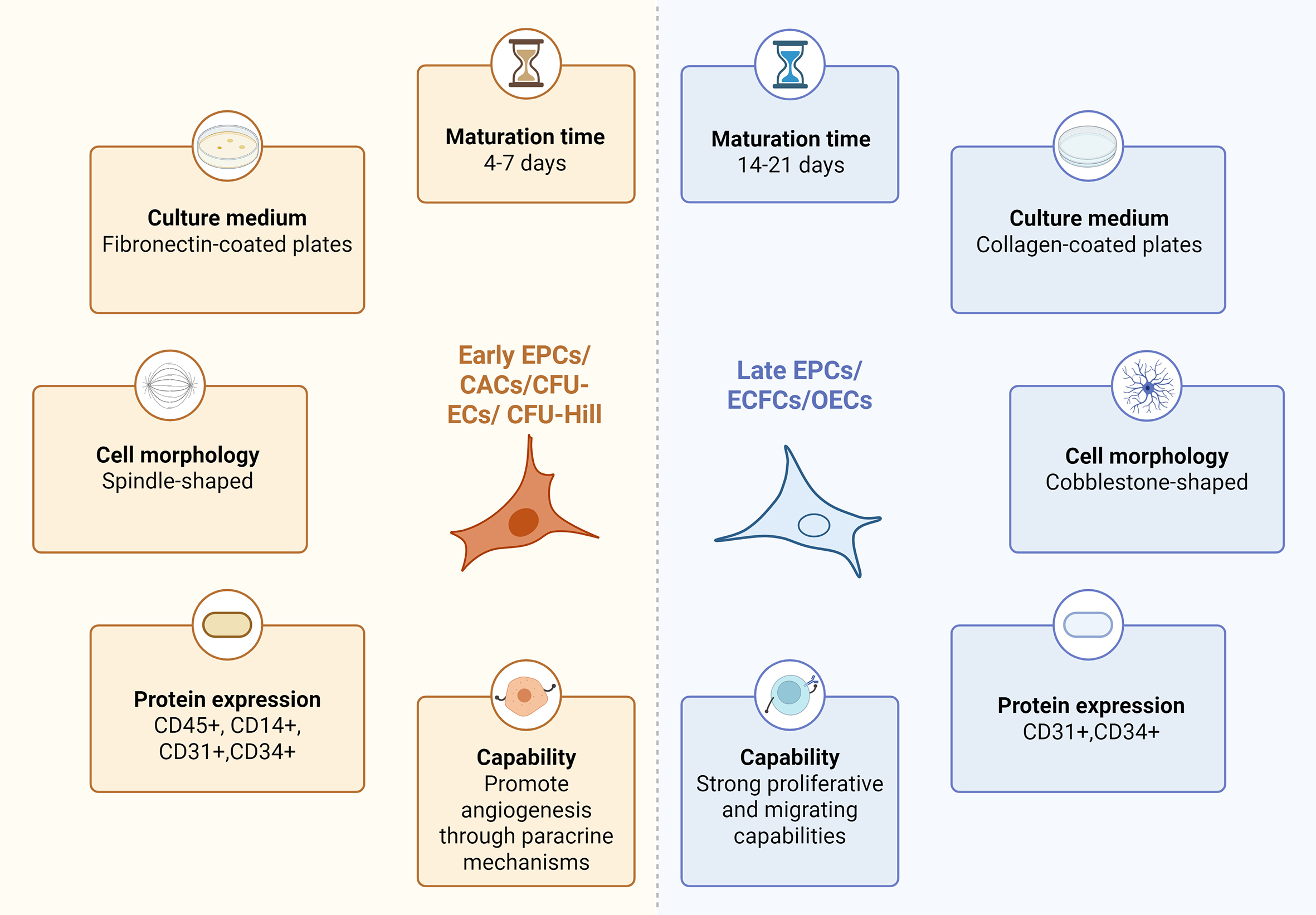
Based on morphological characteristics observed during different culture stages, there are two different subtypes of EPCs in vitro: early EPCs and late EPCs[14]. Early EPCs, also called circulating angiogenic cells (CACs) or colony-forming unit ECs (CFU-ECs)[18], peak in proliferation between weeks 2 and 3 and undergo apoptosis after 4 weeks. While they do not integrate into existing vessels or form colonies in vitro, they enhance angiogenesis through paracrine signaling[14] and recruit mature ECs for tubular formation. Early EPCs are negative for CD146, CD133, and Tie2[19]. In contrast, late EPCs, known as endothelial colony-forming cells (ECFCs)[18], proliferate rapidly between weeks 4 and 8 and are viable for about 12 weeks. ECFCs can locate injured blood vessels and become ECs to support vascular formation[20]. They are negative for VEGFR2, CD45, von Willebrand factor (vWF), vascular endothelial cadherin (VE-cadherin), CD146, and CD14[21]. We have provided a direct comparison of early-stage EPCs and late-stage EPCs in Figure 2.
EPCS ARE INVOLVED IN VASCULAR REMODELING
Classification and functional mechanisms of endothelial progenitor cells from different sources
EPCs may be categorized according to their origin into three distinct types: BM-EPCs, cEPCs, and rEPCs.
BM-EPCs play a pivotal role as a reserve and regulatory center in vascular remodeling, and they represent the primary precursor population for cEPCs. A comparative study examining the ultrastructure of early EPCs sourced from rabbit peripheral blood and bone marrow demonstrated nearly identical ultrastructural features between these two origins[22]. The processes of proliferation, differentiation, and mobilization of BM-EPCs are modulated by various factors. These include vascular endothelial growth factor (VEGF)[23], nitric oxide (NO)[24], the adenosine monophosphate-activated protein kinase (AMPK)/endothelial nitric oxide synthase (eNOS) signaling pathway[25], hypoxia-inducible factor 1 (HIF-1), stromal cell-derived factor 1 alpha (SDF-1α)[26], and the tissue plasminogen activator/matrix metalloproteinase-9 (tPA/MMP-9) axis[27]. In response to injury or ischemic conditions, peripheral tissues secrete VEGF, which activates the PI3K/Akt/eNOS pathway. Simultaneously, elevated NO levels in the bone marrow prompt the migration of EPCs from the bone marrow niche into the circulation, thereby serving as a primary source of cEPCs. These cells subsequently contribute to angiogenesis and wound healing at the tissue level. Under normal physiological conditions, the transforming growth factor-beta (TGF-β) pathway inhibits excessive proliferation of BM-EPCs[28]. However, inadequate activation of this pathway during pathological states, such as metabolic disorders, results in a reduced differentiation capacity of BM-EPCs, thereby exacerbating deficits in vascular repair.
cEPCs predominantly originate from the bone marrow, subsequently entering the bloodstream and migrating chemotactically to sites of tissue injury in response to ischemic or inflammatory signals[29,30]. Through paracrine signaling mechanisms, they secrete factors such as VEGF, stromal cell-derived factor-1 (SDF-1), and interleukin-8 (IL-8), which facilitate EC proliferation, migration, and inhibit apoptosis. A minor subset of these cells may incorporate into the newly formed endothelium.
rEPCs are situated within the vascular wall, including regions such as the adventitia, subendothelium, and perivascular space. In response to vascular injury or hemodynamic changes, these cells become activated and play a direct role in local endothelial repair and angiogenesis[31]. Their involvement in vascular remodeling highlights the importance of spatial specificity and dependence on the microenvironment. Studies suggest that the repair of vascular endothelium following injury is essential for maintaining arterial homeostasis. rEPCs facilitate re-endothelialization by replacing damaged ECs[32], thus inhibiting the formation of a new intima.
In conclusion, EPCs establish a cascade response network transitioning from "bone marrow → circulation → target tissues", collaboratively sustaining vascular homeostasis. In situ EPCs primarily facilitate rapid local repair, cEPCs are responsible for mediating distant repair and paracrine regulation, and bone marrow EPCs function as the principal reserve and regulatory center, playing an essential role in maintaining vascular integrity and responding to injury. Elucidating the mechanisms underlying these distinct EPC subtypes has substantial implications[33] for the development of diagnostic and therapeutic strategies aimed at vascular diseases.
Paracrine effects of EPCs
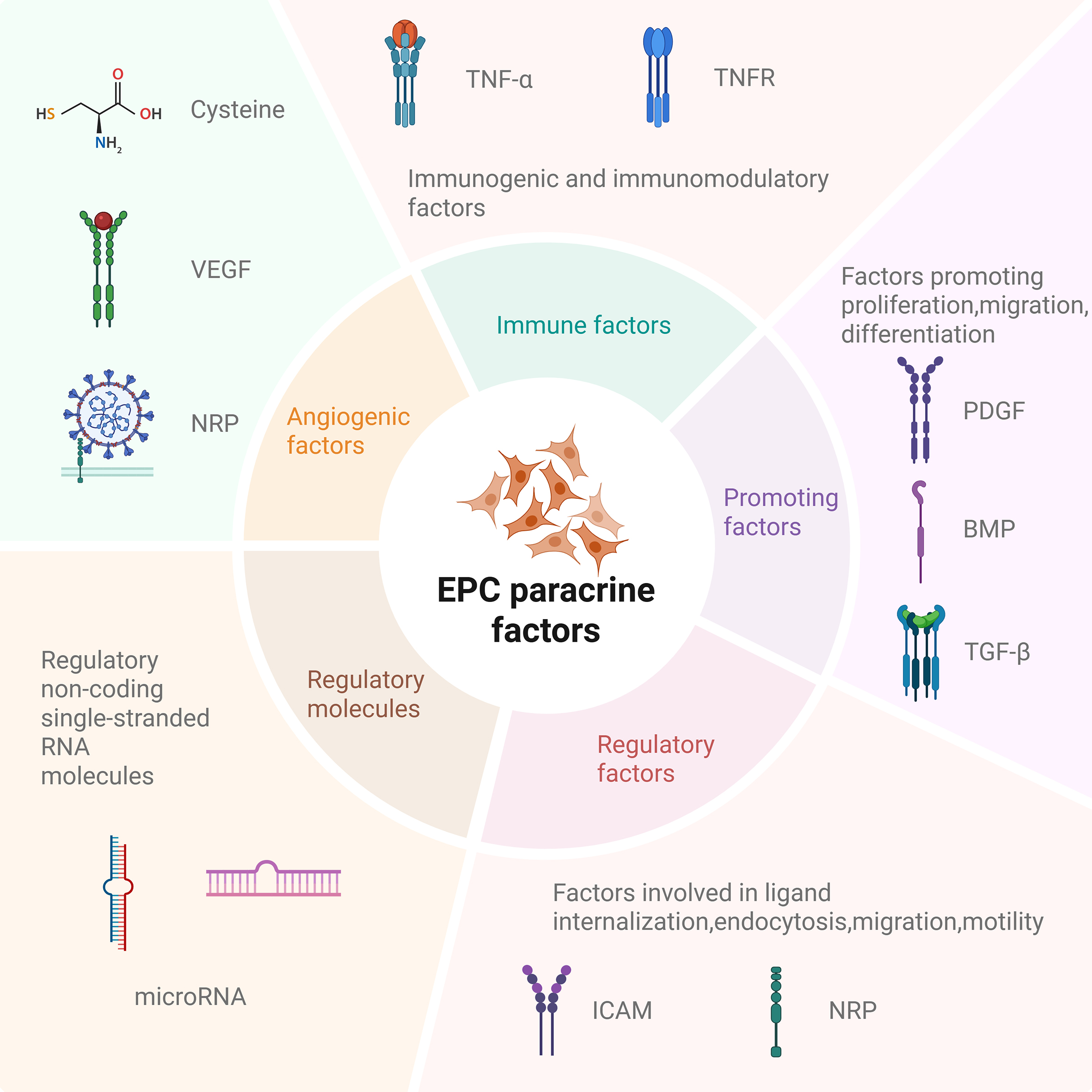
EPCs have the capacity to differentiate into ECs and facilitate vascular repair through the secretion of paracrine factors. The cytokines derived from EPCs can be categorized into five distinct groups, as depicted in Figure 3: (1) Angiogenic factors, which include cysteine and vascular endothelial growth factor receptors (VEGFRs) essential for the formation of blood vessels; (2) Immunogenic factors, which mediate interactions with immune cells, such as tumor necrosis factor-related apoptosis-inducing ligand (TRAIL) and tumor necrosis factor-alpha (TNF-α)[34]; (3) Vascular stem cell factors, comprising platelet-derived growth factor (PDGF), bone morphogenetic protein (BMP), and TGF-β, which are crucial for cell growth and migration[35]; (4) Regulatory RNAs, particularly microRNAs, which modulate EPC functions[36]; and (5) Motility factors, including urokinase-type plasminogen activator (uPA), its receptor (uPAR), and tissue plasminogen activator (tPA), which are involved in cell movement. EPCs employ these factors to facilitate both differentiation and paracrine signaling during the process of vascular repair.
Figure 3. The classification of EPC paracrine factors. TNF-α: Tumor necrosis factor-alpha; TNFR: tumor necrosis factor receptor; VEGF: vascular endothelial growth factor; NRP: neuropilin; PDGF: platelet-derived growth factor; BMP: bone morphogenetic protein; TGF-β: transforming growth factor-beta; ICAM: intercellular adhesion molecule; EPCs: Endothelial progenitor cells.
The role of EPCs in vascular remodeling in cardiovascular diseases
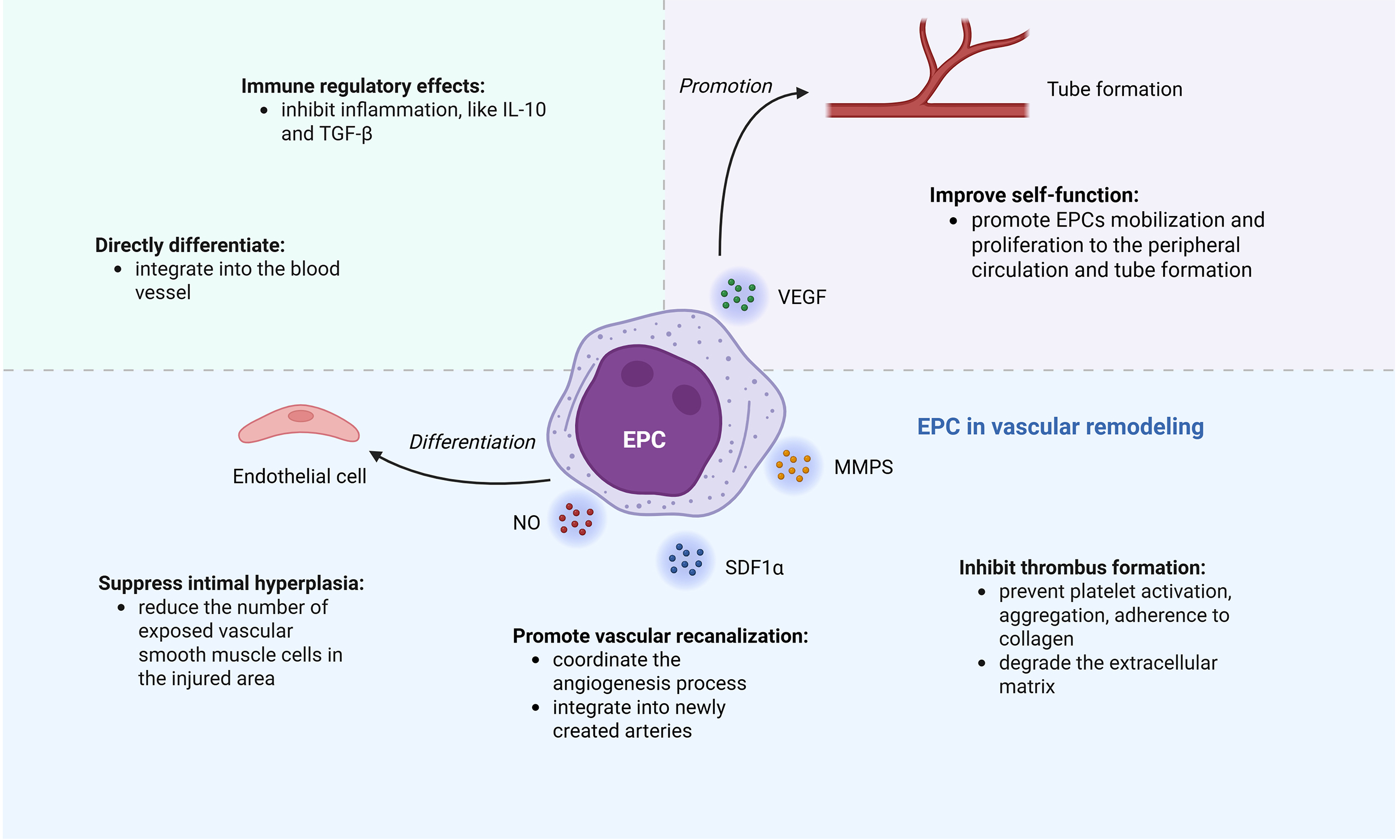
Cardiovascular disease (CVD) is increasingly prevalent as the global population ages and experiences heightened stress levels[37-39]. Atherosclerosis (AS), a chronic inflammatory condition that commences in childhood[40], is characterized by the accumulation of fibrous tissue and lipids within arterial walls. Upon endothelial injury, EPCs are mobilized to facilitate vascular repair and maintain vascular health through cytokine production [Figure 4].
Figure 4. The mechanism of EPCs’ involvement in vascular remodeling. EPCs can directly differentiate into endothelial cells and also secrete cytokines such as NO and VEGF to promote vascular remodeling. VEGF: Vascular endothelial growth factor; NO: nitric oxide; EPCs: endothelial progenitor cells.
A substantial body of research indicates that EPC activity is associated with endothelial damage and various cardiovascular risk factors[13,41,42] [Table 2]. Reduced levels of EPCs are correlated with coronary artery disease, heart failure, and risk factors such as hypertension, diabetes, and smoking. Enhancing the quantity or functionality of EPCs is essential in the management of cardiovascular disease, as alterations in EPC levels may signal underlying health issues.
The influence of various risk factors, including gender, hyperlipemia, smoke, diabetes, hypertension, hyperhomocysteinemia, etc., on EPCs
| Author | Experimental subject | Risk factor | The impact on EPCs |
| Zhuo et al.[43] | A total of 10 randomized, age-matched wild-type (WT) male and female C57BL/6 mice | Gender; Hyperlipemia | Blood EPCs values are high in female hyperlipidemic mice; ROS levels in blood EPCs from male hyperlipidemic mice were significantly elevated |
| He et al.[44] | healthy 4-6 weeks old male C57BL/6J mice | Smoke/Cigarette smoke extract (CSE) | EPCs showed significantly reduced proliferation, adhesion, and secretion capabilities, as well as decreased eNOS protein and mRNA expression levels |
| Eleftheriadou et al.[45] | 50 patients with type 2 diabetes (T2DM) and DPN, 30 patients with T2DM without DPN and 25 healthy individuals | Diabetic complication: diabetic peripheral neuropathy (DPN) | DPN patients have lower SDF-1 levels; DPN may be related to transport damage of EPCs and homing of damaged EPCs to injured endothelium |
| Hill et al.[13] | 45 men who were older than 21 years of age (mean [±SE] age, 50 ± 2), some of whom had conventional cardiovascular risk factors and some of whom did not | High cholesterol; Hypertension; Diabetes | Subjects with elevated serum cholesterol levels, hypertension, and diabetes had significantly reduced numbers of EPCs colony-forming units |
| Nelson et al.[46] | Cystathionine-β synthase (Cbs) mice | Hyperhomocysteinemia (HHcy) | HHcy impairs reendothelialization and inhibits EPCs mobilization, adhesion, migration, and proliferation, as well as β1-integrin expression/activity in EPCs |
EPCs can repair damaged endothelium
Vascular endothelial damage is a critical factor in the pathogenesis of cardiovascular disorders. Traditionally, it was posited that oxidative stress inflicted damage on ECs, necessitating repair through the proliferation of adjacent cells. However, it is now understood that mature ECs possess limited proliferative capacity. Recent investigations have demonstrated that cEPCs play a significant role in endothelial repair. Initially,
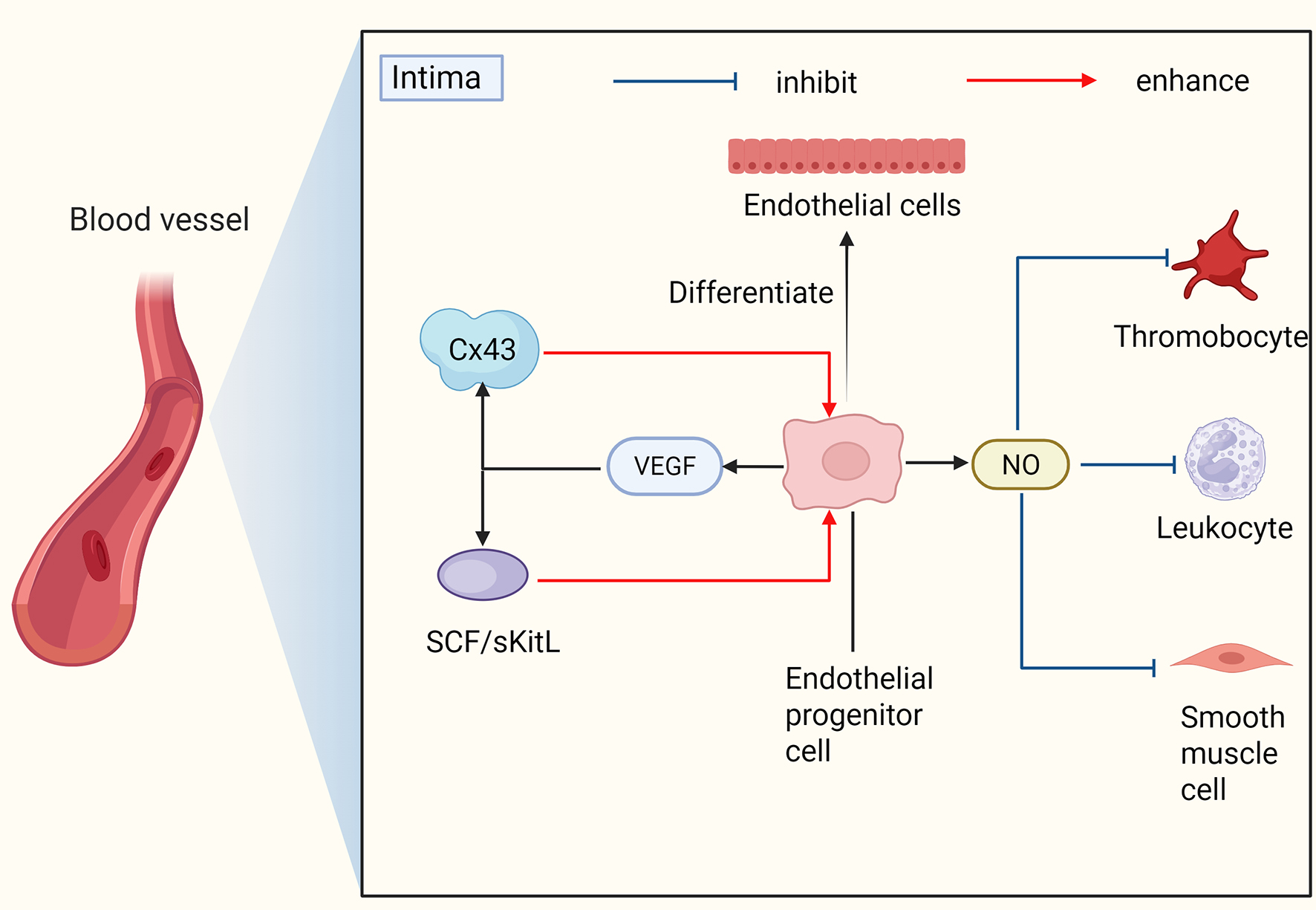
VEGF plays a crucial role in the mobilization, differentiation, and vascular repair of EPCs[49]. It facilitates the activation of MMP-9 within the bone marrow, leading to the release of stem cell factors such as stem cell factor (SCF) and soluble Kit ligand (sKitL)[50]. This release significantly enhances the mobilization and proliferation of EPCs into the circulatory system. Furthermore, VEGF upregulates connexin 43 (Cx43)[51], thereby promoting EPC differentiation and inhibiting neointimal hyperplasia in injured vessels. Both in vitro and in vivo studies have demonstrated that transplantation of VEGF-pretreated EPCs enhances vascular healing[52] and fosters angiogenesis and endothelial repair. Additionally, stromal cell-derived factor 1-alpha (SDF-1α) potentiates the effects of VEGF by activating the SDF-1/CXCR4 (C-X-C chemokine receptor type 4) signaling axis[53,54], which increases VEGF levels and facilitates the maturation of EPCs into ECs at sites of neovascularization.
NO is involved in a multitude of physiological processes. Specifically, in the context of angiogenesis, NO facilitates the mobilization and proliferation of EPCs into the peripheral circulation[55]. Through the regulation of MMP-9, NO induces the mobilization of both hematopoietic stem cells and EPCs. Under hypoxic conditions, NO enhances the expression of integrin α5 and promotes cell-matrix adhesion[56] in EPCs, while simultaneously restoring mitochondrial function and energy homeostasis.
EPCs inhibit thrombus formation and promote vascular recanalization
Atherosclerosis contributes to thrombosis through endothelial damage, which exposes collagen and facilitates platelet adhesion and aggregation. This condition is further aggravated by the rupture of lipid-rich plaques and restenosis after stent placement[57]. Oxidized lipoproteins, including oxidized low-density lipoprotein, exacerbate thrombosis by inducing vascular inflammation[58,59]. EPCs play a crucial role in preventing thrombus formation and promoting vascular recanalization by integrating into the vascular wall, secreting growth factors, and producing exosomes from progenitor cells.
Different sources of EPCs contribute to thrombus recanalization in different ways. When a thrombus forms, cEPCs[60] preferentially home to ischemic areas due to chemokines such as matrix cell-derived factor 1α and monocyte chemotactic protein-1 (MCP-1)[61]. They can reinforce the endothelium layer of both newly formed and existing arteries and integrate into the injured endothelial monolayer to repair damaged vessels. PBMC-derived EPCs[62] interact with platelets via cluster of differentiation 62P (CD62P) to upregulate cyclooxygenase-2 and release prostacyclin, which may prevent platelet activation, aggregation, adherence to collagen, and thrombus formation in vitro. Progenitor cells derived from PBMC that are attracted to ischemic areas[63] do not integrate into newly formed arteries; rather, they secrete a variety of cytokines, such as fibroblast growth factor 2 and VEGF, which may enhance angiogenesis and thereby coordinate the angiogenic process. Additionally, matrix metalloproteinases (MMPs) from EPCs degrade the extracellular matrix, facilitating the movement and growth of ECs, EPCs, and monocytes, while also regulating growth factors and plasminogen.
In recent years, methods for exosome extraction have gradually improved. miR-150 transgenic EPCs[64] may improve EPC homing, thrombus recanalization, and dissolution in vivo, as well as increase migration and tube formation capacity in vitro. Additionally, exosomes with miRNA-136-5p[65] promote thrombolysis by inhibiting the expression of thioredoxin-interacting protein.
EPCs have immune regulatory effects
EPCs can interact with immune cells, and multiple studies have confirmed that EPCs and T cell subsets exhibit close collaboration. The EPC colony center cluster is primarily composed of specific T cell subpopulations[66], named “angiogenic T cells” (CD3+CD31+CXCR4+ T cells). These cells promote EPC differentiation and functional enhancement by secreting angiogenic factors such as VEGF and IL-8, and also participate in angiogenesis by recruiting macrophages and regulating VEGF. Conversely, EPC can also trigger T cell responses[67] under antigen presentation and co-stimulatory signals.
EPCs regulate the immune system in a number of ways. Firstly, in a dose-dependent manner, they can directly inhibit the proliferation of CD4+ and CD8+ T cells[68]. Through paracrine secretion of IL-10 and TGF-β1, EPCs can act on immune cells to participate in the regulation of immune suppression[69]. Additionally, the tumor necrosis factor receptor 2 (TNFR2) signaling system is crucial for immunological regulation. EPCs express TNFR2, which, upon binding TNF-α, activates downstream signaling pathways and enhances their immunosuppressive capacity[70]. Research has also found that ECFCs, derived from vascular endothelium, exhibit unique “immune privilege”[71]: their low expression of major histocompatibility complex (MHC) molecules allows them to evade recognition by the immune system, and they can induce the expansion of regulatory T cells (Tregs) by secreting IL-33, thereby promoting immune tolerance. Simultaneously, they possess the ability to form functional blood vessels in vivo. However, the immunosuppressive effect of ECFCs is independent of Tregs. Furthermore, ECFCs have an adaptive immunological-like "inflammatory memory"[72] that allows them to trigger immune responses linked to the toll-like receptor 3 (TLR3) pathway when exposed to ligands that resemble viruses.
EPCs can suppress intimal hyperplasia
When ECs are damaged, especially after vascular stent implantation, exposed collagen and smooth muscle cells (SMCs) promote platelet adhesion and inflammation[73]. Excess extracellular matrix is then secreted by activated SMCs as they multiply and move to the site of damage. This process, along with immune cell infiltration[74,75], leads to vascular endothelial hyperplasia.
EPCs primarily act on ECs through paracrine secretion of cytokines such as NO and VEGF to inhibit vascular endothelial hyperplasia. VEGF reduces the number of exposed vascular smooth muscle cells (VSMCs) in the injured area by boosting EC proliferation and endothelial repair[76-78]. Of course, EPCs can also directly repair damaged endothelium to restore EC function, enabling them to normally secrete NO, TGF-β, heparin, and heparin-like molecules, which keep SMCs in a quiescent state[79]. As key cells promoting vascular endothelial proliferation, SMCs are also directly targeted by EPCs[53]. They can inhibit angiotensin II-induced proliferation of VSMCs[80] and express Jagged1[81] to inhibit VSMC phenotypic conversion.
Nevertheless, Mause et al.[82] suggested that EPCs can negatively influence SMCs by decreasing their proliferation and migration, which may worsen neointimal accumulation after vascular injury. However, later studies[83] found that EPCs enhance SMC migration via the CXCL12 (C-X-C motif chemokine ligand 12)-CXCR4 signaling pathway, maintaining their contractile phenotype while promoting EC migration and proliferation. In atherosclerosis, EPCs can reverse SMC phenotypic conversion, improve SMC regulation and accelerate reendothelialization, ultimately reducing neointimal formation.
The role of EPCs in vascular remodeling in other diseases
Given the potent vascular repair capabilities of EPCs, they exert therapeutic effects not only in cardiovascular diseases but also in other conditions.
In the context of vascular endothelial injury associated with acute graft-versus-host disease (aGVHD)[42,84,85], EPCs exhibit the capacity to migrate specifically to areas of endothelial damage and differentiate into mature ECs, thereby contributing to the restoration of injured sites. Simultaneously, EPCs play a role in modulating the balance and infiltration of T cell subsets, which aids in alleviating endothelial inflammatory damage. Additionally, they have been shown to upregulate the expression of CD31 and VE-cadherin in compromised endothelium, facilitating the repair of intercellular junctions. This process leads to a reduction in vascular permeability, a decrease in inflammatory cell extravasation and tissue edema, an enhancement of vascular structural stability, and the promotion of vascular remodeling.
In the context of renal pathologies[86,87], EPCs contribute to vascular remodeling not only through direct differentiation but also by augmenting angiogenesis via paracrine mechanisms. Empirical studies have demonstrated that granulocyte colony-stimulating factor (G-CSF) therapy elevates EPC counts and markedly enhances their capacity to form cellular clusters, tubular formations, and network structures[88]. Furthermore, EPCs indirectly facilitate vascular remodeling by attenuating oxidative stress, inflammatory responses, and the progression of fibrosis within renal tissues[89]. This process alleviates the deleterious impact of perivascular fibrosis on vascular architecture, thereby preserving vascular elasticity and patency.
In addition to the diseases listed above, EPCs exhibit considerable potential for vascular remodeling in various other pathological conditions. Notably, they hold promise in the treatment of neurological disorders, particularly stroke, by facilitating re-endothelialization, attenuating inflammatory responses, and reducing cerebral ischemic injury[90-92]. Furthermore, in the context of pulmonary arterial hypertension (PAH), EPCs are pivotal in modulating pulmonary vascular remodeling. EPC transplantation has been explored in both preclinical and clinical trials for PAH, focusing on cell migration and the quantification of transplanted cells[93]. Additionally, EPCs stimulated by riociguat have exhibited protective effects in chronic thromboembolic pulmonary hypertension (CTEPH)[94]. Additionally, EPC transplantation mitigates renal ischemia-reperfusion injury[95], leading to a reduction in serum creatinine and blood urea nitrogen levels. It also enhances peritubular capillary density and safeguards renal function through the regulation of cellular autophagy.
CLINICAL TRANSLATION PROSPECTS AND CHALLENGES OF EPC
Considering the crucial role of EPCs in vascular remodeling, they present significant potential for clinical applications in the treatment of vascular diseases. This potential is primarily realized through three therapeutic strategies: cell therapy, EEPC-derived exosome (EPCs-Exo) therapy, and EPC functional modulation therapy.
Cell therapy
Based on their powerful vascular repair capabilities[96,97], CD34+ progenitor cells are being tested for use in the management of cardiovascular disease[98]. In the treatment of refractory angina pectoris, CD34+ cell injection can decrease the frequency of nitroglycerin use and angina attacks and improve myocardial perfusion and function[99-101]. Patients who had myocardial infarction benefited from increased angiogenesis, decreased infarct size, and enhanced left ventricular function when CD34+ cells were injected close to the infarct site[102-104]. A recent study is examining the use of autologous expanded CD34+ cells delivered by endocardial injection for acute myocardial infarction[105], focusing on increasing cell quantity and improving injection technique.
EPC capture stents, coated with CD34+ antibodies, are used in clinical treatments to capture cEPCs. This promotes endothelial growth, reduces restenosis and stent thrombosis, and decreases the need for anticoagulants compared to traditional PCI[106-109]. According to recent studies, COMBO® stents, coated with sirolimus and CD34 antibodies[110,111], offer benefits for vascular healing.
Furthermore, EPCs can remodel tumor vasculature, making them a potential therapeutic target for cancer; extracellular vesicles derived from EPCs have the potential to be a novel cell-free treatment for amyotrophic lateral sclerosis (ALS) that restores the blood-brain barrier (B-CNS-B) by encouraging EC repair[112]; Rats with preeclampsia-like syndrome can benefit from EPC transplantation[113], which may one day be used to treat pregnancy-related illnesses; patients with favorable prognoses for intracerebral hemorrhage have higher levels of EPCs[114], suggesting that cEPC levels could serve as a biomarker for disease severity.
EPCs-exo therapy
Compared with cell injection, cell-free therapy and EPCs-Exo have more advantages and fewer side effects. For example, enhanced external counterpulsation (EECP) can mobilize endogenous CD34+/CD133+ EPCs, providing an alternative option for patients who cannot undergo invasive injection[115]. In addition, some CD34 cell-derived exosomes[116-118], microRNA 126 and 130a, miR-199a, miR-302b, etc., can also be involved in cardiac therapy and vascular remodeling.
EPCs-Exo inhibits thrombosis and improves blood flow, particularly with miR-126 and miR-136-5P. Additionally, miR-222-3p from EPCs-Exo aids recovery in spinal cord-injured mice by boosting M2-like anti-inflammatory macrophages[119]. miR-182-5p greatly increases the migration and proliferation of ECs in the wound and surrounding tissues, hastening the recovery of diabetic wounds[120]. In addition, exosomes can also regulate the Bcl2 (B-cell lymphoma 2)/Bax (Bcl-2 associated X protein)/Caspase-3 (cysteine-aspartic acid protease 3) pathway, inhibit cell apoptosis, and effectively promote vascular endothelial repair[121] both in vivo and in vitro.
Currently, research on the use of exosomes for disease treatment is widespread. In addition to cardiovascular diseases, exosomes can also ameliorate acute lung injury[122], promote spinal cord injury recovery, accelerate wound healing in diabetes, and even demonstrate potential as a therapeutic target in stroke by transporting miR-126[123]. EPCs and their derived exosomes demonstrate significant potential in enhancing angiogenesis within the contexts of wound healing and tissue engineering[124]. As a viable cellular source for the induction of angiogenesis[125], EPCs have been utilized in numerous tissue engineering models.
Although exosomes have diverse functions and great potential for disease treatment, their clinical application still faces many challenges[126], including the purification of progenitor cell-derived exosomes [Table 3].
Applications of EPC-derived exosomes
| Author | Experimental subject | (Genetically) modified exosomes | Function |
| Yuan et al.[119] | C57BL/L mice | EPCs-EXOs-derived miR-222-3p | Upregulation of miR-222-3P in EPCs-EXOs influences macrophage polarization by participating in the SOCS3/JAK2/STAT3 pathway, promoting functional repair after spinal cord injury in mice |
| Li et al.[120] | Umbilical cord blood | EPCs-EXOs involved in the miR-182-5p/PPARG signaling pathway | EPCs-EXOs participate in the miR-182-5p/PPARG signaling pathway, promoting endothelial cell proliferation and migration in diabetic wounds, which is conducive to wound healing |
| Hu et al.[127] | The rat model of balloon injury | Enriched miR-21-5p EPCs-EXOs | EPCs-EXOs enriched with miR-21-5p inhibit thrombospondin-1 expression and promote endothelial cell repair |
| Xu et al.[128] | Mice | EPCs-derived miR-221-3p | EPCs-EXOs with high expression of miR-221-3p promote wound healing in diabetic mice |
| Liu et al.[129] | A murine model of sepsis | EPCs-EXOs containing miR-382-3p | EPCs-EXOs containing miR-382-3p participate in the regulation of the BTRC and IκBα/NF-κB axis, alleviating immune suppression and organ damage in septic mice |
Functional modulation of EPCs
In diabetic models, overexpression of metallothionein (MT) in EPCs can protect the hypoxia-inducible factor (HIF)-1α/SDF-1/VEGF signaling pathway from oxidative stress by promoting EPC migration and tube formation. Additionally, MT clears reactive oxygen species (ROS), thereby increasing EPC survival under diabetic conditions[130]. Therefore, enhancing MT can improve EPC function and increase the resistance of diabetic populations to ischemic conditions.
The combination of AMD3100 (a CXCR4 antagonist that mobilizes CD34+/CD133+ stem cells by blocking the SDF-1/CXCR4 axis) and low-dose FK506 (tacrolimus, 0.1 mg/kg) activates the BMP pathway to enhance SDF-1 secretion from M2 macrophages, thereby improving stem cell homing[131]. This subcutaneous AF combination enhances wound healing without immunosuppression and may benefit cardiovascular ischemic injury, pending further validation.
Statins represent the most extensively investigated pharmacological agents for the regulation of EPCs. They facilitate mitochondrial uptake via the calcium-dependent CAMK1 (calcium/calmodulin-dependent protein kinase 1)-PINK1 (PTEN-induced kinase 1) signaling pathway, thereby safeguarding EPC proliferation[132]. Furthermore, statins enhance EPC proliferation and migration through the miR-221/VEGFA (vascular endothelial growth factor A) axis[133], leading to an increase in cEPC numbers and providing vascular protective effects in conditions such as atherosclerosis and ischemic heart disease. In addition, pharmacological agents such as sodium-glucose cotransporter 2 (SGLT2) inhibitors and metformin[134,135] have been shown to restore EPC number and function under pathological conditions by modulating metabolic processes and augmenting antioxidant capacity. These findings suggest potential targeted therapeutic strategies for patients with cardiovascular diseases who also present with comorbid risk factors such as diabetes and obesity.
Challenges in clinical translation
Contemporary research on EPCs encounters numerous challenges. The identification and culture standards for EPCs lack consistency, and the functional characteristics of various EPC subpopulations necessitate further elucidation. Additionally, the molecular mechanisms responsible for EPC dysfunction within pathological microenvironments require more comprehensive investigation. The integration of advanced technologies, including biomaterials, gene editing, and targeted delivery, holds the potential to improve the therapeutic efficacy and safety of EPC-mediated vascular remodeling. Such advancements could expedite the transition of EPC research from fundamental studies to clinical applications.
CONCLUSION
EPCs, which serve as precursors to vascular ECs, are integral to the neovascularization of ischemic tissues and the repair of damaged vasculature. These cells possess the capability to integrate into existing blood vessels, differentiate directly into ECs, and secrete cytokines that facilitate vascular repair. Notably, EPCs derived from various sources exhibit distinct functional characteristics. In the context of cardiovascular disorders, EPCs can be mobilized and recruited to sites of vascular injury, where they differentiate into ECs to restore vascular integrity. Concurrently, they release MMPs, NO, SDF-1α, VEGF, and other factors. These paracrine factors not only inhibit thrombosis and intimal hyperplasia but also modulate immune responses. Additionally, they enhance the proliferation, mobilization, adhesion, and angiogenic capabilities of EPCs. Beyond their role in cardiovascular diseases, EPCs have demonstrated therapeutic potential in other conditions, including diabetes, aGVHD, and renal diseases. In clinical applications, EPC-based treatments have shown promising results. For example, CD34+ cell injections can improve myocardial perfusion and function. EPC-captured stents significantly reduce the likelihood of restenosis after stent implantation, progenitor cell exosomes enhance the specificity and efficiency of their functions, and certain drugs, such as statins, can increase the number of EPCs and enhance their function.
PERSPECTIVE
EPCs, due to their significant potential in vascular remodeling, have emerged as a central focus in the study of various vascular diseases. Despite substantial advancements in this area, numerous questions remain that require further exploration.
At the fundamental research level, the precise classification and functional characterization of EPC subpopulations remain significant challenges. Although early-stage and late-stage EPCs have been differentiated based on in vitro culture characteristics and functional disparities, the heterogeneity of EPCs derived from various sources - such as bone marrow, peripheral blood, and in situ - and their specific regulatory mechanisms within distinct pathological microenvironments are not yet fully understood. Advances in single-cell multi-omics technologies, including scRNA-seq and single-cell transposon-accessible chromatin sequencing (scATAC-seq), are anticipated to further elucidate the unique transcriptomic profiles and epigenetic modifications of EPC subpopulations, thereby expanding opportunities for targeted interventions. Additionally, exploring the interactions between EPCs and other cellular components within the vascular microenvironment - such as immune cells, VSMCs, and fibroblasts - will enhance our understanding of the specific roles of EPCs in vascular remodeling and aid in identifying novel targets for modulating EPC function.
In the domain of translational medicine, despite significant advancements in EPC transplantation therapy, several challenges remain, such as low post-transplantation cell survival rates, limited homing capacity, and issues related to immune rejection. Future research should prioritize the optimization of cell expansion protocols to improve the in vitro proliferation capacity and stability of EPCs. Simultaneously, enhancing the targeting efficiency of EPCs, potentially through the use of modified biomaterials or ligand-receptor-mediated targeting technologies, is essential to augment the homing efficiency of transplanted EPCs to pathological sites. Compared to cell therapy, EPCs-exo presents notable advantages in mitigating immune rejection and minimizing safety risks. Nonetheless, critical challenges for their clinical application include the isolation, purification, and long-term storage of exosomes, as well as the enhancement of their targeting and therapeutic efficacy through genetic modification. Furthermore, the functional modulation of endogenous EPCs via pharmacological agents (e.g., statins, SGLT2 inhibitors) or gene editing technologies represents a promising non-invasive therapeutic approach. Nonetheless, significant challenges for clinical application persist, notably in the isolation, purification, and long-term storage of exosomes, alongside the need to enhance their targeting and therapeutic efficacy through genetic modification. Furthermore, the functional modulation of endogenous EPCs using pharmacological agents (such as statins and SGLT2 inhibitors) or gene editing technologies presents non-invasive therapeutic avenues. However, additional clinical trials are necessary to substantiate their long-term safety and efficacy across diverse patient populations.
In summary, EPCs represent not only a key focus in basic medical research but also a critical breakthrough direction in clinical medicine, holding immense promise for disease treatment. Continued in-depth exploration of EPC biological characteristics and optimization of translational strategies will undoubtedly propel EPC-based therapies from preclinical research to clinical application, offering novel approaches for treating vascular remodeling-related diseases.
DECLARATIONS
Acknowledgements
Figures 2-4 and Graphical abstract were created with BioRender.com.
Authors' contributions
Made significant contributions to the conception and design of the article, conducted data collection and analysis, and authored the paper: Dai Y, Fan T
Provided guidance on article design and revisions: Xu Q, Huangfu N, Chen T
All authors have read and agreed to the published version of the manuscript.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
Not applicable.
Financial support and sponsorship
This work was supported by a grant from the National Key Research and Development Program of China (2023YFC3606500); two grants from the National Natural Science Foundation of China (Nos. 82470428, 81873484); grants from the Natural Science Foundation of Zhejiang Province (LZ26H020001, No. LMS25H020003); a grant from the Medical Science Research Foundation of the Health Department of Zhejiang Province (No. WKJ-ZJ-2312); and by the Key Laboratory of Precision Medicine for Atherosclerotic Diseases of Zhejiang Province, China (Grant No. 2022E10026).
Conflicts of interest
Chen T is a Youth Editorial Board Member of the journal Vessel Plus, but was not involved in any steps of the editorial process, notably including reviewer selection, manuscript handling, or decision-making, while the other authors have declared that they have no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Rajendran P, Rengarajan T, Thangavel J, et al. The vascular endothelium and human diseases. Int J Biol Sci. 2013;9:1057-69.
2. Asahara T, Murohara T, Sullivan A, et al. Isolation of putative progenitor endothelial cells for angiogenesis. Science. 1997;275:964-7.
3. Bailey AS, Jiang S, Afentoulis M, et al. Transplanted adult hematopoietic stems cells differentiate into functional endothelial cells. Blood. 2004;103:13-9.
4. Fujisawa T, Tura-Ceide O, Hunter A, et al. Endothelial progenitor cells do not originate from the bone marrow. Circulation. 2019;140:1524-6.
5. Timmermans F, Van Hauwermeiren F, De Smedt M, et al. Endothelial outgrowth cells are not derived from CD133+ cells or CD45+ hematopoietic precursors. Arterioscler Thromb Vasc Biol. 2007;27:1572-9.
6. Saito N, Shirado T, Funabashi-Eto H, et al. Purification and characterization of human adipose-resident microvascular endothelial progenitor cells. Sci Rep. 2022;12:1775.
7. Kawasaki T, Nishiwaki T, Sekine A, et al. Vascular repair by tissue-resident endothelial progenitor cells in endotoxin-induced lung injury. Am J Respir Cell Mol Biol. 2015;53:500-12.
8. Mao SZ, Ye X, Liu G, Song D, Liu SF. Resident endothelial cells and endothelial progenitor cells restore endothelial barrier function after inflammatory lung injury. Arterioscler Thromb Vasc Biol. 2015;35:1635-44.
9. Keighron C, Lyons CJ, Creane M, O'Brien T, Liew A. Recent advances in endothelial progenitor cells toward their use in clinical translation. Front Med. 2018;5:354.
10. Chopra H, Hung MK, Kwong DL, Zhang CF, Pow EHN. Insights into endothelial progenitor cells: origin, classification, potentials, and prospects. Stem Cells Int. 2018;2018:9847015.
11. Abdelgawad ME, Desterke C, Uzan G, Naserian S. Single-cell transcriptomic profiling and characterization of endothelial progenitor cells: new approach for finding novel markers. Stem Cell Res Ther. 2021;12:145.
12. Kalka C, Masuda H, Takahashi T, et al. Transplantation of ex vivo expanded endothelial progenitor cells for therapeutic neovascularization. Proc Natl Acad Sci USA. 2000;97:3422-7.
13. Hill JM, Zalos G, Halcox JP, et al. Circulating endothelial progenitor cells, vascular function, and cardiovascular risk. N Engl J Med. 2003;348:593-600.
14. Hur J, Yoon CH, Kim HS, et al. Characterization of two types of endothelial progenitor cells and their different contributions to neovasculogenesis. Arterioscler Thromb Vasc Biol. 2004;24:288-93.
15. Michalska-Jakubus MM, Rusek M, Kowal M, Czop M, Kocki J, Krasowska D. Anti-endothelial cell antibodies are associated with apoptotic endothelial microparticles, endothelial sloughing and decrease in angiogenic progenitors in systemic sclerosis. Postepy Dermatol Alergol. 2020;37:725-35.
16. Kourek C, Briasoulis A, Zouganeli V, Karatzanos E, Nanas S, Dimopoulos S. Exercise training effects on circulating endothelial and progenitor cells in heart failure. J Cardiovasc Dev Dis. 2022;9:222.
17. Salybekov AA, Kobayashi S, Asahara T. Characterization of endothelial progenitor cell: past, present, and future. Int J Mol Sci. 2022;23:7697.
18. Chen Y, Wan G, Li Z, et al. Endothelial progenitor cells in pregnancy-related diseases. Clin Sci. 2023;137:1699-719.
19. Medina RJ, Barber CL, Sabatier F, et al. Endothelial progenitors: a consensus statement on nomenclature. Stem Cells Transl Med. 2017;6:1316-20.
20. Tasev D, Konijnenberg LS, Amado-Azevedo J, van Wijhe MH, Koolwijk P, van Hinsbergh VW. CD34 expression modulates tube-forming capacity and barrier properties of peripheral blood-derived endothelial colony-forming cells (ECFCs). Angiogenesis. 2016;19:325-38.
21. Wang X, Wang R, Jiang L, Xu Q, Guo X. Endothelial repair by stem and progenitor cells. J Mol Cell Cardiol. 2022;163:133-46.
22. Duranova H, Valkova V, Olexikova L, et al. Rabbit endothelial progenitor cells derived from peripheral blood and bone marrow: an ultrastructural comparative study. Microsc Microanal. 2022;28:756-66.
23. Huang S, He P, Peng X, Li J, Xu D, Tang Y. Pristimerin inhibits prostate cancer bone metastasis by targeting PC-3 stem cell characteristics and VEGF-induced vasculogenesis of BM-EPCs. Cell Physiol Biochem. 2015;37:253-68.
24. Peng C, Yang LJ, Zhang C, et al. Low-dose nifedipine rescues impaired endothelial progenitor cell-mediated angiogenesis in diabetic mice. Acta Pharmacol Sin. 2023;44:44-57.
25. Yu JW, Deng YP, Han X, Ren GF, Cai J, Jiang GJ. Metformin improves the angiogenic functions of endothelial progenitor cells via activating AMPK/eNOS pathway in diabetic mice. Cardiovasc Diabetol. 2016;15:88.
26. Ceradini DJ, Kulkarni AR, Callaghan MJ, et al. Progenitor cell trafficking is regulated by hypoxic gradients through HIF-1 induction of SDF-1. Nat Med. 2004;10:858-64.
27. Leu S, Day YJ, Sun CK, Yip HK. tPA-MMP-9 axis plays a pivotal role in mobilization of endothelial progenitor cells from bone marrow to circulation and ischemic region for angiogenesis. Stem Cells Int. 2016;2016:5417565.
28. Zhang XY, Guo LP, Wang YZ, et al. TGF-β inhibition restores hematopoiesis and immune balance via bone marrow EPCs in aplastic anemia. Exp Mol Med. 2025;57:1324-38.
29. Ferrante A, Guggino G, Di Liberto D, et al. Endothelial progenitor cells: are they displaying a function in autoimmune disorders? Mech Ageing Dev. 2016;159:44-8.
30. Ramirez-Velandia F, Mensah E, Salih M, et al. Endothelial progenitor cells: a review of molecular mechanisms in the pathogenesis and endovascular treatment of intracranial aneurysms. Neuromolecular Med. 2024;26:25.
31. Dight J, Zhao J, Styke C, Khosrotehrani K, Patel J. Resident vascular endothelial progenitor definition and function: the age of reckoning. Angiogenesis. 2022;25:15-33.
32. Yang JX, Pan YY, Wang XX, Qiu YG, Mao W. Endothelial progenitor cells in age-related vascular remodeling. Cell Transplant. 2018;27:786-95.
33. Kou F, Zhu C, Wan H, et al. Endothelial progenitor cells as the target for cardiovascular disease prediction, personalized prevention, and treatments: progressing beyond the state-of-the-art. EPMA J. 2020;11:629-43.
34. Kwon YW, Heo SC, Jeong GO, et al. Tumor necrosis factor-α-activated mesenchymal stem cells promote endothelial progenitor cell homing and angiogenesis. Biochim Biophys Acta. 2013;1832:2136-44.
35. Lu W, Li X. Vascular stem/progenitor cells: functions and signaling pathways. Cell Mol Life Sci. 2018;75:859-69.
36. Qu K, Wang Z, Lin XL, Zhang K, He XL, Zhang H. MicroRNAs: Key regulators of endothelial progenitor cell functions. Clin Chim Acta. 2015;448:65-73.
37. Popovic D, Lavie CJ. Stress, cardiovascular diseases and exercise - a narrative review. Heart Mind. 2023;7:18-24.
38. Gill A, Aldosoky W, Ong MBH, Mir T, Dar T, Abohashem S. A narrative review on mental stress and cardiovascular disease: evidence, mechanisms, and potential interventions. Heart Mind. 2023;7:62-9.
39. Munir LZ, Du Toit EF. Impact of chronic psychological stress on cardiovascular disease risk: a narrative review. Heart Mind. 2024;8:268-78.
40. Araujo G, Valencia LM, Martin-Ozimek A, Soto Y, Proctor SD. Atherosclerosis: from lipid-lowering and anti-inflammatory therapies to targeting arterial retention of ApoB-containing lipoproteins. Front Immunol. 2025;16:1485801.
41. Vasa M, Fichtlscherer S, Aicher A, et al. Number and migratory activity of circulating endothelial progenitor cells inversely correlate with risk factors for coronary artery disease. Circ Res. 2001;89:E1-7.
42. Wang W, Ye Y, Du Y, et al. EPC infusion ameliorates acute graft-versus-host disease-related endothelial injury after allogeneic bone marrow transplantation. Front Immunol. 2022;13:1019657.
43. Zhuo X, Bu H, Hu K, et al. Differences in the reaction of hyperlipidemia on different endothelial progenitor cells based on sex. Biomed Rep. 2021;15:64.
44. He Z, Chen Y, Hou C, He W, Chen P. Cigarette smoke extract changes expression of endothelial nitric oxide synthase (eNOS) and p16(INK4a) and is related to endothelial progenitor cell dysfunction. Med Sci Monit. 2017;23:3224-31.
45. Eleftheriadou I, Dimitrakopoulou N, Kafasi N, et al. Endothelial progenitor cells and peripheral neuropathy in subjects with type 2 diabetes mellitus. J Diabetes Complicat. 2020;34:107517.
46. Nelson J, Wu Y, Jiang X, et al. Hyperhomocysteinemia suppresses bone marrow CD34+/VEGF receptor 2+ cells and inhibits progenitor cell mobilization and homing to injured vasculature-a role of β1-integrin in progenitor cell migration and adhesion. FASEB J. 2015;29:3085-99.
47. Hagensen MK, Shim J, Thim T, Falk E, Bentzon JF. Circulating endothelial progenitor cells do not contribute to plaque endothelium in murine atherosclerosis. Circulation. 2010;121:898-905.
48. Hagensen MK, Raarup MK, Mortensen MB, et al. Circulating endothelial progenitor cells do not contribute to regeneration of endothelium after murine arterial injury. Cardiovasc Res. 2012;93:223-31.
49. Asahara T, Takahashi T, Masuda H, et al. VEGF contributes to postnatal neovascularization by mobilizing bone marrow-derived endothelial progenitor cells. EMBO J. 1999;18:3964-72.
50. Heissig B, Hattori K, Dias S, et al. Recruitment of stem and progenitor cells from the bone marrow niche requires MMP-9 mediated release of kit-ligand. Cell. 2002;109:625-37.
51. Li L, Liu H, Xu C, et al. VEGF promotes endothelial progenitor cell differentiation and vascular repair through connexin 43. Stem Cell Res Ther. 2017;8:237.
52. Li B, Sharpe EE, Maupin AB, et al. VEGF and PlGF promote adult vasculogenesis by enhancing EPC recruitment and vessel formation at the site of tumor neovascularization. FASEB J. 2006;20:1495-7.
53. Yang H, He C, Bi Y, et al. Synergistic effect of VEGF and SDF-1α in endothelial progenitor cells and vascular smooth muscle cells. Front Pharmacol. 2022;13:914347.
54. Liu G, Lu P, Li L, et al. Critical role of SDF-1α-induced progenitor cell recruitment and macrophage VEGF production in the experimental corneal neovascularization. Mol Vis. 2011;17:2129-38.
55. Ozüyaman B, Ebner P, Niesler U, et al. Nitric oxide differentially regulates proliferation and mobilization of endothelial progenitor cells but not of hematopoietic stem cells. Thromb Haemostasis. 2005;94:770-2.
56. Behera J, Govindan S, Ramasamy MS. Nitric oxide promotes cell-matrix adhesion of endothelial progenitor cells under hypoxia condition via ITGA5 CpG promoter demethylation. Biochem Biophys Res Commun. 2023;644:162-70.
57. Bianchini E, Bianchini F, Lunardi M, et al. Outcomes, risk factors, and procedural management of acute myocardial infarction caused by stent thrombosis. Vessel Plus. 2024;8:20.
58. Khatana C, Saini NK, Chakrabarti S, et al. Mechanistic insights into the oxidized low-density lipoprotein-induced atherosclerosis. Oxid Med Cell Longev. 2020;2020:5245308.
59. Kashirskikh DA, Guo S, Panyod S, et al. A novel insight into the nature of modified low-density lipoproteins and their role in atherosclerosis. Vessel Plus. 2023;7:3.
60. Meng Q, Li X, Yu X, Lei F, Jiang K, Li C. Transplantation of ex vivo expanded bone marrow-derived endothelial progenitor cells enhances chronic venous thrombus resolution and recanalization. Clin Appl Thromb Hemost. 2011;17:E196-201.
61. Massberg S, Konrad I, Schürzinger K, et al. Platelets secrete stromal cell-derived factor 1alpha and recruit bone marrow-derived progenitor cells to arterial thrombi in vivo. J Exp Med. 2006;203:1221-33.
62. Abou-Saleh H, Yacoub D, Théorêt JF, et al. Endothelial progenitor cells bind and inhibit platelet function and thrombus formation. Circulation. 2009;120:2230-9.
63. Modarai B, Burnand KG, Sawyer B, Smith A. Endothelial progenitor cells are recruited into resolving venous thrombi. Circulation. 2005;111:2645-53.
64. Wang W, Li C, Li W, et al. MiR-150 enhances the motility of EPCs in vitro and promotes EPCs homing and thrombus resolving in vivo. Thromb Res. 2014;133:590-8.
65. Feng Y, Lei B, Zhang H, et al. MicroRNA-136-5p from endothelial progenitor cells-released extracellular vesicles mediates TXNIP to promote the dissolution of deep venous thrombosis. Shock. 2022;57:714-21.
66. Hur J, Yang HM, Yoon CH, et al. Identification of a novel role of T cells in postnatal vasculogenesis: characterization of endothelial progenitor cell colonies. Circulation. 2007;116:1671-82.
67. Raemer PC, Haemmerling S, Giese T, et al. Endothelial progenitor cells possess monocyte-like antigen-presenting and T-cell-co-stimulatory capacity. Transplantation. 2009;87:340-9.
68. Naserian S, Abdelgawad ME, Lachaux J, et al. Development of bio-artificial micro-vessels with immunosuppressive capacities: a hope for future transplantations and organoids. Blood. 2019;134:3610.
69. Proust R, Ponsen AC, Rouffiac V, et al. Cord blood-endothelial colony forming cells are immunotolerated and participate at post-ischemic angiogenesis in an original dorsal chamber immunocompetent mouse model. Stem Cell Res Ther. 2020;11:172.
70. Chen Y, Jiang M, Chen X. Therapeutic potential of TNFR2 agonists: a mechanistic perspective. Front Immunol. 2023;14:1209188.
71. Arakelian L, Lion J, Churlaud G, et al. Endothelial CD34 expression and regulation of immune cell response in-vitro. Sci Rep. 2023;13:13512.
72. Weiss E, Vlahos A, Kim B, et al. Transcriptomic remodelling of fetal endothelial cells during establishment of inflammatory memory. Front Immunol. 2021;12:757393.
73. Tseng CN, Karlöf E, Chang YT, et al. Contribution of endothelial injury and inflammation in early phase to vein graft failure: the causal factors impact on the development of intimal hyperplasia in murine models. PLoS ONE. 2014;9:e98904.
74. Carracedo M, Artiach G, Arnardottir H, Bäck M. The resolution of inflammation through omega-3 fatty acids in atherosclerosis, intimal hyperplasia, and vascular calcification. Semin Immunopathol. 2019;41:757-66.
75. Kijani S, Vázquez AM, Levin M, Borén J, Fogelstrand P. Intimal hyperplasia induced by vascular intervention causes lipoprotein retention and accelerated atherosclerosis. Physiol Rep. 2017;5:e13334.
76. Olsson AK, Dimberg A, Kreuger J, Claesson-Welsh L. VEGF receptor signalling - in control of vascular function. Nat Rev Mol Cell Biol. 2006;7:359-71.
77. Sun H, Morihara R, Feng T, et al. Human cord blood-endothelial progenitor cells alleviate intimal hyperplasia of arterial damage in a rat stroke model. Cell Transplant. 2023;32:9636897231193069.
78. Hutter R, Carrick FE, Valdiviezo C, et al. Vascular endothelial growth factor regulates reendothelialization and neointima formation in a mouse model of arterial injury. Circulation. 2004;110:2430-5.
79. Gilotti AC, Nimlamool W, Pugh R, et al. Heparin responses in vascular smooth muscle cells involve cGMP-dependent protein kinase (PKG). J Cell Physiol. 2014;229:2142-52.
80. Fang L, Chen MF, Xiao ZL, Yu GL, Chen XB, Xie XM. The effect of endothelial progenitor cells on angiotensin II-induced proliferation of cultured rat vascular smooth muscle cells. J Cardiovasc Pharmacol. 2011;58:617-25.
81. Zhang J, Chen J, Xu C, et al. Resveratrol inhibits phenotypic switching of neointimal vascular smooth muscle cells after balloon injury through blockade of Notch pathway. J Cardiovasc Pharmacol. 2014;63:233-9.
82. Mause SF, Ritzel E, Deck A, Vogt F, Liehn EA. Endothelial progenitor cells modulate the phenotype of smooth muscle cells and increase their neointimal accumulation following vascular injury. Thromb Haemostasis. 2022;122:456-69.
83. Mause SF, Ritzel E, Deck A, Vogt F, Liehn EA. Engagement of the CXCL12-CXCR4 axis in the interaction of endothelial progenitor cell and smooth muscle cell to promote phenotype control and guard vascular homeostasis. Int J Mol Sci. 2022;23:867.
84. Cao XN, Kong Y, Song Y, et al. Impairment of bone marrow endothelial progenitor cells in acute graft-versus-host disease patients after allotransplant. Br J Haematol. 2018;182:870-86.
85. Medinger M, Heim D, Gerull S, et al. Increase of endothelial progenitor cells in acute graft-versus-host disease after allogeneic haematopoietic stem cell transplantation for acute myeloid leukaemia. Leuk Res. 2016;47:22-5.
86. Yang CC, Sung PH, Cheng BC, et al. Safety and efficacy of intrarenal arterial autologous CD34+ cell transfusion in patients with chronic kidney disease: a randomized, open-label, controlled phase II clinical trial. Stem Cells Transl Med. 2020;9:827-38.
87. Kundu N, Nandula SR, Asico LD, et al. Transplantation of apoptosis-resistant endothelial progenitor cells improves renal function in diabetic kidney disease. J Am Heart Assoc. 2021;10:e019365.
88. Tang SY, Lee YC, Tseng CW, Huang PH, Kuo KL, Tarng DC. Granulocyte colony-stimulating factor improves endothelial progenitor cell-mediated neovascularization in mice with chronic kidney disease. Pharmaceutics. 2023;15:2380.
89. Awal HB, Nandula SR, Domingues CC, et al. Linagliptin, when compared to placebo, improves CD34+ve endothelial progenitor cells in type 2 diabetes subjects with chronic kidney disease taking metformin and/or insulin: a randomized controlled trial. Cardiovasc Diabetol. 2020;19:72.
90. Shen S, Pan T, Liu P, Tian Y, Shi Y, Zhu W. The mechanisms and applications of endothelial progenitor cell therapy in the treatment of intracranial aneurysm. J Transl Med. 2025;23:377.
91. Zhang Y, Yang Q, Cheng H, Zhang Y, Xie Y, Zhang Q. Extracellular vesicles derived from endothelial progenitor cells modified by Houshiheisan promote angiogenesis and attenuate cerebral ischemic injury via miR-126/PIK3R2. Sci Rep. 2024;14:28166.
92. Yuan JJ, Yang J, Sun SL, Zhang R, Xu YM. Endothelial progenitor cells' classification and application in neurological diseases. Tissue Eng Regen Med. 2017;14:327-32.
93. Liu Y, Zhao X, Ding J, et al. Evidence of accumulated endothelial progenitor cells in the lungs of rats with pulmonary arterial hypertension by 89Zr-oxine PET imaging. Mol Ther Methods Clin Dev. 2020;17:1108-17.
94. Yamamoto K, Nishimura R, Kato F, et al. Protective role of endothelial progenitor cells stimulated by riociguat in chronic thromboembolic pulmonary hypertension. Int J Cardiol. 2020;299:263-70.
95. Kamel NM, Abd El Fattah MA, El-Abhar HS, Abdallah DM. Novel repair mechanisms in a renal ischaemia/reperfusion model: Subsequent saxagliptin treatment modulates the pro-angiogenic GLP-1/cAMP/VEGF, ANP/eNOS/NO, SDF-1α/CXCR4, and Kim-1/STAT3/HIF-1α/VEGF/eNOS pathways. Eur J Pharmacol. 2019;861:172620.
96. Tongers J, Roncalli JG, Losordo DW. Role of endothelial progenitor cells during ischemia-induced vasculogenesis and collateral formation. Microvasc Res. 2010;79:200-6.
97. Riesinger L, Saemisch M, Nickmann M, Methe H. CD34+ circulating cells display signs of immune activation in patients with acute coronary syndrome. Heart Vessels. 2018;33:1559-69.
98. Aries A, Zanetti C, Hénon P, Drénou B, Lahlil R. Deciphering the cardiovascular potential of human CD34+ stem cells. Int J Mol Sci. 2023;24:9551.
99. Losordo DW, Schatz RA, White CJ, et al. Intramyocardial transplantation of autologous CD34+ stem cells for intractable angina: a phase I/IIa double-blind, randomized controlled trial. Circulation. 2007;115:3165-72.
100. Losordo DW, Henry TD, Davidson C, et al. intramyocardial, autologous CD34+ cell therapy for refractory angina. Circ Res. 2011;109:428-36.
101. Henry TD, Losordo DW, Traverse JH, et al. Autologous CD34+ cell therapy improves exercise capacity, angina frequency and reduces mortality in no-option refractory angina: a patient-level pooled analysis of randomized double-blinded trials. Eur Heart J. 2018;39:2208-16.
102. Zhang S, Ge J, Zhao L, et al. Host vascular niche contributes to myocardial repair induced by intracoronary transplantation of bone marrow CD34+ progenitor cells in infarcted swine heart. Stem Cells. 2007;25:1195-203.
103. Wang J, Zhang S, Rabinovich B, et al. Human CD34+ cells in experimental myocardial infarction: long-term survival, sustained functional improvement, and mechanism of action. Circ Res. 2010;106:1904-11.
104. Mackie AR, Klyachko E, Thorne T, et al. Sonic hedgehog-modified human CD34+ cells preserve cardiac function after acute myocardial infarction. Circ Res. 2012;111:312-21.
105. Roncalli J, Roubille F, Meyer N, et al. Transendocardial injection of expanded autologous CD34+ cells after myocardial infarction: Design of the EXCELLENT trial. ESC Heart Fail. 2025;12:1455-63.
106. Co M, Tay E, Lee CH, et al. Use of endothelial progenitor cell capture stent (Genous Bio-Engineered R Stent) during primary percutaneous coronary intervention in acute myocardial infarction: intermediate- to long-term clinical follow-up. Am Heart J. 2008;155:128-32.
107. Chong MS, Ng WK, Chan JK. Concise review: endothelial progenitor cells in regenerative medicine: applications and challenges. Stem Cells Transl Med. 2016;5:530-8.
108. Pelliccia F, Zimarino M, Niccoli G, et al. In-stent restenosis after percutaneous coronary intervention: emerging knowledge on biological pathways. Eur Heart J Open. 2023;3:oead083.
109. Huang YH, Xu Q, Shen T, Li JK, Sheng JY, Shi HJ. Prevention of in-stent restenosis with endothelial progenitor cell (EPC) capture stent placement combined with regional EPC transplantation: An atherosclerotic rabbit model. Cardiol J. 2019;26:283-91.
110. Lu T, Sakuma M, Sohma R, et al. Mobilization of endothelial progenitor cells after implantation of CD34 antibody-covered sirolimus-eluting COMBO® stent: assessment with EPC colony-forming assay. Heart Vessels. 2025;40:435-45.
111. Goto H, Kitahara H, Matsumoto T, et al. Comparison of very early-phase vascular response to the CD34 antibody‑covered sirolimus‑eluting stent versus durable polymer-coated everolimus-eluting stent. Cardiovasc Interv Ther. 2025;40:527-35.
112. Garbuzova-Davis S, Willing AE, Ehrhart J, Wang L, Sanberg PR, Borlongan CV. Cell-free extracellular vesicles derived from human bone marrow endothelial progenitor cells as potential therapeutics for microvascular endothelium restoration in ALS. Neuromolecular Med. 2020;22:503-16.
113. Zhu J, Cheng X, Wang Q, Zhou Y, Wang F, Zou L. Transplantation of endothelial progenitor cells for improving placental perfusion in preeclamptic rats. Arch Gynecol Obstet. 2015;291:1113-9.
114. Pías-Peleteiro J, Pérez-Mato M, López-Arias E, et al. Increased endothelial progenitor cell levels are associated with good outcome in intracerebral hemorrhage. Sci Rep. 2016;6:28724.
115. Tartaglia JT, Eisenberg CA, DeMarco JC, Puccio G, Tartaglia CE, Hamby CV. Mobilization of endogenous CD34+/CD133+ endothelial progenitor cells by enhanced external counter pulsation for treatment of refractory angina. Int J Mol Sci. 2024;25:10030.
116. Sahoo S, Klychko E, Thorne T, et al. Exosomes from human CD34+ stem cells mediate their proangiogenic paracrine activity. Circ Res. 2011;109:724-8.
118. Aries A, Vignon C, Zanetti C, et al. Development of a potency assay for CD34+ cell-based therapy. Sci Rep. 2023;13:19665.
119. Yuan F, Peng W, Yang Y, et al. Endothelial progenitor cell-derived exosomes promote anti-inflammatory macrophages via SOCS3/JAK2/STAT3 axis and improve the outcome of spinal cord injury. J Neuroinflammation. 2023;20:156.
120. Li P, Hong G, Zhan W, et al. Endothelial progenitor cell derived exosomes mediated miR-182-5p delivery accelerate diabetic wound healing via down-regulating PPARG. Int J Med Sci. 2023;20:468-81.
121. Tan W, Li Y, Ma L, et al. Exosomes of endothelial progenitor cells repair injured vascular endothelial cells through the Bcl2/Bax/Caspase-3 pathway. Sci Rep. 2024;14:4465.
122. Wu X, Liu Z, Hu L, Gu W, Zhu L. Exosomes derived from endothelial progenitor cells ameliorate acute lung injury by transferring miR-126. Exp Cell Res. 2018;370:13-23.
123. Forró T, Bajkó Z, Bălașa A, Bălașa R. Dysfunction of the neurovascular unit in ischemic stroke: highlights on microRNAs and exosomes as potential biomarkers and therapy. Int J Mol Sci. 2021;22:5621.
124. Schwarz N, Yadegari H. Potentials of endothelial colony-forming cells: applications in hemostasis and thrombosis disorders, from unveiling disease pathophysiology to cell therapy. Hamostaseologie. 2023;43:325-37.
125. Ju Y, Hu Y, Yang P, Xie X, Fang B. Extracellular vesicle-loaded hydrogels for tissue repair and regeneration. Mater Today Bio. 2023;18:100522.
126. Gurunathan S, Kang MH, Jeyaraj M, Qasim M, Kim JH. Correction: Gurunathan, S. et al. Review of the isolation, characterization, biological function, and multifarious therapeutic approaches of exosomes. Cells 2019, 8, 307. Cells. 2021;10:462.
127. Hu H, Wang B, Jiang C, Li R, Zhao J. Endothelial progenitor cell-derived exosomes facilitate vascular endothelial cell repair through shuttling miR-21-5p to modulate Thrombospondin-1 expression. Clin Sci. 2019;133:1629-44.
128. Xu J, Bai S, Cao Y, et al. miRNA-221-3p in endothelial progenitor cell-derived exosomes accelerates skin wound healing in diabetic mice. Diabetes Metab Syndr Obes. 2020;13:1259-70.
129. Liu Y, Luo T, Li H, Zhao X, Zhou M, Cheng M. Protective effect of endothelial progenitor cell-derived exosomal microRNA-382-3p on sepsis-induced organ damage and immune suppression in mice. Am J Transl Res. 2022;14:6856-73.
130. Wang K, Dai X, He J, et al. Endothelial overexpression of metallothionein prevents diabetes-induced impairment in ischemia angiogenesis through preservation of HIF-1α/SDF-1/VEGF signaling in endothelial progenitor cells. Diabetes. 2020;69:1779-92.
131. Qi L, Ahmadi AR, Huang J, et al. Major improvement in wound healing through pharmacologic mobilization of stem cells in severely diabetic rats. Diabetes. 2020;69:699-712.
132. Yang J, Sun M, Cheng R, et al. Pitavastatin activates mitophagy to protect EPC proliferation through a calcium-dependent CAMK1-PINK1 pathway in atherosclerotic mice. Commun Biol. 2022;5:124.
133. Sun L, Zhang Y, Zhang J, Wang J, Xing S. Atorvastatin improves the proliferation and migration of endothelial progenitor cells via the miR-221/VEGFA axis. Biosci Rep. 2020;40:BSR20193053.
134. Nandula SR, Kundu N, Awal HB, et al. Role of canagliflozin on function of CD34+ve endothelial progenitor cells (EPC) in patients with type 2 diabetes. Cardiovasc Diabetol. 2021;20:44.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data















Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.