Targeting ferroptosis in photoaging: mechanisms and therapeutic potential of adipose-derived stem cell exosomes
Abstract
Skin photoaging is a form of premature cutaneous aging caused by chronic ultraviolet (UV) exposure, manifesting as wrinkling, laxity, and pigmentary disturbances. UV radiation generates reactive oxygen species and upregulates matrix metalloproteinases, leading to extracellular matrix degradation. Emerging evidence implicates ferroptosis - an iron-dependent form of regulated cell death characterized by lipid peroxidation - in ultraviolet B (UVB)-induced photodamage. Studies show that glutathione peroxidase 4 (GPX4)-dependent antioxidant defenses are compromised in UV-exposed skin cells, and ferroptosis inhibitors can partially attenuate UV-induced cellular damage. Exosomes derived from adipose-derived stem cells (ADSC-Exos) have emerged as a promising cell-free regenerative strategy for skin rejuvenation. These 30-150 nm extracellular vesicles offer advantages over whole-cell therapy, including low immunogenicity, reduced tumorigenic concerns compared with replicating cells, and easier standardization. Through paracrine signaling, ADSC-Exos deliver bioactive proteins and non-coding RNAs that promote collagen synthesis, angiogenesis, and anti-inflammatory effects. Circular RNAs (circRNAs) are highly enriched and stable in exosomes due to their covalently closed structure. Notably, hypoxic-preconditioned ADSC-Exos deliver circ-Ash1l into UVB-damaged skin cells, where it sequesters miR-700-5p and upregulates GPX4. This circ-Ash1l/miR-700-5p/GPX4 axis inhibits ferroptotic cell death, reduces oxidative damage, and attenuates UVB-induced photoaging phenotypes in vitro and in vivo. This review summarizes the mechanisms of UVB-mediated photoaging and ferroptosis, the therapeutic potential of adipose-derived stem cell-derived exosomes, the role of exosomal circRNAs, and engineering strategies to enhance exosome therapy. We also discuss clinical translation challenges, including manufacturing, quality control, and regulatory considerations.
Keywords
INTRODUCTION
Human skin aging is classified into intrinsic (chronological) and extrinsic forms; long-term sunlight exposure is the major extrinsic driver of cutaneous aging, termed photoaging[1,2]. Chronic ultraviolet (UV) exposure leads to premature aging with wrinkles, pigmentary changes, and loss of elasticity[3]. Classical mechanisms of photoaging include oxidative stress and extracellular matrix degradation via upregulated matrix metalloproteinases (MMPs)[4,5]. In addition to these established pathways, growing evidence implicates ferroptosis - a lipid peroxidation-driven, iron-dependent form of regulated cell death - in UV-induced cutaneous injury and photoaging contexts[6,7]. Ferroptosis is defined as an iron-dependent regulated cell death characterized by lipid peroxidation-mediated membrane damage[8,9].
Among regenerative strategies, exosomes derived from adipose-derived stem cells (ADSC-Exos) have emerged as promising cell-free therapeutics for photoaging, improving cutaneous structure and function in ultraviolet B (UVB) models[10-12]. Recent clinical scholarship has further outlined the therapeutic potential of exosomes in plastic surgery for skin rejuvenation and wound healing[13]. These nanoscale vesicles carry diverse bioactive cargos, including circular RNAs (circRNAs)[14]; notably, hypoxia-preconditioned ADSC-Exos deliver circ-Ash1l to modulate the miR-700-5p/glutathione peroxidase 4 (GPX4) axis and inhibit ferroptosis, thereby attenuating UV-induced skin damage[15]. This review is organized around a translational framework that progresses from pathophysiological understanding to therapeutic application. We first delineate the mechanistic intersection between ferroptosis and photoaging, establishing why this cell death pathway represents a rational therapeutic target (see Section “Mechanisms of Photoaging and Ferroptosis”). We then examine how adipose-derived stem cell (ADSC)-derived exosomes modulate these pathways, with particular emphasis on the emerging role of exosomal circRNAs (exo-circRNAs) as functional mediators - using the circ-Ash1l/miR-700-5p/GPX4 axis as a paradigmatic case study (see Section “ADSC Exosomes as Anti-Ferroptosis Therapeutics”). The review subsequently addresses engineering strategies that may enhance therapeutic potential and overcome delivery barriers inherent to topical exosome applications (see Section “Therapeutic Applications of ADSC-Derived Exosomes”), before confronting the regulatory landscape and clinical translation challenges that currently limit the progression of exosome therapeutics from bench to bedside (see Section “Clinical Translation and Regulatory Considerations”). Throughout, we critically evaluate the quality of existing evidence and identify knowledge gaps that must be addressed to advance this field. This framework positions ferroptosis-targeted exosome therapy not as an established treatment modality, but as a promising hypothesis requiring rigorous validation through standardized preclinical studies and well-designed clinical trials.
MECHANISMS OF PHOTOAGING AND FERROPTOSIS
UV exposure and photoaging
Photoaged skin is characterized by coarse wrinkles, roughness/dryness, laxity, telangiectasia, and mottled pigmentation including solar lentigines[2,16,17]. At the histological level, chronic UV exposure produces dermal connective-tissue degeneration with solar elastosis and abnormal accumulation of elastin and glycosaminoglycans, classically termed dermatoheliosis[18,19]. Human skin aging comprises intrinsic and extrinsic components, with UV radiation being the dominant extrinsic driver of premature cutaneous aging[16].
Ultraviolet A (UVA) and UVB both contribute through distinct yet complementary mechanisms: UVA penetrates into the dermis and promotes reactive oxygen species (ROS)-driven signaling and inflammation, whereas UVB - absorbed largely in the epidermis - directly induces cyclobutane pyrimidine dimers (CPDs) and also increases ROS[16,20-22]. UVA/UVB exposure upregulates matrix-degrading metalloproteinases - most prominently matrix metalloproteinase 1 (MMP-1) (collagenase-1) and matrix metalloproteinase 3 (MMP-3) (stromelysin-1), with induction of gelatinases matrix metalloproteinase 2 (MMP-2) and matrix metalloproteinase 9 (MMP-9) shown in vivo - which degrade type I/III collagen and other extracellular-matrix components underlying dermal elasticity loss and wrinkle formation[2,23-25].
Acute in vivo UV irradiation of human skin induces MMP-1 and MMP-3 and concurrently suppresses new collagen synthesis; repeated sub-erythemal exposures sustain MMP elevation and cumulatively drive collagen breakdown and wrinkle development[2,23,26]. Epidemiologically, photoaging is highly prevalent in sun-exposed populations; in a Queensland community study, 83% of adults aged 20-54 exhibited premature skin aging on clinical assessment and surface microtopography[27]. Chronic UV exposure also impairs cutaneous immune function by reducing Langerhans cell density and antigen-presenting capacity, contributing to local immunosuppression[28-30]. Although melanocyte numbers decline overall with age, long-term sun-exposed skin shows higher melanocyte density and activity than photoprotected sites, helping explain hyperpigmented lesions such as solar lentigines despite age-related melanocyte loss[31,32].
Ferroptosis in UVB-induced skin damage
Beyond classical oxidative damage and extracellular matrix (ECM) breakdown, recent research has identified ferroptosis as an additional mechanism underlying UVB-induced photoaging[6,7,33]. Ferroptosis is a non-apoptotic, iron-dependent form of regulated cell death characterized by excessive lipid peroxidation[34]. Unlike apoptosis (which is caspase-driven and involves orderly dismantling of the cell), ferroptosis results from uncontrolled oxidative damage to membrane lipids, particularly polyunsaturated fatty acid (PUFA)-containing phospholipids[34,35]. Cells undergoing ferroptosis show distinctive morphological changes including shrunken mitochondria with increased membrane density and loss of mitochondrial cristae, with no chromatin condensation or apoptotic bodies[35,36].
The key molecular players regulating ferroptosis are GPX4 and ferroptosis suppressor protein 1 (FSP1), both of which reduce lipid peroxides to non-toxic lipid alcohols. GPX4 uses glutathione as a cofactor to reduce phospholipid hydroperoxides directly in membranes, whereas FSP1 acts independently via regeneration of coenzyme Q10 to scavenge lipid radicals[9,37]. When these protective systems are overwhelmed or suppressed, lipid peroxides accumulate, and lipid membranes become unstable, and cell death ensues[35]. UVB radiation impairs these defenses in skin cells. Studies show that human keratinocytes exposed to UVB exhibit glutathione dysregulation with accumulation of ferroptosis-specific oxidized phospholipids and death that is preventable by ferroptosis inhibitors; UVB also promotes autophagy-dependent GPX4 degradation in keratinocytes[6].
The mechanistic link between UVB and ferroptosis includes several pathways. UVB-driven oxidative stress consumes glutathione, limiting GPX4 activity[6,38]. In parallel, tumor protein p53 (p53) can suppress the cystine-glutamate antiporter (system Xc-) by repressing solute carrier family 7 member 11 (SLC7A11), thereby reducing cysteine uptake needed for glutathione (GSH) synthesis and promoting ferroptosis [Figure 1][9]. UVB exposure is also associated with altered iron handling in skin, including increased transferrin receptor 1 and decreased ferroportin, leading to intracellular iron accumulation[33]. Excess intracellular iron catalyzes hydroxyl radical production via the Fenton reaction, exacerbating lipid peroxidation[39]. Importantly, inhibiting ferroptosis with chemical inhibitors - Ferrostatin-1 (a lipid radical trap) or deferoxamine (an iron chelator) - can partially protect cultured skin cells from UVB-induced injury and photoaging-related markers[6,7,40]. In mouse models of UVB-induced skin injury, topical Ferrostatin-1 reduced pro-inflammatory cytokine expression and dermal immune-cell infiltration compared with vehicle controls[33]. These findings collectively suggest that ferroptosis actively contributes to photoaging pathology. Targeting ferroptosis may thus represent a novel therapeutic avenue for photoprotection and skin rejuvenation, complementing antioxidant strategies and matrix-protective interventions[33,41].
Figure 1. UV-induced ferroptosis in skin cells and its link to photoaging. Ultraviolet irradiation increases ROS and p53 activity, which suppresses SLC7A11/SLC3A2 (system Xc-) and limits cystine import for GSH synthesis (via GCL/GSS), thereby weakening GPX4-mediated reduction of PUFA-OOH. Meanwhile, iron is imported through Tf/TfR1, reduced by STEAP3, transported by DMT1, mobilized via NCOA4-mediated ferritinophagy, and exported by FPN1/SLC40A1; increased LIP drives Fenton chemistry. Lipid peroxidation is promoted by membrane PUFA-PL remodeling (ACSL4 → LPCAT3) and lipoxygenases (e.g., ALOX15), culminating in ferroptosis. Parallel antioxidant systems such as FSP1-CoQ10 (and mitochondrial DHODH) counteract phospholipid peroxidation independently of GPX4. The downstream consequences in skin include MMP-1/-3/-9 up-regulation, collagen degradation, and loss of elasticity, contributing to photoaging. Created with BioRender.com. ROS: Reactive oxygen species; SLC7A11: solute carrier family 7 member 11; SLC3A2: solute carrier family 3 member 2; GSH: glutathione; GCL: glutamate-cysteine ligase; GSS: glutathione synthetase; GPX4: glutathione peroxidase 4; PUFA-OOH: polyunsaturated fatty acid hydroperoxide; Tf: transferrin; TfR1: transferrin receptor 1; STEAP3: six-transmembrane epithelial antigen of prostate 3; DMT1: divalent metal transporter 1; NCOA4: nuclear receptor coactivator 4; FPN1: ferroportin 1; SLC40A1: solute carrier family 40 member 1; LIP: labile iron pool; PUFA-PL: polyunsaturated fatty acid-phospholipid; ACSL4: acyl-CoA synthetase long-chain family member 4; LPCAT3: lysophosphatidylcholine acyltransferase 3; ALOX15: arachidonate 15-lipoxygenase; FSP1: ferroptosis suppressor protein 1; CoQ10: coenzyme Q10; DHODH: dihydroorotate dehydrogenase; MMP: matrix metalloproteinase.
Despite these promising findings, several critical limitations warrant consideration before ferroptosis can be established as a validated therapeutic target in human photoaging. The evidence linking ferroptosis to photoaging derives predominantly from acute, high-dose UVB exposure models in cell culture or short-term animal studies, which may not faithfully recapitulate the pathophysiology of chronic human photoaging that develops over decades of cumulative sub-erythemal UV exposure. Whether the ferroptotic signatures observed in acute injury models persist or accumulate in chronically photoaged human skin remains to be systematically investigated. Furthermore, the specificity of ferroptosis markers in photoaging contexts requires careful interpretation. Lipid peroxidation and iron dysregulation also occur in general oxidative stress responses, and distinguishing ferroptosis-specific contributions from broader oxidative damage in the complex milieu of UV-exposed skin presents methodological challenges. The reported efficacy of ferroptosis inhibitors such as Ferrostatin-1 in vitro and in mouse models has not yet been validated in human clinical studies. Practical considerations, including percutaneous penetration, metabolic stability, and potential off-target effects on normal iron-dependent processes in the skin, also remain unaddressed. Finally, the relative contribution of ferroptosis vs. other cell death modalities (apoptosis, necroptosis, pyroptosis) to photoaging pathology has not been quantitatively delineated, making it difficult to predict the therapeutic impact of ferroptosis-selective interventions.
ADSC EXOSOMES AS ANTI-FERROPTOSIS THERAPEUTICS
The therapeutic potential of ADSC-derived exosomes
ADSCs exert regenerative effects primarily through paracrine secretion of exosomes, 30-150 nm extracellular vesicles (EVs) that deliver bioactive proteins and regulatory RNAs - including circRNAs increasingly implicated in ferroptosis modulation - to recipient cells[42-46].
Compared with live-cell therapies, ADSC-Exos offer key translational advantages: acellular nature, low immunogenicity, reduced tumorigenic concerns, and amenability to lyophilized off-the-shelf manufacturing[47-51]. These properties position ADSC-Exos as promising vehicles for delivering anti-ferroptotic cargo to UV-damaged skin. A note on nomenclature: throughout this review, we use “exosome” for consistency with the terminology adopted in the majority of cited studies; however, unless endosomal biogenesis has been experimentally demonstrated, these preparations are more precisely classified as small extracellular vesicles (sEVs) per Minimal Information for Studies of Extracellular Vesicles (MISEV)2023 guidelines. Where cited studies characterized vesicles only at the EV level (e.g., by nanoparticle tracking analysis and tetraspanin markers without confirming endosomal origin), readers should interpret “exosome” as a conventional rather than a strictly biogenesis-defined term.
In photoaging models, ADSC-Exos attenuate UVB-induced oxidative damage, suppress pro-inflammatory cytokines, and limit MMP-mediated matrix degradation, collectively improving skin hydration and reducing wrinkle formation[48]. These antioxidant and cytoprotective effects are mechanistically relevant to ferroptosis, given that oxidative stress, glutathione depletion, and lipid peroxidation constitute the core drivers of ferroptotic cell death. A representative selection of recent exosome-based studies in photoaging [Table 1] illustrates that these therapeutic effects have been consistently observed across diverse experimental models. The majority of investigations utilize ADSC- or other mesenchymal stem cell-derived exosomes in both
Representative studies of exosome-based therapies for photoaging: mechanistic pathways and outcomes
| Author | Year | Primary skin outcomes | Mechanistic pathway | EV/Exosome source |
| Liu et al.[52] | 2023 | Vascular injury ↓; collagen degradation ↓; inflammation ↓ | GLRX5 delivery and ferroptosis inhibition | hADSCs |
| Wang et al.[53] | 2025 | - | Hypothesis: GSH/ROS pathway | hADSCs |
| Wang et al.[11] | 2025 | Epidermal thickness ↓; collagen fibers ↑ (Masson); skin damage ↓ | PINK1/Parkin-mediated mitophagy pathway | hADSCs |
| Gao et al.[54] | 2023 | MMPs production ↓; procollagen type I synthesis ↑; epidermal thickening ↓; collagen degradation ↓ | LncRNA H19/miR-138/SIRT1 axis (gain) | ADSCs |
| Wang et al.[55] | 2020 | Skin injury ↓ (oxidative stress) | NRF2 defense system | MSCs |
| Wu et al.[56] | 2021 | - Skin inflammation ↓ - Skin cell regeneration ↑ - DNA damage markers (γH2AX, 8-OHDG) ↓ - Redness ↓ - Scaling ↓ - Inflammatory cell infiltration ↓ | 14-3-3ζ/SIRT1 axis (gain) | hucMSCs |
| Zhang et al.[57] | 2022 | SA-β-gal activity ↓; collagen synthesis ↑; elastin synthesis ↑; MMP-1 ↓; MMP-3 ↓; cathepsin K ↓ | circ_0011129/miR-6732-5p axis (gain); p53 signaling pathway activation | hADSCs |
Critical appraisal of these representative studies reveals several patterns and gaps that should inform future research directions. Regarding experimental models, the majority of investigations employed acute UVB exposure paradigms (single or short-term repeated irradiation) rather than chronic photoaging models, limiting the extrapolation of findings to the clinical reality of cumulative decades-long sun exposure. Sample sizes in preclinical studies were generally modest (typically n = 3-6 per group for in vivo experiments), and long-term follow-up data beyond 4-8 weeks post-treatment are notably absent, leaving questions about the durability of exosome-mediated effects unanswered. From a mechanistic standpoint, the studies demonstrate considerable heterogeneity in proposed pathways, ranging from canonical transforming growth factor-beta (TGF-β)/Smad and mitogen-activated protein kinase (MAPK)/activator protein 1 (AP-1) signaling to more recently implicated pathways including sirtuin 1 (SIRT1)-mediated deacetylation, nuclear factor erythroid 2-related factor 2 (Nrf2)-dependent antioxidant responses, and mitophagy regulation. Strikingly, ferroptosis-related mechanisms remain underexplored in the exosome-photoaging literature; among the representative studies summarized in Table 1, only entry 1 (Liu et al.[52]) directly investigated ferroptosis inhibition, while entry 2 (Wang et al.[53]) examined the related GSH/ROS pathway. This underrepresentation suggests that ferroptosis modulation may be an underexplored aspect of exosome-mediated photoprotection that warrants systematic investigation. The exosome sources also vary substantially - while ADSC-Exos predominate, several studies employed umbilical cord mesenchymal stromal/stem cells (MSCs), bone marrow MSCs, hair follicle MSCs, dermal fibroblasts, and even plant-derived nanovesicles, complicating direct comparisons across studies. Standardization of cell culture conditions, exosome isolation methods [ultracentrifugation vs. size exclusion vs. tangential flow filtration (TFF)], and characterization protocols according to MISEV guidelines would greatly enhance the reproducibility and comparability of findings in this rapidly evolving field.
Importantly for anti-ferroptosis mechanisms, hypoxia-preconditioned ADSC-Exos can deliver circ-Ash1l, which sponges miR-700-5p to upregulate GPX4, a core ferroptosis-suppressing enzyme. Together with established evidence that GPX4 activity prevents ferroptosis by reducing lipid hydroperoxides, these data support a ferroptosis-modulating role for ADSC-Exos under UV stress[58].
Exo-circRNAs and the circ-Ash1l/miR-700-5p/GPX4 axis
A rapidly emerging area in exosome research is the functional role of circRNAs. These non-coding RNAs are characterized by a covalently closed loop lacking 5’ and 3’ ends, which contributes to resistance to exonucleolytic decay and confers remarkable stability compared with linear RNAs[59,60]. CircRNAs arise via back-splicing, in which a downstream splice donor is joined to an upstream splice acceptor to produce a circular transcript[61]. Large-scale transcriptomic surveys established that circRNAs are broadly expressed and conserved in mammals and can be abundant relative to their linear counterparts in specific tissues (notably brain)[62]. Functionally, many circRNAs act as competing endogenous RNAs (ceRNAs) or “sponges” for microRNAs - classically exemplified by circular RNA sponge for miR-7/cerebellar degeneration-related protein 1 antisense (ciRS-7/CDR1as) that sequesters miR-7 and derepresses its targets[63]. From a kinetic standpoint, endogenous circRNAs show prolonged lifetimes (population averages around ~20-24 h, with several species reaching ≥ 48 h), consistent with their general resistance to exonucleases[60].
Exosomes (sEVs, per MISEV terminology) are conspicuously enriched for circRNAs. RNA sequencing (RNA-seq) analyses demonstrated that exo-circRNAs are increased by at least ~2-fold in exosomes relative to producer cells, with circRNA:linear RNA ratios also elevated (reported up to ~6-fold), indicating selective incorporation and/or retention[64,65]. Moreover, the lipid bilayer shields exosomal RNAs from RNase degradation, promoting stability in biofluids and efficient cargo delivery to recipient cells[66,67]. These properties make exo-circRNAs attractive candidates for intercellular communication and therapeutic delivery, although current guidelines (MISEV2023) recommend precise EV characterization and cautious use of the term “exosome” unless biogenesis is demonstrated[68].
In the context of cutaneous photoaging, hypoxia-preconditioned adipose-derived stem cell exosomes (HExo) mitigate UV-induced skin injury by delivering a specific circRNA, circ-Ash1l[15]. In vitro and in vivo, HExo treatment reversed UVB-provoked oxidative stress and inflammatory readouts and alleviated tissue injury metrics; high-throughput sequencing identified circ-Ash1l as a key effector enriched in HExo; and downregulating circ-Ash1l attenuated these therapeutic benefits[15]. Mechanistically, luciferase reporter assays and perturbation studies support a circ-Ash1l/miR-700-5p/GPX4 axis in which circ-Ash1l acts as a molecular sponge for miR-700-5p, thereby sustaining GPX4 expression and constraining ferroptosis-linked lipid peroxidation[15]. Notably, miR-700-5p is annotated exclusively in mouse (Mir700; MGI:3629655), with no direct human ortholog identified to date[69]. This species restriction has important implications for clinical translation of the circ-Ash1l-mediated anti-ferroptotic mechanism. While the circ-Ash1l/miR-700-5p/GPX4 axis provides compelling proof-of-concept that exo-circRNAs can modulate ferroptosis in photoaged skin, the precise molecular circuitry in human cells likely involves different microRNA (miRNA) intermediaries.
Several human miRNAs have been validated to directly target GPX4 through binding to its 3’ untranslated region (3’UTR), including miR-214-3p[70], miR-324-3p[71], and miR-182-5p[72], though their expression patterns in UV-exposed human skin and their amenability to circRNA sponging remain largely unexplored. Future studies should prioritize identifying human-relevant circRNA/miRNA axes that converge on ferroptosis-regulatory nodes. This knowledge gap does not diminish the therapeutic potential of ADSC-Exos for human photoaging. Indeed, the cargo diversity of exosomes suggests that they likely deliver multiple bioactive molecules beyond circ-Ash1l. However, the specific molecular mechanisms validated in murine models require independent verification in human systems before extrapolation to clinical applications.
These mechanistic links are biologically coherent with ferroptosis biochemistry: GPX4 is the principal enzyme that detoxifies phospholipid hydroperoxides in membranes using glutathione, and downregulating GPX4 - or blocking cystine uptake/GSH synthesis upstream - licenses ferroptotic death; conversely, sustaining GPX4 curbs ferroptosis[73]. Thus, elevating GPX4 via the circ-Ash1l/miR-700-5p axis offers a plausible route by which HExo blunt UVB-exacerbated lipid peroxidation and tissue damage in skin[15,73,74].
Experimental validation spans gain- and loss-of-function across the axis. Overexpressing circ-Ash1l increases GPX4 and reduces oxidative stress markers under UVB; knockdown of circ-Ash1l diminishes HExo-mediated protection; miR-700-5p elevation impairs, whereas miR-700-5p inhibition rescues, GPX4-linked readouts; and luciferase reporter analysis demonstrates the regulatory relationship between miR-700-5p and GPX4 within the axis[15]. Collectively, these lines of evidence implicate the circ-Ash1l/miR-700-5p/GPX4 pathway as a mechanistic contributor to how ADSC-Exos suppress ferroptosis-associated injury in photoexposed skin[15,73,74].
Beyond circ-Ash1l, additional exo-circRNAs are being explored as anti-photoaging payloads. For example, human ADSC-Exos engineered (or enriched via 3D culture) to carry circular RNA hsa_circ_0011129 (circ_0011129) enhanced anti-photoaging efficacy in UV models, reducing senescence markers [senescence-associated β-galactosidase (SA-β-gal); p53/cyclin-dependent kinase inhibitor 1A (p21)/cyclin-dependent kinase inhibitor 2A (p16)] and restoring matrix components (collagen I, elastin) more robustly than unloaded exosomes, highlighting how circRNA cargo selection can potentiate sEV-based interventions[75]. These and related studies across wound repair and dermatologic indications underscore a broader paradigm wherein exo-circRNAs orchestrate redox balance, extracellular matrix homeostasis, and stress responses in skin biology[67,75].
Consistent with MISEV2018/2023, future work should (i) report EV isolation/characterization in detail; (ii) validate circRNA identity (RNase R resistance, back-splice junction specificity) and functional readouts with appropriate controls [e.g., small interfering RNA (si)/antisense oligonucleotide (ASO) targeting the back-splice junction, rescue with miRNA antagomiR]; and (iii) acknowledge species-specificity (e.g., mmu-miR-700-5p) when extrapolating to human contexts[68,69].
THERAPEUTIC APPLICATIONS OF ADSC-DERIVED EXOSOMES
Engineering strategies to enhance exosome therapy
While native ADSC-Exos show promise, researchers are actively exploring engineering strategies to improve efficacy, targeting, and clinical translatability, typically grouped into parental cell modifications (preconditioning/priming), cargo-loading techniques, and surface engineering [Figure 2][76,77].
Figure 2. Workflow for engineering and administration of autologous MSC/ADSC-derived exosomes. (1) Collection of autologous progenitor/stromal cells from the donor; (2) Cell preparation/expansion and optional lineage priming to enable exosome production; (3) Exosome production and purification from conditioned medium; (4) Cargo loading and/or surface engineering of exosomes (e.g., miRNA/siRNA loading; expression or conjugation of tissue-specific membrane ligands); (5) Quality control and administration: vesicles are characterized for size/count and markers, then formulated and delivered according to the intended route. Created with BioRender.com. MSC: Mesenchymal stem cell; ADSC: adipose-derived stem cell; miRNA: microRNA; siRNA: small interfering RNA; HSC: hematopoietic stem cell.
Preconditioning and priming of parent cells
The composition and potency of exosomes depend on the physiological state of the producer cells; subjecting ADSCs/MSCs to defined cues can remodel exosomal cargo and enhance therapeutic properties[76]. Hypoxic preconditioning of human ADSC-Exos upregulates circ-Ash1l and protects UVB-challenged dermal fibroblasts via the circ-Ash1l/miR-700-5p/GPX4 axis[15]. Beyond hypoxia, immunological priming with cytokines can imprint immunomodulatory features: interferon-gamma (IFN-γ) increases miR-125a/miR-125b levels in MSC-Exos and augments anti-inflammatory efficacy in colitis models, while combined IFN-γ/tumor necrosis factor-alpha (TNF-α)/interleukin-1 beta (IL-1β) priming elevates programmed cell death protein 1 (PD-1) ligands on EVs and improves outcomes in graft-versus-host disease (GVHD) models[78,79]. IL-1β-primed MSC-Exos are enriched in let-7c and suppress p21-activated kinase 1 (PAK1)/nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) to drive macrophage M2 polarization, thereby reducing fibrosis[80]. Growth-factor priming can also skew exosomal content toward matrix-supportive effects; EVs released by TGF-β-stimulated MSCs modulate dermal fibroblast functions relevant to skin aging and ECM remodeling[81]. Small-molecule priming provides further control: curcumin-primed ADSC small EVs show enhanced chondro-protection with reduced oxidative stress and apoptosis, and histone deacetylase (HDAC)-inhibitor (valproate) preconditioning alters the MSC secretome in ways consistent with increased therapeutic activity[82,83]. Three-dimensional culture (spheroids/scaffolds) likewise reshapes exosome/secretome profiles; 3D-cultured MSCs secrete EVs enriched for pro-angiogenic factors and angiogenesis-related miRNAs compared with 2D cultures[84,85]. Finally, partially differentiating MSCs toward an endothelial-like lineage before harvesting exosomes can yield vesicles enriched in pro-angiogenic miRNAs and ephrin A1 (EFNA1), thereby enhancing endothelial activation and tissue repair[86].
Direct cargo loading
In addition to modifying the parent cells, exosomes can be directly loaded with therapeutic molecules post-isolation. This approach allows precise control over the delivered cargo, and commonly used post-loading techniques include electroporation, sonication, and membrane-permeabilizing agents such as saponin[87,88].
Electroporation applies brief electric pulses that transiently permeabilize exosome membranes, enabling entry of exogenous nucleic acids or small molecules; it has been widely used to load small interfering RNA (siRNAs) or miRNA mimics into exosomes for functional gene silencing in recipient cells[88]. In oncology models, exosomes electroporated with antitumor or anti-inflammatory miRNAs/siRNAs have shown target gene knockdown and therapeutic effects[88]. In dermatologic applications, analogous miRNA-loading strategies are being explored for skin disease models[88]. However, electroporation can have limitations: high electric fields may compromise vesicle integrity and can induce aggregation/precipitation of RNA, and loading efficiency can vary across preparations[87,89].
Sonication has been explored as an alternative that transiently pores membranes and can achieve high loading efficiency for certain cargos, though excessive sonication may disrupt vesicle structure or reduce vesicle yield[90]. Detergent-based methods (e.g., low concentrations of saponin) create reversible pores by interacting with membrane cholesterol, facilitating cargo entry but requiring careful optimization to balance loading with vesicle integrity and cytotoxicity[91].
Another approach is to use cell-penetrating peptides (CPPs) or hydrophobic anchors conjugated to the therapeutic molecule to promote association with, or insertion into, the exosome membrane or lumen via spontaneous membrane interactions during simple incubation; cholesterol-conjugated siRNAs and cell-penetrating peptide (CPP)-polyethylene glycol (PEG)-lipids are representative strategies that enable EV loading under mild conditions[92,93]. These strategies typically require conjugation chemistry and are not universally applicable to all cargos[92,93].
An alternative to post-isolation loading is genetic/producer-cell engineering, in which exosome-producing cells are engineered to enrich specific cargos or display targeting moieties so that they are packaged during biogenesis. For example, fusing therapeutic or targeting peptides to exosome-associated proteins such as lysosome-associated membrane protein 2B (LAMP2B) or to the C1C2 domain of lactadherin can enrich those peptides/proteins in exosomes and direct them to desired targets[87,94,95]. Endogenous (cell-based) loading inherently modifies the EV-producing cells to incorporate therapeutic agents during natural EV biogenesis[87].
However, the optimal loading strategy for anti-photoaging applications has not been systematically compared, and whether post-isolation loading alters the intrinsic therapeutic properties of native ADSC-Exos (potentially disrupting endogenous bioactive cargos) remains an open question. Despite the absence of systematic head-to-head comparisons for photoaging applications, general principles can guide strategy selection. Preconditioning approaches (see Section “Preconditioning and Priming of Parent Cells”) offer the advantage of preserving native vesicle integrity and leveraging endogenous sorting mechanisms, but provide limited control over specific cargo enrichment and require optimization for each therapeutic target. Post-isolation loading methods such as electroporation and sonication enable precise cargo selection and higher loading efficiency for exogenous molecules, yet may compromise membrane integrity, induce cargo aggregation, or disrupt endogenous bioactive components. Surface modification strategies (see Section “Surface Modification and Targeting”) can enhance targeting specificity and cellular uptake but add manufacturing complexity and potential immunogenicity concerns. For clinical translation of anti-photoaging exosome therapies, the choice of engineering strategy will likely depend on the specific therapeutic goal, with preconditioning favored for applications leveraging native ADSC-Exo properties and post-isolation loading preferred when delivery of defined molecular payloads is required.
Surface modification and targeting
To improve the specificity and efficiency of exosome uptake by target cells, surface engineering strategies are being developed. One approach is to decorate the exosome surface with targeting ligands that bind receptors enriched on the desired cell type. For instance, engineering exosomes to display an epidermal growth factor receptor (EGFR)-binding peptide (GE11) or integrin-binding peptides such as internalized Arg-Gly-Asp (iRGD, a tumor-penetrating cyclic peptide) has yielded preferential uptake by the intended cancer cells and greater tumor accumulation in vivo compared with unmodified vesicles[96-98]. This has been demonstrated in cancer models, where peptide-displaying exosomes showed selective delivery and antitumor effects over non-targeted controls[96-98].
Surface modification can be performed post-isolation via bio-orthogonal coupling (e.g., copper-free click chemistry) or high-affinity biotin-streptavidin bridges, by first functionalizing the exosomal membrane and then conjugating a ligand[99-101]. Alternatively, lipophilic anchors - such as cholesterol-modified oligonucleotide/aptamer tethers - can spontaneously insert into the exosome lipid bilayer and display the attached ligand on the surface, enabling modular retargeting without altering parental cells[102].
For topical skin applications, additional modifications and formulations that enhance traversal of the stratum corneum are often required. Chemical penetration enhancers used in dermatologic formulations (e.g., ethanol, propylene glycol) and skin-penetrating peptides have been shown to transiently increase skin permeability, and are commonly leveraged in topical delivery systems[103-105]. Current evidence suggests that intact stratum corneum significantly limits the deep penetration of free exosomes; therefore, pairing exosomes with penetration enhancers, microneedles, or hydrogel/ethosomal vehicles is typically needed to increase dermal exposure[106].
While these targeting strategies show promise in oncology applications, their utility for topical skin applications is less clear, given that the primary barrier to exosome efficacy in photoaging treatment is likely stratum corneum penetration rather than cell-type selectivity within the dermis.
Lyophilization and formulation
For clinical and commercial viability, exosomes need to be stable during storage and easy to handle. Lyophilization (freeze-drying) with disaccharide cryoprotectants (e.g., trehalose, sucrose) helps preserve vesicle integrity and activity upon reconstitution[107-109]. Studies report that appropriately lyophilized EVs retain size/markers and functional activity after storage at room temperature for weeks to months and at refrigerated temperatures, supporting development of off-the-shelf products[107-109].
Formulation into delivery vehicles is another key consideration. For topical use, exosomes can be incorporated into hydrogels or related semisolid bases that protect vesicles and sustain release; for injection-based treatments (e.g., intradermal/subcutaneous), biocompatible depots such as hyaluronic-acid or alginate-based hydrogels can prolong local retention and reduce dosing frequency relative to free exosomes in saline[110,111].
Scaling up production
For exosome therapies to be practical, especially for topical cosmetic use which could demand large volumes, manufacturing processes must be scalable and cost-effective. Traditional ultracentrifugation, while effective at lab scale, is less suitable for scale-up and often underperforms in yield and time/cost/scalability compared with newer methods. Instead, techniques such as TFF and size-exclusion chromatography (SEC) are being employed for scalable exosome purification [Figure 3][112-115]. These methods can process large volumes of culture supernatant and have been shown to remove abundant soluble protein contaminants (for example, albumin and lipoprotein complexes) while retaining vesicle integrity[112,113,115,116]. A combination workflow - using TFF for volume reduction followed by SEC for fine purification - is increasingly adopted in EV manufacturing[114,117].
Figure 3. Transdermal delivery and scalable manufacturing/quality control of ADSC-derived exosomes. Left: A coated microneedle patch breaches the stratum corneum and deposits exosomes into the viable epidermis/papillary dermis for uptake by keratinocytes and fibroblasts. Right (workflow): (1) Culture/priming of ADSCs; (2) expansion in a microcarrier or hollow-fiber bioreactor; (3) TFF for clarification and volume reduction (e.g., 300 kDa cassette); (4) size-exclusion chromatography (SEC/qEV or FPLC) for polishing; (5) lyophilization of the exosome-coated microneedle formulation with protectants (e.g., trehalose/sucrose ± surfactant); (6) labeled storage (2-8 °C liquid or -80 °C frozen). Quality control is applied across steps, including NTA (size/count), TEM, EV markers (CD63/CD81/CD9, TSG101), and assessment of protein/lipoprotein carryover. Created with BioRender.com. ADSC: Adipose-derived stem cell; TFF: tangential flow filtration; SEC: size-exclusion chromatography; FPLC: fast protein liquid chromatography; NTA: nanoparticle tracking analysis; TEM: transmission electron microscopy; EV: extracellular vesicle; CD: cluster of differentiation; TSG101: tumor susceptibility gene 101.
Upstream, shifting from static 2D cultures to bioreactor systems (such as microcarrier-based stirred-tank or hollow-fiber/perfusion platforms) can substantially increase EV output per run. For MSCs, microcarrier spinner/3D systems and TFF have delivered ≥ 3-fold increases in particle yield (e.g., 7-fold over 3D-UC and ~20-fold over 2D-UC), while maintaining characteristic EV markers and strong bioactivity relative to conventional preparations[118]. Bioreactor platforms also offer controlled operation (continuous perfusion/feed and process control) that facilitates consistency across batches[115].
However, scale-up introduces quality considerations such as hydrodynamic/shear stress in bioreactors or during filtration that may affect EV properties; processes should be tuned to minimize shear (e.g., gentle TFF settings and streamlined processing) to help preserve vesicle integrity[119].
CLINICAL TRANSLATION AND REGULATORY CONSIDERATIONS
Regulatory and safety considerations
The regulatory landscape for exosome therapeutics is still evolving. As of 2024, no exosome-based therapy has received FDA approval[120]. Regulatory agencies such as the Food and Drug Administration (FDA) currently classify human-derived exosomes intended for therapeutic use as drugs/biological products, subject to stringent requirements for safety, purity, and potency testing[120]. The lack of a clear, universally adopted categorization creates complexity in determining whether exosomes represent cell therapy, gene therapy, or a novel class altogether, which can complicate the approval pathway[120].
In the European Union (EU), debate continues regarding whether exosome products fall under the definition of an Advanced Therapy Medicinal Product (ATMP) or as a biological medicinal product, depending on their source/engineering status[120]. If exosomes are loaded with recombinant nucleic acids or drugs, they may fall under the EU definition of a gene therapy medicinal product, requiring demonstration of safety for all components[120]. Regulatory bodies emphasize issues such as immunogenicity, biodistribution, and long-term safety of exosome treatments[120].
While ADSC-Exos are often described as low-immunogenic - e.g., lacking major histocompatibility complex (MHC) class II and with low MHC class I - repeated or systemic administration still necessitates comprehensive toxicity evaluations[121]. Key safety questions include risks of unintended coagulation or complement activation, off-target biodistribution, and potential systemic effects even after topical application. Current evidence suggests exosomes are largely cleared by macrophages and accumulate primarily in the liver/spleen after systemic dosing, with generally favorable toxicity profiles in animals; however, rigorous acute/chronic dosing studies remain essential[122-124].
Tumorigenicity is another consideration. Although exosomes cannot divide, they carry nucleic acids and proteins; quality control must ensure they do not transfer oncogenic cargo or promote a pro-tumor microenvironment[125]. Studies report both anti-tumor and pro-tumor effects of MSC exosomes depending on context, underscoring the need to characterize RNA/DNA cargo carefully during manufacturing[125-127].
Regulators will require comprehensive Chemistry, Manufacturing, and Controls (CMC) documentation to ensure each batch meets defined criteria (e.g., size distribution, identity/surface markers, sterility, and acceptable endotoxin levels) and that production is reproducible under Good Manufacturing Practice (GMP)[120,128]. Given these hurdles, developing standardized production protocols and reference materials is crucial. International initiatives led by International Society for Extracellular Vesicles (ISEV) (e.g., MISEV2023 guidance and EV reference-material efforts) are establishing frameworks that aid regulatory standardization[68].
The absence of approved exosome therapeutics as of 2024 reflects not only regulatory uncertainty but also the inherent challenges of standardizing a biologically complex product; until these issues are resolved, the clinical translation of ADSC-Exos for photoaging will remain constrained regardless of preclinical efficacy.
Future directions and clinical perspectives
Looking ahead, several perspectives can be outlined for advancing exosome-based photoaging treatment. Well-designed clinical trials are needed to rigorously assess the efficacy of ADSC-Exos treatments in human photoaging[129]. Early clinical applications have already appeared in aesthetic dermatology as adjuvants to procedures (e.g., microneedling or fractional laser), where split-face randomized trials reported benefit when exosome formulations were added to standard care[130,131]. Beyond these adjuvant applications, Wan et al. recently reported a prospective study evaluating exosome therapy combined with microneedling for androgenetic alopecia, demonstrating a mean increase of 35 hairs/cm2 at 12-month follow-up with high patient satisfaction[132]. Although targeting a different indication, this study exemplifies the methodological rigor - objective outcome measures, standardized protocols, and adequate follow-up - needed to advance exosome therapeutics toward clinical validation in photoaging contexts. Initial studies can test an exosome serum or mask aimed at improving visible photoaging signs in adults with moderate facial photoaging[130].
Endpoints should include objective measures - standardized photography/3D imaging and wrinkle grading, biomechanical skin elasticity (e.g., Cutometer), and histologic assessment of dermal collagen from biopsies - together with patient- or investigator-reported outcomes on texture, hydration, and pigmentation[130,131]. Such studies can also explore dosing schedules (e.g., session intervals over 8-12 weeks) and quantify whether exosomes yield tangible cosmetic benefits[130,131]. If successful, subsequent trials could evaluate medical outcomes related to chronic photodamage (for example, prevention of precancerous changes such as actinic keratoses)[47].
Customizing exosome formulations for distinct photoaging phenotypes is a logical direction. An anti-wrinkle product could be enriched for factors that stimulate fibroblast collagen synthesis[133,134], whereas an anti-pigmentation product might incorporate miRNAs that suppress melanogenesis pathways in melanocytes[135,136]. This framework enables personalized exosome-based regimens targeting wrinkles, laxity, or dyschromia[133-136].
Combining exosome therapy with barrier-disruptive modalities may yield synergistic effects. In practice, fractional lasers and microneedling are used to induce dermal remodeling; applying ADSC-Exos solutions immediately after these procedures, when micro-channels are open and repair programs are activated, has improved outcomes in randomized split-face studies and reduced post-procedure erythema/downtime[130,131]. Mechanistically, topical exosomes have limited penetration through an intact stratum corneum and mainly act within that layer; micro-channeling (microneedling) facilitates delivery to viable epidermis/dermis and may thereby enhance efficacy[137].
From a mechanistic research perspective, further work should map how exosome cargo intersects aging pathways and which skin cell types are principal recipients in vivo after topical/adjuvant use. Open questions remain regarding relative targeting of keratinocytes, fibroblasts, and immune cells in photodamaged skin; current evidence shows extensive EV-mediated crosstalk among these compartments but requires deeper in situ resolution in humans[138-140].
It is also critical to confirm that effects observed in acute/subacute UV models translate to chronic photoaging. Many mechanistic studies - including those on the circ-Ash1l/miR-700-5p/GPX4 axis - have used acute UVB injury models; chronic photoaging instead reflects decades of repetitive low-dose UV exposure with cumulative repair failure[15,47]. Exosomal interventions will likely need sustained administration and concomitant photoprotection to impact chronically photoaged skin[47]. In acute UV models, hypoxia-primed ADSC-exosomes protect via delivery of circ-Ash1l and downstream ferroptosis control (miR-700-5p/GPX4)[15]; whether similar temporal dynamics operate during long-term photoaging requires testing in extended animal or ex vivo human-skin models[15,47].
Limitations of current evidence
Several limitations of the current literature should be acknowledged when interpreting the therapeutic potential of ADSC-Exos for photoaging.
Regarding experimental model fidelity, the overwhelming majority of mechanistic studies employ acute, high-dose UVB exposure paradigms that induce rapid cellular damage within hours to days. This contrasts sharply with human photoaging, which develops insidiously over decades of cumulative sub-erythemal UV exposure, allowing for compensatory adaptations and repair processes that may fundamentally alter the pathophysiological landscape. Whether the molecular signatures and therapeutic responses observed in acute injury models translate to chronically photoaged human skin remains an assumption rather than an established fact. Additionally, murine skin differs substantially from human skin in thickness, hair follicle density, and immune cell composition, potentially limiting the predictive validity of mouse photoaging models.
From a mechanistic standpoint, the circ-Ash1l/miR-700-5p/GPX4 axis - while providing elegant proof-of-concept for circRNA-mediated ferroptosis regulation - derives primarily from a single research group and awaits independent replication. The species-specificity of miR-700-5p (present in mouse but absent in humans) necessitates identification of functionally analogous human miRNA targets before this pathway can be therapeutically exploited in clinical settings. More broadly, the relative contribution of ferroptosis vs. other cell death modalities (apoptosis, necroptosis, pyroptosis) to photoaging pathology has not been quantitatively established, making it difficult to predict the clinical impact of ferroptosis-selective interventions.
Methodological heterogeneity across studies also complicates evidence synthesis. Exosome isolation techniques (ultracentrifugation, size exclusion chromatography, TFF, precipitation kits) yield preparations of varying purity and bioactivity, yet many studies do not report characterization data consistent with MISEV2023 guidelines. This variability, combined with differences in cell culture conditions, passage numbers, and preconditioning protocols, may account for some of the inconsistencies in reported outcomes and limits the comparability of findings across laboratories.
Finally, clinical evidence remains sparse. The few published human studies are limited to small, short-term, split-face trials evaluating exosomes as adjuncts to microneedling or laser procedures, with follow-up periods typically extending only 8-12 weeks. Long-term efficacy, durability of effects, optimal dosing regimens, and safety profiles with repeated administration have not been systematically evaluated in human subjects.
CONCLUSION
ADSC-Exos have emerged as a cutting-edge biologic approach for skin rejuvenation, leveraging the naturally packaged toolkit of regenerative molecules from stem cells. They address multiple hallmarks of photoaging including oxidative damage, matrix degradation, inflammation, and ferroptosis in a coordinated manner that single-molecule drugs might struggle to achieve. Challenges remain in production scalability, delivery optimization, and regulatory approval, and while ongoing advancements in bioengineering and standardization are promising, significant hurdles must still be addressed before clinical translation can be realized. Realizing the therapeutic potential of ADSC-Exos will require rigorously validating safety and efficacy through well-designed clinical trials, aligning manufacturing processes with regulatory expectations, and establishing standardized protocols for exosome characterization and quality control[141]. If these challenges can be overcome, patients may eventually benefit from non-invasive, cell-free regenerative treatments that ameliorate photodamaged skin, improving both cosmetic appearance and skin health and resilience.
As the field moves forward, interdisciplinary collaboration between cell biologists, bioengineers, dermatologists, and regulatory experts will be essential for translating the promise of exosomal regenerative therapy into widely accessible treatment for photoaging and other degenerative skin conditions.
DECLARATIONS
Authors’ contributions
Conceptualization, writing - original draft, visualization, project administration: Shi X
Literature review, data curation, writing - review and editing: Ji J
Literature review, writing - review and editing: Cheng W
Data curation, writing - review and editing: Pang Q
Writing - review and editing, supervision: Lin C
Conceptualization, supervision, funding acquisition: Wei P
All authors read and approved the final manuscript.
Availability of data and materials
Not applicable.
AI and AI-assisted tools statement
During the preparation of this manuscript, the AI-assisted image generation tool Nanobanana Pro was used solely for generating the Graphical Abstract. The tool did not influence the study design, data collection, analysis, interpretation, or the scientific content of the work. All authors reviewed and finalized the generated content and take full responsibility for the accuracy, integrity, and originality of the manuscript.
Financial support and sponsorship
This research was supported by Zhejiang Provincial Natural Science Foundation of China (Grant No. LQN26H110003).
Conflicts of interest
All authors declared that there are no conflicts of interest.
Ethical approval and consent to participate
Not applicable.
Consent for publication
Not applicable.
Copyright
© The Author(s) 2026.
REFERENCES
1. Wong QYA, Chew FT. Defining skin aging and its risk factors: a systematic review and meta-analysis. Sci Rep. 2021;11:22075.
2. Fisher GJ, Wang ZQ, Datta SC, Varani J, Kang S, Voorhees JJ. Pathophysiology of premature skin aging induced by ultraviolet light. N Engl J Med. 1997;337:1419-28.
3. Krutmann J, Schikowski T, Morita A, Berneburg M. Environmentally-Induced (Extrinsic) Skin Aging: Exposomal Factors and Underlying Mechanisms. J Invest Dermatol. 2021;141 Suppl:1096-103.
4. Zhang S, Duan E. Fighting against Skin Aging: The Way from Bench to Bedside. Cell Transplant. 2018;27:729-38.
5. Kim DJ, Iwasaki A, Chien AL, Kang S. UVB-mediated DNA damage induces matrix metalloproteinases to promote photoaging in an AhR- and SP1-dependent manner. JCI Insight. 2022:7.
6. Vats K, Kruglov O, Mizes A, et al. Keratinocyte death by ferroptosis initiates skin inflammation after UVB exposure. Redox Biol. 2021;47:102143.
7. Zhang PC, Hong Y, Zong SQ, et al. Variation of Ferroptosis-Related Markers in HaCaT Cell Photoaging Models Induced by UVB. Clin Cosmet Investig Dermatol. 2023;16:3147-55.
8. Teng Y, Tang H, Tao X, Huang Y, Fan Y. Ferrostatin 1 ameliorates UVB-induced damage of HaCaT cells by regulating ferroptosis. Exp Dermatol. 2024;33:e15018.
9. Tang D, Chen X, Kang R, Kroemer G. Ferroptosis: molecular mechanisms and health implications. Cell Res. 2021;31:107-25.
10. Xu P, Xin Y, Zhang Z, et al. Extracellular vesicles from adipose-derived stem cells ameliorate ultraviolet B-induced skin photoaging by attenuating reactive oxygen species production and inflammation. Stem Cell Res Ther. 2020;11:264.
11. Wang Y, Liao W, Wang Y, et al. Human adipose-derived stem cell exosomes reduce mitochondrial DNA common deletion through PINK1/Parkin-mediated mitophagy to improve skin photoaging. Stem Cell Res Ther. 2025;16:365.
12. Liang C, Yi Y, Li J, et al. Unveiling exosomes in combating skin aging: insights into resources, mechanisms and challenges. Stem Cell Res Ther. 2025;16:474.
13. Park SY, Yi KH. Exosome-mediated advancements in plastic surgery: navigating therapeutic potential in skin rejuvenation and wound healing. Plast Reconstr Surg Glob Open. 2024;12:e6021.
14. Zhang F, Jiang J, Qian H, Yan Y, Xu W. Exosomal circRNA: emerging insights into cancer progression and clinical application potential. J Hematol Oncol. 2023;16:67.
15. Zha J, Pan Y, Liu X, Zhu H, Liu Y, Zeng W. Exosomes from hypoxia-pretreated adipose-derived stem cells attenuate ultraviolet light-induced skin injury via delivery of circ-Ash1l. Photodermatol Photoimmunol Photomed. 2023;39:107-15.
16. Gromkowska-Kępka KJ, Puścion-Jakubik A, Markiewicz-Żukowska R, Socha K. The impact of ultraviolet radiation on skin photoaging - review of in vitro studies. J Cosmet Dermatol. 2021;20:3427-31.
17. Langton AK, Ayer J, Griffiths TW, et al. Distinctive clinical and histological characteristics of atrophic and hypertrophic facial photoageing. J Eur Acad Dermatol Venereol. 2021;35:762-8.
18. Boyd AS, Naylor M, Cameron GS, Pearse AD, Gaskell SA, Neldner KH. The effects of chronic sunscreen use on the histologic changes of dermatoheliosis. J Am Acad Dermatol. 1995;33:941-6.
19. Widgerow AD, Napekoski K. New approaches to skin photodamage histology-Differentiating ‘good’ versus ‘bad’ Elastin. J Cosmet Dermatol. 2021;20:526-31.
20. Bernerd F, Passeron T, Castiel I, Marionnet C. The damaging effects of long UVA (UVA1) rays: a major challenge to preserve skin health and integrity. Int J Mol Sci. 2022;23:8243.
21. Cadet J, Douki T, Ravanat JL. Oxidatively generated damage to cellular DNA by UVB and UVA radiation. Photochem Photobiol. 2015;91:140-55.
22. Lawrence KP, Douki T, Sarkany RPE, Acker S, Herzog B, Young AR. The UV/visible radiation boundary region (385-405 nm) damages skin cells and induces “dark” cyclobutane pyrimidine dimers in human skin in vivo. Sci Rep. 2018;8:12722.
23. Fisher GJ, Datta SC, Talwar HS, et al. Molecular basis of sun-induced premature skin ageing and retinoid antagonism. Nature. 1996;379:335-9.
24. Koivukangas V, Kallioinen M, Autio-Harmainen H, Oikarinen A. UV irradiation induces the expression of gelatinases in human skin in vivo. Acta Derm Venereol. 1994;74:279-82.
25. Quan T, Qin Z, Xia W, Shao Y, Voorhees JJ, Fisher GJ. Matrix-degrading metalloproteinases in photoaging. J Investig Dermatol Symp Proc. 2009;14:20-4.
26. Fisher GJ, Datta S, Wang Z, et al. c-Jun-dependent inhibition of cutaneous procollagen transcription following ultraviolet irradiation is reversed by all-trans retinoic acid. J Clin Invest. 2000;106:663-70.
27. Green AC, Hughes MC, McBride P, Fourtanier A. Factors associated with premature skin aging (photoaging) before the age of 55: a population-based study. Dermatology. 2011;222:74-80.
28. Seité S, Zucchi H, Moyal D, et al. Alterations in human epidermal Langerhans cells by ultraviolet radiation: quantitative and morphological study. Br J Dermatol. 2003;148:291-9.
29. Dumay O, Karam A, Vian L, et al. Ultraviolet AI exposure of human skin results in Langerhans cell depletion and reduction of epidermal antigen-presenting cell function: partial protection by a broad-spectrum sunscreen. Br J Dermatol. 2001;144:1161-8.
30. Norval M, Halliday GM. The consequences of UV-induced immunosuppression for human health. Photochem Photobiol. 2011;87:965-77.
32. Gilchrest BA, Blog FB, Szabo G. Effects of aging and chronic sun exposure on melanocytes in human skin. J Invest Dermatol. 1979;73:141-3.
33. Le J, Meng Y, Wang Y, et al. Molecular and therapeutic landscape of ferroptosis in skin diseases. Chin Med J. 2024;137:1777-89.
34. Dixon SJ, Lemberg KM, Lamprecht MR, et al. Ferroptosis: an iron-dependent form of nonapoptotic cell death. Cell. 2012;149:1060-72.
35. Battaglia AM, Chirillo R, Aversa I, Sacco A, Costanzo F, Biamonte F. Ferroptosis and cancer: mitochondria meet the “iron maiden” cell death. Cells. 2020;9:1505.
36. Lei P, Bai T, Sun Y. Mechanisms of ferroptosis and relations with regulated cell death: a review. Front Physiol. 2019;10:139.
37. Bersuker K, Hendricks JM, Li Z, et al. The CoQ oxidoreductase FSP1 acts parallel to GPX4 to inhibit ferroptosis. Nature. 2019;575:688-92.
38. Xiao T, Liang J, Li M, et al. ATG5-mediated keratinocyte ferroptosis promotes M1 polarization of macrophages to aggravate UVB-induced skin inflammation. J Photochem Photobiol B. 2024;257:112948.
39. Pourzand C, Albieri-Borges A, Raczek NN. Shedding a new light on skin aging, iron- and redox-homeostasis and emerging natural antioxidants. Antioxidants. 2022;11:471.
40. Kramer-Stickland K, Edmonds A, Bair WB 3rd , Bowden GT. Inhibitory effects of deferoxamine on UVB-induced AP-1 transactivation. Carcinogenesis. 1999;20:2137-42.
41. Wang K, Lin Y, Zhou D, et al. Unveiling ferroptosis: a new frontier in skin disease research. Front Immunol. 2024;15:1485523.
42. Gurung S, Perocheau D, Touramanidou L, Baruteau J. The exosome journey: from biogenesis to uptake and intracellular signalling. Cell Commun Signal. 2021;19:47.
43. Stoorvogel W, Kleijmeer MJ, Geuze HJ, Raposo G. The biogenesis and functions of exosomes. Traffic. 2002;3:321-30.
44. Joshi BS, de Beer MA, Giepmans BNG, Zuhorn IS. Endocytosis of extracellular vesicles and release of their cargo from endosomes. ACS Nano. 2020;14:4444-55.
45. Abels ER, Breakefield XO. Introduction to extracellular vesicles: biogenesis, RNA cargo selection, content, release, and uptake. Cell Mol Neurobiol. 2016;36:301-12.
46. Liu X, Wei Q, Lu L, et al. Immunomodulatory potential of mesenchymal stem cell-derived extracellular vesicles: targeting immune cells. Front Immunol. 2023;14:1094685.
47. Hajialiasgary Najafabadi A, Soheilifar MH, Masoudi-Khoram N. Exosomes in skin photoaging: biological functions and therapeutic opportunity. Cell Commun Signal. 2024;22:32.
48. Zhang H, Xiao X, Wang L, et al. Human adipose and umbilical cord mesenchymal stem cell-derived extracellular vesicles mitigate photoaging via TIMP1/Notch1. Signal Transduct Target Ther. 2024;9:294.
49. Tan F, Li X, Wang Z, Li J, Shahzad K, Zheng J. Clinical applications of stem cell-derived exosomes. Signal Transduct Target Ther. 2024;9:17.
50. Ahmadian S, Jafari N, Tamadon A, Ghaffarzadeh A, Rahbarghazi R, Mahdipour M. Different storage and freezing protocols for extracellular vesicles: a systematic review. Stem Cell Res Ther. 2024;15:453.
51. El Baradie KBY, Nouh M, O’Brien Iii F, et al. Freeze-dried extracellular vesicles from adipose-derived stem cells prevent hypoxia-induced muscle cell injury. Front Cell Dev Biol. 2020;8:181.
52. Liu Y, Wang Y, Yang M, et al. Exosomes from hypoxic pretreated ADSCs attenuate ultraviolet light-induced skin injury via GLRX5 delivery and ferroptosis inhibition. Photochem Photobiol Sci. 2024;23:55-63.
53. Wang Y, Shen X, Song S, et al. Evaluation of the effect of exosomes from adipose derived stem cells on changes in GSH/ROS levels during skin photoaging. Photodermatol Photoimmunol Photomed. 2025;41:e70009.
54. Gao W, Zhang Y, Yuan L, Huang F, Wang YS. Long non-coding RNA H19-overexpressing exosomes ameliorate UVB-induced photoaging by upregulating SIRT1 via sponging miR-138. Photochem Photobiol. 2023;99:1456-67.
55. Wang T, Jian Z, Baskys A, et al. MSC-derived exosomes protect against oxidative stress-induced skin injury via adaptive regulation of the NRF2 defense system. Biomaterials. 2020;257:120264.
56. Wu P, Zhang B, Han X, et al. HucMSC exosome-delivered 14-3-3ζ alleviates ultraviolet radiation-induced photodamage via SIRT1 pathway modulation. Aging. 2021;13:11542-63.
57. Zhang Y, Zhang M, Yao A, et al. Circ_0011129 encapsulated by the small extracellular vesicles derived from human stem cells ameliorate skin photoaging. Int J Mol Sci. 2022;23:15390.
58. Wu J, Li Z, Wu Y, Cui N. The crosstalk between exosomes and ferroptosis: a review. Cell Death Discov. 2024;10:170.
59. Santer L, Bär C, Thum T. Circular RNAs: a novel class of functional RNA molecules with a therapeutic perspective. Mol Ther. 2019;27:1350-63.
60. Holdt LM, Kohlmaier A, Teupser D. Circular RNAs as therapeutic agents and targets. Front Physiol. 2018;9:1262.
61. Zhang XO, Dong R, Zhang Y, et al. Diverse alternative back-splicing and alternative splicing landscape of circular RNAs. Genome Res. 2016;26:1277-87.
62. Jeck WR, Sorrentino JA, Wang K, et al. Circular RNAs are abundant, conserved, and associated with ALU repeats. RNA. 2013;19:141-57.
63. Hansen TB, Jensen TI, Clausen BH, et al. Natural RNA circles function as efficient microRNA sponges. Nature. 2013;495:384-8.
64. Li Y, Zheng Q, Bao C, et al. Circular RNA is enriched and stable in exosomes: a promising biomarker for cancer diagnosis. Cell Res. 2015;25:981-4.
65. Han Z, Chen H, Guo Z, et al. Circular RNAs and their role in exosomes. Front Oncol. 2022;12:848341.
66. Tenchov R, Sasso JM, Wang X, Liaw WS, Chen CA, Zhou QA. Exosomes-nature’s lipid nanoparticles, a rising star in drug delivery and diagnostics. ACS Nano. 2022;16:17802-46.
67. Kalluri R, LeBleu VS. The biology, function, and biomedical applications of exosomes. Science. 2020;367:eaau6977.
68. Welsh JA, Goberdhan DCI, O’Driscoll L, et al; MISEV Consortium. Minimal information for studies of extracellular vesicles (MISEV2023): from basic to advanced approaches. J Extracell Vesicles. 2024;13:e12404.
69. Baldarelli RM, Smith CL, Ringwald M, Richardson JE, Bult CJ; Mouse Genome Informatics Group. Mouse Genome Informatics: an integrated knowledgebase system for the laboratory mouse. Genetics. 2024;227:iyae031.
70. He GN, Bao NR, Wang S, Xi M, Zhang TH, Chen FS. Ketamine induces ferroptosis of liver cancer cells by targeting lncRNA PVT1/miR-214-3p/GPX4. Drug Des Devel Ther. 2021;15:3965-78.
71. Hou Y, Cai S, Yu S, Lin H. Metformin induces ferroptosis by targeting miR-324-3p/GPX4 axis in breast cancer. Acta Biochim Biophys Sin. 2021;53:333-41.
72. Ding C, Ding X, Zheng J, et al. miR-182-5p and miR-378a-3p regulate ferroptosis in I/R-induced renal injury. Cell Death Dis. 2020;11:929.
74. Alves F, Lane D, Nguyen TPM, Bush AI, Ayton S. In defence of ferroptosis. Signal Transduct Target Ther. 2025;10:2.
75. Zhang Y, Zhou F, Nie G, et al. 3D-cultured hADSCs-derived exosomes deliver circ_0011129 to synergistically attenuate skin photoaging. Front Genet. 2025;16:1627472.
76. Noronha NC, Mizukami A, Caliári-Oliveira C, et al. Priming approaches to improve the efficacy of mesenchymal stromal cell-based therapies. Stem Cell Res Ther. 2019;10:131.
77. Zhang M, Hu S, Liu L, et al. Engineered exosomes from different sources for cancer-targeted therapy. Signal Transduct Target Ther. 2023;8:124.
78. Hackel A, Vollmer S, Bruderek K, Lang S, Brandau S. Immunological priming of mesenchymal stromal/stem cells and their extracellular vesicles augments their therapeutic benefits in experimental graft-versus-host disease via engagement of PD-1 ligands. Front Immunol. 2023;14:1078551.
79. Yang R, Huang H, Cui S, Zhou Y, Zhang T, Zhou Y. IFN-γ promoted exosomes from mesenchymal stem cells to attenuate colitis via miR-125a and miR-125b. Cell Death Dis. 2020;11:603.
80. Chen YH, Dong RN, Hou J, et al. Mesenchymal stem cell-derived exosomes induced by IL-1β attenuate urethral stricture through Let-7c/PAK1/NF-κB-regulated macrophage M2 polarization. J Inflamm Res. 2021;14:3217-29.
81. Vu DM, Nguyen VT, Nguyen TH, et al. Effects of extracellular vesicles secreted by TGFβ-stimulated umbilical cord mesenchymal stem cells on skin fibroblasts by promoting fibroblast migration and ECM protein production. Biomedicines. 2022;10:1810.
82. Xu C, Zhai Z, Ying H, Lu L, Zhang J, Zeng Y. Curcumin primed ADMSCs derived small extracellular vesicle exert enhanced protective effects on osteoarthritis by inhibiting oxidative stress and chondrocyte apoptosis. J Nanobiotechnology. 2022;20:123.
83. Işıldar B, Özkan S, Koyutürk M. Preconditioning of human umbilical cord mesenchymal stem cells with a histone deacetylase inhibitor: valproic acid. Balkan Med J. 2024;41:369-76.
84. Rajendran RL, Gangadaran P, Oh JM, Hong CM, Ahn BC. Engineering three-dimensional spheroid culture for enrichment of proangiogenic miRNAs in umbilical cord mesenchymal stem cells and promotion of angiogenesis. ACS Omega. 2024;9:40358-67.
85. Rovere M, Reverberi D, Arnaldi P, Palamà MEF, Gentili C. Spheroid size influences cellular senescence and angiogenic potential of mesenchymal stromal cell-derived soluble factors and extracellular vesicles. Front Bioeng Biotechnol. 2023;11:1297644.
86. Song Y, Liang F, Tian W, Rayhill E, Ye L, Tian X. Optimizing therapeutic outcomes: preconditioning strategies for MSC-derived extracellular vesicles. Front Pharmacol. 2025;16:1509418.
87. Han Y, Jones TW, Dutta S, et al. Overview and update on methods for cargo loading into extracellular vesicles. Processes. 2021;9:356.
88. Almeida C, Teixeira AL, Dias F, Morais M, Medeiros R. Extracellular vesicles as potential therapeutic messengers in cancer management. Biology. 2023;12:665.
89. Liu Q, Li D, Pan X, Liang Y. Targeted therapy using engineered extracellular vesicles: principles and strategies for membrane modification. J Nanobiotechnology. 2023;21:334.
90. Haney MJ, Klyachko NL, Zhao Y, et al. Exosomes as drug delivery vehicles for Parkinson’s disease therapy. J Control Release. 2015;207:18-30.
91. Kooijmans SAA, Stremersch S, Braeckmans K, et al. Electroporation-induced siRNA precipitation obscures the efficiency of siRNA loading into extracellular vesicles. J Control Release. 2013;172:229-38.
92. Haraszti RA, Miller R, Didiot MC, et al. Optimized cholesterol-siRNA chemistry improves productive loading onto extracellular vesicles. Mol Ther. 2018;26:1973-82.
93. Huang T, Sato Y, Kuramochi A, et al. Surface modulation of extracellular vesicles with cell-penetrating peptide-conjugated lipids for improvement of intracellular delivery to endothelial cells. Regen Ther. 2023;22:90-8.
94. Alvarez-Erviti L, Seow Y, Yin H, Betts C, Lakhal S, Wood MJ. Delivery of siRNA to the mouse brain by systemic injection of targeted exosomes. Nat Biotechnol. 2011;29:341-5.
95. Delcayre A, Estelles A, Sperinde J, et al. Exosome Display technology: applications to the development of new diagnostics and therapeutics. Blood Cells Mol Dis. 2005;35:158-68.
96. Ohno S, Takanashi M, Sudo K, et al. Systemically injected exosomes targeted to EGFR deliver antitumor microRNA to breast cancer cells. Mol Ther. 2013;21:185-91.
97. Zhou Y, Yuan Y, Liu M, et al. Tumor-specific delivery of KRAS siRNA with iRGD-exosomes efficiently inhibits tumor growth. ExRNA. 2019;1:28.
98. Choi H, Yim H, Park C, et al. Targeted delivery of exosomes armed with anti-cancer therapeutics. Membranes. 2022;12:85.
99. Morishita M, Takahashi Y, Nishikawa M, et al. Quantitative analysis of tissue distribution of the B16BL6-derived exosomes using a streptavidin-lactadherin fusion protein and iodine-125-labeled biotin derivative after intravenous injection in mice. J Pharm Sci. 2015;104:705-13.
100. Susa F, Limongi T, Dumontel B, Vighetto V, Cauda V. Engineered extracellular vesicles as a reliable tool in cancer nanomedicine. Cancers. 2019;11:1979.
101. Liang Y, Duan L, Lu J, Xia J. Engineering exosomes for targeted drug delivery. Theranostics. 2021;11:3183-95.
102. Srivatsav AT, Kapoor S. The emerging world of membrane vesicles: functional relevance, theranostic avenues and tools for investigating membrane function. Front Mol Biosci. 2021;8:640355.
104. Ramadon D, McCrudden MTC, Courtenay AJ, Donnelly RF. Enhancement strategies for transdermal drug delivery systems: current trends and applications. Drug Deliv Transl Res. 2022;12:758-91.
105. Hmingthansanga V, Singh N, Banerjee S, Manickam S, Velayutham R, Natesan S. Improved topical drug delivery: role of permeation enhancers and advanced approaches. Pharmaceutics. 2022;14:2818.
106. Haykal D, Wyles S, Garibyan L, Cartier H, Gold M. Exosomes in cosmetic dermatology: a review of benefits and challenges. J Drugs Dermatol. 2025;24:12-8.
107. Yuan F, Li YM, Wang Z. Preserving extracellular vesicles for biomedical applications: consideration of storage stability before and after isolation. Drug Deliv. 2021;28:1501-9.
108. Yu H, Zhang J, Yang L, et al. MSC-derived exosomes injectable hyaluronic acid hydrogel for enhanced chronic wound healing. J Control Release. 2025;385:113985.
109. Prasai A, Jay JW, Jupiter D, Wolf SE, El Ayadi A. Role of exosomes in dermal wound healing: a systematic review. J Invest Dermatol. 2022;142:662-78.e8.
110. Di Rocco G, Baldari S, Toietta G. Towards therapeutic delivery of extracellular vesicles: strategies for in vivo tracking and biodistribution analysis. Stem Cells Int. 2016;2016:5029619.
111. Smyth T, Petrova K, Payton NM, et al. Surface functionalization of exosomes using click chemistry. Bioconjug Chem. 2014;25:1777-84.
112. Van Deun J, Mestdagh P, Sormunen R, et al. The impact of disparate isolation methods for extracellular vesicles on downstream RNA profiling. J Extracell Vesicles. 2014;3:24858.
113. Böing AN, van der Pol E, Grootemaat AE, Coumans FA, Sturk A, Nieuwland R. Single-step isolation of extracellular vesicles by size-exclusion chromatography. J Extracell Vesicles. 2014;3:23430.
114. Visan KS, Lobb RJ, Ham S, et al. Comparative analysis of tangential flow filtration and ultracentrifugation, both combined with subsequent size exclusion chromatography, for the isolation of small extracellular vesicles. J Extracell Vesicles. 2022;11:e12266.
115. Busatto S, Vilanilam G, Ticer T, et al. Tangential flow filtration for highly efficient concentration of extracellular vesicles from large volumes of fluid. Cells. 2018;7:273.
116. Kapoor KS, Harris K, Arian KA, et al. High throughput and rapid isolation of extracellular vesicles and exosomes with purity using size exclusion liquid chromatography. Bioact Mater. 2024;40:683-95.
117. Kawai-Harada Y, Nimmagadda V, Harada M. Scalable isolation of surface-engineered extracellular vesicles and separation of free proteins via tangential flow filtration and size exclusion chromatography (TFF-SEC). BMC Methods. 2024;1:9.
118. Haraszti RA, Miller R, Stoppato M, et al. Exosomes produced from 3D cultures of MSCs by tangential flow filtration show higher yield and improved activity. Mol Ther. 2018;26:2838-47.
119. Sharma A, Yadav A, Nandy A, Ghatak S. Insight into the functional dynamics and challenges of exosomes in pharmaceutical innovation and precision medicine. Pharmaceutics. 2024;16:709.
120. Wang CK, Tsai TH, Lee CH. Regulation of exosomes as biologic medicines: regulatory challenges faced in exosome development and manufacturing processes. Clin Transl Sci. 2024;17:e13904.
121. Baharlooi H, Azimi M, Salehi Z, Izad M. Mesenchymal stem cell-derived exosomes: a promising therapeutic ace card to address autoimmune diseases. Int J Stem Cells. 2020;13:13-23.
122. Imai T, Takahashi Y, Nishikawa M, et al. Macrophage-dependent clearance of systemically administered B16BL6-derived exosomes from the blood circulation in mice. J Extracell Vesicles. 2015;4:26238.
123. Wiklander OP, Nordin JZ, O’Loughlin A, et al. Extracellular vesicle in vivo biodistribution is determined by cell source, route of administration and targeting. J Extracell Vesicles. 2015;4:26316.
124. Gupta D, Wiklander OPB, Wood MJA, El-Andaloussi S. Biodistribution of therapeutic extracellular vesicles. Extracell Vesicles Circ Nucl Acids. 2023;4:170-90.
125. Lyu C, Sun H, Sun Z, Liu Y, Wang Q. Roles of exosomes in immunotherapy for solid cancers. Cell Death Dis. 2024;15:106.
126. Vakhshiteh F, Atyabi F, Ostad SN. Mesenchymal stem cell exosomes: a two-edged sword in cancer therapy. Int J Nanomedicine. 2019;14:2847-59.
127. Lin Z, Wu Y, Xu Y, Li G, Li Z, Liu T. Mesenchymal stem cell-derived exosomes in cancer therapy resistance: recent advances and therapeutic potential. Mol Cancer. 2022;21:179.
128. Welsh JA, van der Pol E, Bettin BA, et al. Towards defining reference materials for measuring extracellular vesicle refractive index, epitope abundance, size and concentration. J Extracell Vesicles. 2020;9:1816641.
129. Domaszewska-Szostek A, Krzyżanowska M, Polak A, Puzianowska-Kuźnicka M. Effectiveness of extracellular vesicle application in skin aging treatment and regeneration: do we have enough evidence from clinical trials? Int J Mol Sci. 2025;26:2354.
130. Park GH, Kwon HH, Seok J, et al. Efficacy of combined treatment with human adipose tissue stem cell-derived exosome-containing solution and microneedling for facial skin aging: a 12-week prospective, randomized, split-face study. J Cosmet Dermatol. 2023;22:3418-26.
131. Kwon HH, Yang SH, Lee J, et al. Combination treatment with human adipose tissue stem cell-derived exosomes and fractional CO2 laser for acne scars: a 12-week prospective, double-blind, randomized, split-face study. Acta Derm Venereol. 2020;100:adv00310.
132. Wan J, Kim SB, Cartier H, et al. A prospective study of exosome therapy for androgenetic alopecia. Aesthetic Plast Surg. 2025;49:3151-6.
133. Hu L, Wang J, Zhou X, et al. Exosomes derived from human adipose mensenchymal stem cells accelerates cutaneous wound healing via optimizing the characteristics of fibroblasts. Sci Rep. 2016;6:32993.
134. Lee JH, Won YJ, Kim H, et al. Adipose tissue-derived mesenchymal stem cell-derived exosomes promote wound healing and tissue regeneration. Int J Mol Sci. 2023;24:10434.
135. Qiu Z, Zhong Z, Zhang Y, Tan H, Deng B, Meng G. Human umbilical cord mesenchymal stem cell-derived exosomal miR-335-5p attenuates the inflammation and tubular epithelial-myofibroblast transdifferentiation of renal tubular epithelial cells by reducing ADAM19 protein levels. Stem Cell Res Ther. 2022;13:373.
136. Wang XY, Guan XH, Yu ZP, et al. Human amniotic stem cells-derived exosmal miR-181a-5p and miR-199a inhibit melanogenesis and promote melanosome degradation in skin hyperpigmentation, respectively. Stem Cell Res Ther. 2021;12:501.
137. Rodriguez C, Porcello A, Chemali M, et al. Medicalized aesthetic uses of exosomes and cell culture-conditioned media: opening an advanced care era for biologically inspired cutaneous prejuvenation and rejuvenation. Cosmetics. 2024;11:154.
138. Tienda-Vázquez MA, Hanel JM, Márquez-Arteaga EM, et al. Exosomes: a promising strategy for repair, regeneration and treatment of skin disorders. Cells. 2023;12:1625.
139. Wang M, Wu P, Huang J, et al. Skin cell-derived extracellular vesicles: a promising therapeutic strategy for cutaneous injury. Burns Trauma. 2022;10:tkac037.
140. Nasiri G, Azarpira N, Alizadeh A, Goshtasbi S, Tayebi L. Shedding light on the role of keratinocyte-derived extracellular vesicles on skin-homing cells. Stem Cell Res Ther. 2020;11:421.
Cite This Article
How to Cite
Download Citation
Export Citation File:
Type of Import
Tips on Downloading Citation
Citation Manager File Format
Type of Import
Direct Import: When the Direct Import option is selected (the default state), a dialogue box will give you the option to Save or Open the downloaded citation data. Choosing Open will either launch your citation manager or give you a choice of applications with which to use the metadata. The Save option saves the file locally for later use.
Indirect Import: When the Indirect Import option is selected, the metadata is displayed and may be copied and pasted as needed.
About This Article
Copyright
Data & Comments
Data





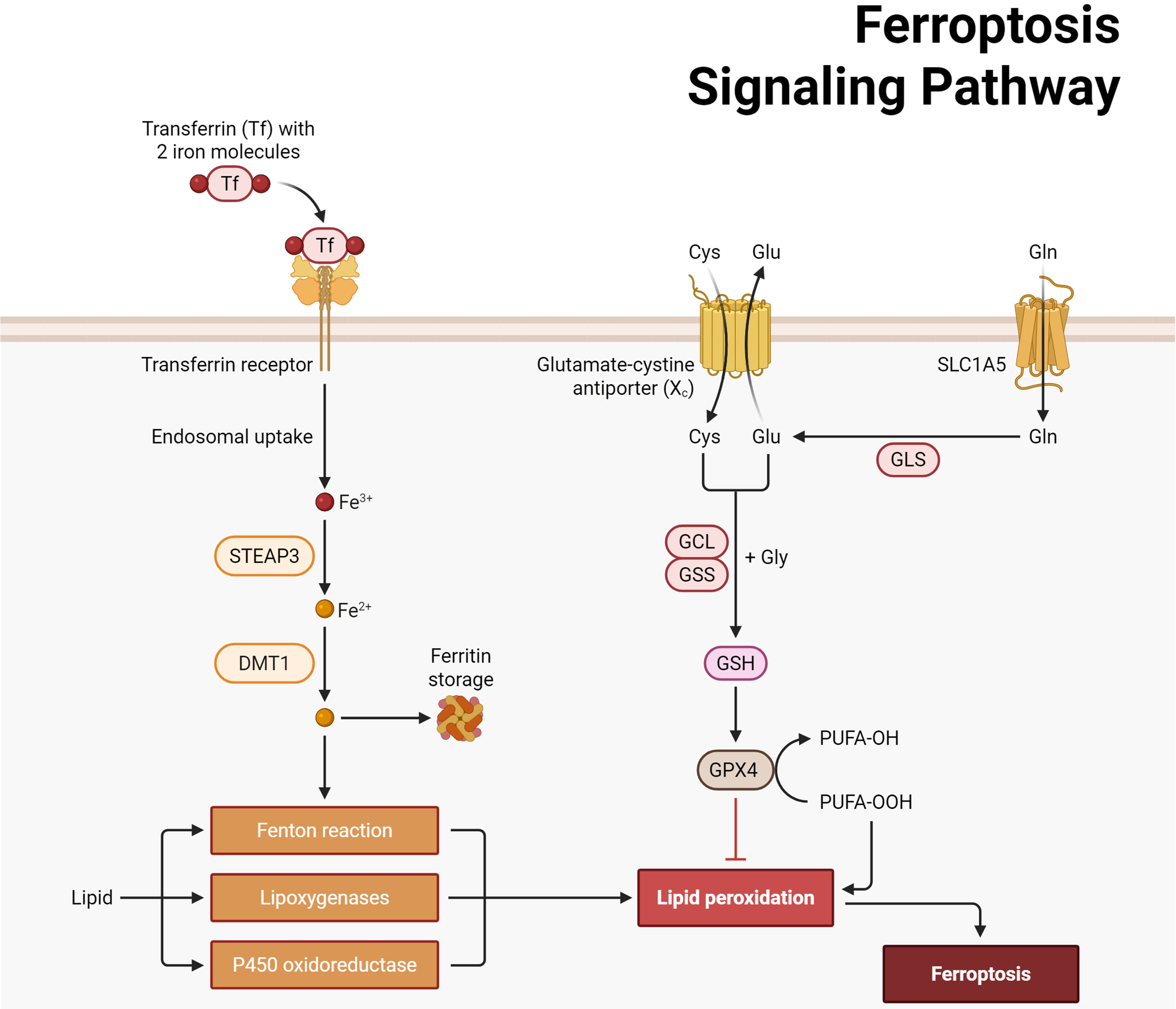
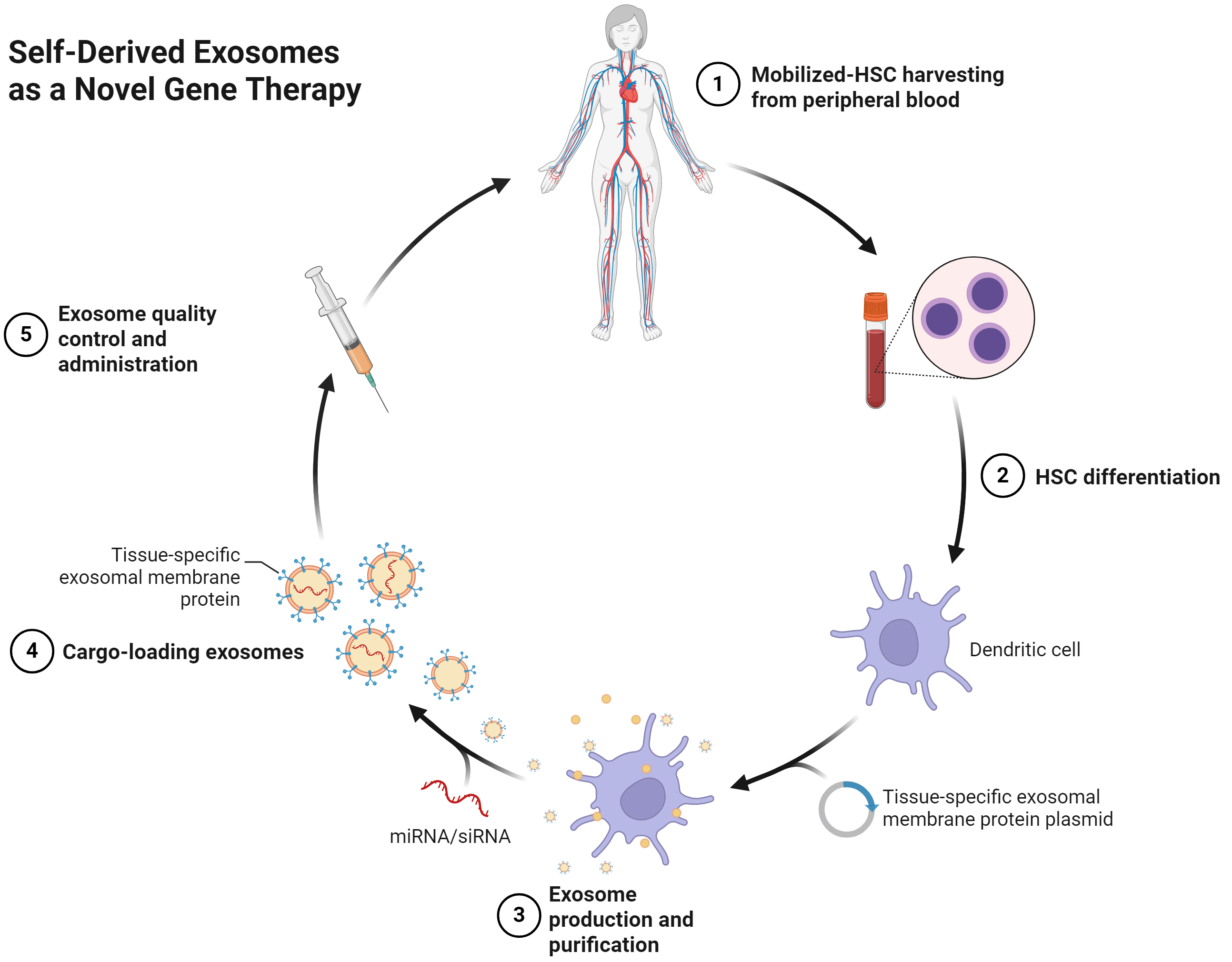
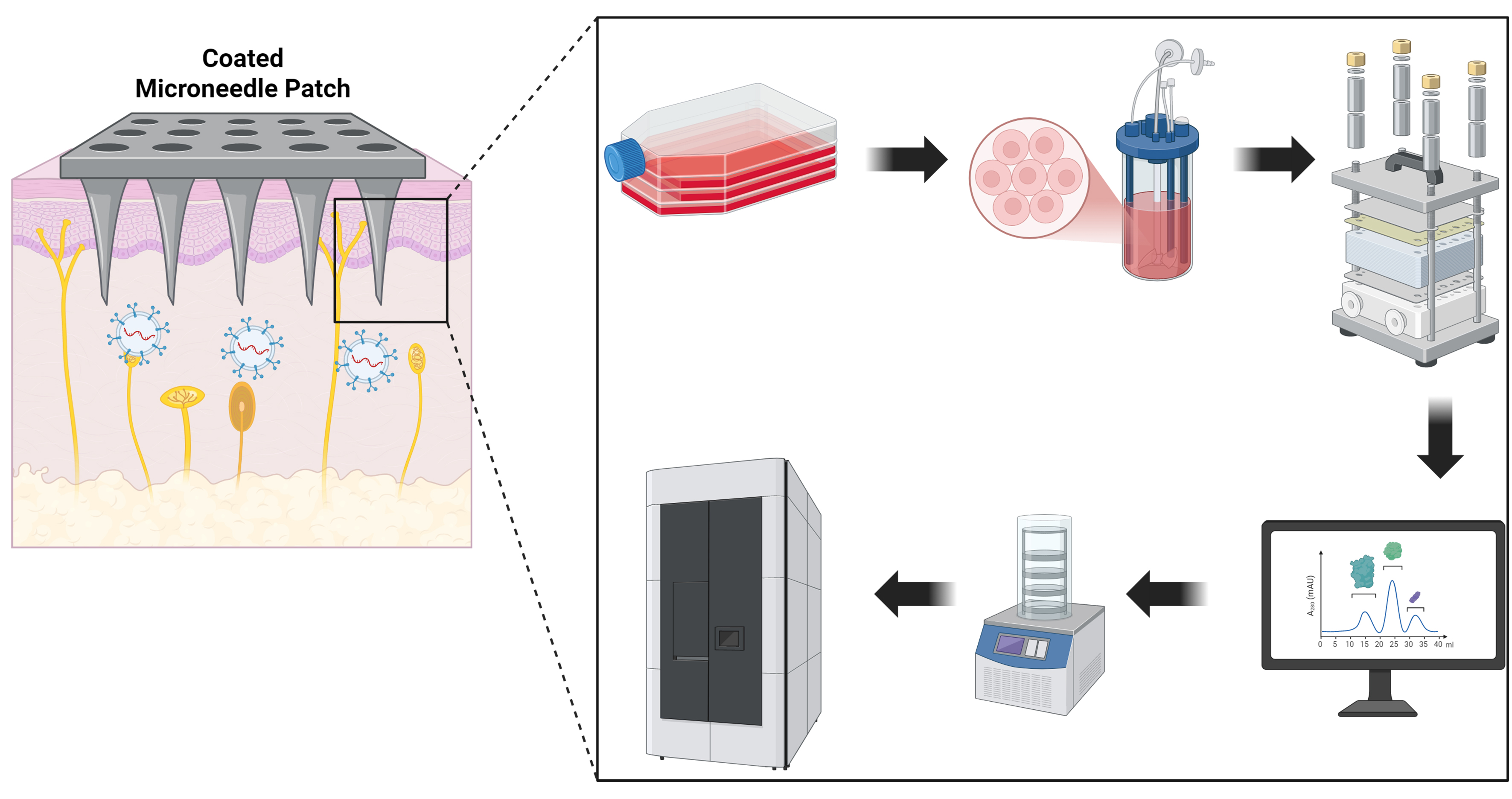











Comments
Comments must be written in English. Spam, offensive content, impersonation, and private information will not be permitted. If any comment is reported and identified as inappropriate content by OAE staff, the comment will be removed without notice. If you have any queries or need any help, please contact us at support@oaepublish.com.